Posts Tagged: grape
Tree and vine fall weed management
The 2023 post-harvest season is upon us and it's time to prep for your fall/winter weed control...
Study offers insights on reducing nitrate contamination from groundwater recharge
Light irrigation before flooding stimulates microbes to remove nitrates from soil
With California enduring record-breaking rain and snow and Gov. Gavin Newsom recently easing restrictions on groundwater recharge, interest in “managed aquifer recharge” has never been higher. This process – by which floodwater is routed to sites such as farm fields so that it percolates into the aquifer – holds great promise as a tool to replenish depleted groundwater stores across the state.
But one concern, in the agricultural context, is how recharge might push nitrates from fertilizer into the groundwater supply. Consumption of well water contaminated with nitrates has been linked to increased risk of cancers, birth defects and other health impacts.
“Many growers want to provide farmland to help recharge groundwater, but they don't want to contribute to nitrate contamination of the groundwater, and they need to know how on-farm recharge practices might affect their crops,” said Matthew Fidelibus, a University of California Cooperative Extension specialist in the UC Davis Department of Viticulture and Enology.
A recently published study by UC scientists sheds new light on how nitrates move through an agricultural recharge site and how growers might reduce potential leaching. Researchers analyzed data from two grapevine vineyards at Kearney Agricultural Research and Extension Center in Fresno County – one flooded for two weeks, and other for four.
Understanding initial nitrate levels crucial
A key factor in mitigating contamination is understanding how much nitrate is in the soil at the outset, said study author Helen Dahlke, a UC Davis hydrologist and leader of UC Agriculture and Natural Resources' strategic initiative on water. In areas with little precipitation and cropping systems that require greater amounts of synthetic fertilizer, the accumulation of residual nitrate – resulting from nitrogen in the fertilizer not taken up by the plants – can be quite high.
“The percentage of nitrates in some soils can really increase over the years, particularly if you have many dry years in a row where you don't have access to irrigation water or natural precipitation flushing some of those nitrates out of the soil,” Dahlke said.
While intense rains in recent weeks have helped dilute nitrate concentrations naturally, farmers looking to participate in recharge during the dry years ahead should consider flooding their fields with greater volumes of water.
“If you're doing this for the first time – on-farm recharge in the winter – check your residual soil nitrate levels because if they're very high, you should apply a lot of water in order to make sure that the residual nitrate is diluted down,” said Dahlke, who also added that growers should check their soil properties for suitability of recharge projects.
She recommended using, as a “good first approximation,” the online Soil Agricultural Groundwater Banking Index map, a project led by Toby O'Geen, a UC Cooperative Extension soil resource specialist.
Researchers looking at other ways to reduce nitrates
Even before flooding the fields for recharge, there are several practices that can lower initial nitrate levels and risk of leaching. Cover crops such as alfalfa and triticale, for example, can help take up residual nitrates that accumulate from fertilizing a main crop over time.
Dahlke and Fidelibus – a co-author of the San Joaquin Valley vineyard study – both pointed to pre-flooding irrigation that encourages denitrification, a process in which soil microbes transform nitrates into gaseous forms of nitrogen.
“Those denitrifying microbes need to be stimulated to do the work,” said Dahlke. “What we have found is that if you do a little bit of irrigation before you start the flooding, increasing the soil moisture can get those microbes started and they can take out more nitrate from the soil.”
The timing and quantity of fertilizer applications are also major factors in reducing leaching. Although more growers are following high-frequency, low-concentration practices to maximize uptake by crops, Dahlke said there needs to be more emphasis on incorporating nitrogen transformation processes – such as denitrification – in the nutrient management guidelines that farmers follow.
“Implementing thoughtful nutrient management plans will play a particularly important role in participating farms,” Fidelibus added.
A more holistic view of groundwater recharge
In short, choices made during the growing season can affect those in the winter recharge season – and vice versa. For example, applying compost or other organic amendments to soil can give microbes the “fuel” they need for sustained denitrification.
“What we have found is that our denitrifying bacteria often run out of steam because they don't have enough carbon to do the work,” Dahlke said. “Like us, microbes need energy to do the work, and for microbes this energy comes from soil carbon.”
Then, adding moisture via recharge to that field with high organic content can stimulate mineralization and nitrification, processes in which microbes transform the organic nitrogen into ammonium – and subsequently nitrates – that the plants can then take up. Those naturally occurring nitrates would thus reduce the need for the grower to apply synthetic fertilizer.
“The winter on-farm recharge experiments have shown that altering the moisture regime in the winter has consequences for the nitrogen budget in the summer growing season,” Dahlke explained. “Theoretically, what we need to be doing is better integrating both seasons by keeping an eye on the soil-nitrogen balance across the whole year so that we can ensure, at the end of the growing season, the residual nitrate in the soil is minimized.”
The study, published in the journal Science of The Total Environment, was part of the post-doctoral work of former UC Davis researcher Elad Levintal. In addition to Fidelibus and Dahlke, other authors are Laibin Huang, Cristina Prieto García, Adolfo Coyotl, William Horwath and Jorge Rodrigues, all in the Department of Land, Air and Water Resources at UC Davis.
The Achemon Sphinx Moth: A Lovely Beast, Indeed
It is indeed a “lovely beast,” as lepidopterist Art Shapiro, UC Davis...

Eumorpha achemon, the Achemon Sphinx, is a "lovely beast," says UC Davis distinguished professor Art Shapiro. Ann Sievers, owner, grower and miller Il Fiorello Olive Oil Co., recently found this one the wall of her patio. (Photo by Ann Sievers)

What's for lunch? A chicken in one of Ann Sievers' flocks at Il Fiorello Olive Oil Co., eyes the photographer. Chickens eagerly eat lots of bugs--if they're available. The larvae of the Achemon Sphinx moth feed on grape leaves. (Photo by Kathy Keatley Garvey)

Naturalist-photographer Greg Kareofelas took this image of an Eumorpha achemon larva in his yard in Davis several years ago. It was feeding on native grape, Vitus californica. (Photo by Greg Kareofelas)

An adult Eumorpha achemon, photographed by naturalist-photographer Greg Kareofelas in his yard in Davis several years ago. It was feeding on native grape, Vitus californica. (Photo by Greg Kareofelas)
Grapes in Distress
Advice for the Home Gardener from the Help Desk of the
UC Master Gardener Program of Contra Costa County
Help Desk Request: I'm delighted you're available to help diagnose garden problems! Below are four pictures of a grape leaf problem. I have four old vines - Flame and Thompson seedless - that grow against a south-facing solid fence. The vines are close enough that they are co-mingled across the fence. The problem currently is located in one area of the vines and was not visually noticeable three weeks ago. All the vines are vigorous and seem healthy except for the affected leaves. I have cut most of the leaves and vines that are visibly distressed and placed them in my green waste bin. In the 15+ years I've had the grapes I have never had this problem; however, this year I was VERY, VERY late in pruning back the vines, and did not complete the pruning until the new vines had grown a couple feet long. Perhaps the evil villain over-wintered because of my tardy pruning?
Any idea what the problem is - fungus, bug or insect critter, bacteria? I have not seen anything crawling on either side of the leaves. And, what is your recommended approach and treatment? Thanks in advance for your help!
Submitted Representative Images of Affected Grape Leaves (below)




MGCC Help Desk Response: Thank you for contacting the UC Master Gardener Help Desk with your issue of ‘bumps and holes' on your grape leaves.
Thank you for the pictures and note that the blister is on the top of the leaf and the bottom shows fuzzy white. We were able to diagnose simply from the damage itself. The bumps are the result of mites – specifically grape erineum mites. The good news here is that even though the mites infest the leaves, no management is necessary as they rarely lead to any crop losses.
Erinium mites are wormlike, light yellowish white and microscope so you really would need magnification to see the actual mites themselves.
Young leaves show bright pinkish or reddish swellings on upper surfaces. Corresponding areas on lower surfaces are concave and densely lined with a felty mass of plant hairs
The mites overwinter under outer bud scales and move to unfolding leaves in spring. They associate in small groups to feed on lower leaf surfaces; the result is production of masses of enlarged leaf hairs inside a blisterlike area on the leaf. On the undersides of the leaves, beneath the swellings, are concave, densely lined, felty masses of oversized leaf hairs in which the mite populations develop. As the population increases, some move to new areas or to other leaves. From mid-August to leaf drop, there is a movement back to the overwintering site underneath the bud scales.
You indicated that this is new to you and that you were a bit late pruning. I cannot explain for sure why this year and not prior years and I cannot be sure the late pruning added to this situation.
Following is a link to a UC Pest Management website on the subject but it does not really add to the information I have provided above. http://ipm.ucanr.edu/PMG/GARDEN/FRUIT/PESTS/grerineummte.html
Good luck and enjoy those grapes, and do not hesitate to contact us again with garden problems.
Help Desk of the UC Master Gardener Program of Contra Costa County (EDC)
Note: UC Master Gardeners Program of Contra Costa's Help Desk is available almost year-round to answer your gardening questions. Except for a few holidays (e.g., last 2 weeks December), we're open every week, Monday through Thursday for walk-ins from 9:00 am to Noon at 2380 Bisso Lane, Concord, CA 94520. We can also be reached via telephone: (925) 608-6683, email: ccmg@ucanr.edu, or on the web at http://ccmg.ucanr.edu/Ask_Us/. MGCC Blogs can be found at http://ccmg.ucanr.edu/HortCoCo/ You can also subscribe to the Biog.
Biorational control options for the western grapeleaf skeletonizer, a re-emerging pest in California
The western Grapeleaf skeletonizer (WGLS), Harrisina metallica Stretch (Lepidoptera: Zygaenidae), previously known to cause severe defoliation to vineyards and backyard grapevines appears to be re-emerging in California. Since its first detection in San Diego in 1941, WGLS spread through commercial vineyards and backyard grapes becoming a serious problem. Although two biological control agents from Arizona and Mexico were introduced in California for WGLS control, a naturally occurring granulovirus (Harrisina brillians granulovirus) nearly eradicated WGLS populations and kept them under control. WGLS has not been a problem especially in conventional vineyards. However, based on some unpublished observations, WGLS populations are emerging in organic vineyards and backyard grapevines.
WGLS lives up to its name by skeletonizing and defoliating grape leaves. Organic vineyards are especially at risk and uncontrolled populations can destroy vineyards resulting in significant losses. Metallic bluish or greenish black moths lay barrel shaped yellowish eggs on the lower side of the leaves. There are five larval instars. Early instars are cream colored and develop black and purple bands in later stages. Pupation occurs in a whitish cocoon. Upon hatching, larvae start feeding side by side in a row on the lower side of leaf. Damage by younger larvae appears as whitish leaf area containing veins and the upper cuticle, which eventually turn brown. Older larvae skeletonize leaves leaving larger veins. Larvae may also feed on fruit leading to bunch rot. Severe damage can cause defoliation and sunburn of the exposed fruit.
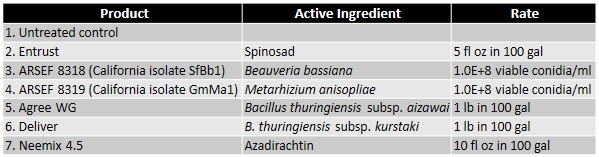
Methodology
A study was conducted to evaluate the efficacy of six non-chemical control options that included formulations of spinosad, two subspecies of Bacillus thuringiensis, and a botanical insecticide/growth regulator along with two unformulated entomopathogenic fungal isolates native to California. Larvae were collected from an infested, untreated backyard grapevine and maintained in one gallon plastic tubs with screened lids on infested leaves. Fresh, untreated grape leaves from uninfested vines were provided daily for 3 days before starting the assay. For each treatment, five 4-5 instar larvae were placed on a grape leaf disc (rinsed in water and dried) in a Petri plate (100 mm dia) with a moist filter paper. Larvae were treated by spraying 1 ml of the treatment solution (containing Dyne-Amic as a surfactant at 0.125% vol/vol). Application rates for commercial formulations were determined based on label recommendations for 100 gallons of spray volume. Entomopathogenic fungal concentrations were also determined based on the label rates for similar commercial products. Treatments were replicated four times and the assay was conducted twice. Larval mortality was observed daily and dead larvae were removed and incubated separately. Fresh leaf discs were provided as needed to the remaining larvae. Actual and corrected (for control mortality) total mortality were calculated.Data were arcsine-transformed for statistical analysis and significant means were separated using Tukey's HSD test.
Results
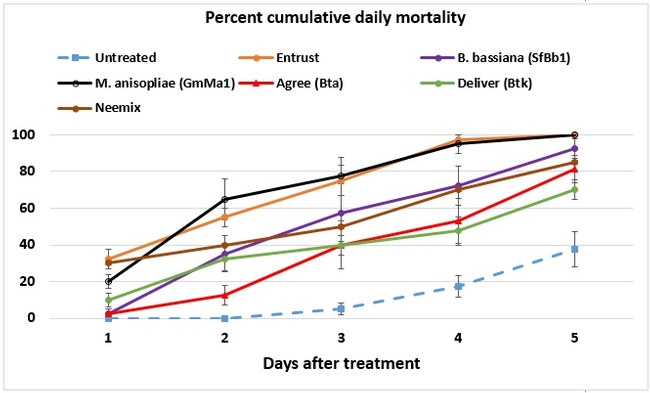
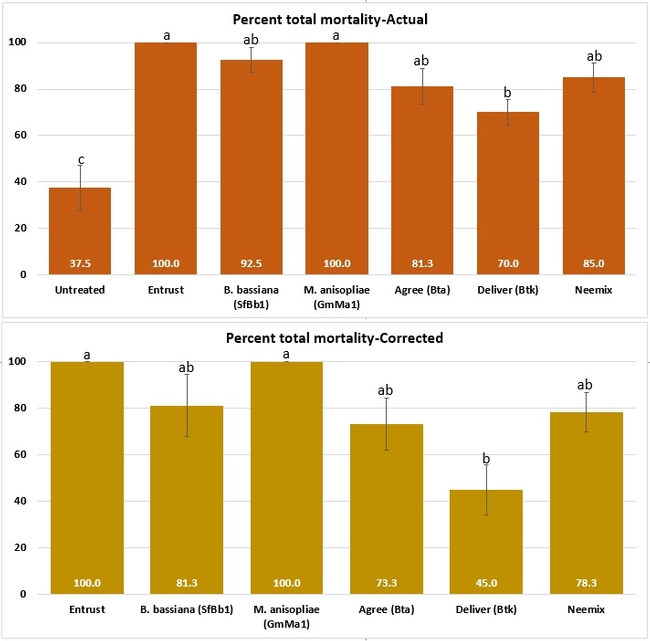
Both cumulative daily mortality and total mortality significantly (P < 0.0001) differed among treatments. Entrust and M. anisopliae resulted in the highest mortality followed by B. bassiana, Neemix, and Agree. In general, feeding reduced or ceased in all larvae following treatment and could have contributed to a lower mortality in B. thuringiensis treatments. Entomopathogenic fungi emerged from all the cadavers from respective treatments. Microbial and botanical options provided good control of WGLS. These non-chemical alternatives can be effectively used in both organic and conventional vineyards. California isolates of B. bassiana and M. anisopliae demonstrated good control efficacy and the potential to be developed as microbial pesticides.
Acknowledgements: Thanks to the technical assistance of Alor Sahoo in carrying out these assays, and Certis and Corteva for providing the pesticide formulations.
References
Federici, B. A. and V. M. Stern. 1990. Replication and occlusion of a granulosis virus in larval and adult midgut epithelium of the western grapeleaf skeletonizer, Harrisina brillians. J. Invertebr. Pathol. 56: 401-414.