- Author: Ben Faber
Soil Biodiversity in California Agriculture: Framework
and Indicators for Soil Health Assessment
Prepared by: California Department of Food and Agriculture Below Ground Biodiversity Advisory Committee
Soil health depends on soil biodiversity.
However, external pressures from land-use change, climate change and certain agricultural practices threaten the biotic networks that underpin the delivery of soil's many ecosystem services. Yet measuring soil biodiversity is a complex task, with a wide variety of possible indicators, and methodologies that are evolving with recent technological advances. This report, prepared by the Belowground Biodiversity Advisory Committee (BBAC) convened by the California Department of Food and Agriculture (CDFA), focuses on how best to assess soil biodiversity in the context of working lands and considers current and future challenges faced by California agricultural producers, policy makers, governing agencies, and related stakeholders. The report presents information on the taxonomic and functional diversity of soil organisms, ecosystem services they provide, threats to soil biodiversity, assessment frameworks, and biodiversity indicators. Examples of how biodiversity indicators can be applied to specific use cases provide insights for soil health, sustainable and climate-smart agriculture, and biodiversity conservation in California.
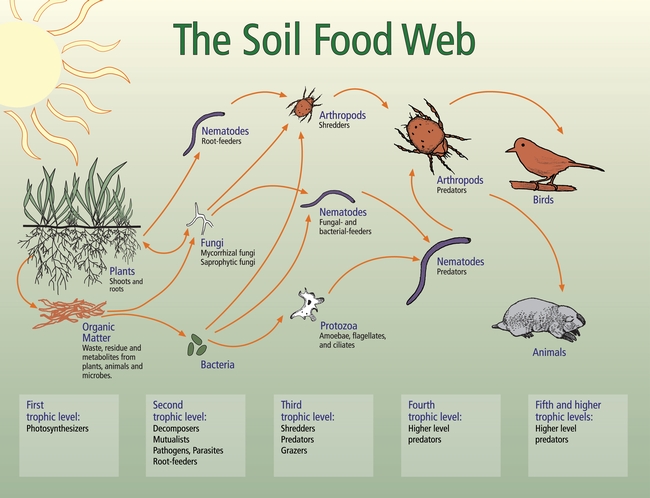
Soil biodiversity is the interconnected ‘social' network of numerous species of living organisms that contribute to soil functioning. As these organisms grow, die, and interact with soil's abiotic components, they perform essential functions in carbon, water and nutrient cycling and plant growth, collectively described as multifunctionality, benefiting ecosystems and humans alike. Comprehensive assessment of soil biodiversity involves measurements of organism abundance, identity, and functional diversity or traits, ideally in tandem with measurements of soil processes, as well as interactions among organisms. Soil biodiversity and soil processes vary in space and time due to factors like location, climate, vegetation, and land management practices across California's diverse landscapes.
Soils are incredibly biodiverse habitats, containing a vast array of organisms ranging from macroscopic organisms like gophers to microscopic worms, fungi, and billions of bacterial cells. The physical and chemical properties of soils – soil texture, pH, water and oxygen content, salinity, organic matter inputs, and nutrients – determine the types of organisms found in a particular habitat. The array of organisms inhabiting soil spans over six orders of magnitude in size, and includes microorganisms (viruses, bacteria, archaea, and fungi); microfauna (protists, nematodes, and tardigrades); mesofauna (mites and springtails); and macrofauna (earthworms). Life in soil exists in ecological communities that are complex and interconnected. These interconnections provide stability to soil functions. Soil organisms are critical to regulation of greenhouse gases, both by consuming and producing gases such as nitrous oxide, carbon dioxide, and methane. Mycorrhizal fungi in symbiosis with most plant species promotes root growth and availability of water and nutrients. A broad range of soil organisms mediate the decomposition of organic inputs and enhance nutrient cycling. Other functions of biodiverse soils include soil structure formation, organic matter formation, carbon storage, water regulation, and pathogen suppression. But despite these critically important functions, the diversity and complexity of soil biodiversity makes it challenging to decipher these intricate relationships and understand the impact of human activities.
Soil biodiversity faces many of the major threats from human activities and global change that also impact soil health and sustainability of California's agroecosystems. Land use changes, intensive agriculture, climate change, pollution, invasive species, overexploitation, and loss of habitat connectivity all pose risks. These threats disrupt soil biological networks, reduce biodiversity, impair ecosystem functions, and degrade soil structure and fertility. Soil biodiversity loss reduces multifunctionality and the provision of ecosystem services, highlighting the need to recognize the value of belowground communities to overcome challenges such as climate change, land degradation, and overall biodiversity loss. Addressing these challenges through sustainable land management, agroecological approaches, and awareness campaigns is crucial for preserving belowground biodiversity to maintain provision of essential ecosystem services.
READ ALL ABOUT SOIL DIVERSITY in the Report:
https://www.cdfa.ca.gov/oefi/biodiversity/docs/Soil_Biodiversity_California_Ag_July_2023.pdf
- Author: Ben Faber
I am frequently asked if I can recommend a book on Soils. And yes, I can. It is Soils: An Introduction by Michael Singer and Donald Munns. The sixth edition recently came out so there's a lot of older used copies floating around on the wed for under $10. This book takes a different tack on describing soils. Instead of tacking the tack of a chapter on Nitrogen another on Calcium etc., it weaves a story of how all the parts are related.
- Author: Ben Faber
CDFA has released a report on Belowground Biodiversity which provides targeted recommendations for policymakers and stakeholders to enhance soil health and conserve biodiversity, which plays an important role in climate resilience and food production.
An advisory committee of soil scientists authored the report, led by Dr. Kate Scow, Distinguished Professor Emeritus of Soil Science and Microbial Ecology at UC Davis: Dr. Eoin Brodie, Deputy Director of the Climate and Ecosystem Sciences Division at Lawrence Berkeley National Laboratory; and Dr. Margaret Smither-Kopperl, Manager, Lockeford Plant Materials Center, United States (US) Department of Agriculture, Natural Resources Conservation Service..
“The State of California contains more than 2,500 types of soil – they host a wealth of life that makes vital contributions to the global food supply,” said Dr. Scow. “Improved understanding of belowground biodiversity is essential to achieving a carbon-neutral future and tackling climate change.”
The report provides recommendations including using soil biodiversity as a key metric for assessing and prioritizing soil health and multi-functionality, integrating a soil biodiversity assessment into CDFA's Healthy Soils Program, and developing an adaptive management framework that incorporates soil biodiversity assessment.

- Author: Ben Faber
I am frequently asked if I can recommend a book on Soils. And yes, I can. It is Soils: An Introduction by Michael Singer and Donald Munns. The sixth edition recently came out so there's a lot of older used copies floating around on the wed for under $10. This book takes a different tack on describing soils. Instead of tacking the tack of a chapter on Nitrogen another on Calcium etc., it weaves a story of how all the parts are related.

- Author: Ben Faber
The Soil Science Society of America (SSSA) started a blog several years ago that covers all aspects of soils from water to organic matter to soil temperature to soil formation to building a retaining wall to ………..the list goes on and on. Something to read on a rainy day, if that ever comes.
The top posts are:
- What do nutrients do for plants?
- What are alluvial soils?
- What type of soil is good for a foundation for buildings or houses?
- When does rock become soil?
- Why do farmers use fertilizers?
- How does soil moisture impact our lives?
- What are sources of phosphorus for crops?
- What is soil temperature?
- How long does soil take to form?
- How can I fix my falling retaining wall?
But it doesn't stop there. Check out the site and sign up for the blogs by email when they become available.




