- Author: Michael D Cahn, Ph.D.
The California Irrigation and Management Information System (CIMIS) operates and maintains more than 145 weather stations throughout California. The CIMIS program is funded by the California Department of Water Resources. Most stations are located on or near agricultural land, and provide measurements of reference evapotranspiration (ETo), which can be used to estimate how much water to apply to crops. Hourly, daily, and monthly averages of data are available through the CIMIS web site (http://www.cimis.water.ca.gov). The website includes an option to automatically email data from selected stations on a daily or weekly schedule. In addition to ET data, CIMIS stations record precipitation, relative humidity, air and soil temperature, solar radiation, wind speed, and dew point. Besides irrigation management, weather data can be used for plant disease forecasts, for calculating insect and crop degree-days, and for determining wind speeds during spray operations. Water management agencies use historical reference ET data to determine pumping demands, and to estimate future ground water supplies using mathematical computer simulation models.
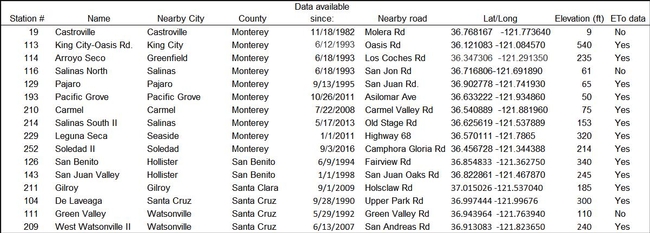
The Central Coast region currently has 16 active CIMIS stations (Table 1). Stations are located in Monterey, San Benito, Santa Cruz, Santa Clara counties. The newest station is #252 (Figure 1), located near Soledad CA on the east side of the Salinas Valley. CIMIS is a cooperative program, requiring collaboration between a local entity to provide land, maintain the site, and provide periodic servicing of the station. In some cases, stations are owned by the CIMIS program, but in many situations the weather stations are purchased through grant funding obtained by a local agency. Funding for station 252 was from a proposition 84 grant administered by the Coastal Conservation and Research Inc. The installation of the station was a partnership among Monterey County Resource Conservation District, UC Cooperative Extension (Monterey County), Dole Food Company Inc., CIMIS, Monterey Bay National Marine Sanctuary, and the Central Coast Wetlands Group (CCWG). Because CIMIS stations need to be sited on well-watered grass to provide accurate estimates of reference ET, funds from this grant were also used to establish 2-acres of grass surrounding the weather station and to install an underground sprinkler system.
Table 1. CIMIS stations located in the Central Coast region.
Figure 1. CIMIS station 252 located near Camphora-Gloria Rd, Soledad CA.
Even if a ranch has a private weather station, CIMIS data can still be useful. Many private weather stations are not instrumented to monitor ET or are not sited on a well-watered reference crop. Frequently private weather stations are located near a building, parking lot, or tree that can confound micro-climatic measurements. Over time instrumentation on weather stations can malfunction and record inaccurate data. The CIMIS system uses both an automated and manual quality assurance program to flag data that appear inaccurate or outside the normal range. CIMIS staff also service and check that the instruments are working properly. Additionally, CIMIS data are archived so that historical data can be accessed by users. CIMIS weather station data also contributes to Spatial CIMIS, a hybrid ET product that uses weather station and satellite data to provide reference ET estimates at approximately a 1-mile resolution. Spatial CIMIS reference ET data are also available through the CIMIS website.
During the past decade, significant progress has been made in adding new CIMIS weather stations or revitalizing old stations with improved site maintenance on the Central Coast. In addition to the Soledad station, Station 209 was established in West Watsonville, Station 211 was installed in Gilroy, and Station 210 was located in the Carmel Valley. Permanent grass was planted at station 129 in Pajaro and also at station 214 in South Salinas. Although progress has been made to increase the number and accuracy of CIMIS stations, weather stations are still lacking in some important Central Coast growing regions. Closer to the coast, the Castroville (#19) and North Salinas (#116) Stations are no longer reporting reference ET because the sites do not have sufficient grass cover to accurate measure ET. Also, the Green Valley road station (#111), which represents a warmer zone of the Pajaro valley no longer reports reference ET data due to insufficient grass cover at the site.
Having reliable long-term weather data from the main growing regions on the Central Coast is becoming more important for our region. As water demands continue to increase on the Central Coast, the agriculture community is under increased pressure to demonstrate efficient irrigation practices. Online irrigation scheduling tools such as CropManage and the Satellite Irrigation Management Support (SIMS), use CIMIS data to help growers quickly determine crop water needs. These tools can also help growers justify water needs of their crops. To comply with the Sustainable Groundwater Management Act (SGMA), water management agencies will need accurate reference ET data for developing ground water extraction plans. My hope is that through partnerships among local and state agencies, private land owners, and grower groups, we can add new or revitalize existing CIMIS stations so that all growing regions on the Central Coast have accurate weather data.
- Author: Michael D Cahn, Ph.D.
Agricultural Order Public Scoping Meeting
Monday, August 7th 9 am – 11:30 am
Monterey County Agriculture Conference Room
1432 Abbott St.
Salinas CA
The current Agricultural Order which regulates water quality for irrigated agriculture on the Central Coast will expire on March 8, 2020. Ahead of this deadline, The Central Coast Regional Water Quality Control Board (CCRWQCB) staff must develop a new Agricultural Order. Water Quality Control Board members and staff will be hosting several listening sessions where attendees will have the opportunity to share their perspective and experiences regarding the pros, cons, and ideas for potential improvement of the Ag Order. Discussion topics will include the structure of the Ag Order, such as tiering and enrollment, as well as monitoring and reporting requirements.
The current agenda is:
1. Current Agricultural Order Staff will present a summary. Public will have the opportunity to comment on the current Ag Order structure, including enrollment; tiering; electronic notice of intent form; annual compliance form.
2. Water Quality Monitoring Public will have the opportunity to comment on the surface water and groundwater monitoring requirements; cooperatives conducting monitoring such as the Preservation Inc.'s Cooperative Monitoring Program and the Central Coast Groundwater Coalition.
3. Total Nitrogen Applied Public will have the opportunity to comment on the total nitrogen applied requirement.
5. Tier 3 Ranch Requirements Public will have the opportunity to comment on Ag Order requirements for tier 3 ranches, including individual surface water discharge monitoring; irrigation and nutrient management plan reporting; water quality buffer plans.
6. Missing Requirements Public will have the opportunity to comment on the requirements they deem are necessary but currently not in the Ag Order.
A quorum of Central Coast Water Board members may be present; however, the Central Coast Water Board will not be taking any action at the scoping meeting.
The meeting will be conducted in English, Spanish translation will be provided for attendees sharing a comment in Spanish.
Esta reunión se va a llevar a cabo en inglés, pero va a haber traducción al español disponible para aquellas personas que quieran dar testimonio en español.
If you cannot attend this meeting, there will be another opportunity in Watsonville at the Public library on August 15th. Please see the attached flyers for more information.
Flyer Spanish
Flyer English
- Author: Laura Tourte
- Author: Richard Smith
- Author: Jeremy Murdock
- Author: Daniel A Sumner
Studies that examine crop production and harvest costs, as well as capital costs and net returns, can be helpful to growers in making production decisions, preparing budgets, and evaluating production loans. Cost and return analyses can also assist growers in considering risk and in determining the potential profitability of a crop. Two recently completed studies estimating costs and returns for Central Coast wrapped iceberg lettuce and fresh market bunched broccoli are now available at https://coststudies.ucdavis.edu. Titled Sample Costs to Produce and Harvest Iceberg Lettuce 2017 and Sample Costs to Produce and Harvest Broccoli 2017, these studies were a collaborative effort that included UC Cooperative Extension farm advisors, local growers and industry, and UC Agricultural Issues Center economists and researchers.
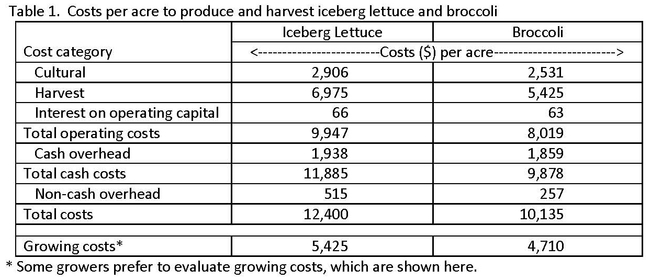
Each study begins with a narrative that describes how the costs and returns were calculated. Following this text, an in-depth analysis of the estimated production and harvest costs are presented in a series of tables. Per acre costs are shown for land preparation, fertilization and pest management practices, and material inputs and labor, all of which fall under the category cultural costs. Harvest practices and costs are also detailed. Cash or business overhead costs are estimated, and include items such as land rent, insurance, food safety and regulatory programs. Finally, non-cash or investment costs are discussed and included in each study. Table 1 summarizes per acre costs for iceberg lettuce and broccoli. Greater detail on the sequence of operations and costs contained in each broad cgory can be found in the studies.
Monthly cash cost tables are presented in each study, which can be helpful in managing cash flow and understanding when production loans may be needed. In addition, the tables can be used to evaluate months with peak labor demands and assist with scheduling.
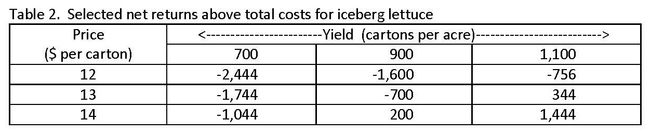
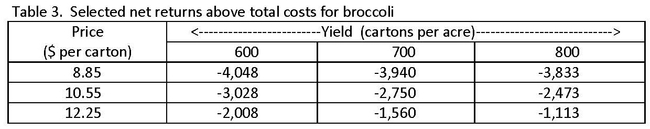
A ranging analysis is also included in each study, which estimates net returns per acre above operating, cash, and total costs for several alternative yield and price combinations. For lettuce, net returns are calculated for yields ranging from 600 to 1,200 cartons per acre (24 film wrapped heads weighing 42 pounds) and prices ranging from $9 to $17 per carton. For broccoli, net returns are estimated using a yield range of 550 to 850 cartons per acre (14 bunches weighing 21 pounds) and prices ranging from $8 to $13.10 per carton. Tables 2 and 3 below contain a selection of the estimated net returns above total costs for lettuce and broccoli.
At the lower yields and prices for iceberg lettuce (table 2), growers operate at a net loss per acre. As yields and prices improve, however, there is potential for a modest positive net return per acre. Growers of bunched broccoli operate at a net loss per acre regardless of yield or price scenario (table 3). The loss is smaller as yields and prices improve. Though net returns to growers can vary substantially from crop to crop and depend on many production and market conditions, the results from both studies—most notably for bunched broccoli—show that there is currently substantial pressure for growers to increase net returns. To improve profitability, growers may use different pack types and market strategies. High labor costs and low product prices, at least in part, help explain the results shown here. An expanded analysis can be found in each study.
Although technology has begun to play a larger role in the production and harvest of fresh market crops than it has in the past, a reliable and productive labor force is still critical to bring a crop to market. Labor costs are discussed and calculated for each crop, with hourly wages set at $21.85 for equipment operators, $17.80 for irrigators, and $16.90 for field workers. These rates include overhead costs of 41 percent. Irrigation, weed management, and harvest are prominent “high cost” labor practices.
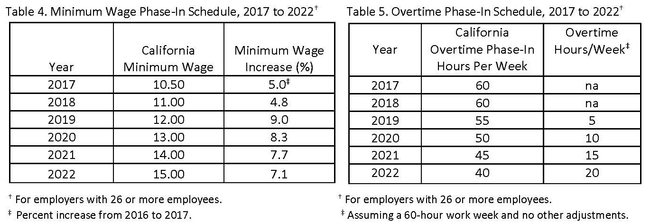
New minimum wage and overtime laws that were passed in California in 2016 will have significant implications for agriculture. Although wage rates used in the studies are already higher than the minimum wage shown below for 2017, tables 4 and 5 provide information on the phase-in schedules for the new laws.
As table 4 indicates, minimum wage will steadily increase each year, reaching a $15 base wage per hour by 2022. Table 5 shows that the overtime law will gradually decrease the number of hours employees can work on a weekly basis before overtime wages are required. Prior to its passage agricultural workers could work up to 10 hours per day or 60 hours per week without overtime wages; by 2022 the requirement will be lowered to 8 hours per day or 40 hours per week for employers with 26 or more employees. The overtime law may change wages and scheduling of work in complicated ways as it is phased in.
The full impact of the two new laws on prevailing agricultural wages is not yet clear. But the studies recognize that growers may already pay wages that are different than those used to calculate the lettuce and broccoli costs, and that agricultural wages are currently in flux. The studies also note that growers may choose to use a farm labor contractor or the H-2A guest worker visa program to employ workers and assure a reliable supply of labor.
Vegetable and other cost and return studies for this area, and for other regions in California, are updated periodically, and are always available at https://coststudies.ucdavis.edu. Questions about these or other studies can be directed to either Laura Tourte or Richard Smith at UC Cooperative Extension.
- Author: Timothy K Hartz
- Author: Richard Smith
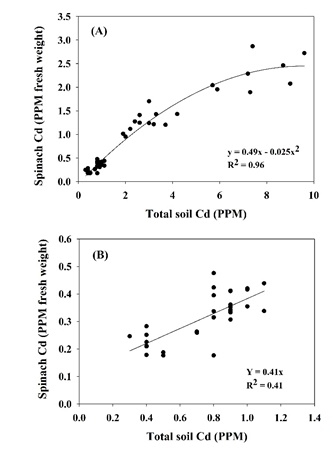
The factors controlling cadmium (Cd) uptake by leafy greens, particularly spinach, have been the focus of intensive study since 2014. By far the largest influence is the soil test Cd level (Fig. 1A). Coastal soils vary from in total Cd content from less than 0.5 PPM to more than 8 PPM; across this range spinach Cd concentration varies by at least a factor of 10. This is the reason why spinach should not be grown on soils of very high Cd content, at least not without some remediation effort to reduce Cd uptake by the crop. However, if one looks at the relationship between soil and spinach Cd concentrations over a narrower range of soil Cd content it becomes clear that factors other than soil Cd content affect spinach Cd uptake (Fig. 1B). Among those factors is the concentration of chloride (Cl) in the soil.
In soils of neutral to alkaline pH like those of coastal California, much of the Cd in the soil is precipitated in compounds of low solubility, or adsorbed on the surface of soil minerals; this reduces the plant availability of soil Cd. Chloride has been shown to enhance the solubility (and therefore, the plant availability) of soil Cd; in this regard Cl is different from the other common anions in the soil (sulfate or nitrate). The main factors governing the amount of Cl in soil are the Cl concentration of irrigation water, and the degree of leaching for salinity control.
To determine how significant an influence soil Cl can be on spinach Cd uptake a pot trial was conducted in 2016. A loam soil with a total Cd content of 1.5 PPM was used. Large pots containing approximately 7 pounds of soil were seeded with ‘Tambourine' spinach seed. Different Cl treatments were imposed by watering the pots with one of four levels of irrigation water Cl concentration. The irrigation water Cl concentrations evaluated were 1, 4, 7 and 10 milliequivalents per liter (meq/L), equal to approximately 35, 140, 250 and 360 PPM Cl. For context, irrigation water on the central coast typically ranges between 1-7 meq/L, occasionally higher. These solutions were made by adding Cl to a low Cl concentration water. A mixture of sodium and calcium chloride was used to maintain a reasonable cation balance. All pots were germinated with the low Cl water, with the different Cl treatments beginning 7 days after seeding. Watering was controlled to ensure at least 10% of applied water leached from the pots, to maintain a relatively constant soil Cl concentration over the trial period. The spinach was allowed to grow for approximately a month, and then the entire vegetative biomass was harvested, oven-dried, ground and analyzed for Cd concentration. The experimental design of the trial was a randomized complete block, with 4 replicate pots per irrigation Cl treatment.
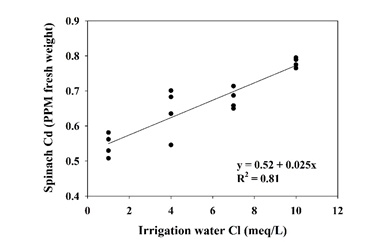
Spinach Cd concentration increased linearly with increasing irrigation water Cl concentration (Fig. 2). The slope of the regression line showed that each meq/L increase in irrigation water Cl corresponded to a 0.025 PPM increase in fresh tissue Cd concentration. This suggested that irrigation water quality can be a potentially significant factor in spinach Cl uptake. All other factors being equal, a field irrigated with water containing 7 meq/L Cl (250 PPM) may produce spinach with a Cd concentration more than 25% higher than if lower Cl irrigation water was used. These results may actually understate the effect of Cl. In this trial all treatments were germinated with low Cl water, meaning that the initial Cl status of the soil was low across treatments; it may have been well into the growth cycle before soil Cl concentrations fully reflected the treatment differences. In a commercial field situation, irrigating with high Cl water would set a baseline of soil Cl concentration at a relatively high level, affecting the crop from germination onward.
We realize that growers often do not have a choice of irrigation wells to use on a particular field. Where a choice of irrigation water exists, using the lower Cl water would minimize spinach Cd uptake. More broadly, it seems appropriate to use irrigation water Cl concentration as one factor in the determination of which fields to use for spinach production.
- Author: Steven T. Koike
- Author: Mark Bolda
Finally. Under the guidance of USDA-ARS and other researchers, and with the help of the California Strawberry Commission and other supporters, the UC Cooperative Extension diagnostic laboratory in Salinas finally has joined the 21st century regarding molecular diagnostic technologies. A DNA-based test is now being implemented to detect and confirm important soilborne diseases of coastal crops. This method can complete the diagnostic process in a few hours, compared to the conventional culture method, which usually takes several days.
The Need. Rapid, accurate, and dependable diagnosis of plant diseases is an essential part of agriculture production. Without knowing the precise agent responsible for the disease, growers, pest control advisers, and other field personnel are hampered when making disease management decisions. Soilborne diseases are particularly challenging to identify. In general, the appearances of the various root rots, crown rots, plant collapses, and vascular wilts all look very similar, making it virtually impossible to diagnose these based on symptoms alone. The UC Cooperative Extension laboratory has some very accurate culture methods to grow out and detect all of these soilborne pathogens. However, such culture techniques are relatively slow and take from three to seven days to complete. For our fast growing coastal crops a more rapid means of disease diagnosis would well serve the growers.
The Method. The new method goes by the name “RPA” which stands for recombinase polymerase amplification. The RPA method, like many other technologies such as the polymerase chain reaction (PCR), is based on DNA components (called primers and probe) that specifically recognize the corresponding DNA found in the target pathogen. However, all PCR methods depend on extracting and purifying the pathogen DNA; getting the DNA out is costly (one needs to buy expensive equipment to analyze the sample) and it requires more time to complete the elaborate procedures. RPA is perfectly suited to an extension laboratory, like the one in Salinas, because DNA purification is not needed as the test uses ground up, crude plant extracts as a source of unpurified DNA (see photos below).
Pathogens Detected. Presently the UC Cooperative Extension laboratory offers RPA diagnostic tests for two pathogens. Phytophthora causes root and crown rots on strawberry, raspberry, and a number of vegetable crops and ornamental/forest plants. Our Phytophthora tests are either genus-specific (it will detect Phytophthora infecting many types of plants) or species-specific for the two pathogens that mostly attack strawberry (P. cactorum and P. fragariae) (Table 1). Our other RPA test detects the specific type of Macrophomina phaseolina that causes severe crown rot and collapse of strawberry. Because this Macrophomina RPA is designed for the strawberry pathogen and not for Macrophomina that infects other crops, this is called a genotype-specific test (Table 1). This summer we anticipate adding tests for the Verticillium wilt pathogen (V. dahliae that infects many crops) and the Fusarium wilt pathogen of strawberry (Fusarium oxysporum f. sp. fragariae). All tests analyze infected plant material only. However, it is hoped that our continuing research will adapt RPA or more sophisticated molecular techniques for detecting these pathogens in soil.
Table 1. RPA tests currently available at the UC Cooperative Extension laboratory.
|
Category of test |
Pathogen |
Comments |
|
Genus-specific |
Phytophthora |
Detects this pathogen in all plants, such as strawberry, raspberry, pepper, asparagus, tomato, avocado, ornamentals, forest species (sudden oak death pathogen) |
|
Species-specific |
Phytophthora cactorum |
Primarily for strawberry but this pathogen can be found on ornamentals such as coffee berry |
|
Species-specific |
Phytophthora fragariae |
For strawberry |
|
Genotype-specific |
Macrophomina phaseolina |
For Macrophomina that infects strawberry |
The Future. Because this RPA method is similar in design to PCR, and PCR is used to develop the DNA tools (primers) to detect any sort of organism, the RPA platform is open ended. It should be possible to readily develop RPA tests for any organism that has DNA, such as other pathogenic fungi and bacteria, nematodes, and even perhaps specialized sub-groups of organisms such as insect biotypes.
Acknowledgements. This advance in rapid, accurate disease diagnostics was developed and implemented by the following research and extension team: Mark Bolda (UC Extension, Santa Cruz Co.), Alyssa Burkhardt (USDA-ARS, Salinas), Oleg Daugovish (UC Extension, Ventura Co.), Steven Koike (UC Extension, Monterey Co.), Frank Martin (USDA-ARS, Salinas), Stacy Mauzey (UC Extension, Monterey Co.), Tim Miles (CSU Monterey Bay), and Cayla Tsuchida (UC Extension, Monterey Co.). This research was supported by the California Strawberry Commission, California Avocado Commission CSU Agricultural Research Institute (grant 5219101A), and the USDA-California Department of Food and Agriculture Specialty Crop Block Grant Program (grants SCB12051 and SCB14052 awarded to F. Martin).
Photo 1: Strawberry samples are examined for symptomatic tissues that indicate possible disease.
Photo 2: Pieces of root, crown, or petiole are macerated and ground up in sample bags containing buffer solutions.
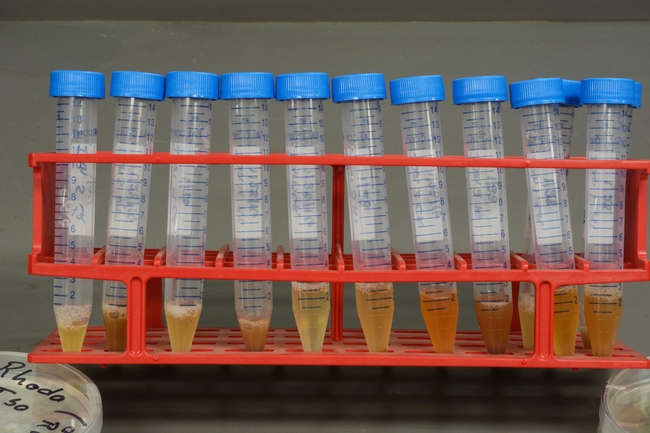
Photo 3: The resulting mixtures are strained and placed into tubes.
Photo 4: Small volumes of the strawberry extracts are loaded into strip tubes.
Photo 5: Loaded strip tubes are placed into the RPA isothermal detection device.
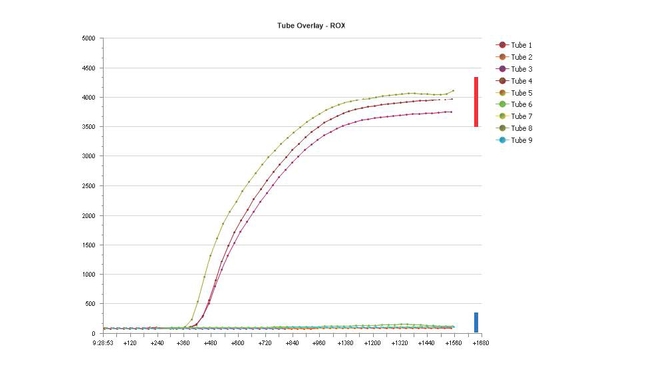
Photo 6: In the computer generated results, samples positive (red bar) for Phytophthora root and crown rot will appear as curved lines moving upwards; negative samples (blue bar) will appear as straight lines mostly remaining flat.