- Author: Steven A. Tjosvold
Roots are often overlooked by horticulturists but deserve to get more attention. Of course, they are usually underground and out of sight so it's somewhat understandable why they can be ignored. But, roots play a critical role in the life of a plant. They anchor the plant to support the shoots above. They absorb water and mineral nutrients and conduct them upwards. They store carbohydrates and other nutrients that are a source of energy for biennials and perennials as they awaken and grow in spring. A root's tip is where most of the action takes place.
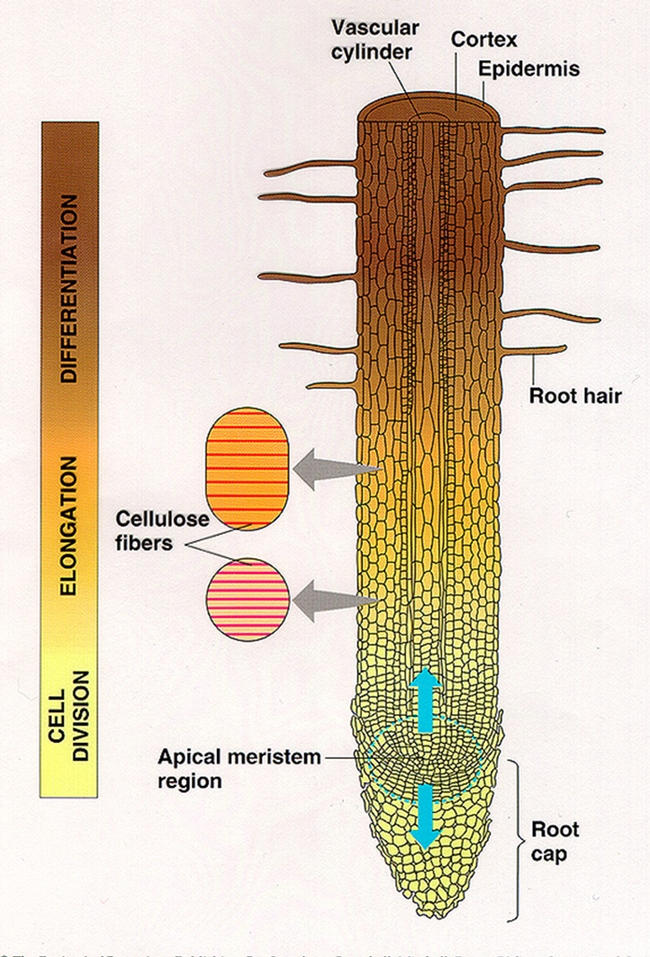
The root tip has overlapping zones: where cells divide, elongate, or form different specialized cells. At the very tip, the root cap protects the rapidly dividing cells known as the meristematic region or meristem (zone of cell division). Behind the meristem, cells elongate and push the meristem and root cap forward into the soil so the root can explore and mine new soil (zone of elongation). And further back, only a fraction of an inch, is the portion where elongation stops and cells become more specialized and functional (zone of differentiation).
Root hairs form in the zone of differentiation and this is where they begin to poke out into the soil to absorb water and mineral nutrients. Root hairs greatly increase the root surface area and therefore increase the ability of a plant to absorb water and nutrients. Vascular tissue (vascular cylinder) is the the piping that helps conduct water and nutrients upward to the shoots. The epidermis forms the protective skin of the roots.
Root hairs are long, thin, single cell extensions from the epidermis. They profoundly increase the overall root surface area and connection with the soil and are responsible for absorbing water and mineral nutrients. Usually they are short-lived, only functional for several days or weeks. So as the root tip advances into virgin soil, new root hairs must be formed continuously. It is important to keep root hairs healthy. The overall vigor of a plant can often be judged by looking at the condition of the root hairs. A nursery scout should remove the pot if possible and look for healthy, usually white, root tips and hairs.
Interveinal chlorosis is indicative of a deficiency in iron in the leaves (and sometimes manganese or zinc deficiencies). But this does not mean that the soil necessarily has low levels of these nutrients. Unhealthy root hairs or the conditions that they are growing might be to blame. Sometimes high soil pH makes iron less available for uptake. Iron is tightly held by soil and must be mined by actively growing roots. When soil is cold in the spring and roots inactive, sometimes iron might not be sufficiently mined and absorbed. Sometimes root diseases such as Pythium and Rhizoctonia might kill root hairs or reduce their functionality.
- Author: Steven A. Tjosvold
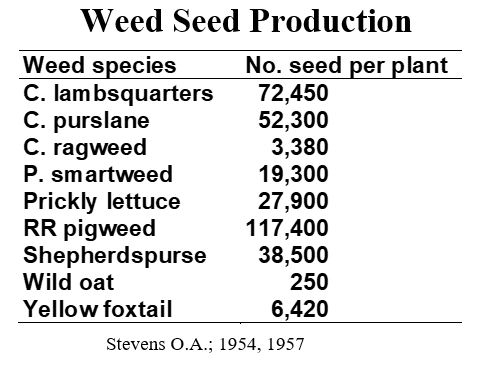
The last post demonstrated the remarkable ability and unique features of aphids that allow them to rapidly boost their numbers and colonize their hosts in favorable conditions. What about weeds? What features give them the ability to rapidly colonize a potted crop or planted field? Many plants become weeds because they have the powerful trick of producing many many seeds. Seeds are dispersed by wind, insects, animals, or by their association with our nursery tools and machinery. Often, these seeds are long-lived in the soil. Consider these statistics:
Probably some unfortunate graduate students or field assistants in 1954 were given the task to count weed seed of hundreds of common weed species from about 50 plant families in North Dakota. The table above is just a sampling (Stevens 1957). In most cases a single plant, judged to be of average size and growing where competition was low, was harvested at maturity or when a maximum number of seeds could be obtained. The plants were air dried for two weeks or more, threshed and cleaned to re-move immature seeds, empty florets, etc. All of the sampling methods are described in the Stevens reference given below.
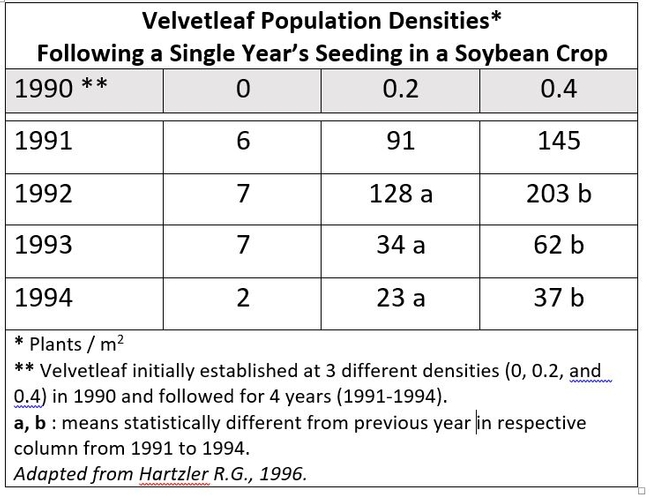
So basically left on their own, weeds have a profound ability to produce seed. Some seed are not viable, some germinate immediately, and some persist, perhaps for years, in the soil as a “seed bank”. This bank represents the holdings of weed seeds in the soil. Place a “deposit” of seed in this bank, and your “interest” is compounded in a big way. An interesting experiment with velvetleaf (Abutilon theophrasti) an important weed in soybean crops demonstrated this (Hartzler, 1996).
Velvetleaf is a prolific seed producer and seeds are long-lived. In 1990, replicated experimental plots were planted with soybean and then with one of three velvetleaf densities: 0, 0.2, and 0.4 plants per square meter. In subsequent years, the experiments were maintained in a corn-soybean rotation. Weed densities were determined at crop harvest for four years. As seen above-- even with competition from the crop plants-- velvetleaf density increased dramatically for years following the very sparse initial planting of the weed. There were even some velvetleaf plants seen in the untreated “0” plots, even though the plots were hand weeded to reduce seed production for 5 years prior to initiating the study.
The number of weed seeds in in the soil can range from near 0 to over 1,000,000 per square yard, and most weed seeds are between 0 and 5 years old. A small number of seed can remain viable for decades or more. With this knowledge, one of the most important principles of weed management is to “never let weeds go to seed”. Never.
References:
Stevens O.A., 1957. Weeds, Vol. 5, No. 1 (Jan.), pp. 46-55
Hartzler R.G. 1996. Velvetleaf (Abutilon theophrasti) Population Dynamics following a Single Year's Seed Rain
- Author: Steven A. Tjosvold
We are all familiar with aphids, especially in the spring when their populations seem to increase so rapidly and may require control before plant damage occurs. Aphids have unique strategies to flourish. The aphid mother you see this spring is actually a grandmother. Her daughters and granddaughters are contained within the same body! She does not need sex to produce offspring. The individuals develop within her body and are deposited as live young. In this way, one female may produce as many as 80 individuals per week. It's a strategy that builds up aphid populations quickly to take advantage of fleeting advantageous environmental conditions and to overwhelm the abundant predators and parasites that can attack them.
Each individual of these 3 generations are genetically alike. Clones might be a disadvantage in a changing environment and pressure from pesticide applications since there is no recombination of genes afforded by sex. But insect ecologists have noted that spontaneous mutations in these clones are possible. If a favorable mutation occurs, then their populations are advanced quickly.
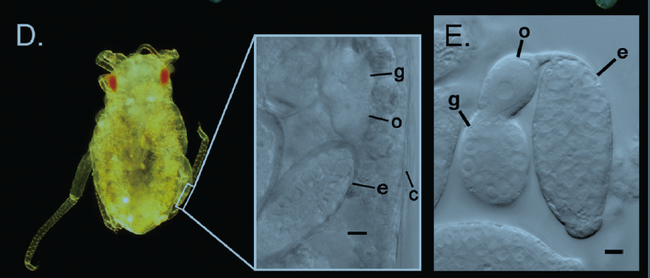
Most aphid species overwinter in the egg stage, and these eggs hatch in the spring into females that produce live young as described above. Several generations like this may be produced during the season. Sometimes winged individuals (see image below) may be produced that migrate off the original host plant species to a different plant species. In the latter part of the season, the aphids migrate back to the original host plant species and a generation consisting of both males and females are produced. The individuals of this generation mate, and the females lay eggs, which overwinter.
Aphid identification: Aphids have soft pear-shaped bodies with long legs and antennae, and may be green, yellow, brown, red, or black depending on the species and the plants they feed on. A few species appear waxy or woolly. Their feeding can cause distorted stems and leaves. They can transmit viruses. They often produce abundant sticky honeydew from their anus that can become colonized by “sooty mold” and become unsightly. Most species have a pair of tube-like structures called cornicles projecting backward out of the hind end of their body (see image below). Cornicles are like the exhaust pipes at the tail end of a hot rod, but in this case, secrete defensive fluid. The presence of cornicles distinguishes aphids from all other insects.
References:
Davis G.K. 2012. Cyclical parthenogenesis and viviparity in aphids as evolutionary novelties. J. Exp. Zool. 318B:448–459.
Dixon A.F.G. 1985. Structure of aphid populations. Ann. Rev. Entomol. 30:155-74
Flint, M.L. 2013. Aphids. Pest Notes. Publication 7404. University of California Statewide Integrated Pest Management Program.