- Author: Steven A. Tjosvold
Container soils should have a good balance between air- porosity and water- holding capacity. But what are the favorable levels of these two parameters? Most horticulturists will recommend providing a soil with “good drainage”. But what exactly does that mean? How do we quantify that? If we know, then soil manufacturers can develop, standardize and describe soil mixes. Growers can select, develop or modify their own soil mixes for their particular needs.
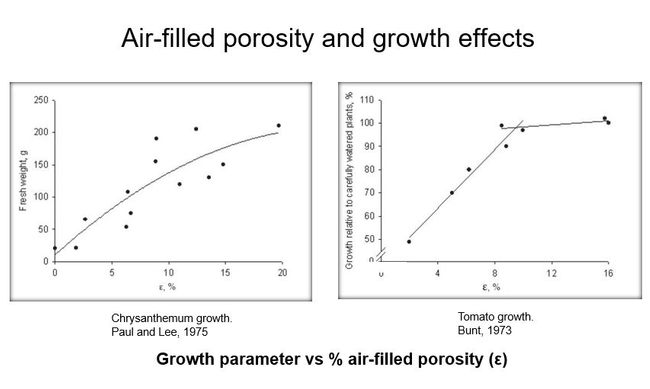
Let's start with air-filled porosity. There have been many experiments demonstrating that air-filled porosity is highly correlated with the growth of various plants. Here are two examples, Fig 1:
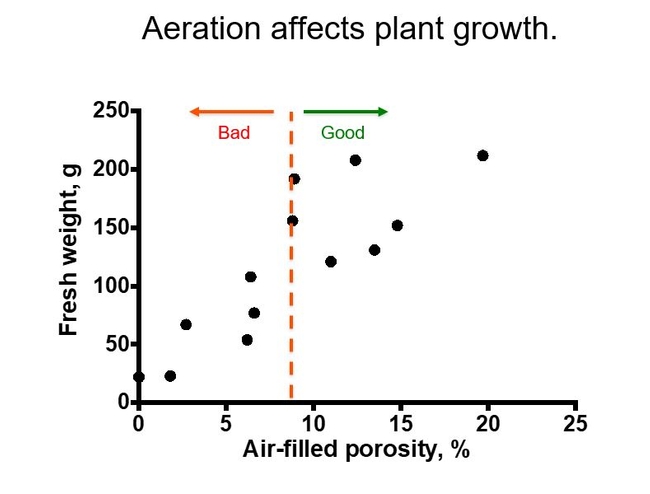
Some researchers reported that the rate of oxygen diffusion through the soil-- rather than air-filled porosity-- is more closely related to plant growth. This might be because the oxygen in the air pores has to move to the roots before it is absorbed and used by the roots. In general, these and similar studies have led to a recommendation that air-filled porosity should be at least 10% and generally no more than 25% of the total soil volume. Fig 2.
So a plants' requirements for soil aeration can vary, and here are some examples. Fig 3.
There are other consequences of having poor soil aeration. Soils with low oxygen or anaerobic conditions can lead to chemical reduction. Reduced forms of some chemicals, such as methane (CH4), hydrogen sulfide (H2S), nitrite (NO2-), and manganese ion (Mn2+), are toxic to plants. The latter is probably the most important. Plants growing in poorly aerated soil can have manganese levels as high as 1200 ppm in their leaves (normal levels are 50-200 ppm). These high levels lead to manganese toxicity, with symptoms of marginal leaf chlorosis (yellowing from loss of chlorophyll, the green pigment in leaves).
What about the water filled portion of the soil? The recommendation is that at least 40% of the total volume should be filled with water at container capacity (after full drainage). In general, container soils with a water-holding capacity of 40% would hold enough water to meet plant demand for about one day. Usually mixes have a higher water-holding capacity than this so that irrigation can be less frequent. Total porosity, air and water porosity combined, must therefore be greater or equal to 50%. These parameters are summarized in Fig 4.
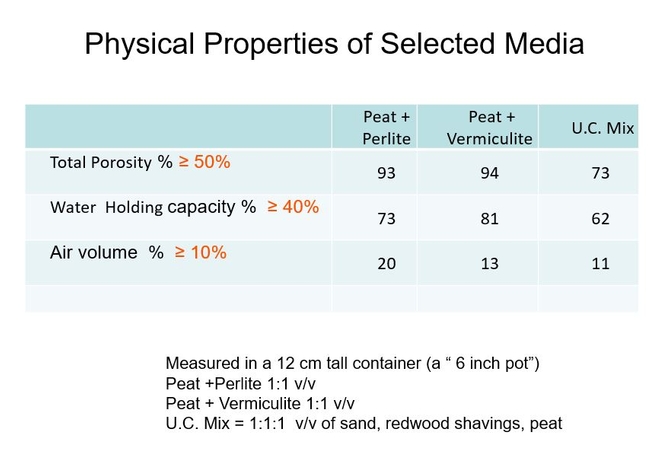
Here are examples of 3 historically important container mixes. The “UC Mix” was the first “soil- less" mix, mostly developed for the outdoor nurseries. It provided the essential requirements for a container mix: Good water holding capacity and aeration, high permeability, relatively disease-free, resistant to salinity buildup, reasonable pH range, good nutrient holding capacity, and provided for enough weight of the container so plants would not blow over in the wind. The “Cornell Lite” mixes containing peat mixed with either vermiculite or perlite are excellent for greenhouse crop production, but they are too light for outdoor nurseries and more expensive because of the relatively high cost of vermiculite and perlite. Fig 5.
These soil physical properties are best measured by commercial or research soil laboratories. If you are buying soil mixes, ask if the provider has these measurements. In a pinch, growers can estimate the values with a relatively simple method that was described by James Altland when he was with Oregon State University. I have provided a simplified outline here. Fig 6. The full description of the method is provided in the attachment at the bottom of the page.
Next: Poor soil aeration can lead to some root diseases.
Figures and tables adapted from: Management of Container Media by Richard Evans, UC Davis, for the class, ENH 120. Porosity determination handout by James Altland (formerly Oregon State University, now with USDA ARS).
Determining porosity a practical guide. Click to download.
- Author: Steven A. Tjosvold
Container soils must have the right balance of air-filled and water-filled pore space. This is especially important right after irrigation. I showed in the previous blog that field soils shouldn't be used in containers because they don't drain well and create a saturated root environment. Roots need air (oxygen) for respiration, so you can't have the roots drowning for very long. You need a good balance of air and water. This blog discusses what is a good balance and explains why container size affects it.
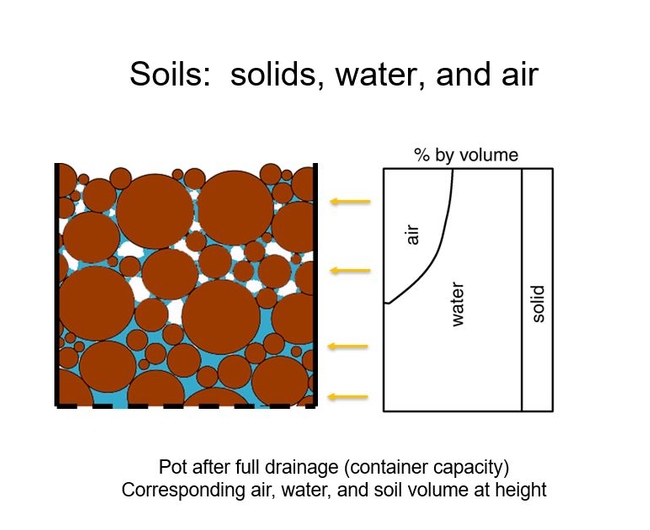
Simply, container soils consist of solids that form the intricate pore space that holds water and air. After full drainage (container capacity), the pore space on the bottom of the pot is saturated with water. Closer to the surface, there are more open pores that hold air. This is seen in Figure 1.
The balance between air and water filled pores in the container can be illustrated more specifically. In Fig 2, water volume (ml. / cc.) in vertically sliced sections are shown on the container with the corresponding air volume in the table. Note that total porosity is constant if the soil components are uniform, and that Air + Water = Total Porosity.
It is useful to define terms to describe the amounts of air and water and total porosity. Total porosity is the percent of the soil volume occupied by pore spaces, water- holding capacity is the percent of the soil volume that contains water, and air-filled porosity is the percent of the soil volume that is filled with air. These parameters can be measured for any soil mix for a designated container size. With standardized parameters, researchers can study how they affect plant growth and root diseases. Soil manufacturers can develop, standardize and describe soil mixes. See figure 3.
These parameters would of course be affected by the particle sizes of the soil components. (See previous blog, Fig 5). In addition, the size of the container-- actually the height of the container-- affects these parameters too. For any given container mix, as the container height decreases, the water- holding capacity increases. As container height increases, the water-holding capacity decreases. This is seen in a very simple way using the pot example given earlier. I have visually "sliced off" the top of the container to show the water that would be held in shorter containers. See figure 4.
And seen more specifically with this example using air-filled porosity. Remember, more water, less air! Fig 5.
So a container soil that works well in a tall container may have too high of a water holding capacity and too little air-filled porosity in a short container. The opposite is true for a short container too. The container height should be considered when selecting the container mix. You probably have seen this. The container media for a propagation flat for vegetative cuttings often is composed of components with large particles (e.g. perlite, scoria, and vermiculite) to minimize the overall water holding capacity and maximize the overall air-filled porosity.
Next: What are favorable levels for water-holding capacity, air-filled porosity and total porosity?
Figures and tables adapted from: Management of Container Media by Richard Evans, UC Davis, for the class, ENH 120. Photo of pot/water relationship by James Altland
- Author: Steven A. Tjosvold
First some basics: A container soil, as with any field soil, must provide a reservoir for moisture, air, mineral nutrients, and anchorage for plants. A container limits the soil volume that roots can develop in and is relatively shallow in depth. As a result, plants grown in containers are subjected to more intense environmental stresses than those grown in a field. That is why growing in containers requires precision in irrigation and fertilizer management and is often fraught with plant problems and even disasters.
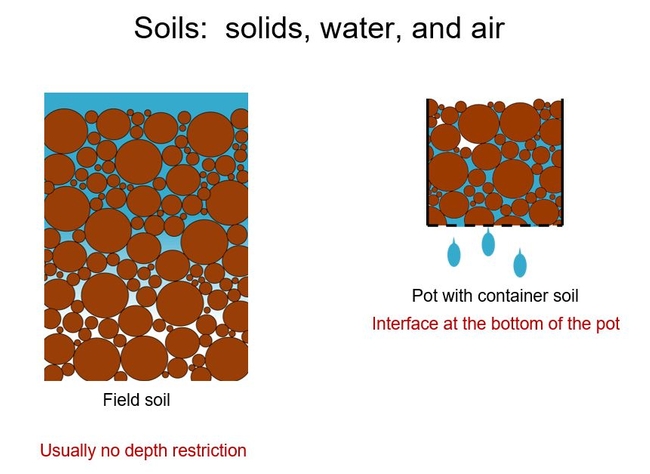
Why not just use field soils in containers? As mentioned, they are relatively heavy; consequently they are expensive to ship and are more difficult to lift and move in the nursery. Moreover, field soils hold too much water in containers. An understanding of the physical forces that are acting on water in the field and the container helps explain this. The first thing to note is the effective depth of the two situations, field versus container. Good agricultural field soils have no physical depth restrictions to a developing root system (e.g. a mature fruit tree root's often only need to colonize the top two or three feet of soil, while the effective soil depth is often greater). However, a container of course limits the depth of the soil and the drainage water interfaces with the ambient air at the bottom of the container.
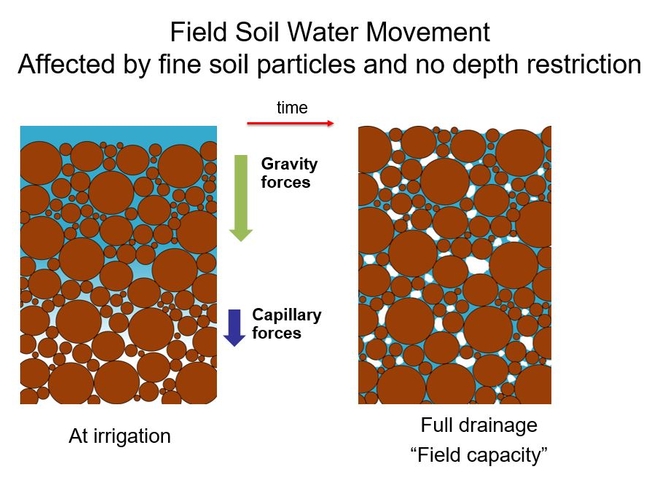
When water is first added to a field soil, gravity forces pull the water in the water-filled pores down. Below the saturated zones, capillary forces help pull water deeper too. (These forces are a result of the small- pores or capillary-like spaces). When full drainage has occurred, water movement is mostly at equilibrium. The soil is said to be at “field capacity”. (This term is useful to describe the point in time when the water holding capacity for a field soil is measured).
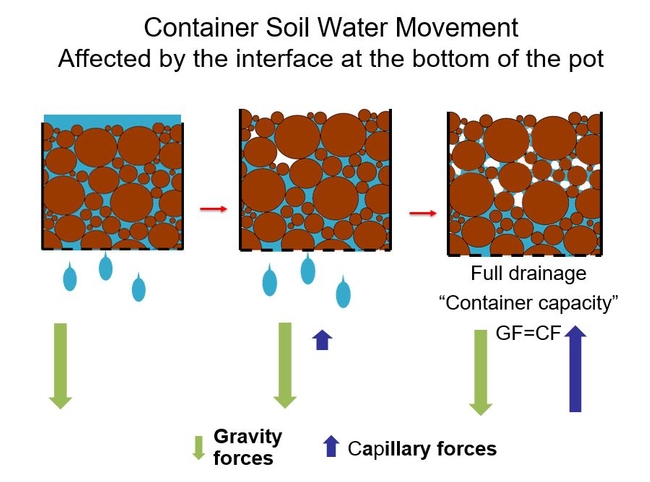
The same forces affect the movement of water in containers but in a different way. At first gravity is pulling the water downward out of the water-filled pores, and water drains out the bottom of the pot. But as water drains, the upper soil profile begins to form capillary-like spaces, and capillary forces begin to tug water upward. As water continues to drain, the same upward capillary forces begin to tug upward at the bottom of the container. After several minutes of drainage, the upward capillary forces on water equals the downward gravity force, and water movement is mostly at equilibrium. The container stops draining. The soil is said to be fully drained or at “container capacity”. (This term describes the point in time at which water and air holding capacity is measured for container soils).
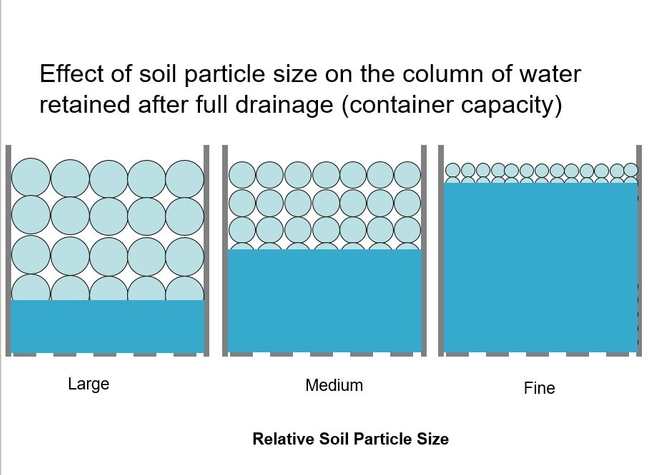
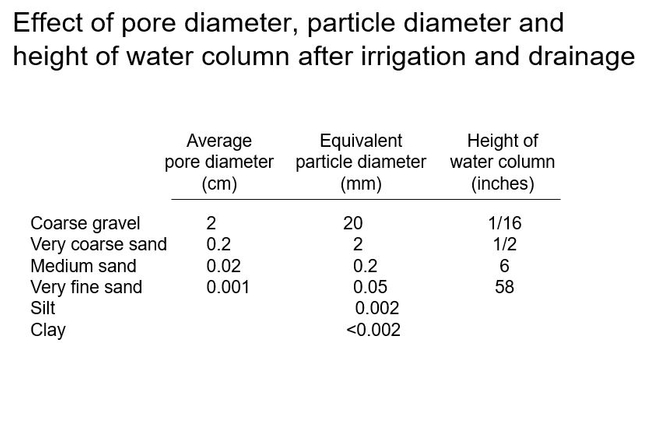
The capillary forces tugging upward is related to the pore diameter. The smaller the pore diameter (the tinier the capillary) and the more the capillary pulls upward. Smaller soil particles make smaller pore diameters and will result in a higher column of water at container capacity. Simply this is seen below.
The rise of the water column can be calculated based on soil particle size. Here are examples of various soil particles and their pore diameter, particle diameter, and resulting height of the water column at container capacity.
Field soils are composed of the smallest particles in this table: clay, silt, and various sizes of sand. This table illustrates that, after irrigation and drainage, even a pure medium sand would create a water column of about six inches and saturate the entire soil to the top of a 6-inch-high (gallon) container. As I mentioned earlier, the soil has to provide air to the roots (beside moisture, etc). And now you can see that field soils would create a saturated root environment in a container after an irrigation. You can't have the roots drowning for very long. For this reason, field soils work poorly for plants grown in containers. Container soils have to contain large soil particles!
Next: Water and air holding capacity of container soils.
*Figures and tables adapted from: Management of Container Media by Richard Evans, UC Davis, for the class, ENH 120.
- Author: Steven A. Tjosvold
Now through the spring is the time to take a close look at crops that are susceptible to downy mildew so that the disease can be detected early. This post follows the previous one that describes the general identification and biology of downy mildew diseases. Here are some more images illustrating the symptoms on various ornamental crops.



Downy mildew on statice ( species) caused by Peronospora statices. (BELOW) Bluish gray sporulation occurs on undersides of leaves. On upper surface of leaves, infections appear as light green areas that turn yellow, then coalesce, and eventually the leaf dies.


Downy mildew on hebe (Hebe species) caused by Peronospora grisea. (BELOW). The disease is usually noticed first by a yellowish discoloration often with purple halos on the lower leaves of the plants. Leaves become curled and distorted. A grayish- brown sporulation may develop on the underside of the leaves. Eventually the affected parts of leaves turn brown and shrivel. Plants can be entirely defoliated and killed.
Downy mildew on snapdragon (Antirrhinum majus) caused by Peronospora antirrhini. (BELOW) Young tip leaves are dull green, severely stunted, and roll downward. Gray‑purple fungus grows on undersides of leaves. Disease is common on seedling phase; large plants are less frequently attacked. Infected plants fail to produce flowers.
- Author: Steven A. Tjosvold
Downy mildew diseases have become increasingly important and common on ornamental plants over the last decade in California. The wet and cool weather we recently have had should promote the development of this disease. Now through the spring is the time to take a close look at susceptible crops. One of the newest introductions of downy mildew in California is on iceplants, and this disease provides an excellent example to talk about downy mildew diseases in general.


In general, downy mildew diseases can be severe in cool or warm (but not hot) temperatures, high humidity, and when liquid water is present on plant tissue for extended periods. Link to information on leaf wetness importance. Downy mildew pathogens are part of a larger taxonomic group of fungi-like micro-organisms that contains Pythium and Phytophthora species. They primarily cause foliar blights and rapidly spread in young green leaf, twig, and fruit tissues. They produce spores on the ends of stalks and the spores can be carried by wind and rain to new infection sites of the same or different plant. The pathogen can be present in soil associated with host and non-host plants and therefore can spread by any means that aids in the movement of soil and/or water from infected plants to non-infected ones. The pathogen can spread through contaminated plant cuttings and transplants. Some are found in seed and can be spread through seed propagation.
Learn which crops that you grow are susceptible to downy mildew diseases, and monitor them closely now through the spring.