- Author: Steven A. Tjosvold
Container soils must have the right balance of air-filled and water-filled pore space. This is especially important right after irrigation. I showed in the previous blog that field soils shouldn't be used in containers because they don't drain well and create a saturated root environment. Roots need air (oxygen) for respiration, so you can't have the roots drowning for very long. You need a good balance of air and water. This blog discusses what is a good balance and explains why container size affects it.
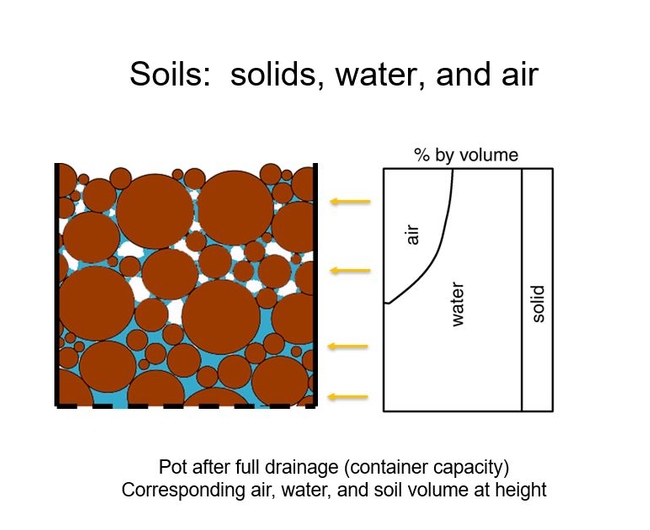
Simply, container soils consist of solids that form the intricate pore space that holds water and air. After full drainage (container capacity), the pore space on the bottom of the pot is saturated with water. Closer to the surface, there are more open pores that hold air. This is seen in Figure 1.
The balance between air and water filled pores in the container can be illustrated more specifically. In Fig 2, water volume (ml. / cc.) in vertically sliced sections are shown on the container with the corresponding air volume in the table. Note that total porosity is constant if the soil components are uniform, and that Air + Water = Total Porosity.
It is useful to define terms to describe the amounts of air and water and total porosity. Total porosity is the percent of the soil volume occupied by pore spaces, water- holding capacity is the percent of the soil volume that contains water, and air-filled porosity is the percent of the soil volume that is filled with air. These parameters can be measured for any soil mix for a designated container size. With standardized parameters, researchers can study how they affect plant growth and root diseases. Soil manufacturers can develop, standardize and describe soil mixes. See figure 3.
These parameters would of course be affected by the particle sizes of the soil components. (See previous blog, Fig 5). In addition, the size of the container-- actually the height of the container-- affects these parameters too. For any given container mix, as the container height decreases, the water- holding capacity increases. As container height increases, the water-holding capacity decreases. This is seen in a very simple way using the pot example given earlier. I have visually "sliced off" the top of the container to show the water that would be held in shorter containers. See figure 4.
And seen more specifically with this example using air-filled porosity. Remember, more water, less air! Fig 5.
So a container soil that works well in a tall container may have too high of a water holding capacity and too little air-filled porosity in a short container. The opposite is true for a short container too. The container height should be considered when selecting the container mix. You probably have seen this. The container media for a propagation flat for vegetative cuttings often is composed of components with large particles (e.g. perlite, scoria, and vermiculite) to minimize the overall water holding capacity and maximize the overall air-filled porosity.
Next: What are favorable levels for water-holding capacity, air-filled porosity and total porosity?
Figures and tables adapted from: Management of Container Media by Richard Evans, UC Davis, for the class, ENH 120. Photo of pot/water relationship by James Altland
- Author: Steven A. Tjosvold
The previous post described the importance of inspecting incoming plants and keeping a clean nursery to avoid pest introductions. Many important root pathogens and other pathogens, weeds, and insects can be introduced in contaminated soil and growing media, and so the focus of this post will be on ways to prevent introductions from these potential sources.
Soil- inhabiting plant pathogens can be found in growing media and associated root and crop debris. Anything that has contacted the ground, such as equipment, tools, irrigation hoses, and workers' hands and shoes could be contaminated. Pots should not sit directly on the ground. Phytophthora infecting the roots of just one potted plant can produce thousands of propagules that can move in water draining from the pot and infect roots of nearby plants.
Likewise, during a vigorous rain storm, these propagules can be splashed from the contaminated pot or ground onto nearby plants. Benches or similar structures that support plants above the ground can eliminate or minimize this. In greenhouse structures, concrete floors or other impervious surfaces are ideal for walkways between benches. Floor surfaces should be kept clean of plant debris, soil, or growing media. After a crop cycle, benches should be cleaned of plant debris, washed and dried. Drying can kill sensitive plant pathogens.
Benches can be sprayed with diluted chlorine bleach (0.5 % sodium hypochlorite solution) or other suitable disinfectant. Potting media and plant debris will inhibit the activity of most disinfectants.
There are many clever ways to raise pots and containers off the ground.




The bottom of clean shoes can be sanitized with disinfectants such as quaternary ammonium compounds .


Store and handle growing media so it does not come in contact with the ground or water runoff. Cover the media when not in use.