- Author: Steven A. Tjosvold
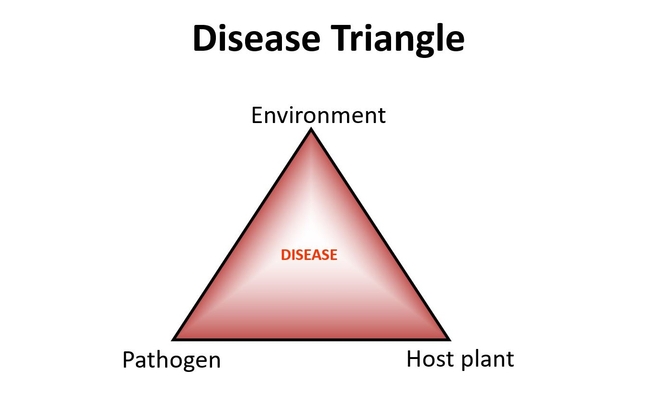
Plant diseases– their occurrence and severity– result from the impact of three factors: the host plant, the pathogen, and the environmental conditions. This is represented with the disease triangle.
If any one of the three factors is missing, the triangle is not complete, no disease will occur. Simply, plant disease will not occur if there is no viable pathogen, or no susceptible host plant, or the environmental conditions are not favorable. The severity of disease depends on the favorable level of each factor. How susceptible is the plant? How virulent is the pathogen? How conducive are the existing environmental conditions in supporting disease and pathogen spread?
The triangle also helps illustrate that the 3 factors are interacting with each other. The clearest example of this is how the environment factor interacts with the pathogen and host factors. Previous blogs illustrate the importance of leaf wetness on pathogen infection and disease severity. Long durations of free water on a susceptible plant can increase pathogen infection and disease severity. At the same time, the low- sunlight conditions, when these wet periods could occur (e.g. winter), could also be stressful to the plant, and the plant is less likely to mount defensive reactions to fend off infection.
Learn about the biology for any disease you are managing. Consider the disease triangle and the three interacting factors, and how management practices might help weaken or break the triangle's bond. For example, could you grow non-susceptible plant varieties or species? Could you eliminate the pathogen through judicious sanitation practices? Could you manage leaf wetness and relative humidity to create unfavorable environmental conditions for disease?
Could you grow non-susceptible plant varieties or species?
Although these three plant hosts all have rust diseases, each host is infected with a unique plant pathogen that attacks its specific host. In managing these diseases, a strategy of host resistance might be employed, where crops of non-susceptible hosts are rotated into the infested area. Conceivably these three hosts could be rotated.

Sanitation is a key management practice that employs the strategy of eliminating the pathogen from the growing area. Here, sanitizing shoes before entering the greenhouse. Cleaning soil from tools and sanitizing them before use. Properly covering and disposing of rogued plants or cut flowers (not shown here!).
Could you manage leaf wetness and relative humidity to create unfavorable environmental conditions for disease?
- Author: Steven A. Tjosvold
As you walk quickly down the nursery walkway on the way to the sales team meeting, you glance over at the 1- gallon stock that's almost ready to sell. Then you stop; something is wrong with the Chamaecyparis lawsoniana. Yikes, the older portions of the branches are browning on many plants. So you take a closer look.
Left, what you might see quickly walking by. Looks pretty good!
Left, a closer look, without a hand lens. Something is wrong!
If you are like me, I've got my field hand lenses ready to pull out to take a closer look. I have got a 10 X and a 20 X (magnification) lens. The 10X is the most common magnification used in the field, and sufficient to help see and generally identify most of the common adult insect pests such as aphids, whiteflies, scales, mealybugs, fungus gnats and two-spotted spider mites.
When I see symptoms that appear to be caused by much smaller pests, such as eriophyid or broad mites, I'll pull out my 20X lens and take even a closer look. Sometimes I might see them. The 20 X lens has a very limited field of view, short depth of field, and is crazy hard to hold still. For these pests, and many other tiny immature insect life stages, it is best to take a sample back to the office or lab and use a stereo-microscope (binocular microscope), which works well at higher magnifications (20 X to 40X). But for me the 20X gives me a first look in the field, and may help pinpoint an infested sample to take back for the "scope".
You get the best view with a hand lens by holding it close to the eye. Use whatever hand and eye combination that seems comfortable. (Often right-handers use the right eye). You can keep glasses on if it works for you. Brace your lens holding hand on your cheek to stabilize it. Now with the other hand, move the specimen-- leaf, branch, stem-- to your lens until it is focused. With a 10X lens this will be about 1 inch away from the lens; with a 20X lens, about 1/2 inch away. With the subject in focus, position your body so that ambient light can illuminate the specimen. No hunching over the specimen like it is some big secret or something; let the light in.
Field hand lenses come in various forms and quality. "Hastings triplet" lenses have the best clarity, least distortion, and larger field of views than lower quality lenses. These high quality lenses are only about $35.
And back to the Chamaecyparis lawsoniana. What did the grower see associated with the necrotic branches when viewed with a field hand lens? Surprise, greenhouse thrips. Adults are black with pale wings. This slow-moving species lives in groups, unlike the solitary, fast-moving blackish predatory thrips. Larvae are white or yellow. At the tip of their abdomen they often carry a droplet of dark excrement. (Below. Adult circled in red on left image. Light colored larva and one adult on right image).
Don't forget to subscribe to the blog so that you receive an email notification when a new post goes up.
- Author: Steven A. Tjosvold
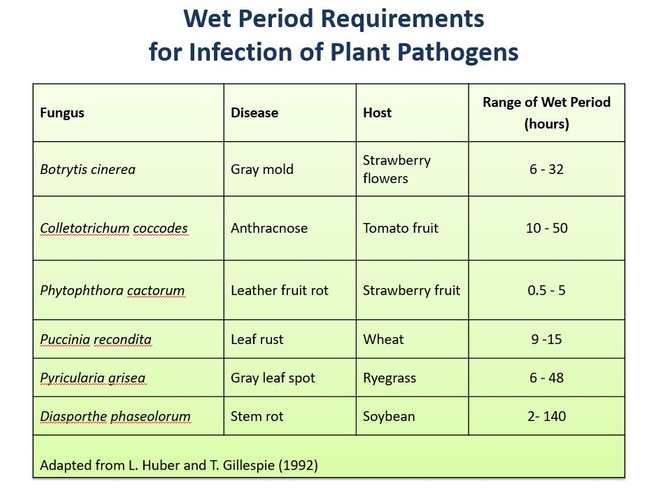
The majority of fungi, aerial nematodes and bacteria that cause plant diseases require liquid “free” water on the plant surfaces before they can infect the plant. Free water, sufficient for disease infection, commonly occurs in the form of rain, fog, and dew. Or it can even occur after a sprinkler irrigation , syringing , or pesticide spray. Moreover, many fungi need high relative humidity to produce spores, and abundant free water can contribute to high relative humidity. Dew formation is triggered when the surface temperature of the plant surface drops below the dew point temperature of the surrounding air (See previous blog post). This typically occurs at night in greenhouses that are not ventilated and heated properly, or outside on calm clear nights. Plant pathogens have different leaf wetness requirements for infection to occur (as seen below).
Many diseases will get worse with longer leaf wetness periods. In the table below you can see that the severity of gray mold (Botrytis cinerea) on rose increases with longer wet periods.
Commercial disease prediction models exist for apple scab, cedar apple rust, potato late blight, tomato early blight, strawberry anthracnose, botrytis fruit rot, citrus brown spot, lettuce downy mildew, grape powdery mildew, among others. Leaf wetness sensors help quantify leaf wetness periods, and models predict disease risk These systems can reduce the number of sprays that are needed for disease control. It has been recently suggested that disease models instead use leaf wetness data based on a simple empirical model using relative humidity. Relative humidity sensors can be standardized and calibrated more easily than leaf wetness sensors.
Many current greenhouse control systems can help collect and organize data from leaf wetness, relative humidity and temperature sensors. Alternatively, a simple environmental monitoring system can be pieced together for an outdoor nursery or greenhouse using readily available sensors and data loggers from various companies (e.g. , Campbell Scientific Inc (Logan, UT), Onset (Bourne, MA), Spectrum Technologies Inc. (Aurora, IL). Most disease risk models have not been tested in ornamental crops but there is no reason why they cannot be wholly or partly used for disease risk monitoring in ornamental crops. Botrytis models have been intensively studied in other crops and should be one of the first to try in ornamental crops. Empirical evaluation of these models in the field is a first step to confirm their usefulness. Models that predict high disease risk could improve scouting efficiency by targeting more intensive scouting during these periods, help reduce fungicide applications by predicting optimal timing of fungicides before infection occurs, and target periods when de-humidification cycles are needed in greenhouses.
- Author: Steven A. Tjosvold
Good air circulation in a crop canopy has many important positive impacts. As described in previous blogs, carbon dioxide utilization and consequently photosynthesis is improved. Moreover, temperatures within the leaf canopy and greenhouse are more uniform, and leaf temperature will be closer to the immediate surrounding air temperature. This has a profound impact on when and where condensation occurs as the greenhouse air temperature falls.
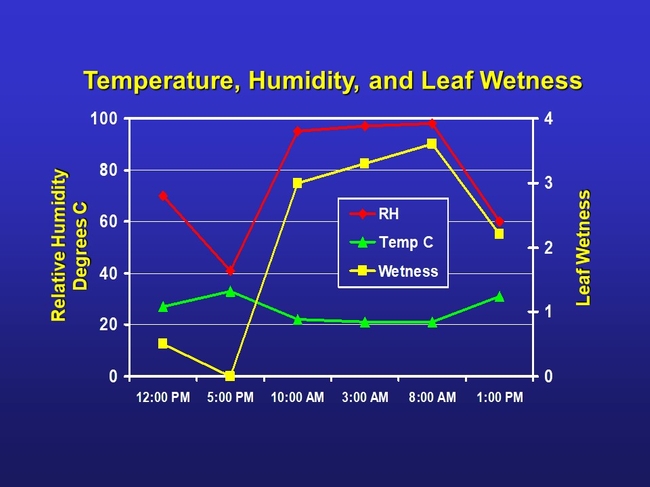
Above are actual temperatures, relative humidity and leaf wetness data I measured in a greenhouse in a 24 hour period. As temperature (green line) climbs in the day the relative humidity (red line) falls. As temperatures fall at night the relative humidity rises. The relative humidity is the amount of moisture in the air compared to what the air can "hold" at that temperature. When the air can't "hold" all the moisture, then it condenses as dew. Leaves get wet when water-laden air cools at night and falls to dew point temperatures. So you can see as relative humidity gets high, approaching 100%, then leaf wetness also increases as dew is formed.
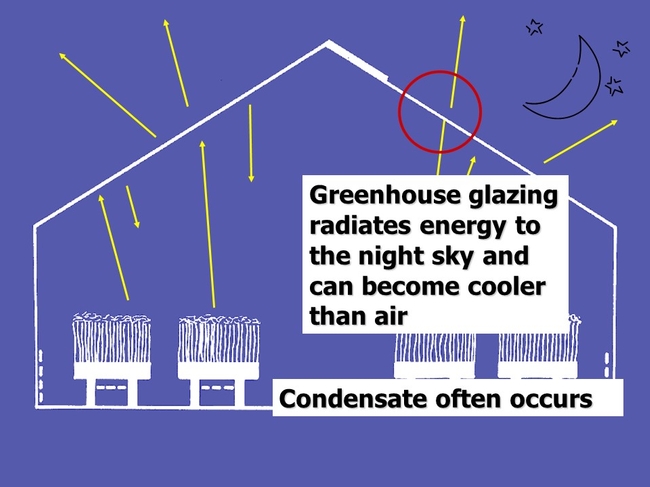
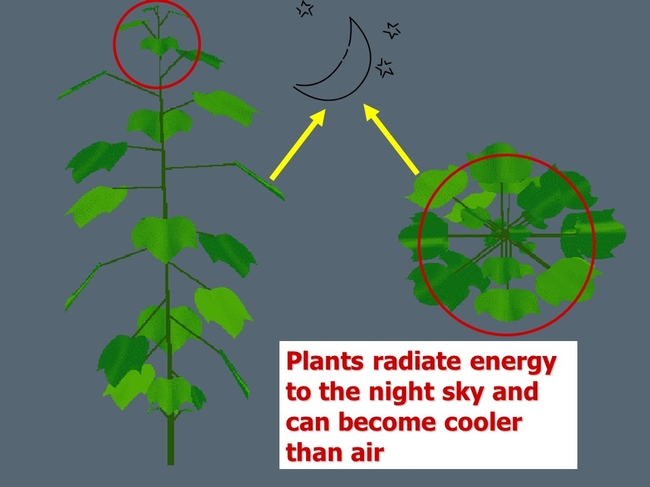
Water condenses first on the coolest surfaces such as the greenhouse structure, and usually crop leaves and flower buds too. Greenhouse and plant surfaces that face the night sky are radiating and losing energy, and can become cooler than the ambient air temperature. Thus they are prone to become surfaces that support condensation as illustrated below.
Condensation occurs first on the coolest surfaces as air cools and approaches the dew point. Above, dew formed first on the top of rose leaves near the top of the rose plant (which faces the night sky).
Moving air within the leaf canopy at night helps move the relatively warmer greenhouse air around the cooler leaf and flower surfaces. Even very small temperature effects matter. The result is that condensation on those important crop surfaces may be prevented or reduced.
This has strong implications for disease control.....which will be described in next week's blog.
- Author: Steven A. Tjosvold
The previous blog post explained how moving air helps maximize photosynthesis by increasing the ambient CO2 diffusion into the leaf. The trick is to efficiently move air around the greenhouse so that can happen.
Since 1967, when the Horizontal Air Flow (HAF) concept was introduced by Dr. Jay Koths, from the University of Connecticut , it has become the standard method for air circulation in greenhouses. Over the years, changes to fan design, installation techniques and efficiency have brought some improvements to the original design. But the HAF concepts and benefits have not changed at all. I scanned one of the original newsletter articles that Dr Koths wrote in 1985 (available below). It explains the concepts and practical design features as well as anyone ever could.
Horizontal Air Flow