- Author: Ben Faber
WESTMINSTER, Colorado - November 23, 2018 - Herbicides have been instrumental in managing Italian ryegrass, a weed that frequently competes with perennial crops in California. Herbicide-resistant populations have become increasingly commonplace, though, including paraquat-resistant Italian ryegrass found recently in a California prune orchard.
A team of scientists set out to determine if the paraquat-resistant population might also be resistant to other postemergence herbicides. Seven other herbicides commonly used in fruit tree and nut tree crops were included in the study, including clethodim, fluazifop-P-butyl, glufosinate, glyphosate, pyroxsulam, rimsulfuron and sethoxydim.
Researchers found the paraquat-resistant population was also resistant to both clethodim and glyphosate. Among the remaining herbicides, glufosinate, rimsulfuron and sethoxydim were found to deliver the best postemergence control. Unfortunately, though, other populations of Italian ryegrass have developed resistance to the three herbicides, indicating their effectiveness may be short-lived.
"Overreliance on postemergence herbicides from a variety of chemical classes can result in weed populations that exhibit multiple resistances," says Caio Augusto Brunharo, a postdoctoral research fellow at the University of California, Davis. "Effective herbicide-resistance management programs are necessary for sustainable weed control."
The researchers recommended a number of preemergence herbicides as control options for Italian ryegrass in fruit and nut tree crops, including tank mixes containing indaziflam and flumioxazin.
###
Full text of the article, "Multiple Herbicide-Resistant Italian Ryegrass (Lolium multiflorum) in California Perennial Crops: Characterization, Mechanism of Resistance and Chemical Management" is now available in Weed Science Volume 66, Issue 6.
About Weed Science
Weed Science is a journal of the Weed Science Society of America, a nonprofit scientific society focused on weeds and their impact on the environment. The publication presents peer-reviewed original research related to all aspects of weed science, including the biology, ecology, physiology, management and control of weeds. To learn more, visit http://www.

- Author: Travis Bean
Although the main objective of herbicide use in avocado orchards (and all crops) is to manage weed populations, sometimes unintentional injury of the crop itself can occur when herbicides are incorrectly applied. Herbicide injury in avocado can reduce yield, decrease fruit, reduce plant vigor, increase susceptibility to diseases and pests, and sometimes result in plant death. Common situations resulting in injury include spray drift, tank contamination, application of the wrong herbicide or rates, and herbicide carryover from a previous crop. The extent of herbicide damage on avocado can vary widely according to factors such as herbicide mechanism of action (MOA) and application rate, route of exposure, plant size and growth stage, soil properties, and weather.
Herbicide injury can be difficult to diagnose properly and is often confused with disease, insect damage, nutrient deficiencies, and other environmental stresses. It is recommended that trained researchers or Pest Control Advisers, who may utilize plant tissue, make diagnoses or soil samples along with plant symptoms, injury progression, and other plant species affected, orchard herbicide use history, weather conditions, and other factors to confirm or rule out injury from herbicides or other causes.
Where the injury occurs can also be an indication of herbicide injury. For example, if injury is on just one side of a tree or trees near another field, it may be an indication of spray drift. If it occurs only along the edge of the skirts, it may be a hint that an uneven ground spray was applied.
The majority of herbicides for use in avocado orchards in California fall into eight MOAs as defined by the Weed Science Society of America. MOAs describe the specific biological processes that are disrupted by a group of herbicides. These processes control the growth and development of plants and when interfered with, can result in plant injury or death.
Table 1: Common herbicides used in avocado, their mechanism of action, and possible injury symptoms
|
WSSA Group |
Mechanism of Action |
MOA description1 |
Example herbicides |
Possible injury symptoms1 |
|
1 |
Acetyl CoA Carboxylase (ACCase) Inhibitors |
Inhibits lipid creation in grasses, preventing production of plant cell membranes |
Fluazifop-P-Butyl (Fusilade DX), Sethoxydim (Poast) |
Chlorosis, necrotic spots, leaf crinkling, leaf distortion |
|
3 |
Mitosis Inhibitors |
Inhibits cell division in germinating seedlings and lateral roots |
Oryzalin (Surflan) |
Thickened, shortened lower stems and small, crinkled leaves |
|
5 |
Photosystem II Inhibitors |
Prevents the transfer of energy generated during photosynthesis, causing a buildup of reactive molecules that damage chlorophyll and cell membranes |
Simazine (Princep 4L) |
Chlorosis, necrosis progressing from leaf margins toward the center of the leaves, foliar applications will appear as leaf burn |
|
9 |
Enolpyruvyl Shikimate-3-Phosphate (EPSP) Synthase Inhibitors |
Inhibits the production of three aromatic amino acids and the enzymes and proteins built from them |
Glyphosate (Roundup) |
Leaves of trees and vines become chlorotic 3 to 7 days after exposure, and margins of new leaves become necrotic |
|
12 |
Carotenoid Biosynthesis Inhibitors |
Inhibits production of carotenoid pigments, which harvest light and protect chlorophyll from reactive molecules |
Norflurazon (Solicam DF) |
Plant foliage turns white and appears bleached
|
|
14 |
Protoporphyrinogen Oxidase (PPO) Inhibitors |
Blocks the production of chlorophyll and causes a buildup of reactive molecules that damage existing chlorophyll, carotenoids, and cell membranes |
Oxyfluorfen (Goal 2XL), Carfentrazone (Shark EW), Flumioxazin (Chateau) |
Drift injury will appear as speckling on leaf tissue. The necrotic spots are sometimes surrounded by a reddish colored ring. Injury from soil applications or residues appears as a mottled chlorosis and necrosis. |
|
21 |
Cellulose Inhibitors |
Inhibit cell wall synthesis and plant growth |
Isoxaben (Gallery 75 DF) |
Chlorosis, necrosis, leaf crinkling, leaf distortion, purpling of the leaf, and stunting |
|
22 |
Photosystem I Inhibitors |
Disrupts photosynthesis, forming reactive molecules that destroy cell membranes |
Paraquat (Gramoxone SL) |
Drift injury will appear as speckling or necrotic spots on leaf tissue |
1Not a complete list. Symptoms listed are likely for established orchards. For detailed descriptions of MOAs and injury symptoms, as well as a searchable database of specific injury images (e.g., “chlorosis, necrosis, stem swelling, etc.” visit http://herbicidesymptoms.ipm.ucanr.edu.
References:
Al-Khatib, K. 2015. University of California Integrated Pest Management Herbicide Symptoms. http://herbicidesymptoms.ipm.ucanr.edu (accessed 09/05/18)
Faber, B.A., C.A. Wilen, B.D. Hanson. 2016. Weeds. Pages 107-124 in University of California Integrated Pest Management Guidelines for Avocado. http://ipm.ucanr.edu/PMG/selectnewpest.avocado.html (accessed 09/05/2018)
Sosnoskie, L.M., B.D. Hanson. 2013. Understanding herbicide mechanisms (modes) of action and how they apply to resistance management in orchards and vineyards. UC Weed Science Blog Post. http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=9383 (accessed 09/05/2018)
Weed Science Society of America. Summary of Herbicide Mechanism of Action
According to the Weed Science Society of America. https://wssa.net/wp-content/uploads/WSSA-Mechanism-of-Action.pdf (accessed 09/05/2018)
Photo: Sometimes weeds are tasty, like amaranth and purslane

- Author: Ben Faber
With the rains, and in those area where fire took out the competition, weeds are coming back in their glory. Mustard has painted the hills yellow. The question comes up, what to do about all that wild growth. Mechanical control, such as discing or whipping can work great. Sometimes chemical control is the only answer. A recent request for an alternative to glyphosate (Round-up) control of marestail (horseweed, Conyza canadensis) which is similar to hairy fleabane (Conyza bonariensis), came to the office from a lemon grower. Glyphosate just wasn't controlling it. And it's been a problem for a while, even in tank mixes with paraguat and old-line weed killer. The alternative might be a newer material, such as saflufenacil (Treevix) which has been recently added to the herbicides that can be used on citrus.
As always before doing "vegetation management" it's best to identify the plant that is the problem
Identify the problem plant (weed)
http://wric.ucdavis.edu/information/weedid.htm
or if you know what the plant is, go directly to a listing of the weeds
Listing of weeds, their biology and control
http://wric.ucdavis.edu/information/info_spec_weed.htm
- Susceptibility of Winter Weeds to Herbicide Control (9/08)
- Susceptibility of Spring/Summer Weeds to Herbicide Control (9/08)
Or you can go in reverse order and look at your tree crop and see what herbicides are listed
Listing of herbicides by tree crops, including avocado and citrus
http://wric.ucdavis.edu/PDFs/T&V_herbicide_registration_chart.pdf

- Author: Ben Faber
Plant-out-of-place photo galleries:
http://wric.ucdavis.edu/photo_gallery/photo_gallery.htm
http://ipm.ucanr.edu/PMG/weeds_intro.html
Horseweed - Conyza canadensis
Herbicide treatment table for citrus:
http://ipm.ucanr.edu/PMG/r107700411.html
And if you are wondering what herbicide damage might look like on various plant species (this is heavily weighted to annuals and landscape plants):
http://herbicidesymptoms.ipm.ucanr.edu/index.cfm
Blueberry herbicide damage
- Author: Brad Hanson
Glyphosate is one of the most widely used herbicides in the world and is extremely important in many of our orchard, vineyard, and annual crops as well as in non-crop and home situations. However, it can be confusing to understand some of the differences among various formulations of glyphosate herbicides.
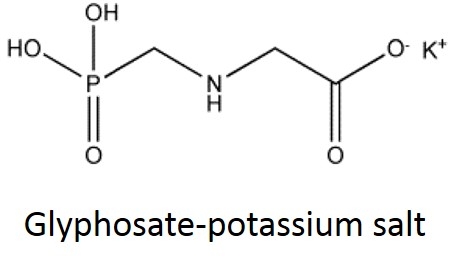
I'll paraphrase a recurring extension question as “I'm trying to compare the rates and cost-effectiveness of two glyphosate herbicides. One lists the active ingredient as ‘41% glyphosate as the isopropylamine salt' and the other as 48.7% glyphosate as the potassium salt'. How do I compare these two herbicides?”
First important point, glyphosate is a weak acid herbicide. The various salt formulations have major impacts on how the herbicide behaves in the jug, and to some degree on how it gets into the plant. But once in the plant, it is the glyphosate acid that binds to the target enzyme in susceptible plants and causes the herbicidal effect.
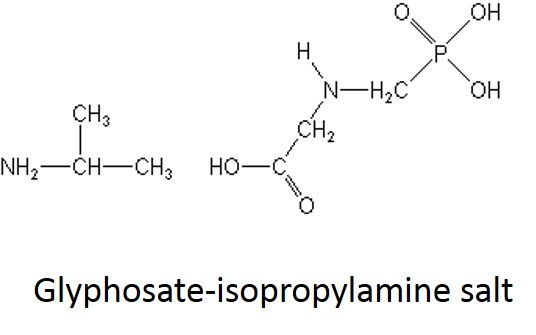
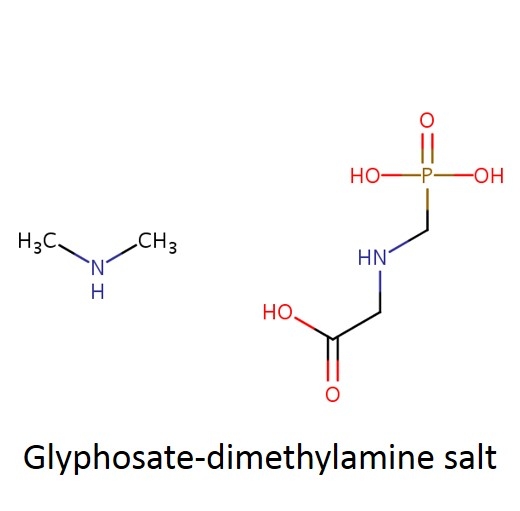
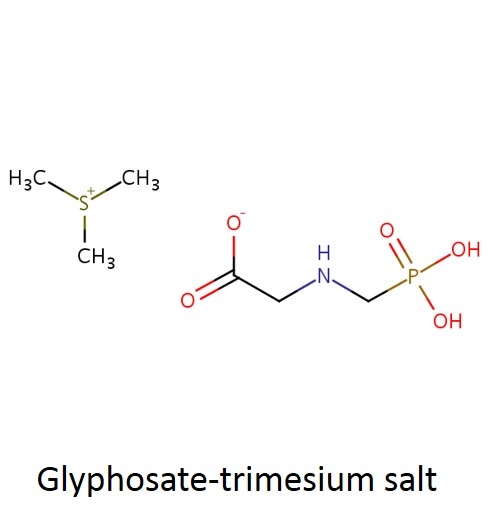
What is a salt? From a chemistry perspective, a salt is simply a compound formed by ionic bonding of two ions of opposite charge. Glyphosate acid has a weak negative charge and the salt is formed when the glyphosate acid is bound to a base that has a positive charge. In the cartoon below, this is illustrated a little incorrectly - you can see the negatively charged glyphosate acid on the left (C3H8NO5P); however, the "salt+" tagged on the right should really be labeled "base+" (the combined molecule is actually the "salt").
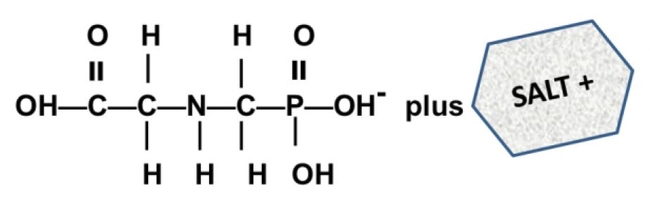
In case it's been a while since your last organic chemistry class, here's a quick refresher on nomenclature. In the isopropylamine salt, that simply means it as a 3-carbon chain (that's “propyl”) and amine group (NH), and the chain is connected in the middle (the “iso” position) rather than on one end. Dimethylamine indicates two (di) methyl groups and an amine. Trimesium is a shortened name for trimethylsulfonium which means three methyl groups and a sulfur. Potassium means, well that one's pretty straightforward and means potassium (K).
How does the salt formulation affect the herbicide? The salt formulations are those of us managing weeds for a couple of important reasons.
- First, the different glyphosate salts have different solubility in water (or other solutions). This doesn't have much, if any, effect when we have the herbicide diluted in water to make a spray application (eg. quarts of product in 10 or more gallons of water). However, it has a big impact on how concentrated the herbicide can be in the formulated product. (It's useful here to note that pure glyphosate is actually a solid crystal that is dissolved in liquid to make the products we use in the field.) In the example I started with, that is the big driver behind why one product has 41% active ingredient and the other has 48.7%. From a packaging, shipping, storing, and handling perspective, it's far more efficient to have more concentrated materials (eg less water). So, we can assume the potassium salt is quite a bit more soluble than the isopropylamine salt form of this herbicide.
- Second, the different glyphosate salts have different weights. Remember the periodic table of the elements? The atoms that make up the salts (and any other chemical structure for that matter) have markedly different weights on an atomic scale. For our purposes, we can think of this as a carbon (C, atomic number 6) weighing six times a hydrogen atom (H, atomic number 1) and a potassium (K, atomic number 19) weighs about three times what carbon does. Of course, the glyphosate acid weight is the same C3H8NO5P in each formulation – only the salt is different. So, it is important to remember that in these herbicides the “active ingredient” (AI) is the salt formulation (eg glyphosate isopropylamine) and that each of the different active ingredients has a slightly different weight.
How can I use the percent active ingredient to compare products? I'll be honest here, I find it very confusing to think about the percent AI list on the label of a glyphosate herbicide. As I indicated above, this does not allow a straight across comparison for two different salts (eg a potassium salt glyphosate is not the same weight as a dimethylamine salt). More importantly, the percentage listed on the label is actually on a weight basis (weight of glyphosate salt per weight of formulated herbicide). I don't know about any of you, but I typically measure glyphosate herbicides by volume (fluid ounces, quarts, milliliters) rather than by weight (ounces, pounds, kilos). So knowing the amount of glyphosate per pound of Roundup Powermax isn't very helpful to me when I don't weigh the liquid.
Instead of focusing on the percent active ingredient, look on the label just below where the percentages are listed for the active ingredient and acid equivalent information. (I'm looking at a Roundup PowerMax label and a Credit41 label, but there are dozens of examples). In these two herbicides, you can see:
- Credit41 has 4 lbs per gallon of glyphosate as the isopropylamine salt, which is equivalent to 3 lbs per gallon of glyphosate acid.
- Roundup PowerMax has 5.5 lbs per gallon of the glyphosate potassium salt, which equates to 4.5 lb/gal of the acid.
So, once we're comparing on an acid equivalent (AE) basis, we can see that that one product is 50% more concentrated than the other (3 lbs vs 4.5 lb ae). If you wanted to make an equivalent rate (AE per acre) you'd have to apply 50% more of the 3 lb product to have the same amount of glyphosate acid. Similarly, in making cost comparisons, you should be thinking about it in terms of cost per unit of glyphosate acid. In the above example, if making an equivalent AE rate per acres, the breakeven point would be if the 3 lb material costs 2/3 of what the 4.5 lb material costs. Don't make the mistake of assuming a quart of one glyphosate is the same as another (or that a 2% solution of two different products is equivalent!
Check out this publication we did a couple years ago On the second page, there's a chart that illustrates this concept – if you wanted to apply a rate of ¾ lb ae per acre, you'd need to apply 32 fl oz of a 3.0 lb ae/gal material (eg Credit41, Roundup Original) but only 22 fl oz of a 4.5 lb ae/gal material (eg Roundup PowerMax).
What else is different among the formulations? This is a really good question without a really clear answer.
Besides the glyphosate-salt that makes up the active ingredient in each formulation, there are other components of the herbicide product. Some of these influence physical properties that affect handling, storage stability, etc. Others are surfactants and other adjuvants that impact how the herbicide penetrates the leaf surface. The surfactants (types and loading) are proprietary information and are not reported in the same way as the active components are so this can be difficult to ascertain. However, one way that price points can be reduced in this competitive market (lots of generic glyphosate herbicide due to loss of patent protection) is to reduce or change the surfactant packages. Thus, even if the active ingredient is the same between two products, they could have substantially different surfactant loading which can impact weed control efficacy (amount of herbicide getting to the target enzyme in the plants). If you use a low-surfactant-load glyphosate product, you should consider adding appropriate surfactants to make up for it.
I'll wrap this up by saying that I think a weed manager can get similar levels of weed control performance with the wide range of glyphosate herbicides currently available on the market. However, it is really important to make sure you're comparing apples-to-apples in determining rates needed from the various salt formulations and concentrated products. In my experience, at equivalent AE rates, a 3 lb glyphosate plus a good surfactant can perform similarly to a higher AE and higher surfactant load formulations; however, be sure to sharpen your pencils to make sure the savings from the cheaper AI isn't offset by higher required rates and surfactant additions.
N.B. https://www.epa.gov/pesticides/epa-releases-draft-risk-assessments-glyphosate