- Author: Ben Faber
Imagine going to the supermarket to stock up on groceries but coming home empty-handed because you just couldn't figure out how to work the shopping cart or figure out how to get to the ice cream tubs in the freezer aisle.
Welcome to the life of a bumblebee.
Gathering sweet nectar from flowers, it turns out, is much more difficult than one might think, and it requires a lengthy learning process. By the time a bee has figured out how to efficiently pry open the lips of a snapdragon flower, for example, most likely it has made dozens, if not hundreds, of floral visits.
How does a bee in charge of shopping for food needed to raise dozens of hungry larvae back in the hive learn to navigate the multitude of floral architectures it may encounter during an average workday, let alone over the course of its life?
Mostly by what biologists call associative learning, more widely known as trial and error, researchers have found. But while extensive research -- starting with famous bee researcher and Nobel laureate Karl von Frisch a century ago -- has focused on uncovering how bees forage for nectar, much less is known about how bees go about collecting pollen, which constitutes the most important protein source for the developing brood in the hive.
Avery Russell, Stephen Buchman and Daniel Papaj in the Department of Ecology and Evolutionary Biology at the University of Arizona decided to take a closer look. In a new paper published in the journal Behavioral Ecology, they tell a fascinating story of what is involved in a seemingly simple process of a bumblebee visiting a flower to gather pollen. And for the first time, they have untangled the subtle cues that a bee looks for when she visits a flower in search for pollen.
"For a long time, we have known that bees can learn all kinds of cues -- tactile, visual and olfactory -- when going after nectar rewards," says Russell, the study's first author. "When you open a can, you have to use a can opener, then use your fingers to pry the lid open. A bee might have to pop open the flower's petals, and might have to try many times over multiple trips until they get good at it. But not much was known in the context of pollen rewards."
Specifically, Russell and his co-authors wanted to know if bees need to learn in order to collect pollen efficiently from flowers that vary in their form. The research suggests they don't, and they don't need to.
"Our findings suggest that unlike nectar foraging, which requires complex learning behavior, bumblebees already know how to collect pollen," says Russell, who did the research as a doctoral student in the UA's Graduate Interdisciplinary Program in Entomology and Insect Science, "and they do it by switching between two responses that are seemingly hardwired into their brains."
Once a bumblebee touches down on a flower, it wastes no time. If it senses that the anthers are laden with abundant pollen just waiting to be shaken off like ripened apples from a tree, the bee does the obvious: a behavior that bee researchers call "scrabbling." Using its mandibles and legs, the bee brushes the pollen grains onto its body, then combs them off into collection baskets located on each of its hind legs.
"If you picture a happy toddler in a play pit filled with plastic balls, you get the idea of scrabbling," Russell says.
However, some flowers make their pollen grains more difficult to access, or sport intricate anther designs that dispense only a little bit of pollen at a time.
"That way, the plant makes sure pollinators don't eat it all, but carry it to other flowers for pollination instead, and also leave some for other visitors as well, so the flowers aren't limited to a single pollinator," he says.
When visiting some of these trickier flowers, Russell's team found, bumblebees switch to a different behavior called sonication -- or, in more familiar terms, buzzing. Not unlike a sonicating toothbrush that vibrates to shake plaque from teeth, a sonicating bee vibrates vigorously to free pollen grains hidden inside the flower.
The team observed that the bees switched between these two motor regimes depending on chemical and mechanical cues: They scrabbled when pollen was abundant, and sonicated when pollen was scarce, either because the flower already had been depleted or because its pollen is less accessible by design.
To tease apart the cues that trigger each behavior, the researchers made artificial flowers and treated some of them with chemical extracts from natural anthers. Bees visiting a surrogate flower without extract didn't stick around and took off again in search of more rewarding offerings. When they encountered a foam flower without pollen but with the chemical cue, they buzzed them in a futile attempt to harvest the nonexistent pollen. And when they sensed pollen grains, even artificial ones, scrabbling ensued.
"Bumblebees tend to sonicate on pollen-concealing anthers right away, but they also buzz accessible anthers when they can't detect pollen by touch," Russell says. "We think they do that in an effort to collect the dregs from a flower after most of its pollen has been harvested."
Being able to switch between two programmed routines allows bees to effectively collect pollen from flowers in many different shapes and forms, the researchers conclude. This flexibility also may explain a fact that had evolutionary biologists stumped for a long time: Flowers with concealed pollen stores evolved many times independently, suggesting that pollinators must always have had a way to harvest pollen from them, or else the co-evolution between the two would have led to a dead end and not survived.
"Researchers used to think that floral sonication is a behavior only used to collect pollen from concealed pollen stores," Russell says, "but because we often observe bees buzzing on flowers with accessible pollen, we conclude that it's a behavior that has evolved as a general strategy to collect pollen from any type of flower."
To buzz or to scrabble? To foraging bees, that's the question
Presenting bumblebees with various combinations of natural and artificial flowers laced with chemical and mechanical cues, UA biologists have discovered that for a bumblebee, foraging for pollen versus nectar is very different.
photo: honeybee on avocado flower. Is it buzzing or scrabbling?

- Author: Elizabeth Fichtner
Recent advances in understanding the history of olive domestication
Elizabeth Fichtner, Farm Advisor, UCCE Tulare and Kings Counties
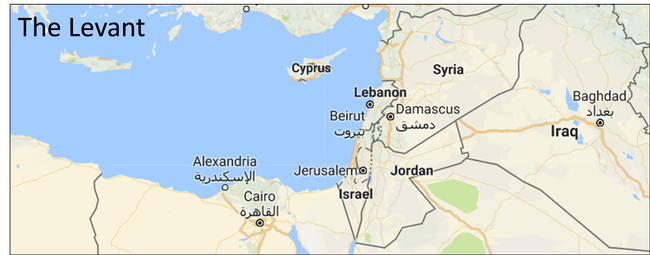 Olives are thought to have first been domesticated in the northeastern Levant, an area near the border of present-day Turkey and Syria. Map captured from Google Maps. |
With the emergence of the California olive oil industry, the state has witnessed a dramatic diversification in the olive cultivars grown commercially. Our mainstay black ripe olive industry, dominated by the ‘Manzanillo' olive, is now combined with increasing acreage of Spanish, Greek, and Italian cultivars used to create high quality, extra virgin oil. The historic table olive industry of California still represents around 18,000 acres of olives in the state, while approximately 40,000 acres are currently devoted to oil production.
Although olive cultivation in California is relatively new (dating back to the historic Spanish Missions established by Franciscan priests), olives are of key importance in the history and culture of the Mediterranean basin. A recent publication by a group of European, American, and North African scientists has re-evaluated the location of the domestication of the olive, providing genetic evidence that domestication occurred in the northeastern Levant, close to the present-day border of Syria and Turkey.
To complete the study, researchers collected plant material from nearly 2000 trees, sampling both wild oleaster populations and domesticated cultivars of olive. World Olive Germplasm Banks in Córdoba (Spain) and Marrakech (Morocco) served as sources of the majority of cultivars included in the study. Researchers utilized the genetic sequences of plastids (ie. chloroplasts) to discern differences between cultivars and wild oleaster populations. Plastids are organelles (structures inside cells) that contain their own DNA. Since plastids are generally inherited from one parent (similar to mitochondria), their genetic sequences are more conserved then that of nuclear DNA, which is contributed by both parents. Since olive is a wind-pollinated crop, nuclear DNA may be disseminated over large distances.
The genetic analysis of wild populations indicates three distinct lineages of olive: the Near East (including Cyprus), the Agean area, and the Straight of Gibralter. These three wild populations are likely linked to refuge areas where populations persisted through historic glaciation events. Interestingly, the geographic distribution of these three populations also corresponds to the subdivisions of the olive fruit fly, suggesting that these regions offered shared refuge habitat for both the host and the pest. The wild oleaster population in the eastern Mediterranean was found to be more diverse than previously thought and ninety percent of the present-day cultivars analyzed in the study matched this group. Common olive cultivars grown in California, including, Sevillano, Arbosana, Arbequina, and Koroneiki, all belong to this group originating in the eastern Mediterranean.
As a result of this study, it is proposed that the initial domestication of olive took place in the northeastern Levant; subsequently, plant material was disseminated to the whole Levant and Cyprus before being spread to the western Mediterranean. After these initial domesticated trees spread throughout the Mediterranean basin, they likely underwent subsequent domestication events by crossing with wild oleasters, thus introducing genetic material from the other two ancient western Mediterranean lineages.
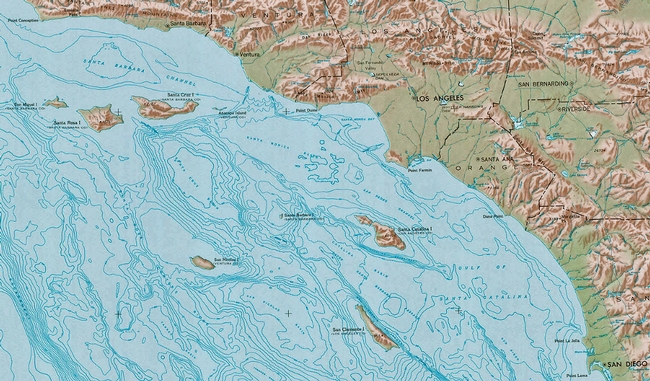
Such studies may appear purely academic; however, they can also address more timely questions and assist in characterizing cultivars. For example, a 2010 study in California made genotypic comparisons between historic olive plantings in Santa Barbara, CA and at Santa Cruz Island, CA. The study elucidated that the olives on Santa Cruz Island, planted in the late 19th century are different than other historic olive plantings in Santa Barbara, CA. Olives planted at the Santa Barbara Mission in the late 18th century are the ‘Mission' cultivar, whereas those on Santa Cruz Island (Figure 3) are generally ‘Redding Picholine.' Interestingly, the olives on Santa Cruz Island are thought to have been planted for oil production, but there are no historic reports of harvest or sale of a crop. Additionally, the Santa Cruz Island olives have become somewhat invasive on the island due to their propensity to establish from seed. As a result of genotypic analysis of these populations and the fact that ‘Picholine' makes an excellent rootstock due to its ease of propagation from seed, it is hypothesized that the ‘Picholine' variety was intended as a rootstock, but the grafts never took. Consequently, maturation of a ‘Picholine' orchard may have just been an accident, a mistake, or simply bad luck. The completion of this local population genetics study may have helped explain the unsolved mystery of the historically unharvested trees on Santa Cruz Island.
Find Santa Cruz Island.
Besnard, G., Khadari, B., Navascués, M., Fernández-Mazuecos, El Bakkali, A., Arrigo, N., Baali-Cherif, D., Brunini-Bronzini de Caraffa, V., Santoni, S., Vargas, P., Savolainen, V. 2013. The complex history of the olive tree: from Late Quaternary diversification of Mediterranean lineages to primary domestication in the northern Levant. Proc R Soc B. 280: 20122833.
Soleri, D., Koehmstedt, A., Aradhya, M.K., Polito, V., Pinney, K. 2010. Comparing the historic olive trees (Olea europaea L.) of Santa Cruz Island with contemporaneous trees in the Santa Barbara, CA area: a case study of diversity and structure in an introduced agricultural species conserved in situ. Genet Resour Crop Evol 57:973-984.

- Author: Ben Faber
The latest edition of Topics in Subtropics newsletter is out, Elizabeth Fichtner as editor. Read on.
TOPICS IN THIS ISSUE:
-
Why has California red scale been so difficult to control?
-
Navel Orange Nitrogen Fertilization
-
Recent Advances in Understanding the History of Olive Domestication
- Upcoming UC Olive Center Events


- Author: Frank Byrne, Akif Eskalen and Joe Morse
Two closely related Ambrosia beetles (Euwallacea sp.) have been identified in commercial avocado groves in California. The polyphagous shot hole borer (PSHB), detected in Los Angeles, Orange counties and recently in Ventura county, and the Kuroshio shot hole borer (KSHB), detected in San Diego and recently in Orange and Santa Barbara counties, are morphologically indistinguishable, but genetically distinct. Already widespread in a variety of reproductive host trees common in the urban landscape (including box elder, willow, several maples, oak and sycamore species), the beetles represent a significant threat to trees in both landscape and agricultural settings. Adult females construct galleries in the xylem system of host trees, where they cultivate symbiotic fungi (Fusarium, Paracremonium and Graphium spp.) as a food source for their developing young. The fungi are taken up by progeny females in specialized organs within their mouthparts, and transported to other sites within the same tree, where new colonies are established, or to newly colonized hosts. The galleries compromise the structural integrity of infested trees, which can represent a serious safety hazard in urban environments, and disrupt the flow of water and essential nutrients within the xylem. In addition to the physical damage, the fungi extract nutrients from the xylem system, further depriving the tree of nutrients essential for healthy growth and fruit production.
An effective biological control agent is not yet available to manage the SHB in California, and so management for now must rely on the use of chemical pesticides. The control of Ambrosia beetles and their associated fungi using
chemical pesticides is complicated because of their location inside the host trees. The application of insecticides to the external surfaces of trees, where the beetles must first alight prior to boring, has the potential to kill beetles by contact activity, and they may also have the potential to control emerging young adults before they can re-infest the trees.
The drawback of surface treatments is that multiple applications are often required because of the relatively short duration of efficacy. In addition, once the beetle burrows inside the tree, surface treatments are become ineffective. One possible solution to this problem may be the use of systemic pesticides, and scientists at UC Riverside are evaluating the use of both systemic insecticides and fungicides in a 2-pronged attack against the symbiotic system.
Systemic pesticides are mobile within the xylem system of plants, and the fungicides could potentially target the fungi growing in the xylem and deprive the beetle larvae of a food source. The insecticides would prevent the beetle from establishing galleries within susceptible tree hosts, and prevent the survival of beetles and their offspring already present within trees. The big problem with systemic pesticides is getting sufficient concentrations of chemicals to the areas within the trees where the beetle and fungus occur. Although there are exceptions, most systemic treatments are administered to the soil for uptake through the roots. However, in mature avocado groves, the high organic matter content of the soil can prevent effective absorption by roots because the pesticide becomes bound to organic components within the soil. Trunk injection of pesticides directly into the vascular system of trees eliminates the potential for binding of pesticides within the soil, and increases the amount of active ingredient inside the tree available to impact the beetle/fungal system. Systemic pesticides must be formulated for trunk injection and so careful evaluation is needed to ensure optimal efficacy. Trials are being conducted with the assistance of avocado industry and grower collaborators in areas where the SHB has been recorded. The chemicals are injected into the trees using commercially available equipment, and the movement of the active ingredients is then monitored over time in wood core samples taken at different heights of the trees. Two methods are being used to confirm the presence of the chemicals. Insecticides are being quantified using ELISAs that are specific for the active ingredients under investigation. Wood cores taken from trees treated with fungicides are placed in direct proximity to the fungal pathogens growing on agar plates to determine if growth of the fungus is inhibited.
The investigations are still at an early stage, but the researchers are optimistic that they will develop effective control strategies for the SHB that growers can incorporate into their overall pest management programs. Laboratory based bioassays have been used to identify several pesticides that are toxic to the beetle and fungi. The objective of the field trials is to determine whether these chemicals can be utilized as trunk injection agents for the protection of avocado trees. Anyone interested in finding out more about the SHB should go to the web site maintained by Dr. Akif Eskalen at:
http://eskalenlab.ucr.edu/avocado.html

- Author: Elizabeth Fichtner and Rachel Elkins
Lime-induced Iron Chlorosis: a nutritional challenge in the culture of several subtropical perennial crops in California
Elizabeth Fichtner, UCCE Tulare County and Rachel Elkins, UCCE Lake and Mendocino Counties
Spring, and new leaves are coming out, but this could, but yellow could be a sign of iron chlorosis, as well.
Although iron (Fe) is the 4th most abundant element in the lithosphere, Fe deficiency is among the most common plant micronutrient deficiencies. Fe deficiency in plants is common in calcareous soils, waterlogged soils, sandy soils low in total Fe, and in peat and muck soils where organic matter chelates Fe, rendering the element unavailable for plant uptake. In California, lime-induced Fe deficiency is often observed in soils and irrigation water containing free lime, and is exacerbated by conditions that impede soil drainage (ie. compaction, high clay content), resulting in reductive conditions. Given that over 30% of the world's soils are calcareous, lime-induced Fe deficiency is a challenge in numerous perennial cropping systems including: grapes, pears, apple, citrus, avocado, pecans, and stone fruit (prune, almond, apricot, peach, nectarine, cherry).
In most soils, Fe oxides are the common source of Fe for plant nutrition. Solubility of Fe oxides is pH dependant; as pH increases, the free ionic forms of the micronutrient are changed to the hydroxy ions, and finally to the insoluble hydroxides or oxides. In calcareous soils, the bicarbonate ion inhibits mobilization of accumulated Fe from roots to foliage and directly affects availability of Fe in soil by buffering soil pH. When irrigation water is also high in bicarbonate, probability of Fe deficiency is enhanced because bicarbonate is continuously supplied to the soil, and more importantly, the roots may become crusted with lime as water evaporates, thus inhibiting root growth and function. Inside the plant, bicarbonate inhibits nutrient translocation from roots to aboveground plant parts. The adverse effects of high bicarbonate levels are exacerbated in very saturated, very dry, or compact soils, where bicarbonate levels increase concurrent with diminished root growth and nutrient uptake.
Symptoms of Fe deficiency in plants
Fe is immobile in plants; therefore, symptoms appear in young leaves. Interveinal chlorosis (Figure 1) is the main symptom associated with Fe deficiency, followed by reduced shoot and root growth, complete foliar chlorosis, defoliation, shoot dieback, and under severe conditions may result in tree mortality. Overall productivity (yield) is reduced, mainly from a reduced number of fruiting points.
Plant Adaptation
Plant species and cultivars vary in their sensitivity to Fe deficiency, and are categorized as either "Fe-efficient" or "Fe-inefficient". Fe-efficient plants have Fe uptake systems that are switched on under conditions of Fe deficiency. Fe-inefficient plants are unable to respond to Fe deficient conditions. All Fe-efficient plants, except grasses, utilize a Fe-uptake mechanism known as Strategy 1. Strategy 1 plants decrease rhizosphere pH by release of protons, thus increasing Fe solubility. Some plants may excrete organic compounds in the rhizosphere that reduce ferric iron (Fe3+) to the more soluble ferrous (Fe2+) forms or form soluble complexes that maintain Fe in solution. Additionally, roots of Strategy 1 plants have specialized mechanisms for reduction, uptake, and transfer of Fe within the plant. Strategy 2 plants (grasses) produce low molecular weight compounds called phytosiderophores which chelate Fe and take up the chelated Fe with a specific transport system.
Amelioration of Fe chlorosis
Planting sites in calcareous soils should be well drained to provide optimal conditions for root growth and nutrient uptake. Waterlogged and compact soils contain
more carbon dioxide, which reacts with lime to form even more bicarbonate. These conditions, as well as very dry soils, also inhibit microbial activity which aids in
solubilization and chelation of Fe. Prior to planting, soils and water should be tested to determine the pH, lime equivalent, and bicarbonate concentration. Bicarbonate concentrations greater than 3 meq/L in irrigation water increase the hazard of lime accumulation on and around roots. If high bicarbonate water must be used, the pH must be adjusted to 6.0-6.5 to dissolve the bicarbonate and prevent it from negating the effects of soil-based treatments. In microsprinker and drip systems, acidification of irrigation water will also reduce the risk of emitter clogging, a common problem at bicarbonate levels over 2 meq/L. The cost of reducing the pH of irrigation water will more than compensate for the savings incurred from avoiding wasted investment in failed soil- and plant-based remedies. Systems can be set up to continuously and safely inject water with acids such as sulfuric, urea-sulfuric, or phosphoric during irrigations. Specific choice and rate will depend on crop, soil type, other nutrient needs, availability, and cost. Downstream pH meters are available to continuously adjust rate of acid use. Acetic and citric acid can be utilized by organic growers.
Soil based pre-plant treatments to reduce pH include elemental sulfur (S) and acids as mentioned above. It is only necessary to treat a limited area near the root zone to ameliorate symptoms because the tree only needs to take up a small amount of Fe. Material can be shanked in or banded and incorporated in the prospective tree row. One ton of elemental sulfur per treated acre is needed to mitigate three tons of lime, and may need to be re-applied every 3 to 5 years after planting. The addition of organic matter such as well-composted manures will benefit poorly drained or compact soils by increasing aeration for better root growth, fostering chelation of nutrient cations, and reducing pH (depending on source material).
If possible, choose a Fe efficient species or cultivar. In perennial systems, lime-tolerant rootstocks may be the first line of defense in combating Fe deficiency. Some rootstocksmentioned are peach-almond and Krymsk-86 for stone fruit, Gisela 5 for cherry, and Pyrus communis for pear. Ongoing research studies in Europe focus on screening rootstocks of grape and olive for lime tolerance.
Once soil and water quality improvements are made, post-plant management strategies may also be implemented to ameliorate lime-induced Fe chlorosis in the short term. Soil can be acidified as described above. Individual trees can be treated by digging four to six 12-24 inch
holes around the drip line and burying a mixture of sulfur and Fe fertilizer. Historically, two principal methods have been utilized: 1) foliar application of inorganic Fe salts (ie. ferrous sulfate), and 2) soil or foliar application of synthetic chelates. Application of Fe salts to foliage may have mixed results due to limited penetration of Fe into leaves and inadequate mobilization within the plant. Use of Fe chelates may be of benefit; however, they are expensive and pose an environmental concern due to their mobility within the soil profile. Because soil lime interferes with Fe mobility with the plant, repeat application of inorganic Fe salts or Fe chelates may be necessary throughout the growing season.
Choice of nitrogen (N) fertilizer may also influence solubility of rhizosphere Fe. When N is applied in the ammonium form (NH4+), the root releases a proton (H+) to maintain a charge balance, thus reducing rhizosphere pH. Alternately, fertilization with nitrate (NO3-) results in root release of hydroxyl ions (OH-), resulting in an increase in rhizosphere pH. Solubility of Fe3+ increases 1000 fold with each one unit decrease in pH; therefore, fertility-induced rhizosphere pH changes may significantly influence Fe availability.
New methods for amelioration of Fe chlorosis are under investigation. For example, container studies have demonstrated that inter-planting sheep's fescue, a Strategy 2 plant, with a Fe-inefficient grape rootstock may ameliorate Fe chlorosis in grape. In this system, the grass produces a phytosiderophore that enhances Fe availability to the grape. Additionally, soil amendment with Fe3(PO4)2• 8H2O), a synthetic iron(II)-phosphate analogous to the mineral vivianite, has been effective at preventing Fe chlorosis in lemon, pear, olive, kiwi, and peach. Vivianite has a high Fe content (~30%) and serves as a slow release source of Fe in calcareous soils.
Figures below: Shoot dieback in citrus and iron chlorosis in avocado