- Author: Emma Coflin
Bee Hotels and Gardens – Applications and Limitations
The model of a ‘Bee Hotel' has been a useful tool for surveying insects in research studies for decades. However, in recent years, the concept of a bee hotel has entered the mainstream; capturing the attention of naturalists, gardeners, and those alike. With this transition, it has gained its name and evolved to take on various forms that hardly resemble the humble wooden box model used in research.
Increased interest in creating spaces for bees is hopeful, as it signifies a positive trend in attention to pollinator livelihood. The UC Berkeley Urban Bee Lab has had great success with simplistic wooden box models of bee hotels in our Costa Rica Bee Project. We share here some of the knowledge we have gained through numerous years of research working with bee hotels in both Costa Rica and California. Additionally, we offer some considerations to ponder when creating your own bee hotel, acknowledging the limitations that they hold, especially in regards to a changing climate.
Read the full story at: http://www.helpabee.org/lab-news/bee-hotels-considerations-and-limitations
A Bee Hotel
A Bee Garden in an Avocado Orchard
- Author: Ben A Faber
Rain is wonderful stuff. If it comes and washes the accumulated salts of the last several years out of the root zones of citrus and avocado, that's a good thing. But what happens if there is a little too much rain? In the winter of 2005, Ventura got over 40” of rain, which is 100% more of what is normal. The last time big rains occurred prior to that was in the winter of 1997-98. That year the rains were evenly spaced on almost a weekly basis through the winter and into the late spring and over 50" fell. That year we had major problems with both citrus and avocados collapsing from asphyxiation. The same occurred in 2005, but not so pronounced.
Most rainfall years are not average, usually it is more or less than average. And sometimes the rains come, and flooding happens throughout California. The winter of 1861/62 saw such flooding that the harbor at Goleta was filled in.
In 2023, we had a lot more rain than we normally see and in Carpinteria it rained 4 inches in July!!!!!! And with the rain we saw mudslides and buried orchards, and with wet soil and winds we saw avocado trees being blown over.
And then came the winter of 2023/24, and there was more rain and more devastation (mudslides, buried orchards, etc.). But in general, trees look good. The consequences of low rainfall and the resulting greater application of salty irrigation water is leaf burn and often Botryosphaerias. After two wet rainy winters, we see much less of this kind of damage.
But there's been more subtle damage going on, especially avocados. It's been particularly hard on young trees with poorly developed root systems and orchards on flat ground. Asphyxiation has happened with the soils being too wet. Excluding air reduces root function, especially in the uptake of iron. It also allows ethylene to accumulate. Ethylene is a plant growth regulator that accelerates maturity. When roots sense ethylene, it causes leaf and fruit drop. The combined lack of oxygen and accumulation of ethylene leads to iron chlorosis and leaf drop. These are characteristic symptoms of asphyxiation and wet soils.
What other problems have we seen with rain, and what can be done to prepare for the unexpected? Learning from the Rain
https://ceventura.ucanr.edu/newsletters/Topics_in_Subtropics102527.pdf

- Author: Ali Montazar
Soil moisture sensor selection. An extensive range of soil moisture sensors/probes have already been commercialized and are available to use in avocado and other orchards. They determine the real-time soil water potential (tension) or volumetric water content and are dominated by a small number of technologies including granular matrix or gypsum block sensors, tensiometers, time domain reflectometry (TDR) sensors, and Frequency Domain Reflectometry (FDR) or capacitance sensors (Table 1 and Fig. 2). Some commonly used soil moisture sensors can be combined with various telemetry devices to access the data through cloud-based data storage applications. Data is automatically uploaded by radio or cell phone communications to cloud-based computer servers and is accessible through apps on smartphones and tablets. These communication advancements greatly improve the convenience of accessing data and can be configured to provide timely alerts when trees require irrigation.
Read more about their use in avocado and other orchard tree cropsl, as well as other tasty topics at:
https://ceventura.ucanr.edu/newsletters/Topics_in_Subtropics102527.pdf

- Author: Ben A Faber
We recently compiled the last year's activities in the Ventura and Santa Barbara offices of UC ANR, check it out:
https://ucanr.edu/sites/ucceventura/files/402039.pdf
https://cesantabarbara.ucanr.edu/Quarterly_Reports/

- Author: Ben A Faber
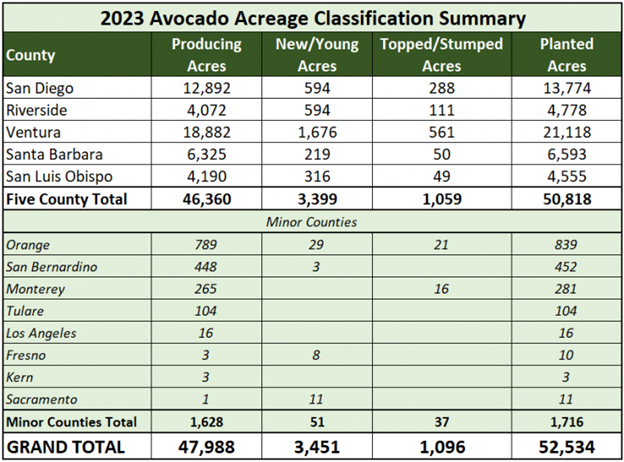
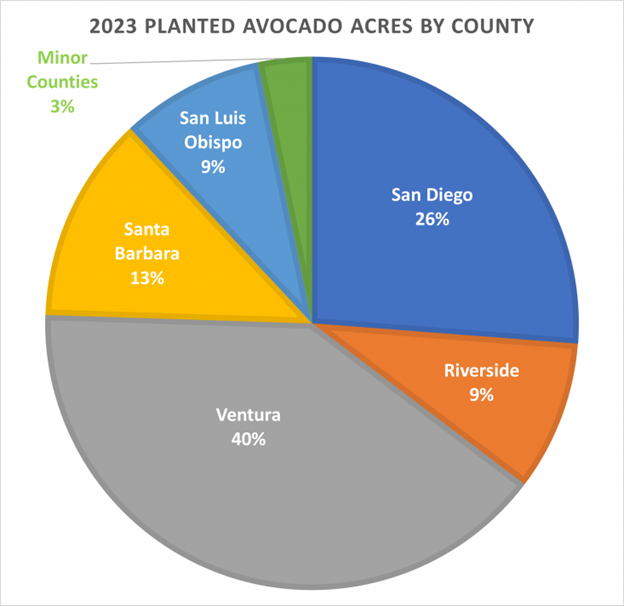
Each year the California Avocado Commission partners with Land IQ to produce a Statewide Avocado Acreage and Condition Analysis report utilizing digital satellite imagery, aerial photography and analytical tools to survey California avocado groves. This data helps the Commission make informed budgeting and marketing decisions and provides industry members with spatial data concerning crop type, location, condition and density. See the full 2023 Report HERE.
According to the report:
- In 2023 there were 52,534 planted avocado acres
- The majority of growing acreage is located in Ventura, San Diego, Santa Barbara, Riverside and San Luis Obispo Counties with a total of 50,818 planted acres
- The five top-producing counties reported 1,059 new/young acres and 3,399 topped/stumped acres
The report also provides acreage data by zip code, county, condition, year planted and density.
The California Avocado Commission's crop estimating team in conjunction with Land IQ uses the latest in remote sensing techniques to assess avocado acreage in production. As technology continues to advance refinements in our fourth generation of remote sensing techniques were applied to satellite imagery collected during spring and summer months. The imagery processing techniques include; segmentation into homogenous polygons, retention of tree crop polygons, calculation of average crop canopy moisture and vegetation indices, analysis of change maps from previous inventories, and classification of avocado groves into four categories; producing, topped/stumped, new/young, and abandoned. Aerial imagery (for a real-world view), and satellite imagery (for spectral and temporal data) are integrated into previously classified avocado acreage and analyzed for current condition of California avocado acreage statewide.
from CA Ag Net: https://californiaagnet.com/2024/07/29/california-avocado-acreage-increases-2023-report-available/