- Author: Ben Faber
Voluntary Best Practices for Growers' Response to Huanglongbing
To provide California citrus growers with a strong toolbox of science-supported strategies and tactics to protect their orchards from Huanglongbing, the Citrus Pest & Disease Prevention Committee endorsed a set of best practices for growers to voluntarily employ in response to HLB in California.
The recommendations – which are grouped based on a grower's proximity to an HLB detection – represent the most effective tools known to the citrus industry at this time and are meant to supplement the California Department of Food and Agriculture's required regulatory response. They were developed by a task force consisting of growers from various regions across the state and scientists, including Dr. Beth Grafton-Cardwell and Dr. Neil McRoberts.
Growers are encouraged to use as many methods as feasible for their operation in order to limit the spread of the Asian citrus psyllid (ACP) and HLB, as the cost to manage the Asian citrus psyllid is far less than any potential costs or loss to the industry should HLB take hold throughout our state.
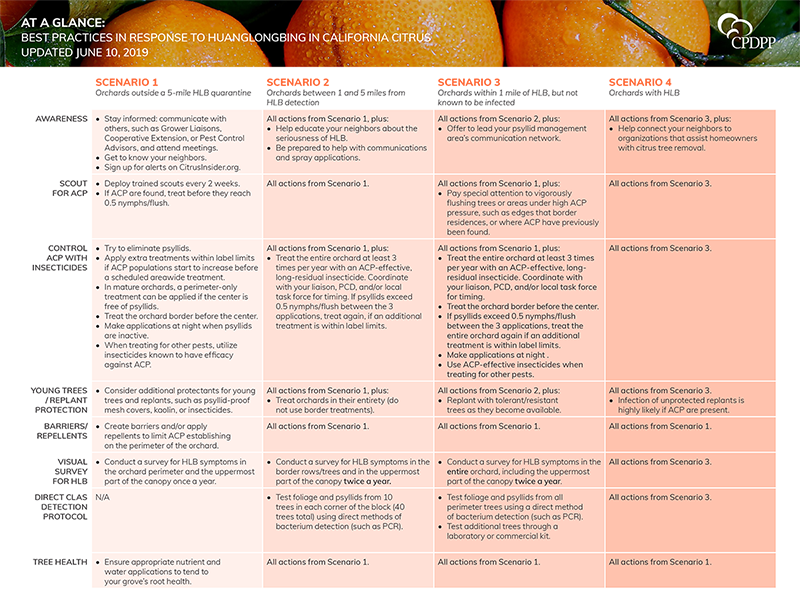
The Best Practices at a Glance
The complete best practices document, which includes the scientific rationale for the best practices, can be downloaded here. The following grid is intended to provide a brief, digestible format of the best practices.

- Author: Ben Faber
Canines can detect trees infected
with the bacterium
that causes huanglongbing
Research by Dr. Tim Gottwald
Article written by Tim Gottwald, Holly Deniston-Sheets and Beth Grafton-Cardwell.
Revised June 13, 2019.
What is the technique?
Canines have a highly sensitive scent detection capability that is significantly better (parts per trillion) than most laboratory instruments and they can be trained to “alert” (either sit or lay) when they detect specific ‘smells' (known as scent signatures). Most people are familiar with their ability to detect bombs, drugs, and plant material at airports. However, canines are also used to detect human pests, such as bed bugs, and agricultural pests, such as stink bugs, date palm weevils and imported fire ants.
With regard to agricultural pathogens, canines have been shown to detect with greater than 98% accuracy the fungal pathogen that causes laurel wilt disease in avocado, the bacterium that causes citrus canker disease in citrus, and plum pox virus in peach orchards.
Researchers have been training and evaluating the efficacy of canines for detecting “Candidatus Liberibacter asiaticus” (CLas), the bacterium that causes huanglongbing (HLB), for 5 years in Florida, and CLas detection efforts with canines have recently begun in California. Dogs have been trained in both the laboratory environment and in the field. Researchers have demonstrated that well-trained canines can detect CLas over 95% of the time in commercial trees and over 92% of the time in residential trees. Researchers did not observe any differences in canine performance between citrus species and varieties. The training that the canines receive is very specific to CLas. When they are taken into citrus orchards infected with citrus tristeza virus, viroids, the fungal pathogen Phytophthora, or the bacterium that causes citrus stubborn, the CLas-trained canines do not respond to these diseases.
Video of canine Maci running a row of trees in the Rio Grande Valley of Texas
The canines provide a significant opportunity to be used as an Early Detection Technology (EDT) in California. In a field study using potted citrus in Florida, dogs could detect CLas in some of the trees as early as 2 weeks after CLas-infected psyllids fed on the trees. In contrast, it can take 1-2 years for CLas to distribute itself in a mature citrus tree sufficiently for the bacterium to be present in sampled the leaves, which are then tested and shown to be infected using laboratory techniques, such as Polymerase Chain Reaction (PCR). Using canines to detect early infections could significantly help reduce disease spread in California, where HLB is currently limited to southern areas of the state and identify areas where increased psyllid control measures are needed
Who is working on the project?
Dr. Tim Gottwald, Research Leader and Epidemiologist at the USDA, U.S. Horticultural Research Laboratory in Fort Pierce, Florida, and additional collaborators with F1K9 laboratories, USDA, North Carolina State University, Texas A&M University and the California Department of Food and Agriculture.
What are the challenges and opportunities?
The volatile scent signature associated with CLas-infection settles from the canopy and simultaneously emanates from root infections pooling at the base of the tree. The detector dog interrogates the tree holistically by alerting in seconds on the scent signature regardless of its origin (i.e., a single leaf, root, stem or the entire tree if systemically infected). Conversely, other detection technologies, like PCR, are reliant on selecting and processing a small amount of tissue from large trees and often miss incipient infections because infected tissue is so rare in newly infected trees. Early detection via dogs is devoid of these sampling issues. Therefore, it is difficult to confirm CLas detections by dogs using currently available molecular or chemical detection methods. Dogs have been tested in hot and cold temperatures and with wind speeds up to 20 MPH with no perceptible degradation in detection.
Human scouts require several minutes per tree to visually examine it for symptoms, then they must collect tissue which must be transported to a diagnostic lab for processing and analysis, which is time consuming and labor-intensive. Whereas, in a residential environment dogs can assess all trees in even large yards in a couple of minutes. The major limitation to the number of trees a dog can assess per day is access to these residential properties and the time required to relocate from property to property. In commercial groves a team of two dogs and one handler can survey a 10 acre planting (~1500 trees) in 1-2 hours depending on the number of infected trees; each positive alert requires rewarding the dog and tagging the infected tree. Dogs usually work 30 min then rest 30 min and can work 6-8 hours a day.
Utilizing dogs, CLas can be detected early in a region, when it is in just a few trees. If these few early infected trees are removed, the establishment and spread of the disease could be greatly reduced.
Like every detection instrument, dogs need to be periodically recalibrated. This is done by resensitizing them to known CLas-positive trees or specially prepared ‘scent pads' that contain the scent signature of CLas to ensure they maintain > 98% accuracy of detection before being redeployed.
Funding source: This project is funded by the USDA Farm Bill, USDA HLB Multiagency Committee (MAC), and USDA ARS program funds.
This article originally posted on the Science for Citrus Health website.
Photo: Canine checking trees at Lindcove Research and Extension Center, Exeter, CA

By John Krist
Chief Executive Officer of the Farm Bureau of Ventura County. Contact him at john@farmbureauvc.com.
Nearly 600 scientists, government agency representatives and members of the agricultural community gathered in Riverside last month for the sixth International Research Conference on HLB. This was the first iteration of the IRCHLB to take place in California, but like its predecessors in Florida, the conference drew a global sampling of smart people trying to figure out an answer to the most pressing question facing the worldwide citrus industry: How do we stop Huanglongbing and the Asian citrus psyllid from destroying it?
As was the case at previous conferences, no definitive answer to that question was provided by any of the hundreds of research presentations and posters. Work continues on every imaginable mechanism for disrupting the lethal vector-host-pathogen complex: breeding HLB-resistant or HLB–tolerant citrus rootstocks and scions; disrupting the ability of ACP to reproduce, feed or acquire the disease-causing bacteria; treatments to cure or reduce symptoms of infection; genetically or biologically based methods for killing ACP more efficiently. The “solution,” however, remains as elusive as it was during all previous biennial conferences.
But there have been important advances. This year, researchers presented results from their use of a tool that did not exist as recently as seven years ago. And it offers the prospect of rapid development of a suite of interventions — HLB-tolerant trees, crippled psyllids, perhaps even a lethal agent that attacks the bacteria themselves — that could change the course of the epidemic.
The new approach is being made possible by a gene-editing technology known as CRISPR. The term (pronounced “crisper”) is an acronym for “Clustered Regularly Interspaced Palindromic Repeats,” and it refers to odd DNA fragments discovered 20 years ago by scientists examining the genome of various families of microbes.
The short, repeated fragments of DNA were distinctive and appeared unrelated to the remainder of the microbial genome. Additional investigation revealed similar structures in the genomes of microbes from vastly different families of life, suggesting that they were not random but performed some function useful to microbial survival.
Research over the next decade revealed that CRISPRs were a feature of bacterial immune systems. Bacteria attacked by viruses (known as bacteriophages, or just phages) evolved a way to duplicate small snippets of the invader's DNA, insert it into their own genome, and pass that modified genetic code along to subsequent generations of bacteria. If attacked again by a phage containing that same snippet of DNA, the bacterial DNA recognizes it and triggers a targeted enzyme response that severs the viral genome at precisely that point, disabling the virus.
These discoveries led eventually to a technique by which researchers can pretty much treat the genome of any organism as a set of Lego blocks, its pieces plugged in, removed or replaced virtually at will. Using tools developed in the lab, but replicating the function of the CRISPR complex in microbes, they can dispatch custom-made genetic packets into living organisms, where they can remove, silence, activate or replace specific genes and their functions.
Since scientists first reported six years ago that this adaptive microbial immune system could be repurposed into a simple and reliable technique for gene editing, thousands of researchers have been exploring its application in a wide variety of fields. One of them is the quest for a solution to HLB. And at this year's conference, several teams reported progress toward using the CRSPR system to modify the genome of all three elements of the pest-host-disease triad: ACP, the HLB bacteria, and citrus trees.
Perhaps the most remarkable of these reports carried an inscrutable title: “BAPC-assisted-CRISPR-Cas9 Delivery into Nymphs and Adults for Heritable Gene Editing (Hemiptera).”
Hidden within that bland language was a dramatic achievement: Two scientists — Wayne Hunter at the U.S. Department of Agriculture's Fort Pierce lab in Florida, and John Tomich of Kansas State University — injected a special variant of the CRISPR package, designed to knock out two genes in the ACP genome, into a female adult psyllid. Her offspring inherited a CRISPR-modified genome in which those two genes — one governing physiological development, and the other eye color — had been deleted.
As nymphs, the genetically modified ACP took much longer to develop and had lower survival rates. As adults, the survivors had strange white eyes and malformed wings, and their lifespan was one-third of normal.
Those would be useful achievements on their own. But the experiment also demonstrated a powerful tool for determining the functions of other ACP genes by disabling them and observing the results on psyllid morphology, physiology or behavior. This could potentially lead to one of the Holy Grails of ACP-HLB research — a psyllid that is incapable of transmitting the disease-causing bacterium.
Driving that genetic trait throughout the ACP population would halt the epidemic in its tracks. Growers could greatly reduce, if not eliminate, their reliance on pesticides as a means of slowing disease spread by suppressing the vector population — currently the only viable strategy.
There were several other presentations about research to use CRISPR or other techniques to genetically modify the citrus genome to improve HLB tolerance — a much more rapid way of generating potential new rootstocks than the years-long process required to screen varieties and rear new trees — and to disable or kill the bacteria themselves.
The speed with which this field of knowledge is evolving, and the remarkable power of the CRISPR technology, left many of us who heard these presentations hopeful. After years of tantalizing research “breakthroughs” that have failed to yield meaningful and deployable interventions, the fight against ACP and HLB may soon be waged on our terms – not the bug's.
Until transgenic psyllids move from the lab to the field, however, we're stuck with our current approach to disease and vector management: Suppress ACP populations with chemicals, identify and remove infected trees as quickly as possible, and replant using disease-free nursery stock. But there was exciting news on this front as well.
From the beginning, the effort to identify and remove HLB-positive trees has been hampered by the long lag time between initial infection and confirmation using the classic DNA testing that serves as the regulatory gold standard. Because the test looks specifically for sequences of DNA that are unique to the bacteria in samples of ACP or plant tissue, it can provide incontrovertible and direct evidence of infection. From a legal standpoint, this is key when regulators act to force removal of positive trees.
The problem, however, is one of sampling. A mature citrus tree may have 200,000 leaves, yet in the early months and even years of infection, the bacteria are not distributed uniformly through the canopy. Typical samples submitted for DNA analysis may consist of as few as 12 leaves per tree. Chances of picking the right leaves are minuscule when the bacterial population is small, and perhaps isolated only in a particular branch. The odds are stacked against the sampling effort from the start.
This is why a major branch of the global research effort is devoted to developing early detection technologies — methods of confirming probable HLB infection based on changes in tree physiology that come about within days or weeks of infection. Being able to identify and remove infected trees before significant numbers of ACP have had the opportunity to feed on them and acquire the bacteria would greatly improve our ability to influence the course of the epidemic.
Several good candidates have been identified and validated, but they require intensive laboratory processing, and capacity limitations have stymied their wide use. But the most promising of these technologies doesn't require a lab at all.
As reported here after the 2017 IRCHLB, a team of dogs trained according to protocols developed by researchers at USDA and North Carolina State University have demonstrated the ability to correctly identify HLB-positive trees as little as two weeks after infection. During field trials, their accuracy rate has been up to 98 percent.
From the outset of this project, which has been funded through a federal grant, lead researcher Tim Gottwald of the USDA theorized that the dogs were identifying a change in the suite of volatile organic compounds emitted by trees. This is known to occur almost immediately upon infection, as the plant mounts a physiological defense against the invading bacteria. Sick trees don't smell like healthy trees, and Gottwald's theory has been that dogs can tell the difference.
At this year's conference Gottwald declared that he'd been wrong. In more recent experiments, his team injected the HLB bacterium into plants that are utterly unlike citrus — periwinkle, tobacco, dodder. Although the dogs had been trained on citrus, they correctly identified the inoculated non-citrus plants even though they smelled nothing like oranges or lemons. He then took it one step further, and ran the dogs past cages containing ACP, some of which had been infected in the lab. The dogs were able to correctly identify infected psyllids as well. And bugs don't smell anything like plants.
To confirm his new hypothesis, he exposed the dogs to an array of bacterial cultures, some of which included the bacteria known to cause HLB. The dogs were able to identify those, too. Gottwald now believes that when the dogs identify citrus tree as infected with HLB, it's because they are picking up the actual scent of the bacteria within it.
If that's the case, it would provide a powerful argument for immediate and mandatory removal of suspect trees very early in the disease process: The dogs would providing direct evidence of infection. From a regulatory standpoint, it should be no different from a positive DNA test result.
The current risk-based HLB survey being conducted throughout the state — consisting of systematic collection of plant tissue and psyllid samples for DNA testing — has been heavily weighted toward urban areas, which is how the exploding epidemic in Los Angeles, Orange and Riverside counties was discovered. The current tally of HLB-positive trees removed there now exceeds 1,200, all of them in urban yards.
Less attention has been paid, however, to rigorous surveying of commercial groves. This places an imperative on arranging for trained dogs and handlers, now based in Florida, to begin their work here. Nothing is more important than finding HLB in commercial groves while it is early enough for tree removal to stop or slow the spread of the epidemic. The CRISPR research has opened the door to an entirely new array of potential weapons against HLB, but the citrus industry needs to survive long enough to use them.
Photo:
HLB deformed fruit

- Author: Ben Faber
Critical Leaf Nutrient Thresholds to Diagnose Deficiencies in HLB Trees
By Arnold Schumann, Laura Waldo, Tripti Vashisth, Alan Wright and Kelly Morgan
Huanglongbing (HLB) disease severely impacts the nutrient status of citrus trees, particularly by stunting the feeder roots and causing measurable deficiencies of nutrients in the roots and canopies. Visible symptoms of nutrient deficiencies on citrus foliage are characteristic but not diagnostic of HLB disease.
Recent HLB research efforts have focused on manipulating nutrient deficiencies with fertilizer applications to mitigate HLB symptoms and help keep affected trees alive and productive. Efficient diagnosis of nutrient imbalances in HLB-affected trees (both deficiencies and excesses) is necessary to make fertilizer remedies cost-effective and productive. Published thresholds defining deficient, low, optimal, high and excess ranges of nutrient concentrations in citrus leaves were derived long before HLB spread in Florida. Therefore, we conducted a series of survey studies to verify and improve those existing thresholds in HLB-endemic Florida citrus groves.
Traditionally, nutrient thresholds were obtained empirically from long-term replicated fertilization trials conducted decades ago, most notably where “missing nutrient” experiments would, over many years, cause those nutrients to become deficient in trees not receiving certain fertilizers. By comparing treatment plots to fully fertilized control plots, the fruit yield reduction and other symptoms caused by the nutrient deficiency could be quantified.
Critical threshold concentrations (CTCs) derived for each nutrient from those studies were then published and used as thresholds to diagnose leaf tissue samples collected from commercial groves. In the current study, we used a sample survey method and the Diagnosis and Recommendation Integrated System (DRIS) instead of “missing nutrient” experiments in order to save considerable time.
STUDY SETUP
Leaf tissue samples were collected quarterly from 2016 to 2018 in three citrus-growing regions of Florida (11 locations from the Central Ridge, five from the east coast Indian River and six from the southwest Flatwoods). Soil samples were collected annually, and tree canopy measurements, leaf size, starch content, quantitative polymerase chain reaction (qPCR) assay and other measurements were conducted periodically for the same sites. In this article, we will focus only on the leaf tissue nutrients of sweet orange trees and assume that most of the sampled trees were HLB-positive, based on the qPCR analyses.
ABOUT DRIS
In an April 2009 Citrus Industry article, DRIS was introduced as a promising method for interpreting nutrient levels in plant tissues. See “Potential use of DRIS for leaf nutrient diagnosis in Florida citrus” (www.crec.ifas.ufl.edu/extension/trade_journals/trade_journals2009.shtml).
The DRIS method produces an index for each plant nutrient calculated from a series of leaf concentration ratios of that nutrient with all other measured nutrients, relative to a set of nutrient norms (standards) that were previously calculated from a high-yielding grove. We obtained the DRIS norms from leaf samples collected in the summer of 2003, from a 22-year-old block of Hamlin orange trees on Cleopatra mandarin rootstock near Fort Meade, Florida, spaced 20 by 25 feet and with an average fruit yield over four years of 770 boxes per acre. The leaf sampling for DRIS norm development coincided with a peak yield of 970 boxes per acre in the 2003–04 season, prior to Hurricane Charlie and the appearance of HLB in Florida.
DETERMINING DEFICIENCIES
The application of DRIS to diagnose nutrient deficiencies in leaf tissue from HLB-affected trees is best done in conjunction with the CTCs. The CTCs are the thresholds for each nutrient that determine whether a given tree or grove would be responsive to fertilizer supplying that nutrient. Traditionally the CTCs occupy a spot on the plant response curve for each nutrient concentration of about 90 percent maximum yield or growth.
When multiple deficiencies occur, as is often the case with HLB-affected trees, the DRIS indices provide information about the relative severity and ranking of the deficiencies, which the CTCs do not accomplish. Dealing with multiple nutrient deficiencies efficiently is important because of the biological stoichiometry that determines the ideal proportions of nutrients in a plant, just as, for example, a water molecule consists of exactly two atoms of hydrogen and one of oxygen.
Liebig's law of the minimum applied to plant growth, and often visualized as a leaking barrel, (Figure 1) demonstrates that it is the most limiting nutrient that limits growth and yield, regardless of the amounts or types of other nutrients available. Another analogy is that a gasoline engine will not run faster if only the gasoline flow is increased without proportionately increasing the air (oxygen) supply, such as if the choke control is active. The engine may flood and stall if too much gasoline is supplied without increasing the most limiting ingredient for combustion at that time (oxygen).
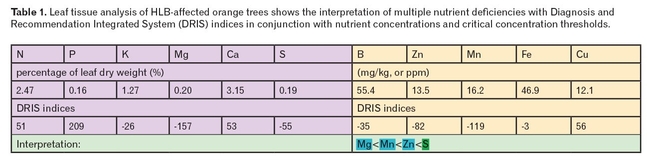
For example, consider the results in Table 1 of leaf tissue analysis from an HLB-affected tree.
DRIS indices have a theoretical optimum at zero, with more negative values indicating possible nutrient deficiency, and increasing positive values indicating possible nutrient excess. In the Table 1 example, Mg<Mn
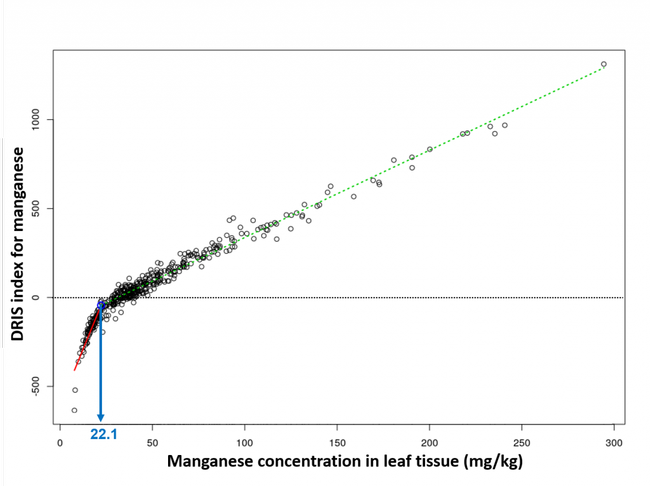
Figure 1. Scatter plot of leaf manganese Diagnosis and Recommendation Integrated System (DRIS) indices versus manganese concentrations in HLB-affected citrus. The breakpoint between red and green regression lines indicates the location of the critical threshold concentration (CTC = 22.1 mg/kg).
CALCULATING CTCs
To examine the validity of currently available CTCs for nutrient diagnosis in HLB-endemic Florida citrus, we used scatter diagrams of the DRIS indices for each nutrient plotted against the nutrient concentrations. This technique has been used before in other crops to determine CTCs from nutrient survey data, including loblolly pine, cotton and signalgrass. Figure 2 shows that the scatter plot for manganese in our survey of HLB-affected groves follows two data trends of different slopes, with the breakpoint indicating the CTC for that nutrient on the x-axis. We used segmented linear regression to determine the correct breakpoint.
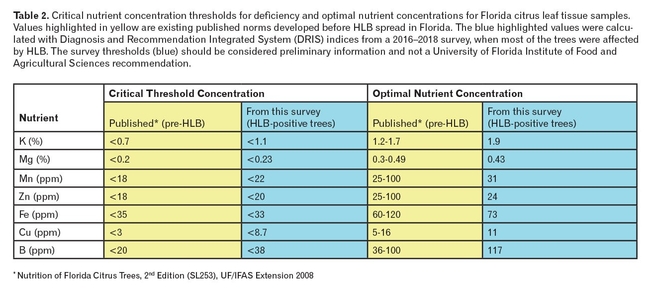
The process was repeated for each nutrient in order to determine the preliminary CTCs listed in Table 2. The optimal nutrient ranges in Table 2 were estimated from the intercept of the regression line with zero on the y-axis (DRIS). By comparing our new CTCs with published CTCs, it appears that deficiency diagnoses for copper, boron, potassium, manganese and zinc could be underestimated by currently available CTCs when used for HLB-affected trees.
The optimal nutrient concentrations calculated from this study are mostly in agreement with ranges previously published by the University of Florida Institute of Food and Agricultural Sciences (UF/IFAS), except that boron and potassium optimal levels were estimated slightly higher in the survey than those previously published. These results are preliminary, and in this study, CTCs could only be calculated for those nutrients where deficiencies occurred in the survey data. We plan to augment this survey with more leaf nutrition data to better define and expand CTC estimates to be used for diagnosing HLB-affected citrus for all nutrients.
Please contact us if you have leaf nutrient data from your groves that you are willing to share in this study, or if you would like to use the DRIS program which will be published online to a UF/IFAS web page.
SUMMARY
In this article we provided updates on research efforts to improve the nutrition of HLB-affected Florida citrus by revising the diagnostic CTC of nutrients in leaf tissue. We also revisited the DRIS method for improving diagnosis of multiple nutrient deficiencies that are common in HLB-affected groves, including potassium, magnesium, manganese, zinc, iron, boron and copper. Additional leaf nutrient data will be obtained for an expanded survey, to be analyzed and corroborated with results of recently completed micronutrient fertilization experiments.
Acknowledgements: We thank the grower cooperators who offered their groves for our nutrient surveys, including Gapway Groves, Ben Hill Griffin Groves, OrangeCo, Brent Shirard, Michael Monroe and English Brothers. We are grateful to the Citrus Research and Development Foundation for financial support of this project.
Arnold Schumann (schumaw@ufl.edu) and Laura Waldo are soil scientists, and Tripti Vashisth is a horticulturist, all at the UF/IFAS Citrus Research and Education Center in Lake Alfred. Alan Wright is a soil scientist at the UF/IFAS Indian River Research and Education Center in Fort Pierce. Kelly Morgan is a soil scientist and center director at the UF/IFAS Southwest Florida Research and Education Center in Immokalee.
Table 1. Leaf tissue analysis of HLB-affected orange trees
Figure 1. Manganese concentration of leaf tissue
Table 2. Critical nutrient concentration thresholds
- Author: Ben Faber
News from the Ventura County ACP-HLB Task Force
Winter 2019 area-wide treatment cycle has begun
This is a reminder that the Winter 2019 area-wide management (AWM) treatment window opened Jan.7. You are encouraged to file pesticide use reports (PURs) electronically through CalAgPermits, Agrian, or another system that allows treatments reports to be rapidly filed and recognized. Treatment reminders will be emailed (mailed for those without an email record) about 2-3 weeks prior to the treatment window. In some instances, our contact list may only have contact information for your farm manager, pest control advisor, or pest control operators. If you are not receiving emails, please contact one of your grower liaisons (see contact information below) to receive information about ACP, HLB and your citrus.
Website redesigned
The Citrus Pest & Disease Program (CPDPP) launched a redesign of its website to provide members of California's citrus industry easier access to the key maps, regulatory updates and events they need to stay informed on the fight against HLB in California. The website is at www.citrusinsider.org.
HLB confirmations continue to increase
At least 1,024 residential trees had been confirmed as infected with HLB as of January 2019. No HLB-positive trees have been found in commercial groves. The HLB quarantine boundaries and the latest tally of HLB confirmations, updated weekly, is available online at https://citrusinsider.org/maps-and-quarantines/.
Report neglected and abandoned citrus
Help prevent neglected and abandoned citrus from serving as a breeding ground for ACP and the spread of HLB by reporting its location County Agricultural Commissioner's office at (805) 388-4222. If your citrus is not worth the resources required to protect it from ACP and HLB, it may be a good time to consider removing the trees. Tree-removal assistance is offered to small growers through the California Citrus Mutual and Bayer's ACT NOW program. For more information contact Joel Reyes at jreyes@cacitrusmutual.com or (559) 592-3790.
Calendar
UCR Citrus Day (Jan. 29)
This year's UC Riverside Citrus Day will be on Tuesday, Jan. 29. Agenda and registration information can be found by clicking here.
International Research Conference on HLB/Citrus Virologist Conference (March 10-15)
The joint International HLB and Citrus Virologist conference will be at the Riverside Convention Center in Riverside, CA. Registration and more information can be found here.
Citrus Pest and Disease Prevention Committee meeting (March 12)
The CPDPC is charged with advising the state on management of the Citrus Pest and Disease Prevention Program. The CPDPC and subcommittee meetings are open to the public, and options for participation include in person or by webinar and conference call. Click here to view and register for upcoming committee and subcommittee meetings. Attendance is free.
esources
University of California ACP area-wide materials list and ACP monitoring protocols
Movement of bulk citrus materials list
Ventura County AWM maps and schedule
Citrus Pest and Disease Prevention Program
Ventura County ACP-HLB Task Force mailing list signup
Contact your grower liaisons if you have additional questions:
Sandra Zwaal
(949) 636-7089
Cressida Silvers
(805) 284-3310