- Author: Drew Lyon
- Posted by: Gale Perez

- A WSU Wheat Beat Podcast episode, “Crossing the Boundaries of Herbicide Resistance” with Drs. Ian Burke and Nick Bergmann, highlights the role of social science in tackling the problem of herbicide resistance. In their conversation with me, they share how social scientists can facilitate more meaningful discussion between growers, scientists, and government agencies to identify possible solutions for managing herbicide resistance. Listen to the podcast and share your thoughts on the approach they discuss.
- Ian Burke and Drew Lyon are members of the GROW (Getting Rid of Weeds) network, a network of weed scientists from across the U.S. focused on herbicide resistance management. GROW just published a new page about the Basics of Herbicide Resistance. The page covers how herbicide resistance develops, how to identify herbicide resistance, and what future threats might be evolving, such as metabolic resistance. The information on this page provides a strong foundation for understanding herbicide resistance. We welcome your thoughts on the page, including what additional information you would like to see on the topic.
- Italian ryegrass is the poster child for herbicide resistance in the inland PNW. Several PNW colleagues and I just published PNW778, Italian Ryegrass Management in Inland Pacific Northwest Dryland Cropping Systems. If Italian ryegrass plagues your farming operation, or you just want to learn more about this particularly adaptable weed, download a free copy of the publication for your library, and let us know if you find the information helpful.
The problems created by herbicide resistance will likely get worse as we rely more on a fewer number of still effective herbicides to manage troublesome weeds. To stay up-to-date on the latest resources, visit Herbicide Resistance Resources regularly, and please let us know what additional information you are looking for. Thanks!
Drew Lyon
Endowed Chair in Small Grains Extension and Research, Weed Science
Washington State University
Original source: Weeders of the West blog :: May 30, 2024
- Author: Ryan Hill
- Author: Marcelo Moretti
Introduction:
Pollinator insects are essential to produce many economically and nutritionally important crops grown in the western USA. These crops include blueberries, almonds, sunflowers, cucurbits, and many others. Almond pollination in California plays a vital role in the apiary industry, driving beekeepers to haul huge numbers of bee colonies to California for the few weeks in late winter when almonds bloom. Bees are selective of the pollen and nectar they forage, and diverse floral resources can allow bees to forage according to their nutritional needs (Leponiemi et al. 2023). Planting pollinator habitat in natural areas, gardens, and agricultural land is one method of supporting bee health. Irrigated agricultural land in the western USA can be an excellent resource for bees during the dry summer when flowers are rarer. However, the resident weeds in these settings are often not of high nutritional quality for hungry pollinators. To make matters worse, pollinator habitat in agricultural fields can be choked out by competition from weeds. Our control plots from these studies (Figure 1) demonstrate that point effectively.
Figure 1: Control plots at all three experimental sites were extremely weedy. This sometimes meant that none of the planted species could grow, as seen in the pictures above.
Objective:
The studies described here attempt to use herbicides to improve the chances for success in pollinator habitat establishment.
Methods:
Three locations in Oregon's Willamette Valley were selected for studies. Two were hazelnut orchards watered with drip irrigation, and one was a field plot set up for sprinkler irrigation. Each location received different soil preparation. The first orchard location (Corvallis) was not tilled, and soil compaction issues were present. The second orchard location (Amity) was power-harrowed, so the top two inches of soil were loosened. The third location (Lewis-Brown Research Farm) was plowed and disked.
All three locations were seeded in the fall with a set of flowering species with potential for pollinator habitat (Table 1).
|
Table 1: Species and seeding rates used for pollinator habitat establishment in Oregon's Willamette Valley. |
||
|
Common Name |
Scientific name |
Lb/Acre |
|
Hairy vetch |
Vicia villosa |
60 |
|
Lacy Phacelia |
Phacelia tanacetifolia |
12 |
|
California poppy |
Eschscholzia californica |
8 |
|
Farewell to spring |
Clarkia amoena |
2 |
|
Globe Gilia |
Gilia capitata |
2 |
|
Sweet alyssum |
Lubularia maritima |
2 |
These species were planted in rows, and herbicide treatments were applied over the top perpendicular to planting rows (Table 2). Four herbicides were applied post-emergence, and the rest were applied one day after planting (pre-emergence). Glyphosate treatments were only included in the orchard trials. Experimental plots were set up as a randomized complete block design with four replicates, and each species was treated as a separate experiment. A crop oil concentrate at 1% v/v was included for Motiff (mesotrione) and Basagran (bentazon), while a nonionic surfactant at 0.25% was included for Matrix (rimsulfuron) and Quinstar 4L (quinclorac). All post-emergent treatments (and glyphosate) included ammonium sulfate (AMsol 1% v/v).
|
Table 2: Trade name, active ingredient, and rate of herbicides applied to pollinator habitat species. Pre-emergent herbicides were applied at planting, and post-emergent herbicides were applied 30 days after crop emergence. |
||
|
Trade name |
Active Ingredient |
Rate (product/A) |
|
Pre-emergent treatments |
|
|
|
Cornerstone Plus |
Glyphosate |
3 qt |
|
Alion |
Indaziflam |
4 fl oz |
|
Trellis SC |
Isoxaben |
21 fl oz |
|
Devrinol 2XT |
Napropamide |
8 qt |
|
Chateau SW |
Flumioxazin |
6 oz |
|
Prowl H2O |
Pendimethalin |
6.3 pt |
|
Princep |
Simazine |
4 qt |
|
Motiff |
Mesotrione |
6 fl oz |
|
Post-emergent treatments |
||
|
Motiff |
Mesotrione |
6 fl oz |
|
Matrix |
Rimsulfuron |
4 oz |
|
Quinstar |
Quinclorac |
12.6 fl oz |
|
Basagran |
Bentazon |
2 pt |
In Amity, competition from perennial grasses resulted in poor stand establishment. A grass-selective herbicide (clethodim) was used, and the site was reseeded six months after the initial planting when soil conditions were appropriate.
Results and discussion:
Site differences.
Drastic differences were seen between sites. Table 3 shows how crop coverage differed between the three sites for each species.
Coverage at the Corvallis site was deficient for all species except hairy vetch.
Several species did very well at the Amity location. Phacelia in the glyphosate plots was exceptionally well established due to glyphosate's good control of perennial grasses that were not killed by the power harrow.
Lewis-Brown (LB) plots had the best crop establishment initially. However, this location had intense pressure from perennial weeds, so the initial crop establishment did not translate to superior pollinator habitat. The plots at LB where Alion was applied produced a good stand of Canada thistle (Cirsium arvense) by the end of the trial, which the bees loved.
|
Table 3: Crop Coverage for each species at each location is shown here. The values reported are from the treated plots with the highest coverage. |
|||||||
|
% Crop coverage (Best treatment) |
Site |
Vetch |
Phacelia |
Poppy |
Gilia |
Clarkia |
Lobularia |
|
Plowed and Disked |
Lewis-Brown Research Farm (LB) |
100 |
100 |
75 |
84 |
97 |
98 |
|
Power harrow |
Amity |
55 |
81 |
47 |
47 |
34 |
47 |
|
No tillage |
Corvallis |
89 |
11 |
28 |
0 |
15 |
0 |
Pre-emergent treatments
Pre-emergent herbicides often had inconsistent pollinator species safety; however, several combinations seemed safe. Napropamide was safe for Phacelia, Gilia, Clarkia, and Lobularia, while flumioxazin and pendimethalin were safe for poppy (Table 5). All five species only had adequate crop establishment at two of the three locations. Hairy vetch establishment was improved by simazine applications at all three locations, but crop coverage was not significantly different from the untreated control for this species (Table 5). Figure 2 shows the treatment by species combinations that were sometimes safe versus the combinations that were consistently safe for the planted species.
At the two orchard sites, glyphosate treatments were the best for Gilia, Phacelia, and poppy establishment (Table 5).
All three trials were conducted on fine soils with organic matter content ranging from 2-7% (USDA-NCSS soil survey). The safety of pre-emergent herbicides for pollinator species establishment may vary depending on soil characteristics.
Post-emergent treatments
Post-emergent (POST) applications were challenging to evaluate for safety. Weed control efficacy was inadequate, and so often, crop establishment was not good enough to confidently assess crop injury.
One exception was hairy vetch. This species exhibited good tolerance to a post-emergent application of Basagran, a result seen at all three locations. The results from two trials suggest that Clarkia tolerated POST applications of Quinstar. Not enough data were collected to conclude the other four species. See Table 4 for crop coverage data.
Conclusions
Site preparation was an essential consideration in our study. Soil compaction and perennial weed pressure must be addressed to have a successful pollinator habitat planting. It was also clear that pre-emergent herbicides can improve habitat establishment, but safety must be adequately established. This is especially true of different soil types and environments. In California's Central Valley, pendimethalin has been seen to occasionally cause injury in poppy plantings, which is in contrast with this study.
|
Table 4: Spring crop coverage (%) from plots treated with post-emergent herbicides one month after planting, which happened the prior October. Missing data from Phacelia and Lobularia at LB is due to crop loss from frost injury. |
|||||||
|
Crop coverage (%) |
|||||||
|
|
Corvallis |
Amity |
LB |
|
Corvallis |
Amity |
LB |
|
Hairy Vetch |
|
|
|
Gilia |
|
|
|
|
Untreated |
29 |
28 |
98 b |
Untreated |
0 |
8 |
54 b |
|
Mesotrione |
21 |
3 a |
0 a |
Mesotrione |
0 |
0 |
0 a |
|
Rimsulfuron |
18 |
1 a |
0 a |
Rimsulfuron |
0 |
4 |
78 b |
|
Quinclorac |
34 |
0 a |
28 a |
Quinclorac |
0 |
0 |
6 a |
|
Bentazon |
50 |
26 b |
94 b |
Bentazon |
0 |
7 |
47 b |
|
Phacelia |
|
|
|
Clarkia |
|
|
|
|
Untreated |
0 |
53 ab |
|
Untreated |
3 a |
0 |
83 b |
|
Mesotrione |
0 |
42 ab |
|
Mesotrione |
0 a |
0 |
34 b |
|
Rimsulfuron |
5 |
17 a |
|
Rimsulfuron |
4 ab |
0 |
55 b |
|
Quinclorac |
5 |
64 b |
|
Quinclorac |
15 b |
0 |
77 b |
|
Bentazon |
2 |
48 ab |
|
Bentazon |
6 ab |
0 |
0 a |
|
Poppy |
|
|
|
Lobularia |
|
|
|
|
Untreated |
2 |
11 |
2 |
Untreated |
0 |
0 |
|
|
Mesotrione |
3 |
11 |
0 |
Mesotrione |
0 |
0 |
|
|
Rimsulfuron |
6 |
0 |
0 |
Rimsulfuron |
0 |
0 |
|
|
Quinclorac |
2 |
5 |
3 |
Quinclorac |
0 |
0 |
|
|
Bentazon |
2 |
9 |
0 |
Bentazon |
0 |
0 |
|
|
Table 5: Spring crop coverage for pre-emergent herbicide treatments applied just after planting, which happened the prior October. Phacelia and Lobularia experienced winter kill at the LB location, so reported data is coverage from December for that location. |
||||||||
|
Crop coverage (%) |
||||||||
|
|
Corvallis |
Amity |
LB |
|
Corvallis |
Amity | LB | |
|
Hairy Vetch |
|
|
|
Gilia |
|
|
|
|
|
Nontreated |
29 |
28 ad |
98 b |
Nontreated |
0 |
8 a |
54 c |
|
|
Glyphosate |
23 |
46 bd |
|
Glyphosate |
0 |
47 b |
|
|
|
Indaziflam |
6 |
3 d |
79 b |
Indaziflam |
0 |
3 a |
36 bc |
|
|
Isoxaben |
11 |
17 ab |
83 b |
Isoxaben |
0 |
5 a |
31 ac |
|
|
Napropamide |
8 |
19 ab |
91 b |
Napropamide |
0 |
38 b |
56 c |
|
|
Flumioxazin |
5 |
36 bd |
90 b |
Flumioxazin |
0 |
18 a |
2 c |
|
|
Pendimethalin |
6 |
38 bd |
98 b |
Pendimethalin |
0 |
0 a |
13 ab |
|
|
Simazine |
34 |
55 d |
98 b |
Simazine |
0 |
44 b |
6 ab |
|
|
Mesotrione |
6 |
25 abc |
0 a |
Mesotrione |
0 |
3 a |
8 ab |
|
|
Phacelia |
|
|
|
Clarkia |
|
|
|
|
|
Nontreated |
0 a |
53 bc |
96 b |
Nontreated |
3 |
0 a |
83 b |
|
|
Glyphosate |
11 b |
92 c |
|
Glyphosate |
5 |
34 b |
|
|
|
Indaziflam |
0 a |
13 ab |
90 b |
Indaziflam |
0 |
0 a |
74 b |
|
|
Isoxaben |
0 a |
0 a |
82 b |
Isoxaben |
0 |
8 a |
88 b |
|
|
Napropamide |
2 a |
81 c |
98 b |
Napropamide |
0 |
25 b |
86 b |
|
|
Flumioxazin |
0 a |
30 ab |
57 ab |
Flumioxazin |
0 |
5 a |
20 a |
|
|
Pendimethalin |
0 a |
0 a |
15 a |
Pendimethalin |
0 |
2 a |
67 b |
|
|
Simazine |
0 a |
75 c |
56 ab |
Simazine |
0 |
30 b |
0 a |
|
|
Mesotrione |
2 a |
54 bc |
99 b |
Mesotrione |
0 | 0 a | 25 a | |
|
Poppy |
|
|
|
Lobularia |
|
|
|
|
|
Nontreated |
2 a |
11 |
2 a |
Nontreated |
0 |
0 a |
70 c |
|
|
Glyphosate |
30 b |
42 |
|
Glyphosate |
0 |
21 ab |
|
|
|
Indaziflam |
2 a |
8 |
8 a |
Indaziflam |
0 |
0 a |
16 ab |
|
|
Isoxaben |
2 a |
3 |
69 b |
Isoxaben |
0 |
0 a |
0 a |
|
|
Napropamide |
6 a |
14 |
0 a |
Napropamide |
0 |
47 c |
98 d |
|
|
Flumioxazin |
2 a |
31 |
63 bc |
Flumioxazin |
0 |
15 a |
34 b |
|
|
Pendimethalin |
2 a |
40 |
64 b |
Pendimethalin |
0 |
0 a |
0 a |
|
|
Simazine |
0 a |
46 |
28 ac |
Simazine |
0 |
44 bc |
5 a |
|
|
Mesotrione |
0 a |
7 |
8 a |
Mesotrione |
0 | 3 a | 3 a | |
Figure 2: Crop coverage pictures from two months after planting the Lewis-Brown research farm show that the planted species (rows) tolerated several pre-emergent herbicides (columns). A black outline surrounds successful combinations seen in at least one of the other two trials. Combinations that were never seen to be successful again are surrounded by a red outline.
References:
Leponiemi, M., Freitak, D., Moreno-Torres, M. et al. (2023). Honeybees' foraging choices for nectar and pollen revealed by DNA metabarcoding. Sci Rep 13, 14753. https://doi.org/10.1038/s41598-023-42102-4
Soil Survey Staff, Natural Resources Conservation Service, United States Department of Agriculture. Web Soil Survey. Available online at the following link: http://websoilsurvey.sc.egov.usda.gov/.
- Author: Drew Lyon
- Posted by: Gale Perez
In my previous position as the Extension Dryland Cropping Systems Specialist with the University of Nebraska Panhandle Research and Extension Center in Scottsbluff, I focused my research efforts on intensifying and diversifying the winter wheat-fallow cropping system. The addition of summer crops into the rotation was a first step. Inserting summer crops such as proso millet, sunflower, or corn reduced the frequency of summer fallow from every other year to once every three years. Summer crops also helped in the management of winter annual grass weeds such as downy brome, jointed goatgrass, and feral rye. As I looked for ways to eliminate summer fallow from the rotation, my attention turned to forage crops.
Forage crops are typically harvested at about 50% heading (grass crops) or flowering (broadleaf crops). This is often six or more weeks earlier than if the crops were harvested for grain. This earlier harvest provides a couple of benefits. First, it decreases soil water depletion. Water use by crops is typically greatest from the boot or flower bud stage through early grain fill. By harvesting the crop shortly after the start of this heavy water use time, valuable soil water is conserved for the following grain crop, which in western Nebraska, was often winter wheat. The earlier harvest also prevented seed production in many weeds growing in the forage crop. Taken together, these two benefits made forage crops a good alternative for summer fallow. However, there were obstacles to the use of forage crops such as a lack of forage harvesting and handling equipment and limited local markets.
When I arrived in Pullman, WA in 2012, the focus of my program shifted to weed management in wheat production systems. One of the first new weed species I learned about was Italian ryegrass. Italian ryegrass biotypes have developed resistance to eight different herbicide modes of action, which makes managing Italian ryegrass with herbicides very difficult. Some growers have turned to forage crops to help them manage Italian ryegrass.
At a Washing State University (WSU) Weed Science Field Day a few years back, we had a grower tell us that one of his approaches for managing Italian ryegrass was to plant forage barley and harvest the forage barley and Italian ryegrass before they headed and sell it in small bales to horse owners. Italian ryegrass is an excellent forage, and the bales fetched a good price. I thought that was an excellent out-of-the-box approach for managing this very troublesome weed. I could see a similar approach working for feral rye or wild oat management.

I have also noticed that some growers with Italian ryegrass issues have planted alfalfa to manage the problem. Here, Roundup Ready alfalfa could help in the establishment year, but after that, the simple act of harvesting the alfalfa before Italian ryegrass sets seed is all that would be needed. A perennial crop like alfalfa also brings other crop rotation benefits for weed control. Once established, alfalfa competes strongly with annual weeds. The seed of most annual grasses do not survive long in the soil, so keeping alfalfa in for just three or four years will significantly decrease the soil seedbank of annual grass weeds and allow a return to annual cropping with much lower grass weed pressure.
Despite the obstacles to the adoption of forage crops in our dryland cropping systems, forage crops do offer several benefits for weed management. As herbicide resistance becomes more problematic throughout the region, non-chemical approaches to weed management will become more important. I would love to hear how you have used forage crops in your rotation to manage weeds. Please leave a comment so that others can learn from your experience.
Thanks!
Dr. Drew Lyon
Endowed Chair Small Grains Extension and Research, Weed Science
Washington State University
(drew.lyon@wsu.edu)
Original source: Weeders of the West blog | April 25, 2024
- Author: Jorge Antonio Angeles
- Posted by: Gale Perez
Silverleaf nightshade, Solanum Elaeagnifolium, is perennial weed that is native to South America, Mexico, American Southwest and Southern States. This herbaceous and woody summer weed belongs to Solanaceae just like other weeds such as black nightshade (Solanum nigrum), hairy nightshade (Solanum physalifolium) and horsenettle (Solanum carolinense). It can be found throughout California and in grows in desert and semi-arid areas. Silverleaf nightshade is often found growing in different cropping systems, rangeland, pastures, roadsides, and disturbed areas. Silverleaf nightshade is highly adaptable and can tolerate a wide range of soil and climatic conditions such as high temperatures, low rainfall, saline and drought conditions. The leaves and berries produced by silverleaf nightshade plants have glycoalkaloid compounds that can be toxic to livestock and humans if consumed (UC IPM) (Boyd 1982).


Life Cycle

Impacts
Silverleaf nightshade is becoming a problematic weed in some young pistachio orchards in Tulare, Kings and West Fresno. This perennial weed is mostly found in young orchards that are planted on west Fresno and Kings County by interstate 5 highway (Figure 3). In Tulare County, silverleaf nightshade is found pistachio orchards that were planted in unmanaged fallow land or in fields near roadsides where it's commonly found. One of the major impacts of silverleaf nightshade is that it competes with young pistachio orchard for resources such as water, light and nutrients. If left unmanaged, silverleaf nightshade can also interfere with irrigation operations and potentially reduce crop vigor in young pistachio trees (Figure 2). Since silverleaf nightshade can adapt to alkaline and saline soils, it will also outcompete many of the summer annual weeds and become the dominant weed in the population.
Management
Silverleaf nightshade can be a weed that is difficult to control with the available management methods in orchards. Tillage is not recommended as that is one of the ways that rhizomes can be spread across the orchard (Ensby 2011). Mechanical control methods such mowing can be an effective control method to prevent weeds from setting seed. Flail mowers are often used in orchards and vineyards to mow weeds in between tree rows. It is important to note that, new silverleaf nightshade shoots will potentially sprout from the root system after the tops are mowed (Stanton 2011). Even though mowing can be an effective weed control method during the growing season, most commercial mowers will miss the weeds that grow in between trees that directly compete with young trees. Hand weeding can be used to remove some of the weeds around the trees, but extra precaution needs to be taken. Mature silverleaf nightshade plants are covered in reddish prickles that can be harmful if weeds are handled with bare hands. Weeding tools such as shovels and hula hoes can damage surface drip hoses if the user is not careful.
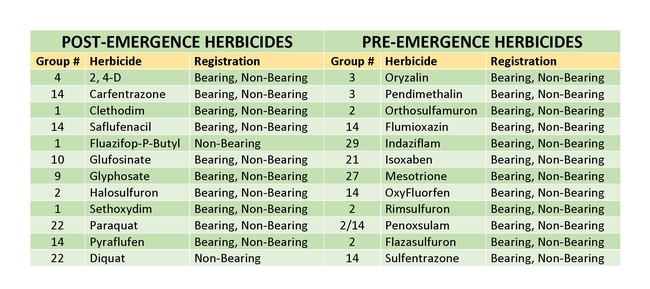
Herbicides can be an effective weed management method to control silverleaf nightshade in different tree crops. There are a twelve pre-emergent and thirteen post-emergent herbicides with different sites of action that are registered for use in pistachios (Table 1). Pre-emergent herbicides are normally applied during the dormant season and most only control weeds before they germinate. Pendimethalin, rimsulfuron, mesotrione, flumioxazin, isoxaben, and flazasulfuron are herbicides that have great control over black and hairy nightshade. Isoxaben can suppress silverleaf nightshade, but cannot be used in pistachios that have not been established for at least three years. Pre-emergent herbicides will not control silverleaf nightshade that emerges from rhizomes in the summer. Post-emergent herbicides can be used to control silverleaf nightshade that emerges in the summer and early fall before harvest. Glyphosate, glufosinate, pyraflufen, and carfentrazone are post-emergent herbicides with different sites of action that can be used to control different weeds and can be used up to two weeks before harvest. 2, 4-D is another post-emergent herbicide that is registered for use in pistachios, but needs to be applied to trees that have been established for at least one year and has a pre-harvest interval of 60 days. Research work from other researchers has shown that glyphosate and 2, 4-D have excellent control of silverleaf nightshade (DiTomaso 2013) (Gitsopoulos 2017). Glyphosate is a systemic herbicide that can potentially kill the root system of silverleaf nightshade, when applied at the correct timing and rate. Since Silverleaf nightshade has extensive rhizome root systems, the root systems need to be killed to fully control this weed. In the summer months, a combination of mowing and the use of post-emergent herbicides can kill the aboveground tissues of silverleaf nightshade weeds and deplete the root bank in the soil (Heap 2018). Post-emergent herbicides need adjuvants such as nonionic surfactants, crop and seed oils, to increase their efficacy. Furthermore, always consult the herbicide labels on information regarding information on the required adjuvants, pre-harvest intervals, application rates and maximum applications per seasons.
The weed management tools to control silverleaf nightshade in pistachio orchards are limited. Developing and maintaining field records before planting is a great way to determine the history of a field before planting. Conducting weed surveys in the winter and spring can help determine what weed species are present in a field. Silverleaf nightshade populations can be reduced by mowing in between tree rows and applying herbicides to weeds present in between trees. These management practices need to be done before the weeds set seeds to contribute to reduce the seedbank. To avoid introducing silverleaf nightshade to other fields, it is important to sanitize tractor equipment and manage the weeds that grow on the field edges or near irrigation canals (. To have an effective silverleaf nightshade management program, a combination of herbicides with different modes of action are needed decrease the possibility of it developing herbicides resistance. Silverleaf nightshade is difficult to control because of its tolerance to many herbicides (Gitsopoulos 2017). During the summer months, it is best to use a combination of systemic and contact post-emergent herbicides to get higher levels of control for silverleaf nightshade. To enhance the efficacy and herbicide absorption, post-emergent herbicides require adjuvants such as methylated seed oils, non-ionic surfactants, crop-oil concentrates, and nitrogen-based fertilizers.
Cited Literature
Boyd, J.W. and Murray, D.S. 1982. Growth and development of silverleaf nightshade (Solanum elaeagnifolium). Weed Science 30, 238-43.
DiTomaso, J.M., G.B. Kyser et al. 2013. Weed Control in Natural Areas in the Western United States. Weed Research and Information Center, University of California. 544pp.
Ensbey R. 2011. Noxious and environmental weed control handbook – A guide to weed control in non-crop, aquatic and bushland situations. 6th ed. New South Wales: Department of Trade and Investment, Regional Infrastructure and Services.
Gitsopoulos, Thomas & Damalas, Christos & Georgoulas, Ioannis. 2017. Chemical options for the control of silverleaf nightshade (Solanum elaeagnifolium). Planta Daninha. 35. 10.1590/s0100-83582017350100064.
Heap, J. & Wu, H. 2018. Silverleaf Nightshade: Australian best practice management manual. Primary Industries and Regions SA & NSW Department of Primary Industries: Adelaide.
Roberts J., Florentine. 2022. Biology, distribution and management of the globally invasive weed Solanum elaeagnifolium Cav. (silverleaf nightshade): A global review of current and future management challenges. Weed Res. 2022;62:393–403. doi: 10.1111/wre.12556.
Stanton, R. & Wu, Hanwen & Lemerle, D. 2011. Root regenerative ability of silverleaf nightshade (Solanum elaeagnifolium Cav.) in the glasshouse. Plant Protection Quarterly. 26. 54-56.
- Posted by: Gale Perez

Job #JPF01902
Location: Riverside, CA
APPLICATION WINDOW
Open date: April 17, 2024
Next review date: Wednesday, Jun 12, 2024 at 11:59pm (Pacific Time)
Apply by this date to ensure full consideration by the committee.
Final date: Sunday, Jun 30, 2024 at 11:59pm (Pacific Time)
Applications will continue to be accepted until this date, but those received after the review date will only be considered if the position has not yet been filled.
Job Description: https://aprecruit.ucr.edu/JPF01902
POSITION DESCRIPTION
The Department of Botany and Plant Sciences at the University of California Riverside invites applications for an Assistant Professor of Extension (Assistant Specialist in Cooperative Extension) in the area of weed management in agronomic crops. This is a fiscal year position available July 1, 2024 with an 80% Cooperative Extension and 20% Organized Research Appointment in the Agricultural Experiment Station (http://cnas.ucr.edu/about/aes/). The position will be housed at the University of California Riverside campus in Riverside, CA. The successful applicant will develop an internationally recognized, extramurally funded basic and applied research program on the management of non-aquatic weeds using chemical and non-chemical strategies that decrease weed competition, minimize environmental impacts from weed control practices, and reduce the economic costs of weed management. Preference will be given to candidates conducting research at the forefront of their field who have a strong working knowledge of weed management and control and a desire and ability to work with diverse clientele. Cooperative extension (CE) responsibilities include the development and implementation of educational programs on weed management in agronomic crops, which may include extension publications, newsletters, web pages, blogs, field days, participation in CE Advisor meetings, organizing workshops, and contributing to the University of California Agriculture and Natural Resources (UC ANR) workgroups and teams.
The current base pay range for the Assistant Professor of Extension position is $84,100 to $110,100 and the Agronomist/ Organized Research position is $86,500 to $112,800. Additional off-scale to be commensurate with market value, qualifications and experiences.
Required qualifications for this position that must be met by the date of application include: a Ph.D. in Weed Science, Agronomy, Plant Sciences or a related discipline, demonstrated experience in applied research, and excellent communication and interpersonal skills.
Preferred qualifications for this position include: Postdoctoral experience, experience in research and outreach with demonstrated productivity in publishing in journals related to weed management, and a track record of success in obtaining extramural funding.
Advancement through the Professorial ranks at the University of California is through a series of structured, merit-based evaluations, occurring every 2-3 years, each of which includes substantial peer input.
To apply: submit the following to https://aprecruit.ucr.edu/apply/JPF01902.
- Cover Letter – short cover letter describing background and interests
- Curriculum Vitae
- Statement of Teaching and Mentoring - This statement should be a concise description of the applicant's teaching and mentoring philosophy and include past experience and future teaching and mentoring plans (2-page maximum)
- Statement of Research interests and goals (3-page maximum)
- Statement of Extension interests and philosophy (2-page maximum)
- Statement of Past and/or Planned Future Contributions to Advancing Diversity and Inclusive Excellence (2-page maximum)
- 3 Letters of Reference – At time of submission, applicants should provide contact information and request letters of recommendation from 3 referees to be submitted through AP Recruit, Letters will need to be submitted to AP recruit from the letter writers by June 12, 2024.
- Up to 3 select reprints of publications or manuscripts in press (optional)
Review of applications will commence on June 12, 2024, and proceed until position is filled. For full consideration, applicants should submit their complete applications prior to the above date.
For the FULL JOB DESCRIPTION: https://aprecruit.ucr.edu/JPF01902
For more information about this position, please contact Dr. Milt McGiffen, Chair of the Search Committee, Department of Botany and Plant Sciences, at milt@ucr.edu. For questions on application procedures and requirements, please contact Mrs. Naudia Samuels Johnson, Academic Personnel, at naudia.samuelsjohnson@ucr.edu.