- Author: Trina Kleist, UC Davis Department of Plant Sciences
Weeds abound: On farmland, pastures, rangelands, in natural areas, in our yards and along our sidewalks. Managing those pesky plants costs plenty: California farmers alone spend more than $900 million annually on weed control, according to a University of California study. Rangeland managers reported spending as much as $5 million to manage weeds on their land, according to a survey.
“Weed management is a critical component of managing California lands,” said Brad Hanson, a professor of Cooperative Extension in the UC Davis Department of Plant Sciences. That's why research on the impacts of weed control tactics on the targets, on crops and on other plants is one of the missions of weed researchers.
Some of the latest findings and updates on ongoing research were presented during the UC Davis Weed Day 2024, hosted by the department and the UC Weed Research and Information Center. Topics included the potential damage caused by herbicide drift, the benefits of using native plants to combat invasive weeds, impacts of using endothall in irrigation water, new weed detection and treatment technologies, and other issues related to weed management.
Herbicide drift explored
When herbicides are applied on California rice fields, breezes can blow the chemicals into adjacent fields. Growers and others are concerned about the damage herbicide drift could cause to other crops nearby. In a field west of campus, postdoctoral scientist Deniz Inci showed rows of corn, beans, squash, tomatoes and sunflowers that had been exposed to benzobicyclon, sold under the name Cliffhanger. “Due to California's diverse cropping systems, off-target herbicide drift into nearby fields of annual crops can be a significant problem,” Inci said.
In mid-June, Inci simulated this problem in the field by exposing annual crops to different concentrations of benzobicyclon, including anticipated drift rates plus higher concentrations. Injury is still being evaluated weekly, and the results will be shared with California rice growers during the Winter Rice Grower Meetings in January, Inci said.
As part of the extension program, rice herbicide drift studies are presented annually at different locations across the Sacramento Valley. For updates and more information about drift, visit the UC Rice Research and Information Center and the Weed Research and Information Center.
Inci recently graduated with a Ph.D. from the lab of Kassim Al-Khatib, in the Department of Plant Sciences.
Native plants and soil interactions offer clues
The interaction of native plants and the soil they occupy could provide clues to both fighting weeds and restoring drought-resistant plants to landscapes. Justin Valliere, an assistant professor of Cooperative Extension in the department, explained work in native grasslands that shows promise.
The key may be using late-season summer annuals: In a pilot study, they appeared to help restore bunchgrasses and prevent re-invasion of non-native annual grasses, Valliere said. Feedbacks between plants and tiny things living in the soil may play a key role in facilitating restoration. When non-natives take over, they may drastically change soil microbial communities. That, in turn, can pose challenges for re-introducing natives.
“We are currently exploring how seeding different mixes of native annual plant species may help recover soil microbial communities and improve restoration success,” Valliere wrote.
Restoration of native California bunchgrasses is generating interest because they have deep and wide fibrous root systems that allow them to tolerate drought, reduce water run-off and resist erosion. They provide good forage for livestock, plus provide food and habitat for other creatures, store significant amounts of carbon and improve air quality.
Endothall in irrigation water
Aquatic weeds that grow in thousands of miles of irrigation canals in California are commonly managed using herbicides. In one part of the state, growers reported crop damage in almonds, annual crops and alfalfa after using herbicide-treated irrigation water, and they worry the chemical could be the culprit. Stephen Chang, a master's student in the lab of Brad Hanson, is working on a multi-year experiment to dig into these claims.
“We are testing applications of two formulations of the aquatic herbicide endothall in a young almond orchard and an older almond orchard to see how trees of differing ages will react,” Chang wrote. More work is coming: “Additionally, we are looking to investigate the effect of endothall in several annual crops commonly grown in California.”
Chang took first place in a poster contest where he outlined his research. The contest was held during a meeting of the California Weed Science Society in Santa Barbara in January.
Weed-zapper works in organic almonds
As part of his Ph.D. studies, Tong Zhen is looking at how well an electrical device can zap weeds growing in almond orchards. The device, which generates electricity using a power take-off generator, shoots electrical current through plants as the tractor slowly drives along. As the current passes through the plant stems and roots, resistance generates heat, which damages plant cells.
Zhen and the research team found the electrical weed control system didn't bother the growth of young almond trees, measured by height and trunk diameter, in an organic orchard at a UC Davis research site. The almond trees and a nearby blueberry planting where weeds were treated with the electrical weeder grew similarly to plots where weeds were managed with hand-weeding, hoeing and spot-spraying with an organically-approved herbicide. Plus, soil respiration also was about the same, suggesting little impact on soil microbial activity.
At Weed Day, Zhen demonstrated the device on an open field infested with lovely, but pernicious, field bindweed. Most of the plants were toast soon after treatment and the smell of crispy greens hung in the air.
- Author: Lynn M. Sosnoskie
Field bindweed (Convolvulus arvensis) is a perennial plant in the Convolvulaceae family (which is also the family of dodder (Cuscuta spp.), morningglories (Ipomoea spp.), and alkaliweed (Cressa truxillensis)). The species possesses an extensive root network (vertical roots are reported to reach depths of 10 to 20 feet or more), although the majority of the underground biomass is in the top 1 to 2 feet of soil. Individual field bindweed plants produce multiple creeping vines that can grow up to 6 feet in length.
Figure 1. Field bindweed vines.
Native to the Mediterranean region and Western Asia, field bindweed is presumed to have been brought to the United States (in 1739) as a seed contaminant. The species moved westward and was officially documented in the state of California (San Diego County) in 1850. By the first quarter of the twentieth century, EW Hilgard (The Weeds of California, 1891) and FT Bioletti (The Extermination of Morning-glory, 1911) had proclaimed the species to be the most troublesome weed in the state.
Figure 2. Field bindweed (lower right) is in the same family as dodder (top) and alkaliweed (lower left). Photo by Lynn M. Sosnoskie.
Bindweed reproduces both by seed and vegetatively. Plants produce white to pinkish/purple trumpet-shaped flowers (1 inch in diameter to 1 inch in length) beginning in April and continuing through September in California (depending on latitude). Flowers open during the day and close tightly at night into a twisted tube. Seeds are approximately 1/8th of an inch long, black in coloration, shaped like an orange wedge, and produced in a papery capsule. Although estimates vary dramatically, it has been reported that bindweed infestations can produce between 20,000 and 20,000,000 seeds per acre.
Figure 3. Field bindweed flowers can be white to pinkish/purple in coloration. Photo by Lynn M. Sosnoskie.
Figure 4. Field bindweed seeds are dark brown to black in color, shaped not unlike orange wedges, and are held in papery capsules. Photo by Lynn M. Sosnoskie.
The germinability of freshly produced bindweed seed is highest 20-30 days after pollination; changes in seed moisture content and the permeability of the seed coat result in a dormancy that requires scarification to overcome. This hard-seededness is one reason bindweed is so enduring in fields. Although viability does diminish with time, field bindweed seed has been shown to persist in the soil for 20 to 30 years. Bindweed seed has been shown to germinate under a wide range of temperature conditions (between 41 F and 104 F with an optimum temperature around 86 F. Field bindweed germination and emergence is also impacted by burial depth: results from multiple studies have suggested that most new plants are developing from depths of 2 inches or less. Field bindweed plants begin to develop their extensive system within 4 to 6 weeks of emergence. This includes the development of latent buds that generate rhizomes from which new crowns arise.
New bindweed plants can also develop following the fragmentation of the root system of a parent vine. Roots are brittle and infrequent mechanical disturbance may serve to disseminate rhizome pieces around a field. According to published reports, the most regenerative root and rhizome tissues appear to arise from pieces derived from the top 12 inches of soil. Fragment size can also influence regenerative success; root portions longer than 1" in length will enhance establishment potential.
Figure 5. A newly germinated bindweed seedling (note the presence of cotyledons (seed leaves)). Photo by Lynn M. Sosnoskie.
Figure 6. Exhumed root system of a young field bindweed plant displaying lengthening and differentiating root buds and the development of a new crown. Photo by Lynn M. Sosnoskie.
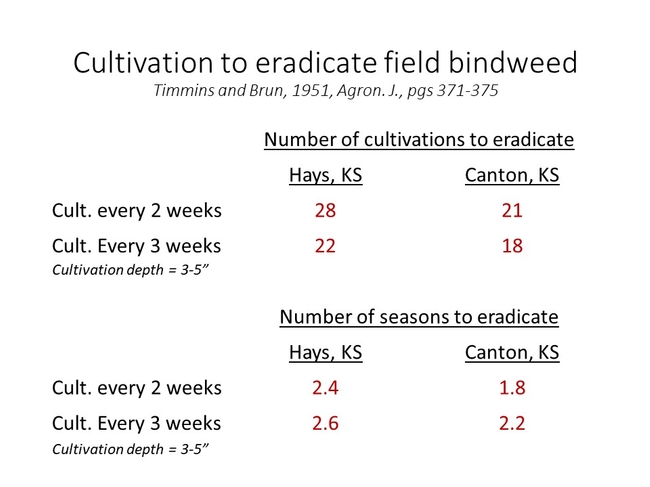
The biology of field bindweed can directly impact how easily it is controlled by physical and chemical control measures. The extensive root system and regenerative potential of field bindweed necessitate frequent/continuous cultivation to exhaust nutrient reserves. Results from Kansas trials conducted in the mid 20th century suggest that soil disturbance every 2 to 3 weeks for 2 years is required to eradicate the perennial vines.
Figure 7. Results from studies conducted in the mid 20th century still form the basis for today's recommendations regarding field bindweed control with cultivation. Disturbance events need to occur every 2-3 weeks for up to 2+ years to exhaust the nutrient reserves stored in underground root and rhizome tissues.
The efficacy of foliar applications of glyphosate are also impacted by the species' roots...or, more importantly, the movement of photosynthates from above ground tissue to storage organs. The results of a study published in 1986 indicated that bindweed was more susceptible to glyphosate during the late spring/early summer months (when plants were flowering) as compared to early spring and late summer/early fall. There are multiple reasons for the differences observed in sensitivity, one of which is that the phloem mobile glyphosate was more readily translocated to meristematic tissues (including those in the root) where the herbicide inhibits the synthesis of aromatic amino acids.
Figure 8. Field bindweed is more readily controlled with glyphosate when it is vigorously growing and flowering. Glyphosate is phloem mobile and moves with photosynthates to the plants growing points (including underground meristems) where it inhibits the synthesis of aromatic amino acids.
Field bindweed is an increasing concern of growers in the Central Valley of California, especially those that are producing crops in reduced tillage/drip-irrigated systems. With a limited number of effective herbicides available (i.e. trifluralin in processing tomatoes, glyphosate in glyphosate-resistant agronomic commodities), the problem is likely not going to abate in the short term. Knowing what we do about the biology of bindweed (and how biology affects our ability to control this species), where do we go from here? The following considerations are just my opinions
• We need to discover/evaluate new strategies to increase the disruption/disturbance experienced by rhizomes. We need to do this both within and across crops (i.e. crop rotation).
• Can we manipulated bindweed biology to make the dormant root buds more sensitive to control measures?
• How does bindweed adopted to California and the southwestern United States differ from bindweed in the rest of the country? Should we assume that data generated in midwestern environments is appropriate for a California climate.
• Speaking of climate, how will variable weather impact bindweed growth, rhizome and bud dormancy, and subsequent control going forward?
This information was derived from a presentation presented at an extension event hosted by UCCE Fresno at the Westside Research and Extension Center in Five Points, CA, on August 15, 2018. A link to the pdf of the original slides is provided below.
2018 Westside Bindweed Talk
- Author: Lynn M. Sosnoskie
You've got trouble with a capital "T" that rhymes with "B", which stands for bindweed.
My apologies to Meredith Wilson (I just couldn't pass up the opportunity...). But seriously, if you've got bindweed, you've probably got trouble. Field bindweed (Convolvulus arvensis) is a deep-rooted (and drought-tolerant) perennial vine that has become a significant concern in reduced-tillage, drip-irrigated, processing tomato systems.
Figure 1. Have you got trouble? Field bindweed at the UC Davis research farm.
Field bindweed, a native of Europe and Asia, is a member of the Convolvulaceae, or morning-glory, family. The species is characterized by creeping or climbing (when it has support) vines with spirally-arranged, arrow-like leaves and white to pink trumpet-shaped flowers. The root system of field bindweed can be quite extensive, reaching depths of up to 20 feet. Roots are brittle and infrequent mechanical disturbance may only serve to disseminate rhizome pieces around a field. Bindweed seeds are particularly long-lived; some reports indicate that they (the seeds) may even remain viable for 60 years. Although seedlings are easy to control with chemical and physical control measures, mature plants are significantly more resilient.
Figure 2. Rhizome pieces as short as two inches in length can become perennialized.
For the past two years, I've looked at bindweed responses to herbicides in order to maximize weed suppression in summer annual cropping systems. Specifically, I've evaluated pre-plant PP burndown, residual pre-plant incorporated (PPI) and pre-emergence (PRE), and post-emergence (POST) and shielded (SHIELD) herbicides for bindweed control in processing tomatoes.
Pre-plant burndown:
One of the main tenets of IPM is to start off clean! This means planting into fields with reduced weed densities and/or no emerged weed vegetation that can compete with a newly emerged/establishing crop. Results from 2013/2014 field trials show that an application of Roundup Powermax (1.75 qt/A) to actively growing vines (ten days prior to transplanting) can reduce field bindweed cover by more than 50% for up to eight weeks after transplanting (by which time the crop canopy should be starting to close in).
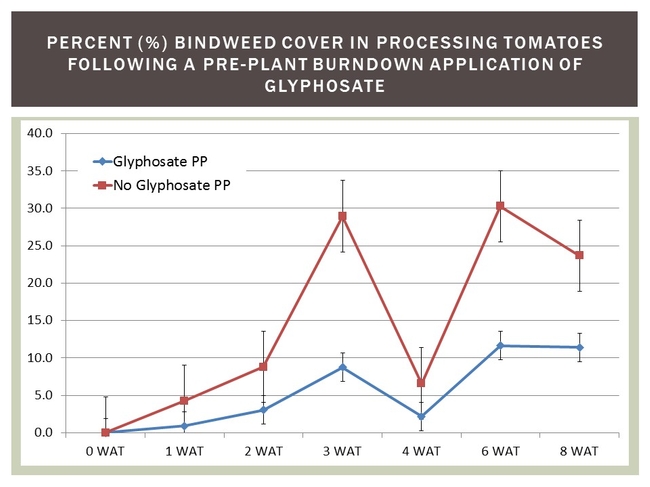
It is important to realized that field bindweed response to the active ingredient in Roundup can vary significantly in response to plant growth stage at the time of application, environmental factors and their influence on cuticle development, spray volume and natural variability among bindweed biotypes to glyphosate. Even under the best of application circumstances, a single application of Roundup (or any other herbicide, really) is unlikely to eradicate field bindweed.
Figure 4. Bindweed regrowth following an application of Roundup Powermax.
Pre-plant incorporated and pre-emergence herbicides:
The use of residual herbicides may help growers 'buy some time' against bindweed in severely infested fields. By this, I mean that growers can suppress vine emergence and growth while the tomato transplants become established. Research conducted by Tom Lanini (Professor Emeritus, UCD) and others has demonstrated that Treflan (trifluralin) is the most (consistently) effective product for managing bindweed in tomatoes. In my experience, Matrix (rimsulfuron) and Zeus (sulfentrazone) may also be effective, although rotation restrictions limit their use. Tank mixes with Dual Magnum may be advantageous if nutsedges are also a problem. One word of caution: crop injury can occur if transplant root balls are not positioned below the herbicide-treated zone! I've tended to see more injury with Zeus than with the other products, but. Regardless, make sure that you are applying the right product at the right rate with respect to your local environmental/soil conditions to maximize current and future crop safety!
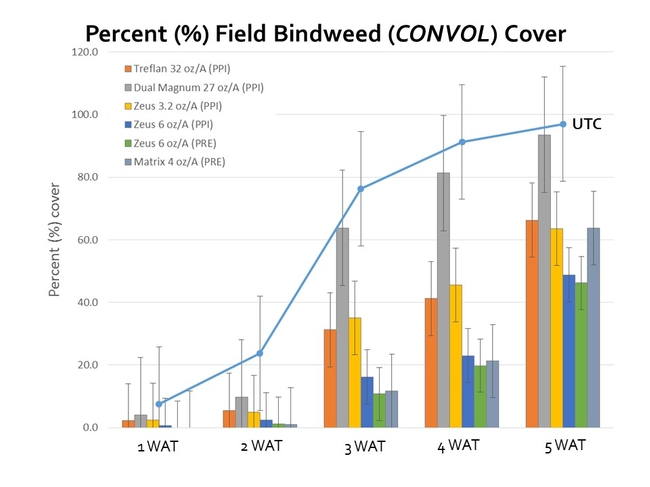
Post-emergence and shielded herbicides:
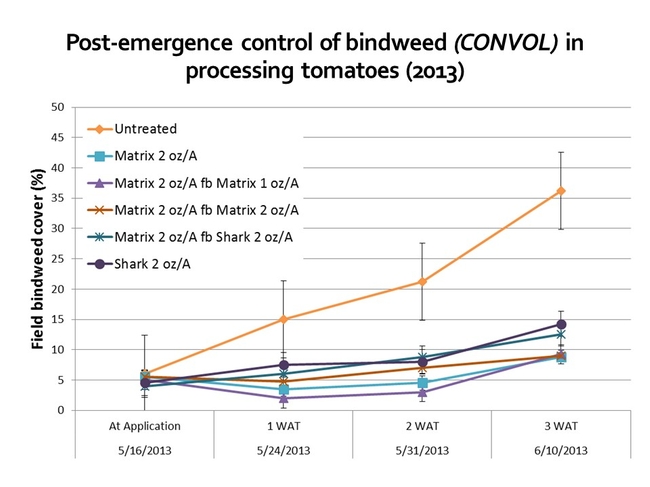
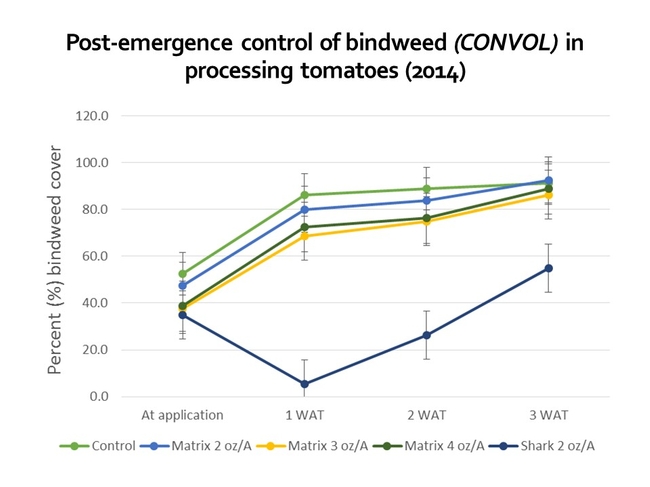
Recent research conducted in our lab has found that the application of Matrix POST or Shark (carfentrazone) SHIELD can reduce bindweed cover, relative to an untreated control, although extensive, vigorous vines are more likely to recover from herbicide applications. Matrix can 'yellow-up' tomatoes, although the injury is fairly transient. Shark must not come into contact with leaves, branches, and stems or serious crop injury can occur!
Figures 6 and 7. Results from research trials to look at Matrix and Shark efficacy for the control of field bindweed. Results suggest that POST and SHIELD applications may be more effective at managing the species when the starting weed cover is low (i.e. 5% in 2013 as compared to 40% in 2014). NOTE: The rates applied in this study were chosen for experimental reasons (several are NOT labeled for use on tomatoes); review all labels and select herbicide doses appropriate for your growing conditions.
Cultivation, crop rotation, and post-harvest weed control:
Chemicals aren't the only strategy for dealing with bindweed in tomatoes; frequent in-crop cultivation can also be suppressive. Rotating to corn or wheat, which would diversify the type and timing of weed control practices may also be beneficial. Another tool for managing field bindweed is engaging in weed control following crop harvest. I think that this could be very important, especially after wheat when the vines can grow unimpeded, although I have not, personally, evaluated summer vs. fall vs. spring herbicide applications to bindweed in crop stubble (that is on the 2015/2016 research agenda).
Figure 8. Cultivation following a PPI Treflan application.
More information about bindweed and bindweed management can be found at the UC Pest Notes page and the UC Crop Management page. Your farm adviser should also be able to provide you with more information about weed control, including field bindweed suppression, in tomatoes that is appropriate for your region. Herbicides and rates included in this study were chosen for experimental purposes; some are not legally registered for use in tomatoes in CA. Be sure to review all labels to ensure that you are applying appropriate products at appropriate rates!