- Author: Pershang Hosseini
- Author: Alison Colwell
- Author: Bradley Hanson
- Posted by: Gale Perez
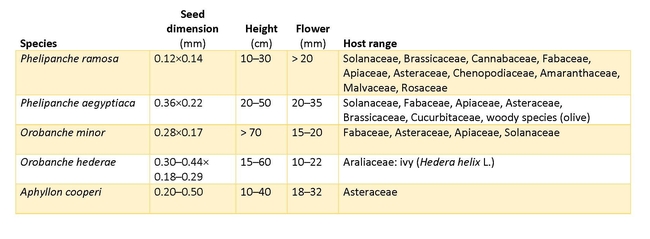
The Orobanchaceae family comprises 270 holoparasitic species that cannot photosynthesize. Instead, they rely entirely on the host plant roots for nutrition, and can produce great numbers of minute, dust-like seeds that last for years in the soil. This family contains some of the most serious agricultural parasites in the genera of Phelipanche and Orobanche some of which are present in California. The different species can be distinguished by the degree of branching of their stems and presence/absence of bracteoles at the base of the flower Orobanche species have unbranched stems and no bracteoles, while Phelipanche species feature branched stems with bracteoles. In this paper, we discuss three economically significant broomrape species, and two that are not crop pests.
Phelipanche ramosa (Branched broomrape)
The natural distribution of branched broomrape (Table 1, Figure 2b) encompasses Europe, the Middle East, West Asia, and North Africa. It is established in southern Africa, Chile, and north America, having been reported in IL, TX, WA, OR, DE, WV, VA since early in the last century. Notable new infestations have been documented in Australia, resulting in several million dollars in annual expenses for quarantine and control efforts across 200,000 hectares. Further economic losses accrue from restrictions on the sale of produce or crop seed from the infested areas; these measures are aimed at preventing the spread of the parasite to other agricultural fields (Warren, 2006). Although reported occasionally in California in past decades, a new infestation of branched broomrape has been documented in California in recent years (Osipitan et al., 2021).
The host range of branched broomrape is exceptionally broad, encompassing various crops in the Solanaceae (tomato, eggplant, tobacco, pepper, and potato), Brassicaceae (rapeseed), Cannabaceae (hemp), Fabaceae (chickpea, clovers, groundnut, faba bean, lentil, pea), Apiaceae (carrot, celery, fennel, parsnip) and Asteraceae (lettuce, sunflower, and many ornamental species). Furthermore, branched broomrape can parasitize wild hosts within a range of families, including Chenopodiaceae, Amaranthaceae, Malvaceae, and Rosaceae. Although there have been reports of it affecting onions, it typically does not infest monocots.
There is evidence of lineages of this species having undergone both host specialization and host extension. For instance, one population initially parasitizing tomatoes was found to also parasitize tobacco and lettuce. In southwestern Germany, some farmers switched from cultivating tobacco to parsley, leading to an infestation of branched broomrape in the parsley crop (Kohlschmid et al., 2011). However, a population originating from tobacco failed to parasitize lettuce (Musselman & Parker, 1982), suggesting the host shift requires some selective adjustments in order to succeed.
Economic Importance: Yield losses in tomato and tobacco are commonly reported to be 30–50% (Parker, 2013). Branched broomrape was found to reduce tomato shoot dry weight by 60-70%, while also causing a concurrent 50% decrease in chlorophyll content and photosynthesis levels (Mauromicale et al., 2008) in pot experiments.
Phelipanche aegyptiaca (Egyptian broomrape)
Branched and Egyptian broomrape (Table 1, Figure 2a) are closely related and easily confused for one another. Egyptian broomrape is a more robust, taller plant than a typical branched broomrape plant but the most easily noted difference is the larger size of the flowers at 20–35 mm long (branched broomrape typically no more than 20 mm). The distribution of Egyptian broomrape overlaps with branched broomrape in South Europe, the Mediterranean, and North Africa but extends much further eastwards into South Asia and China.
Egyptian broomrape can attack most of the same crops affected by branched broomrape. It differs in occurring on a wider range of Brassicaceae, especially various mustard species in India. It is also a more important pest on Cucurbitaceae compared to branched broomrape.
Economic Importance: The effects of Egyptian broomrape on the host are the same as branched broomrape, and damage can be similarly severe.
Orobanche minor (small broomrape)
Small broomrape (Table 1, Figure 2c) is typically a one (or few) stalked, unbranched plant up to 70 cm tall and occasionally 1 m. It is very widely distributed, being native throughout most of Europe, Western Asia and Northern Africa, as far south as Ethiopia and Somalia, while it has also been sporadically introduced to Japan, New Zealand, Australia and several countries in North and South America. This species is a federally listed noxious weed in the United States that has infested red clover fields in Washington and Oregon (Mallory-Smith & Colquhoun, 2012; Osterbauer & Rehms, 2002 ).
The host range for small broomrape is very wide, particularly in Fabaceae (clover, alfalfa, vetch). Small broomrape can also parasitize Asteraceae (lettuce, safflower) and Apiaceae (carrot, celery), Solanaceae, and other families.
Economic Importance: The greatest economic damage is to clover, reported in several countries in Europe. This problem has led to the abandonment of the clover seed industry in some of those countries including the UK.
Orobanche hederae (ivy broomrape)
While the majority of Orobanche species exhibit a broad host range for parasitism, some, such as ivy broomrape (Table 1, Figure 2d), exhibit narrow preferences. In this case, the parisite exclusively attacks species within the Hedera genus, commonly known as ivy. Ivy broomrape is a short-lived monocarpic perennial, reaching heights of up to 60 cm. Its distribution area spans across Western and Southern Europe, North Africa, and Asia Minor. It is a close relative of Orobanche minor, and arguably derived from it.
Economic Importance: None known.
Aphyllon Cooperi or Orobanche Cooperi (desert broomrape)
Desert broomrape (Table 1, Figure 2e) is a biennial broomrape that is native to the desert southwest of the US and Mexico. It parasitizes shrubs of Ambrosia and Encelia (Asteraceae). It was segregated from Orobanche based on chromosome number and 5 calyx lobes instead of 4.
Economic Importance: None known. It has been reported occasionally in tomato fields, but does not appear to persist under cultivation of the crop. It has been grown in potted tomatoes by the second author, but it is slow to reach the flowering stage and the phenology unfolds slowly, so the plant is unlikely to set seed during a tomato crop produciton cycle.
References
Kohlschmid E, Mu¨ller-Sto¨ver D, Sauerborn J. (2011). Ausbreitung des parasitischen Unkrauts Phelipanche ramosa in der deutschen Landwirtschaft. Gesunde Pflanzen, 63:69–74.
Mallory-Smith, C., & Colquhoun, J. (2012). Small broomrape (Orobanche minor) in Oregon and the 3 Rs: regulation, research, and reality. Weed science, 60(2), 277-282.
Mauromicale G, Lo Monaco A, Longo AMG. (2008). Effect of branched broomrape (Orobanche ramosa) infection on the growth and photosynthesis of tomato. Weed Science, 56: 574–581
Musselman LJ, Parker C. (1982). Preliminary host ranges of some strains of economically important broomrapes (Orobanche). Economic Botany, 36:270–273.
Osipitan O, Hanson B, Goldwasser Y, Fatino M, Mesgaran M. (2021). The potential threat of branched broomrape for California processing tomato: A review. Calif Agriculture, 75(2):64-73. https://doi.org/10.3733/ca.2021a0012.
Osterbauer, N. K., & Rehms, L. (2002). Detecting single seeds of small broomrape (Orobanche minor) with a polymerase chain reaction. Plant Health Progress, 3(1), 1.
Parker, C. (2013). The parasitic weeds of the Orobanchaceae. In Parasitic Orobanchaceae: parasitic mechanisms and control strategies (pp. 313-344). Berlin, Heidelberg: Springer Berlin Heidelberg.
Warren, P. (2006). The branched broomrape eradication program in Australia. In: Preston C, Watts JH, Crossman ND (eds) 15th Australian Weeds Conference, Adelaide, September 2006, pp 610–613.



