Workshop: Developing & Marketing Successful Farm Stand and U-Pick Operations
Tuesday May 3, 2022
9am-12pm
Soul Food Farm, 6046 Peasants Valley Road, Vacaville
For more details click HERE
- Author: Margaret Gullette Lloyd
SUMMARY
- Two broccoli plantings immediately prior to growing the verticillium-susceptible crop is recommended for best protection
- Fresh broccoli residue has greater reduction in V. wilt than dry residue
- Field tarping following fresh residue incorporation did not increase (or decrease) efficacy
- Suppression of V. dahliae is specific to broccoli and not provided by other Brassicaceae crops.
- V. dahliae isolates from 15 host crops, including tomato, eggplant, bell pepper, lettuce, potato, watermelon and strawberry, were effectively suppressed by 5 broccoli cultivars
- The most significant reduction in V. dahliae occurs 15 days post-incorporation, and continues to decline over the season.
- More mature broccoli plants have higher levels of volatile antifungal substances
- The mechanisms of action are hypothesized to include: volatile antifungal compounds, changes in the soil microbial communities and serving as a ‘dead-end host'.
- Broccoli has been shown to reduce pathogens causing Verticillium wilt and lettuce drop, but not other soilborne pathogens such as Fusarium spp..
BACKGROUND
In 1999, several UC researchers published foundational research in a paper titled, “Evaluation of broccoli residue incorporation into field soil for Verticillium wilt control in cauliflower.” Since this publication more than 20 years ago, many studies have further investigated this concept and many coastal growers, especially organic producers, have adopted broccoli rotations as a strategy for Verticillium wilt control. Today, typical implementation of this strategy is two broccoli plantings back to back prior to the crop for which Verticillium wilt suppression is desired. While California coastal vegetable production has been the framework for much of this work, the adaptability of this practice to the Sacramento Valley is very promising for management of Verticillium wilt in warm and cool season crops[1].
Verticillium wilt is caused by the soilborne fungal pathogen Verticillium dahliae. Microsclerotia, the fungal inoculum that causes infection, dwell in the soil until root exudates stimulate germination and direct the fungal hyphae towards the root. In susceptible plants, infection occurs when hyphae enter the roots right behind the root tip, and continue growth into the water-conducting vascular tissue, the xylem. Once in the xylem, hyphal growth and sporulation can move the fungus into the upper plant tissue. Plant death triggers the fungus to a reproductive stage, prompting microsclerotia formation. When infected crop residue is incorporated into the soil, microsclerotia in the crop residue are incorporated, too. Management is particularly challenging because the pathogen host range is over 300 crops and the inoculum survive upwards of 13 years (1). To establish control of the pathogen, the key is to reduce inoculum—the number of microsclerotia, below levels damaging to susceptible crops.

Image:Thousands of microsclerotia, small, black propagules of V. dahliae, formed on susceptible crop residue and remained intact post residue incorporation. Photo credit: M. Lloyd
BROCCOLI SUPPRESSES VERTICILLIUM WILT AND DECREASES PATHOGEN PROPAGULES
Broccoli is one of the few non-host vegetables and member of the Brassicaceae family. Bok choy, broccoli raab, Brussels sprouts, cabbage, cauliflower, Chinese cabbage, and rapini are susceptible to V. dahliae, as are black mustard, Indian mustard, oilseed rape, and turnip. In broccoli, no infection to minor infection from V. dahliae has been observed. In the case of minor infections, the pathogen does not progress beyond the roots and microsclerotia formation in the roots is repressed. Apart from the importance of selecting a non-host as a rotation crop, the glucosinolate profile of broccoli, the secondary compounds responsible for the toxic effect, differs from other brassicaceous crops
Following broccoli residue incorporation, research out of Japan demonstrated Verticillium wilt incidence of eggplant decreased by 53 % compared to eggplant without broccoli rotation (2, 3). In California Cauliflower production, disease incidence and severity were both reduced approximately 50% following broccoli residue treatments.
Broccoli did not just decrease disease incidence, but decreased the amount of pathogen inoculum (2), showing promise for longer term management. In a California study, overall reduction in the number of propagules in V. dahliae-infested plots after two broccoli crops was approximately 94%, in contrast to the five-fold increase in the number of propagules after two cauliflower crops (4). These findings corroborate earlier studies showing reductions in the numbers of soilborne microsclerotia of V. dahliae and incidence of wilt on cauliflower that were comparable to reductions caused by chloropicrin and metham sodium treatments (5). Importantly, following broccoli rotations, microsclerotia continue to decline through-out the following cropping season and remain low during the following season. In contrast, propagules in soil fumigated with chloropicrin and metham sodium declined initially but later returned to pre-treatment levels by the end of the cropping season.
MECHANISM OF SUPPRESSION
Shetty et al. (2000) reported that the effects of broccoli in reducing microsclerotia and suppressing disease may be associated with the following mechanisms: (1) production of volatile antifungal substances such as allyl-isothiocyanate (ITC) by broccoli residue, (2) increase in antagonistic microorganisms, and (3) degradation of microsclerotia melanin by ligninase/melaninase produced by soil microorganisms in the presence of broccoli lignin. ITCs are chemically similar to methylisothiocyanate, the active agent from the chemical fumigant metam sodium. Likely associated with the ability to generate these conditions, fresh broccoli residue was shown to be more suppressive than dry residue. During tissue decomposition, the glucosinolates in crucifer crops, the characteristic sulfur-containing constituents of the members of Brassicaceae responsible for their inherent pungent odor, break down to produce sulfides, isothiocyanates, thiocyanates, and nitriles that have either fungistatic or fungicidal properties (6). In addition to release of toxic compounds and microbial activity provided by broccoli residue, the plant may be serving as a ‘decoy', ‘trap crop' or ‘dead end host', further driving population numbers down (2). As described earlier, some V. dahliae infection is observed in broccoli roots, but it does not result in microsclerotia formation. By stimulating inoculum germination and preventing fungal reproduction, the number of viable microsclerotia decrease in the soil.
GROWER IMPLEMENTATION OF RESEARCH FINDINGS
To facilitate greater adaptation of rotations with broccoli in other crops susceptible to V. dahliae, Bhat and Subbarao asked the question whether isolates of V. dahliae originating from different susceptible hosts could cause wilt on broccoli. They evaluated 15 different host isolates against multiple broccoli varieties. This included tomato, eggplant, bell pepper, lettuce, potato, watermelon and strawberry, and found that only isolates from cabbage and cauliflower were weakly pathogenic. Broccoli cultivars Baccus, Greenbelt, Parasol, Patriot, and Symphony showed resistance to Verticillium infection (3). This provides some evidence for the usefulness of this method in other cropping systems.
Implementation of broccoli rotations for Verticillium wilt management is optimized when two successive broccoli crops are grown immediately prior to desired Verticillium wilt reduction. Higher amounts of glucosinolates, specifically glucobrassicin, are found in older plants. Research has reported a complete absence of glucobrassicin in broccoli seedlings, 50% of the total in immature heads (5-10 cm diameter) and the highest levels at fully developed Packman broccoli heads (15-20 cm diameter) (7). These results suggest that glucobrassicin synthesis is active during later stages of broccoli development. Plants should be mowed and finely chopped in order to disrupt the plant cells as much as possible. The greatest reductions in microsclerotia occur at soil temperatures above 68°F, and most of this reduction occurs within 15-30 days of incorporation (8). Variation in efficacy of this method is attributed to multiple factors: fluctuation in climate and cultivation conditions, physical and chemical properties of the soil, soil microbial properties, the type of broccoli cultivar used, differences in pathogen density, and variance in the susceptibility of the following crop host. The types and amounts of glucosinolates vary with the crucifer species and determine the level of plant pathogen growth reduction.
This practice could also have other potential benefits and drawbacks. Growers in California have observed for many years that where broccoli residues from processing plants are dumped onto a field, weed populations are reduced the following year (5). Thus, rotations with broccoli may have multiple pest management benefits. However, in recent years in the Sacramento Valley, crop damage from bagrada bug has been significant. Although these outbreaks have largely occurred in fall, outbreaks have occurred in the spring in this region. Members of the Brassicaceae family are the host plants for bagrada and under favorable environmental conditions would support this pest population.
This management strategy is specific to Verticillium dahliae and is not transferrable to other soilborne pathogens such as Fusarium spp.. Because these two pathogens are common in the Sacramento Valley and above ground symptoms are similar, diagnosis is important. Contact me at any time for disease diagnostic support. All visits and sample analyses are provided free of charge.
SUGGESTED READING
Koike S, Subbarao K. 2000. Broccoli residues can control Verticillium wilt of cauliflower. Calif Agr 54(3):30-33. https://doi.org/10.3733/ca.v054n03p30.
http://calag.ucanr.edu/archive/?type=pdf&article=ca.v054n03p30
REFERENCES
1 Schnathorst, W. C. 1981. Life cycle and epidemiology of Verticillium. Pages 81-111 in: Fungal Wilt Diseases of Plants. M. E. Mace, A. A. Bell, and C. H. Beckman, eds. Aca-demic Press, New York.
2 Ikeda, K., Banno, S., Furusawa, A. et al. Crop rotation with broccoli suppresses Verticillium wilt of eggplant. J Gen Plant Pathol 81, 77–82 (2015).
3 Bhat, R. G., and Subbarao, K. V. 2001. Reaction of broccoli to isolates of Verticillium dahliae from various hosts. Plant Dis. 85:141-146.
4 Xiao, C. L., Subbarao, K. V., Schulbach, K.F., and Koike, S. T. 1998. Effects of crop rotation and irrigation on Verticillium dahliae microsclerotia in soil and wilt in cauliflower. Phytopathology 88:1046-1055.
5 Subbarao, K. V., Hubbard, J. C., and Koike,S. T. 1999. Evaluation of broccoli residue incorporation into field soil for Verticillium wilt control in cauliflower. Plant Dis.83:124-129.
6 Gamliel, A., and Stapleton, J. J. 1993. Characterization of antifungal volatile compounds evolved from solarized soil amended with cabbage residues. Phytopathology 83:899-905.
7 Kushad, M.M., B.P. Klein, M.A. Wallig, E.H. Jeffery, A.F. Brown, and A.C. Kurilich. 1999. Variation of glucosinolates in vegetable crops of Brassicaoleracea. J. Agr. Food Chem. 47:1541–1548.
8 Subbarao, K. V., and Hubbard, J. C. 1996.Interactive effects of broccoli residue and temperature on Verticillium dahliae microsclerotia in soil and on wilt in cauliflower. Phytopathology 86:1303-1310.
[1] Research has also demonstrated suppression of lettuce drop caused by Sclerotinia minor from broccoli residue. Hao J, Subbarao KV, Koike ST (2003) Effects of broccoli rotation on lettuce drop caused by Sclerotinia minor and on the population density of sclerotia in soil. Plant Dis 87:159–166.

- Author: Margaret Gullette Lloyd
The days are long and the temperatures are high. This is a great time to solarize the soil in the Central Valley as a way to manage annual weeds and improve the growth of fall crops.
What is solarization? A non-chemical approach to weed control, soilborne pest management and soil enhancement using solar heating of plastic-covered moist soil. This method allows the sun's radiant energy to be trapped in the soil thereby heating the upper levels, killing and/or suppressing soil pests.
Watch video here: Clip of laying plastic with a tractor for soil solarization.
Why solarize?
- Kills or weakens many crop pests such as plant pathogenic bacteria and fungi, insects, nematodes and weeds within 2-12 inches +/- of the soil surface
- Soil physical and biological benefits
○ Increases levels of soluble, mineral nutrients available to the subsequent crop (nitrogen in the form of ammonium and nitrate, calcium, magnesium)
○ Generally less lethal to beneficial microbes than to plant pests
○ Increases breakdown of organic matter
○ Improves soil tilth
- Effects often last for multiple seasons, though best effects are directly after solarization
- Can work in combination with other pest management techniques such as steam, hot water, fumigants and pesticidal chemicals, biological controls, organic amendments (“biosolarization”), and host resistance
Watch video here: Paul Underhill discusses how has used soilsolarization
Solarization Factors:
Temperature: Solarization should be done in the warmest part of the summer. Usually this is between June and August in California. (See http://ucanr.edu/sites/solarization/california_air_temperature_maps/ for average temperatures for different months and locations in California.)
Timing: 4-6 weeks for maximum benefits (even 1-3 can provide some effects; time may be reduced with combinations such as biosolarization)
Soil preparation: Soil should be as smooth as possible to get maximum soil-plastic contact. This can be achieved by finishing with roller or bedshaper, or working the soil by hand and removing any large debris or clumps.
Plastic*: The most commonly used plastic is transparent polyethylene. Optimal thickness is 025-.03 mm unless solarization is being done in a particularly windy or animal-trafficked environment, in which case slightly thicker plastic should be used. Infrared-transmitting plastic can be purchased to facilitate a slightly higher temperature increase. Plastic with additives to decrease brittleness-causing sun damage can also be purchased.
Moisture: Soil moisture should be at 70% of field capacity in the upper layers of the soil and moist up to 24 inches deep to obtain maximum benefits. It should crumble easily when squeezed. When moistened prior to plastic, soil should be covered as soon as possible to avoid evaporation. When done after, water can be added via drip lines, furrows or hose pipe outlets under the plastic.
Beds: Plastic can be laid in strips over beds (min. 30” beds) or continuously for complete coverage. Strip coverage tends to be less effective than complete coverage because there is a cooling effect on the edge of the beds. For complete coverage, plastic can be installed by hiring a custom applicator, or can be laid in strips and joined by glue.
Bed specifications: For strip solarization, wider bed tops will accumulate more heat. It is best to solarize on strips with north-south orientation rather than east-west, to minimize shading. Beds with no slope or a slight south-facing slope will yield optimal results.
Plastic placement: Plastic can be lain by machinery or by hand. It is important that the edges of the plastic are buried.
Repair: If tearing occurs, plastic can be patched with patching tape.
Possible Disadvantages:
- Challenges associated with plastic disposal
- Perennial weeds, bulbous weeds, and seeds with a hard seed coat are harder to control
- Climate factors, such as fog and wind, may impede solarization
- May decrease root nodulating rhizobial bacteria in the soil
- May increase purple nutsedge under certain circumstances
Further information:
Check out videos, how-to and the science of solarization here.
* Plastic can be sourced locally from Irrigation Supply Co. in Woodland. The price is $137.14 for a 60” x 4000' x 0.001” roll. Plastic is usually in stock, though should be ordered several weeks to months in advance for large orders.
|
Short List of Pests Controlled by Soil Solarization (for a complete list, visit references) |
|||||
|
Plant Diseases |
Nematodes |
Weeds |
|||
|
Fusarium Wilt (tomato, cucumber, strawberry) Pythium, seedling disease Phytophthora root rot Rhizoctonia, seedling disease Verticillium Wilt |
Corky Root (tomato) Lettuce Drop White Rot (Garlic and onions) Southern blight Black root rot (many crops) Crown gall (many crops) Canker (tomato) Scab (potato) |
Ring Stem and bulb Potato cyst Northern root knot nematode Lesion nematode Citrus nematode Dagger nematode |
Velvet leaf Pigweed Fiddleneck Wild oat Black mustard Shepherd's purse Lambsquarter Miner's lettuce Field bindweed (seed only) Horseweed Large crabgrass |
Barnyardgrass Goosegrass Henbit Cheeseweed Branched broomrape Bermuda buttercup Annual bluegrass Purslane |
Common groundsel Prickly sida Black nightshade Hairy nightshade Sowthistle Common chickweed Horse purslane Common cocklebur |
|
Short List of Pests Unpredictably Controlled by Soil Solarization |
|||||
|
Diseases |
Nematodes |
Weeds |
|||
|
Charcoal Rot (many crops) Bacterial wilt |
Southern root knot nematode |
Field bindweed Bermudagrass Yellow nutsedge |
Purple nutsedge Lovegrass Bull mallow |
White sweet clover Johnsongrass |
|
References:
Elmore, C.L., Stapleton, J.J., Bell, C.E., & Devay, J.E. (1997). Soil Solarization: A Nonpesticidal Method for Controlling Diseases, Nematodes, and Weeds. (Publication No. 21377). Oakland: University of California Agricultural and Natural Resources.
Stapleton, J.J. (n.d.). Solarization for Vegetable Weed Control. California Weed Science Society. Retrieved from http://www.cwss.org/uploaded/media_pdf/59-67_2007.pdf
Yaduraju, N.T., & Mishra, J.S. (2004). Soil Solarization: An Eco-Friendly Approach for Weed Management. In Inderjit (Ed.), Weed Biology and Management. (1st ed., pp. 345-362). Dordrecht, Netherlands: Springer Science + Business Media.
- Author: Margaret Gullette Lloyd
Yes, mow it! Mowing works to at least prevent seed spread and regular mowing will weaken rhizomes.
Seed establishment has the greatest potential for the establishment and spread of johnsongrass and must be prevented. Therefore, mowing works to at least prevent seed spread. An individual plant can produce 28,000-30,000 seeds and a single inflorescence can measure 1,240 seeds (Keeley and Thullen, 1979). Viable seed may be produced as early as 2 weeks after flowering begins, so timely control measures throughout the season are needed to prevent seed production. Even though the viability of johnsongrass seed in soil remains high for as long as five years, seed can remain viable in soil up to 10 years.
Regular mowing can be effective in both orchard and non-crop situations. Due to its capacity to regenerate from rhizome fragments, complete control of S. halepense by mowing alone is difficult. Nonetheless, intensive grazing and mowing can be used to reduce a stand of johnsongrass. If grazed or mowed closely for at least two years, the plants become weak and stunted and the rhizomes become concentrated near the soil surface.
Consider mowing johnsongrass stands before seeds set to weaken current stands and avoid the spread through seed dispersal.

REFERENCE
P. E. Keeley, & R. J. Thullen. (1979). Influence of Planting Date on the Growth of Johnsongrass (Sorghum halepense) from Seed. Weed Science, 27(5), 554-558. Retrieved June 30, 2020, from www.jstor.org/stable/4043118
- Author: Margaret Gullette Lloyd
Are you concerned with your plant growth or wondering how your winter and spring activities (compost, cover crops, pre-plant fertilizers) are contributing to plant-available nitrate? If so, now is a great time to consider taking leaf or soil samples.
Here are easy ways to test for plant or soil nitrate.
SOIL NITRATE TESTS will tell you about the current plant-available nitrate levels. This can help you understand how compost, cover crops, pre-plant fertilizers as well as soil organic matter are currently contributing to nitrate in the soil.
Soil nitrate can be determined by taking a soil sample and sending it to a lab or using test strips which can be done in the field for an immediate estimate.
- How to Soil Sample for Nitrate Determination can be found here.
- How to use Soil Nitrate Test Strips can be found here
- Also, here's an instructional video on how to use Soil Nitrate Test Strips in the field.

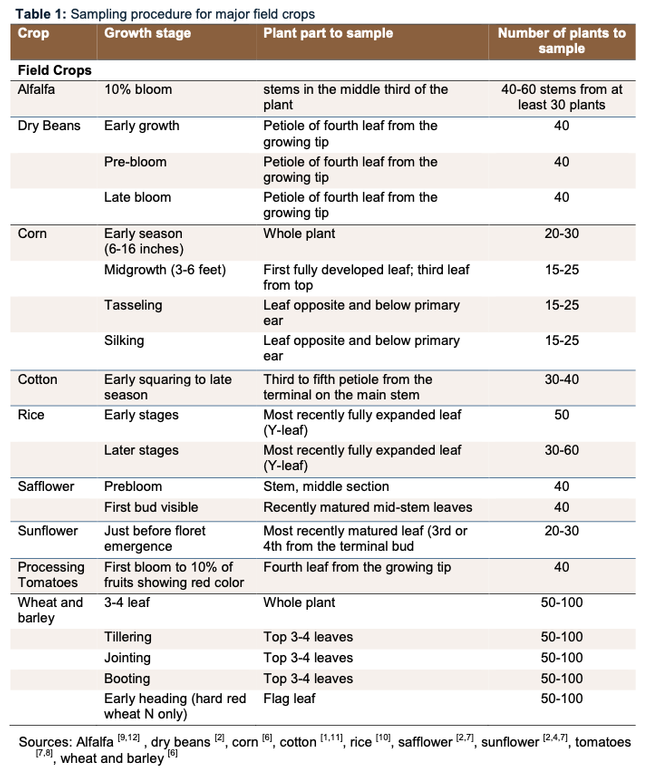
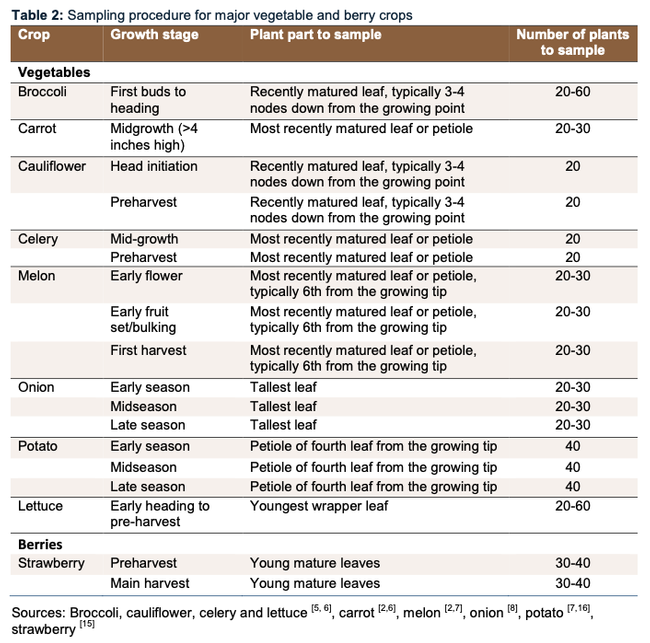
PLANT TISSUE samples are useful for understanding the status of your plant nutrition (NPK), which helps answer the questions: are you meeting the crop demand or should you supplement? This can also help answer questions about whether symptoms you are seeing are due to crop nutrition or something else. Below is a list of the optimal plant tissue to sample for nitrate analysis. Click here for more detailed information.

The timing and type of tissue is unique to each crop. Use these tables as guidelines for your crop of interest.