- Author: Margaret Gullette Lloyd
This post was generated in response to a case of Fusarium crown and foot rot in Sacramento County on 'Lumina' pumpkins.
SYMPTOMS
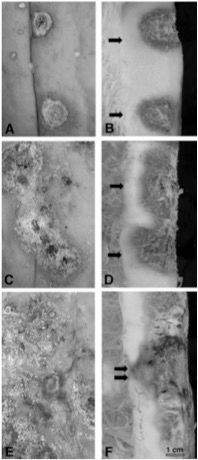
- The first symptoms are wilting of the leaves of the entire plant, followed by plant death within several days (Image 1).
- Early infection symptoms at the crown include light colored, water soaked areas that become progressively darker.
- Advanced disease symptoms show destroyed plant tissue leaving only fibrous vascular strands.
- Examination of the crown shows necrotic rot at or immediately below the soil surface.
- Upon removal of an infected plant, a bad odor coming from the stem lesions is most likely caused by secondary bacteria that have colonized the decaying tissue and are not the cause of plant death.
- Pink to white fungal mycelium is often found on the soil surface beside the lesion. Also, darkened soil around the crown can be evident (Image 2).
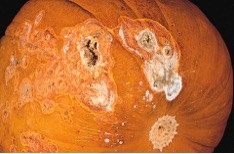
- If soil is the source of inoculum and therefore point of infection, fruits can also be attacked. Where the fruit rests on a moist soil surface, mycelial growth and dry rot may be evident (Image 4).


ABOUT THE PATHOGEN and DISEASE
California produces 25% of the pumpkins in the United States and is the largest supplier of the nation's fresh-market pumpkin. For pumpkin growers on California's central coast, Fusarium foot and crown rot of squash is an economic problem with at least 30% of the pumpkin fruit infected in some fields (4).
Fusarium crown and foot rot is caused by Fusarium solani f. sp. cucurbitae race 1 (Fsc), a fungal pathogen found worldwide that is specific to members of the Cucurbitae family. Fsc has two races, race 2 was identified with sewage water and is not considered a problem for growers. Because there are few morphological features that distinguish Fsc1 from Fsc2 (5), field diagnosis of the disease has been inferred based on whether or not crown and root rot is present.
In the field, the disease is generally a problem only in summer squash, winter squash and certain pumpkin cultivars. Howden, the leading cultivar in pumpkin production (4), is highly susceptible to fruit infection by Fsc. Laboratory studies have shown, however, that most cucurbits are susceptible, including melon and watermelon.
Plants and fruit of any age can be infected. Mature cucumber is much less susceptible than other cucurbits, but all are equally susceptible in the seedling stage. If fruit becomes infected, after Fsc penetrates the fruit rind, and flesh, it can ultimately grow into the seed cavity.
Inoculum can come from the soil and/or the seed. Inoculum from the soil originates from previously infected plants incorporated into the soil. From seed, Fsc can be internally or externally seedborne. It is found in the seed coat and between the seed coat and cotyledons, however, it generally does not invade the cotyledons or reduce seed viability (11). It remains viable in seeds for 1-2 years.
MANAGEMENT
Fusarium crown and foot rot occurs sporadically in most areas, and disease severity is dependent on soil moisture and inoculum density.
- Crop rotation. Because race 1 can live in the soil for 2-3 years (8), recommendations for control of FFR include a 4-year rotation without cucurbits if a field is infested and use of clean seed when replanted (1,3,8,11).
- Pathogen-free seed. Fsc can be externally or internally seedborne. Commercially produced seed that is coated with a contact fungicide (e.g., thiram), can reduce the incidence of disease initiated from infected seed. However, it does not entirely prevent the transmission of F. solani f. sp. cucurbitae-infected seed because the pathogen can remain viable inside the seed coat. Therefore, only pathogen-free seed can prevent the transmission of F. solani f. sp. cucurbitae. However, in the United States, cucurbit seed are sold to growers without any guarantee that the seed are free of Fsc. To treat infested seed, soak in hot water (55°C/131°F) for 15 minutes.
- Remove infected plant debris and associated soil, when possible.
RELATED RESEARCH
Do you save your pumpkin seed? Because clean seed is critical for disease control, UC Davis researchers conducted experiments to determine if visual rating of the fruit at harvest could predict whether seed would be infected. Results showed that no infected seed were recovered from fruit in which the surface was lesion-free or in which a lesion extended less than midway through the fruit flesh. Consequently, a rapid, visual inspection and exclusion of symptomatic fruit should be sufficient to obtain uninfected seed, even in infested fields.
Mehl, H. L., and Epstein, L. 2007. Identification of Fusarium solani f. sp. cucurbitae race 1 and race 2 with PCR and production of disease-free pumpkin seeds. Plant Dis. 91:1288-1292.
Biological control of Fusarium wilt of cucumber by chitinolytic bacteria.
Like the exoskeleton of insects, fungal cell walls are largely composed of chitin. In this research, two chitin-degrading bacterial strains, Paenibacillus sp. 300 and Streptomyces sp. 385, suppressed Fusarium wilt of cucumber caused by Fusarium oxysporum f. sp. cucurbitae in nonsterile, soilless potting medium. A mixture of the two strains in a ratio of 1:1 or 4:1 gave significantly (P< 0.05) better control of the disease than each of the strains used individually or than mixtures in other ratios.
Please note that this work was not done in field soil under field conditions, but in potting medium. Especially for biocontrol work, extrapolating findings to be applicable in the field can be a long stretch. For field production, I would start with a small test plot.

Singh, P. P., Shin, Y. C., Park, C. S., and Chung, Y. R. 1999. Biological control of Fusarium wilt of cucumber by chitinolytic bacteria. Phytopathology 89:92-99.
REFERENCES
1. APSnet Fusarium Crown and Foot Rot of Squash
http://www.apsnet.org/publications/apsnetfeatures/Pages/CrownFootRotSquash.aspx
2. Champaco, E. R., Martyn, R. D., and Miller, M. E. 1993. Comparison of Fusarium solani and F. oxysporum as causal agents of fruit rot and root rot of muskmelon. HortScience 28:1174-1177.
3. Davis, R. M., Turini, T. A., Gubler, W. D., and Stapleton, J. J. 2005. UC IPM Pest Management Guidelines: Cucurbits. Univ. Calif. Agric. Nat. Resour. Publ. 3445.
4. Gaskell, M., and Smith, R. 1997. Pumpkin production in California. Vegetable Production Series. Univ. Calif. Agric. Nat. Resour. Publ. 7222.
5. Matuo, T., and Snyder, W. C. 1973. Use of morphology and mating populations in the identification of formae speciales in Fusarium solani. Phytopathology 63:562-565.
6. Mehl, H. L., and Epstein, L. 2007. Identification of Fusarium solani f. sp. cucurbitae race 1 and race 2 with PCR and production of disease-free pumpkin seeds. Plant Dis. 91:1288-1292.
7. Mehl, H. L., and L. Epstein. 2007. Fusarium solani species complex isolates conspecific with Fusarium solani f. sp. cucurbitae race 2 from naturally infected human and plant tissue and environmental sources are equally virulent on plants, grow at 37°C and are interfertile. Environmental Microbiology 9:2189-2199.
8. Nash, S. M., and Alexander, J. V. 1965. Comparative survival of Fusarium solani f. cucurbitae and F. solani f. phaseoli in soil. Phytopathology 55:963-966.
9. Paternote, S. J. 1987. Pathogenicity of Fusarium solani f. sp. cucurbitae race 1 to courgette. Neth. J. Plant Pathol. 93:245-252.
10. Sumner, D. R. 1976. Etiology and control of root rot of summer squash in Georgia. Plant Dis. Rep. 60:923-927.
11. Toussoun, T. A., and Snyder, W. C. 1961. The pathogenicity, distribution, and control of two races of Fusarium(Hypomyces) solani f. cucurbitae. Phytopathology 51:17-22.
ACKNOWLEDGEMENTS
Thanks for assistance from Dr. Lynn Epstein.