- Author: Kathy Keatley Garvey

Attardo will discuss “The Mating Biology of Tsetse Flies--Insights into the Morphological, Biochemical, and Molecular Responses to Mating Stimuli in a Viviparous Disease Vector.”
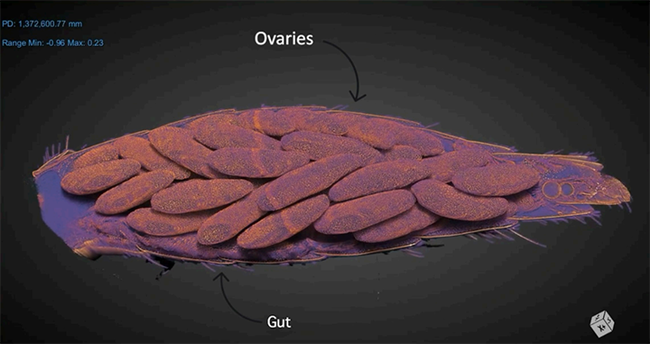
"Research into the reproductive behavior of tsetse flies offers key insights into controlling diseases like African sleeping sickness," he writes in his abstract. "Unique among insects, these flies give birth to live offspring. During mating, males transfer a mix of sperm and other vital substances to the females. This study employs state-of-the-art techniques, including 3D scanning and genetic analysis, to monitor changes in the female fly's reproductive system over a 72-hour period post-mating. Findings indicate that mating sets off a chain of intricate changes in the female, affecting everything from biochemistry to gene activity. These changes prepare her for pregnancy and childbirth. The study opens up new avenues for understanding tsetse fly biology and offers potential strategies for disease control."
The seminar also will be on Zoom. The link:
https://ucdavis.zoom.us/j/95882 849672
The Attardo lab monitors the dynamics of vector insects at the levels of physiology, population genetics and environmental interactions.
Attardo is a global expert on vectorborne diseases, including his groundbreaking work on tsetse flies. He researches the invasive yellow mosquito, Aedes aegypti, which can carry such diseases as dengue, chikungunya, Zika and yellow fever. His work involves predicting insecticide resistance and tracking movements of genetically independent populations of aegypti throughout the state.
Attardo, who holds a doctorate in genetics from Michigan State University, where he researched the molecular biology of mosquito reproduction, joined the UC Davis Department of Entomology and Nematology in 2017 from the Yale School of Public Health's Department of Epidemiology of Microbial Diseases.
He received the 2022 Medical, Urban, and Veterinary Entomology Award from the Pacific Branch, Entomological Society of America, which encompasses 11 Western states, plus parts of Canada and Mexico, and U.S. territories.
For any technical issues regarding Zoom, contact seminar coordinator Brian Johnson at brnjohnson@ucdavis.edu.
Resources:
- Tsetse Fly Research (article featuring Geoffrey Attardo, Feb. 3, 2021, UC Davis Department of Entomology and Nematology
- A Tsetse Fly Births One Enormous Milk-Fed Baby, Deep Look, featurng the work of Geoffrey Attardo

- Author: Kathy Keatley Garvey

The event, free and family friendly, will take place in Room 1124 of the Academic Surge Building, 455 Crocker Lane, UC Davis. Parking is also free.
Among the presenters will be
- Lynn and Bob Kimsey of the UC Davis Department of Entomology and Nematology faculty. Lynn, a hymenopterist, is a UC Davis distinguished professor who teaches general entomology and the biodiversity of California insects and serves as the director of the Bohart Museum, and Bob is a forensic entomologist, specializing in public health entomology; arthropods of medical importance; zoonotic disease; biology and ecology of tick-borne pathogens; tick feeding behavior and biochemistry.
- Carla-Cristina "CC" Melo Edwards, a first-year doctoral student in the laboratory of medical entomologist-geneticist Geoffrey Attardo, associate professor of entomology, UC Davis Department of Entomology and Nematology. She will share her expertise on mosquitoes and show specimens.
- Moriah Garrison, senior entomologist and research coordinator with Carroll-Loye Biological Research (CLBR). She is scheduled to show live ticks and mosquitoes and field questions.
- Educators from the Sacramento-Yolo Mosquito and Vector Control District. They will discuss mosquitoes and their program
- Nazzy Pakpour, UC Davis alumna, Novozymes scientist and author of Please Don't Bite Me
- Jeff Smith, curator of the Bohart Museum's ;Lepidoptera (butterflies and moths) collection. He will display butterfly specimens collected globally. Also on the "Lep crew" are Bohart volunteers Greg Kareofelas and Brittany Kohler.
CC Edwards. In the Attardo lab, CC focuses her research "on investigating the physiological mechanisms underlying pyrethroid resistance in Aedes aegypti (the yellow fever mosquito)." She was a McNair scholar at Baylor University, where she completed her undergraduate degree in cell and molecular biology in May 2021. "I got interested in the mosquito field through my undergraduate research of studying the sensory and oviposition responses of Aedes aegypti in relation to the compound geosmin."
"I went on to do my masters at Texas Tech University under the advisement of Dr. Corey Brelsfoard where I graduated this past summer (2023)," she said. "I investigated the effects of microplastics in relation to the mosquitoes Aedes aegypti and Aedes albopictus (the Asian tiger mosquito). Plastic pollution has been a worldwide problem and research has shown that microplastics have been found in the guts of various organisms. With Aedes species being container inhabiting species, my master's research was focused on investigating if there are alterations of the mosquitoes' microbiomes and their immunity due to the ingestion of microplastics."
"When I am not in the lab, I enjoy getting involved with my local community by helping out and doing outreach," Edwards said. This past summer she helped the city of Lubbock, Amarillo, and the Texas Public Health Department by identifying mosquitoes for West Nile surveillance. She also served as the outreach chair for the Texas Tech Association of Biologists during her masters' degree pursuit and enjoyed being a mentor for first-generation students.

CLBR, involved in insect repellent testing and natural product development, is owned and managed by scientists Scott Carroll and Jenella Loye, a husband-wife team affiliated with UC Davis Department of Entomology an Nematology. Formed in 1989, the company serves the consumer products industry with testing, development and regulatory services for insect repellents, organic products, and low toxicity pesticides. Carroll, president of CLBR, is an evolutionary biologist, while Loye is a medical entomologist and microbiologist. Carroll holds a doctorate in biology/biological sciences from the University of Utah, while Loye holds a doctorate in zoology, microbiology and epidemiology from the University of Oklahoma.
Photography Displays. Professor Attardo, who maintains a lab website on Vector Biology and Reproductive Biology at http://attardo-lab.com, and chairs the Designated Emphasis in the Biology of Vector-Borne Diseases (DEBVBD), will display some of his mosquito images, including a blood-fed Aedes aegypti, and a female and male Culex tarsalis. Alex Wild, a UC Davis doctoral alumnus and curator of entomology, University of Texas, Austin, will display an image of mosquito larvae that currently hangs in Briggs Hall, home of the UC Davis Department of Entomology and Nematology. Wild's insect images can be viewed on his website, https://www.alexanderwild.com.
Petting Zoo. A popular attraction is the live petting zoo; visitors are encouraged to hold or get acquainted with live Madagascar hissing cockroaches and stick insects.
Family Arts and Crafts Activity. The event will be held outside and will highlight two collecting techniques, said Tabatha Yang, education and outreach coordinator.
- Clear Packing Tape Art. "Clear packing tape is a good way to collect small, hard-to-see insects," Yang said. "Glitter will mimic small insects like fleas or bed bugs. Putting the tape on white paper makes it easy to look at them under a microscope and for this craft it will make a pretty card."
- Making insect collecting or "kill" jars. Participants are asked to bring a recycled jar. "This should be a clean and dried glass jar with a wide, metal top--think jam, pickle, peanut butter jars. Four to 16-ounce jars work well. We will have some on hand as well, but recycling is good! We will fill the bottom with plaster of paris and let it dry and teach people how to use it properly, using something like nail polisher remover containing ethyl acetate as the killing agent. A UC Davis Department of Entomology and Nematology video explains the procedure: https://youtu.be/s8yCzFGzbn8?si=71sNmA5l8NyP1zj0



- Author: Kathy Keatley Garvey

Her seminar takes place at 3:30 in 366 Briggs Hall, and also will be on Zoom.
Mack studies Aedes aegypti with a focus on analysis of transcriptomic datasets and 3D imaging datasets. "Throughout my time in graduate school, my projects have considered pyrethroid resistance in Aedes aegypti ;examining the genetic response to this insecticide. As I finish up my dissertation, I hope to pursue a career in industry using the skills I've developed to continue to analyze large datasets!"
Insecticide resistance is a global issue, Mack says in her exit seminar abstract. Ae. aegypti, known as "the yellow fever mosquito," can transmit dengue fever, chikungunya, Zika fever, Mayaro and yellow fever viruses, and other disease agents. The mosquito was first colonized California in 2013 and arrived resistant to pyrethroids. "The pyrethroid target site genotype differs geographically in California and partially infers resistance phenotype, indicating that other mechanisms are at play as well."

"Since their detection in 2013, Aedes aegypti has become a widespread urban pest in California," the co-authors wrote in the abstract. "The availability of cryptic larval breeding sites in residential areas and resistance to insecticides pose significant challenges to control efforts. Resistance to pyrethroids is largely attributed to mutations in the voltage gated sodium channels (VGSC), the pyrethroid site of action. However, past studies have indicated that VGSC mutations may not be entirely predictive of the observed resistance phenotype."
"To investigate the frequencies of VGSC mutations and the relationship with pyrethroid insecticide resistance in California, we sampled Ae. aegypti from four locations in the Central Valley, and the Greater Los Angeles area. Mosquitoes from each location were subjected to an individual pyrethrum bottle bioassay to determine knockdown times. A subset of assayed mosquitoes from each location was then analyzed to determine the composition of 5 single nucleotide polymorphism (SNP) loci within the VGSC gene."
The conclusion:
"Resistance associated VGSC SNPs are prevalent, particularly in the Central Valley. Interestingly, among mosquitoes carrying all 4 resistance associated SNPs, we observe significant heterogeneity in bottle bioassay profiles suggesting that other mechanisms are important to the individual resistance of Ae. aegypti in California."
Mack, who holds a bachelor of science degree (2018) in biology from Creighton University, Omaha, Neb., enrolled in the UC Davis graduate school program in 2018.
Active in the Entomological Society of America, Mack scored second place in student competition at the 2022 joint meeting of the Entomological Societies of America, Canada, and British Columbia, held last November in Vancouver, British Columbia. She entered her presentation, "Three Dimensional Analysis of Vitellogenesis in Aedes aegypi Using Synchrotron X-Ray MicroCT,” in the category, "Graduate School Physiology, Biochemistry and Toxicology: Physiology.
Her abstract: "Traditional methods of viewing the internal anatomy of insects require some degree of tissue manipulation and/or destruction. Using synchrotron-based x-ray phase contrast microCT (pcMicroCT) avoids this issue and has the capability to produce high contrast, three dimensional images. Our lab is using this technique to study the morphological changes occurring in the mosquito Aedes aegypti during its reproductive cycle. Ae. aegypti is the primary global arbovirus vector, present on all continents except Antarctica. Their ability to spread these viruses is tightly linked with their ability to reproduce, as the production of eggs in this species is initiated by blood feeding. Amazingly, this species produces a full cohort of eggs (typically 50-100) in just 3 days' time following a blood meal. This rapid development represents dramatic shifts in physiological processes that result in massive volumetric changes to internal anatomy over time. To explore these changes thoroughly, a time course of microCT scans were completed over the vitellogenic period. This dataset provides a virtual representation of the volumetric, conformational, and positional changes occurring in tissues important for reproduction across the vitellogenic period. This dataset provides the field of vector biology with a detailed three-dimensional internal atlas of the processes of vitellogenesis in Ae. aegypti."
"As for career plans, I am applying to computational biology positions in industry," Mack said. "I'm not filing my dissertation until July so I am still working on this."
- Author: Kathy Keatley Garvey

Nayduch, based in Manhattan, Kansas, will speak at 4:10 p.m., Wednesday, March 1 in 122 Briggs Hall. Her lecture also will be virtual. The Zoom link:
https://ucdavis.zoom.us/j/95882849672
"Dana is doing very cool work with house flies and looking at how bacteria in the fly are trading antibiotic resistance genes amongst themselves," said the seminar host, medical entomologist-geneticist Geoffrey Attardo, assistant professor, UC Davis Department of Entomology and Nematology. "It's an interesting and scary system as antibiotic resistance is so high due to antibiotic usage in livestock rearing."
"House flies (Musca domestica L.) are ubiquitous, cosmopolitan pests inhabiting urban, rural and agricultural environments throughout the world. In these habitats acquire microbes from septic substrates that are used for feeding and reproduction. Flies subsequently harbor and disseminate these microorganisms which may pose a risk to human and animal health," Nayduch says in her abstract. "Our research characterizes and analyzes microbial communities of house flies using culture-based and molecular approaches in order to better understand their roles in the transmission of important bacterial disease agents and/or antimicrobial resistance. Because the microbial communities within house flies represent a snapshot of the microbes found in their local habitat, we also gain valuable insight into existing and emerging microbial threats to humanand animal health through our surveys which can help in predicting and preventing disease."
A pre-seminar will take place from 3:30 to 4:10 in 158 Briggs.
Nayduch, an authority on fly-microbe interactions, joined USDA in August of 2011. She is a member of the USDA-ARS Arthropod-Borne Animal Diseases Research Unit, researching molecular and microbiological studies of Culicoides midges and house flies. She works with several laboratories on comparative transcriptomic and microbiomic studies of Muscid flies.
Nayduch received her bachelor's degree in animal science from Rutgers University and her doctorate in zoology from Clemson University, where she studied house flies as vectors for pathogens. She served as a postdoctoral fellow at Yale University School of Public Health, working on molecular-genetic studies of tsetse flies. She then joined Georgia Southern University (GSU) as an assistant professor of biology in 2004, advancing to associate professor in 2009. At GSU she received NIH-R15 funding to study house fly-microbe molecular interactions.
Nayduch, active in the Entomological Society of America (ESA), is the vice president-elect of the Medical, Urban and Veterinary Entomology (MUVE) Section. A peer reviewer for the Journal of Medical Entomology and an editorial board member and subject editor for Annals for ESA, she organized and edited the first special collection for Annals: “Filth Fly-Microbe Interactions."
The UC Davis Department of Entomology and Nematology's winter seminars are held on Wednesdays at 4:10 p.m. in 122 Briggs Hall. All are virtual. A pre-seminar coffee is held from 3:30 to 4:10 p.m. in 158 Briggs. Urban landscape entomologist Emily Meineke, assistant professor, coordinates the seminars. (See schedule.) She may be reached at ekmeineke@ucdavis.edu for technical issues.

- Author: Kathy Keatley Garvey

The students, along with the three winners in the Arts, Humanities and Social Sciences category, received their awards at a recent ceremony in the UC Davis Shields Library Courtyard.
A paper written by Maram Saada, a student in a longevity class taught by UC Davis distinguished professor James R. Carey, won first place in the SEM category and a $1000 prize. Saada's research paper, “Huntington's Disease: Etiology, Research Models and Treatment,” is now online. It will be published in eScholarship, an open-access scholarly publishing service affiliated with the University of California.
Second place in the SEM category went to Jocelyn Chu, a student in a medical entomology class taught by assistant professor Geoffrey Attardo, for her paper, “Vector-borne Disease Control Plan for West Nile Virus in California.” She received $750.
Two of Carey's students, Jessica Hevener and David Vo, tied for third place in the SEM category. Hevener, of Carey's longevity class, submitted her paper on “The Impact of Maternal Obesity on Maternal and Offspring Health,” while Vo, enrolled in a special study class for advanced undergraduates, entered his paper on “Surviving the Cold: How Circumpolar Peoples Have Adapted to the Extreme Conditions of the Arctic Entomology." They shared the third-prize award of $500.

The Lang Prize competition recognizes students who make exceptional use of library resources and services — such as primary source materials and special collections, online databases and journal articles; inter-library loan services, or consulting with a librarian, according to Alesia McManus, chair of the Lang Prize for Undergraduate Information Research and head of student services, UC Davis Shields Library.
In a earlier news release, McManus announced that “It was a strong pool of applications, with 17 out of the 21 applications meeting the average 39.99 score threshold for being considered for an award." The Lang Prize honors the legacy of professor emeritus and plant biologist Norma Lang, 1931-2015, a member of the faculty from 1963 to 1991.
UC Davis Distinguished Professor James R. Carey. Carey, a member of the UC Davis entomology faculty since 1980, instructs undergraduates in his classes--which usually exceed 200 students--how to research topics, use style sheets, and structure their papers. He has produced 13 videos on how to research and write a research paper, along with a new video on the use of style sheets.
Highly honored by his peers for his teaching expertise, Carey received the Entomological Society of America's 2015 Distinguished Teaching Award; a 2018 Robert Foster Cherry Award from Baylor University, which presents international teaching awards; and the UC Davis Academic Senate's 2014 Distinguished Teaching Award, an honor given to internationally recognized professors who excel at teaching.
Carey is considered the preeminent global authority on arthropod demography. He directed the multidisciplinary, 11-institution, 20-scientist program, “Biodemographic Determinants of Lifespan,” which garnered more than $10 million in funding from the National Institute on Aging from 2003 to 2013.


Assistant Professor Geoffrey Attardo. UC Davis medical entomologist-geneticist Geoffrey Attardo, who joined the UC Davis Department of Entomology and Nematology in 2017 from the Yale School of Public Health's Department of Epidemiology of Microbial Diseases, is a global expert on vectorborne diseases, and renowned for his groundbreaking work on tsetse flies. The Attardo lab monitors the dynamics of vector insects at the levels of physiology, population genetics and environmental interactions.
Attardo, who won the 2022 Medical, Urban, and Veterinary Entomology (MUVE) Award from the Pacific Branch, Entomological Society of America, “excels not only as a researcher, but as a teacher, mentor, scientific illustrator, macro photographer,videographer and science communicator,” said UC Davis distinguished professor Bruce Hammock in his letter of nomination for the MUVE Award. Attardo drew praise for his "highly effective teaching style in helping students build critical thinking skills and confidence."
The 2022 winners of the Lang Prizes in the Arts, Humanities and Social Sciences category are Joyce Do, Annie Miyadi, and Sarah Grimes. (See more on Shields Library website.)




