- Author: Surendra K. Dara
Integrated pest management, commonly referred to as IPM, is a concept of managing pests that has been in use for several decades. The definition and interpretation of IPM vary depending on the source, such as a university, institute, or a researcher, and its application varies even more widely depending on the practitioner. Here are a few examples of its definitions and interpretations:
“IPM is an ecosystem-based strategy that focuses on long-term prevention of pests or their damage through a combination of techniques such as biological control, habitat manipulation, modification of cultural practices, and use of resistant varieties. Pesticides are used only after monitoring indicates they are needed according to established guidelines, and treatments are made with the goal of removing only the target organism. Pest control materials are selected and applied in a manner that minimizes risks to human health, beneficial and nontarget organisms, and the environment.” UC IPM
“Integrated Pest Management, or IPM, is an approach to solving pest problems by applying our knowledge about pests to prevent them from damaging crops, harming animals, infesting buildings or otherwise interfering with our livelihood or enjoyment of life. IPM means responding to pest problems with the most effective, least-risk option.” IPM Institute of North America
“A well-defined Integrated Pest Management (IPM) is a program that should be based on prevention, monitoring, and control which offers the opportunity to eliminate or drastically reduce the use of pesticides, and to minimize the toxicity of and exposure to any products which are used. IPM does this by utilizing a variety of methods and techniques, including cultural, biological and structural strategies to control a multitude of pest problems.” Beyond Pesticides
“IPM is rotating chemicals from different mode of action groups.” A grower
These definitions and interpretations represent a variety of objectives and strategies for managing pests. IPM is not a principle that can/should be strictly and equally applied to every situation, but a philosophy that can guide the practitioner to use it as appropriate for the situation. For example, varieties that are resistant to arthropod pests and diseases are available for some crops, but not for others. Mating disruption with pheromones is widely practiced for certain lepidopteran and coleopteran pests, but not for several hemipteran pests. Biological control is more readily employed for greenhouse pests, but not to the same extent under field conditions. While chemical pesticides should be used as the last resort, in principle, sometimes they are the first line of defense to prevent damage to the transplants by certain pests or area-wide spread of certain endemic or invasive pests and diseases.
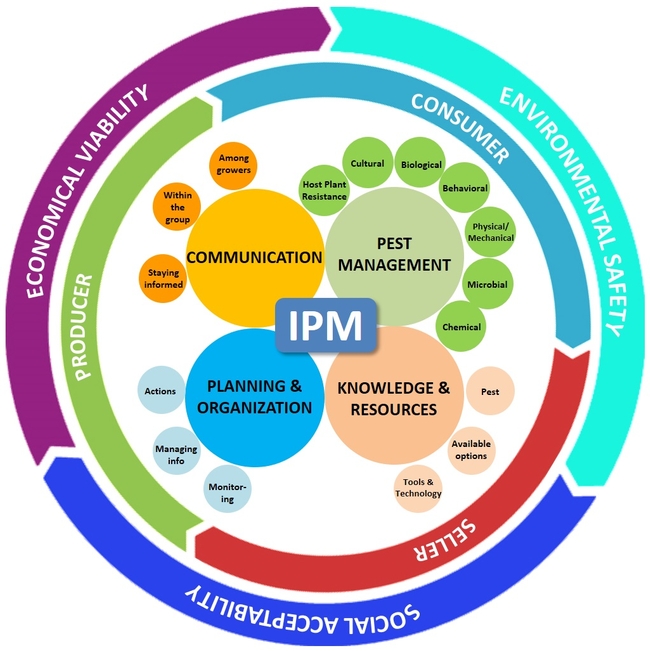
Crop production is an art, science, and business, and by adding environmental and social factors, IPM – an approach used in agriculture – can also be influenced by a number of factors. Each grower has their own strategy for producing crops, minimizing losses, and making a profit in a manner that is acceptable to the society, safe for the consumers, and less disruptive to the environment. In other words, “IPM is an approach to manage pests in an economically viable, socially acceptable, and environmentally safe manner”. Keeping this simple, but loaded, definition in mind and considering recent advances in crop production and protection, communication technology, and globalization of agriculture and commerce, here is the new paradigm of IPM with its management, business, and sustainability aspects.
I. Management Aspect
There are four major components in the IPM model that address the various pest management options, the knowledge and resources the grower has in order to address the pest issue, planning and organization of information to take appropriate actions, and maintaining good communication to acquire and disseminate knowledge about pests and their management.
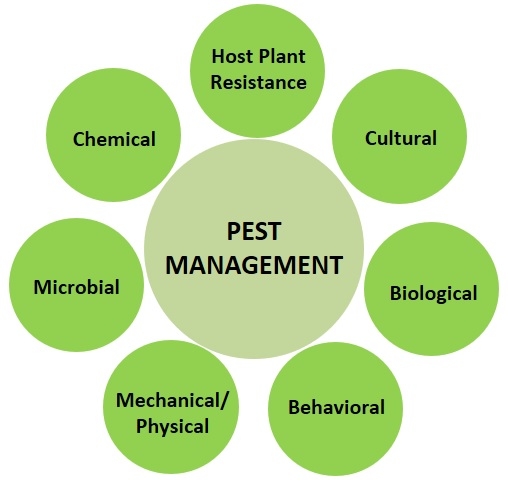
1. Pest Management:
The concept of pest control has changed to pest management over the years knowing that a balanced approach to managing pest populations to levels that do not cause economic losses is better than eliminating for environmental and economic reasons. Although the term control is frequently used in literature and conversations, it generally refers to management. A thorough knowledge of general IPM principles and various management options for all possible pest problems is important as some are preventive and others are curative. It is also essential to understand inherent and potential interactions among these management options to achieve maximum control. The following are common control options that can be employed at different stages of crop production to prevent, reduce, or treat pest infestations. Each of them may provide only a certain level of control, but their additive effect can be significant in preventing yield losses.
a. Host plant resistance: It involves the use of pest resistant and tolerant cultivars developed through traditional breeding or genetic engineering. These cultivars possess physical, morphological, or biochemical characters that reduce the plant's attractiveness or suitability for the pest to feed, develop, or reproduce successfully. These cultivars resist or tolerate pest damage and thus reduce the yield losses.
b. Cultural control: Changing agronomic practices to avoid or reduce pest infestations and damage refers to cultural control. Adjusting planting dates can help escape pest occurrence or avoid most vulnerable stages. Modifying irrigation practices, fertilizer program, plant or row spacing, and other agronomic practices can create conditions that are less suitable for the pest. Destroying crop residue and thorough cultivation will eliminate breeding sites and control soil-inhabiting stages of the pest. Crop rotation with non-host or tolerant crops will break the pest cycles and reduce their buildup year after year. Choosing clean seed and plant material will avoid the chances of introducing pests right from the beginning of the crop production. Sanitation practices to remove infected/infested plant material, regular cleaning field equipment, avoiding accidental contamination of healthy fields through human activity are also important to prevent the pest spread. Intercropping of non-host plants or those that deter pests or using trap crops to divert pests away from the main crop are some of the other cultural control strategies.
c. Biological control: Natural enemies such as spiders, predators, and parasitic wasps can be very effective in causing significant reductions in pest populations in certain circumstances. Periodical releases of commercially available natural enemies or conserving natural enemy populations by providing refuges or avoiding practices that harm them are some of the common practices to control endemic pests. To address invasive pest issues, classical biological control approach is typically employed where natural enemies from the native region of the invasive pest are imported, multiplied, and released in the new habitat of the pest. The release of irradiated, sterile insects is another biological control technique that is successfully used against a number of pests.
d. Behavioral control: Behavior of the pest can be exploited for its control through baits, traps, and mating disruption techniques. Baits containing poisonous material will attract and kill the pests when distributed in the field or placed in traps. Pests are attracted to certain colors, lights, odors of attractants or pheromones. Devices that use one or more of these can be used to attract, trap or kill pests. Pheromone lures confuse adult insects and disrupt their mating potential, and thus reduce their offspring.
e. Physical or mechanical control: This approach refers to the use of a variety of physical or mechanical techniques for pest exclusion, trapping (in some cases similar to the behavioral control), removal, or destruction. Pest exclusion with netting, handpicking or vacuuming to remove pests, mechanical tools for weed control, traps for rodent pests, modifying environmental conditions such as heat or humidity in greenhouses, steam sterilization or solarization, visual or physical bird deterrents such as reflective material or sonic devices are some examples for physical or mechanical control.
f. Microbial control: Using entomopathogenic bacteria, fungi, microsporidia, nematodes, and viruses, and fermentation byproducts of microbes against arthropod pests, fungi against plant parasitic nematodes, and bacterial and fungal antagonizers of plant pathogens generally come under microbial control.
g. Chemical control: Chemical control typically refers to the use of synthetic chemical pesticides, but to be technically accurate, it should include synthetic chemicals as well as chemicals of microbial or botanical origin. Although botanical extracts such as azadirachtin and pyrethrins, and microbe-derived toxic metabolites such as avermectin and spinosad are regarded as biologicals, they are still chemical molecules, similar to synthetic chemicals, and possess many of the human and environmental safety risks as chemical pesticides. Chemical pesticides are categorized into different groups based on their mode of action and rotating chemicals from different groups is recommended to reduce the risk of resistance development. Government regulations restrict the time and amount of certain chemical pesticides and help mitigate the associated risks.
The new RNAi (ribonucleic acid interference) technology where double-stranded RNA is applied to silence specific genes in the target insect is considered as a biopesticide application. Certain biostimulants based on minerals, microbes, plant extracts, seaweed or algae impart induced systemic resistance to pests and diseases, but are applied as amendments without any claims for pest or disease control. These new products or technologies can fall into one or more abovementioned categories.
As required by the crop and pest situation, one or more of these control options can be used throughout the production period for effective pest management. When used effectively, non-chemical control options delay, reduce, or eliminate the use of chemical pesticides.
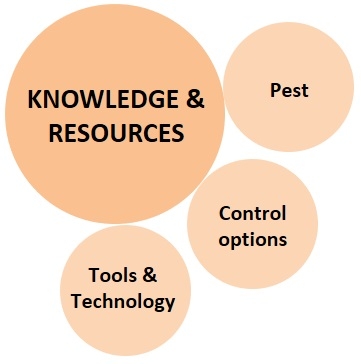
2. Knowledge and Resources:
The knowledge of various control options, pest biology and damage potential, and suitability of available resources enables the grower to make a decision appropriate for their situation.
a. Pest: Identification of the pest, understanding its biology and seasonal population trends, damaging life stages and their habitats, nature of damage and its economic significance, vulnerability of each life for one or more control options, host preference and alternate hosts, and all the related information is critical for identifying an effective control strategy.
b. Available control options: Since not all control options can be used against every pest, the grower has to choose the ones that are ideal for the situation. For example, systemic insecticides are more effective against pests that mine or bore into the plant tissue. Pests that follow a particular seasonal pattern can be controlled by adjusting planting dates. Commercially available natural enemies can be released against some, while mating disruption works well against others. Entomopathogenic nematodes can be used against certain soil pests, bacteria and viruses against pests with chewing mouthparts such as lepidoptera and coleopteran, and fungi against sucking pests.
c. Tools and technology: A particular pest can be controlled by certain options, but they may not all be available in a particular place, for a particular crop, or within the available financial means. For example, the release of natural enemies may be possible in high-value speciality crops, but not in large acreage field crops. A particular pesticide might be registered against a pest on some crops, but not on all. Use of netting or tractor-mounted vacuums can be effective, but very expensive limiting their availability to those who can afford.
This is a critical component where diagnostic and preventive or curative decisions are made based on available and affordable control options.
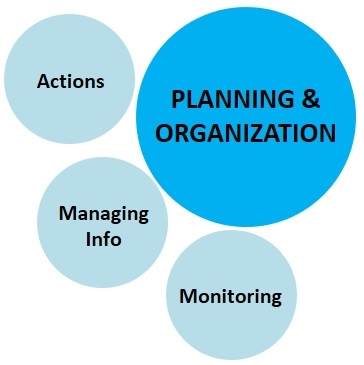
3. Planning and Organization:
This component deals with the management aspect of the of the new IPM model for data collection, organization, and actual actions against pest infestations.
a. Pest monitoring: Regularly monitoring the fields for pest infestation and spread is a basic step in crop protection. Early detection in many cases can help address the pest situation by low-cost spot treatment or removal of pests or infected/infested plant material. When pest infestations continue to grow, regular monitoring is necessary to assess the damage and determine the time to initiate farm-wide control. Monitoring is also important to avoid calendar-based pesticide applications especially at lower pest populations that do not warrant treatments.
b. Managing information: A good recordkeeping about pests, their damage, effective treatments, seasonal fluctuations, interactions with environmental factors, irrigation practices, plant nutrition, and all related information from year to year will build the institutional knowledge and prepares the grower to take preventive or curative actions.
c. Corrective actions: Taking timely action is probably the most important aspect of IPM. Even with all the knowledge about the pest and availability of resources for its effective management, losses can be prevented only when corrective actions are taken at the right time. Good farm management will allow the grower to take timely actions. These actions are not only necessary to prevent damage on a particular farm, but also to prevent the spread to neighboring farms. When pest management is neglected, it leads to area-wide problems with larger regulatory, social, and economic implications.
4. Communication:
Good communication to transfer the individual or collective knowledge for the benefit of everyone is the last component of the new IPM model. Modern and traditional communication tools can be used for outreach as university and private researchers develop information about endemic and invasive pests, emerging threats, and new control strategies.
a. Staying informed: Growers and pest control professionals should stay informed about existing and emerging pests and their management options. Science-based information can be obtained by attending extension meetings, webinars, or workshops, reading newsletter, trade, extension, or scientific journal articles, and keeping in touch with researchers and other professionals through various communication channels. Well-informed growers can be well prepared to address pest issues.
b. Communication within the group: Educating farm crew through periodical training or communication will help with all aspects of pest management, proper pesticide handling, ensuring worker safety, and preventing environmental contamination. Knowledgeable field crew will be beneficial for effective implementation of pest management strategies.
c. Communication among growers: Although certain crop production and protection strategies are considered proprietary information, pests do not have boundaries and can spread to multiple fields when they are not effectively managed throughout the region. Sharing knowledge and resources with each other will improve pest control efficacy and benefit the entire grower community.
In addition to these four components with an IPM model, factors that influence profitable, safe, and affordable food production at a larger scale and their implications for global food security should also be included. There are two layers surrounding these four components addressing the business and sustainable aspects of food production.
II. Business Aspect:
Consumers want nutritious, healthy, and tasty produce that is free of pest damage at affordable prices. Growers try to meet this demand by producing food that meets all the consumer needs, while maintaining environmental and human safety and still being able to make a profit. Sellers evaluate the market demand and strategize their sales to satisfy consumers while making their own profit to stay in the business. In an ideal system, consumer, producer, and seller would maintain a harmonious balance of food production and sale. In such a system, food is safe and affordable to everyone, there will be food security all over the world, and both growers and sellers make a good profit with no or minimal risk to the environment in the process of food production. However, this balance is frequently disrupted due to i) consumers' misunderstanding of various food production systems, their demand for perfectly shaped fruits and vegetables at affordable prices or their willingness to pay a premium price for food items that are perceived to be safe, ii) growers trying to find economical ways of producing high quality food while facing with continuous pest problems and other challenges, and iii) sellers trying to market organic food at a higher price as a safer alternative to conventionally produced food. If growers implement good IPM strategies to produce safe food and consumers are aware of this practice and gain confidence in food produced in an IPM system, then sellers would be able to market what informed-consumers demand.
III. Sustainability Aspect:
As mentioned earlier, IPM is an approach to ensure economic viability at both consumer and producer level (seller is always expected to make a profit), environmental safety through a balanced use of all available pest control options, and social acceptability as food is safe and affordable.
While organic food production is generally perceived as safe and sustainable, the following examples can explain why it is not necessarily true. Organic food production is not pesticide-free and some of the pesticides used in an organic system are as harmful to humans and non-target organisms as some chemical pesticides. Certain organically accepted pesticides have toxins or natural chemical molecules that are very similar to those in synthetic pesticides. In fact, some synthetic pesticides are manufactured imitating the pesticidal molecules of natural origin. Mechanical pest control practices such as vacuuming or tilling utilize fossil fuels and indirectly have a negative impact on the environment. For example, diesel-powered tractors are operated for vacuuming western tarnished bug in strawberry 2-3 times or more each week while a pesticide application typically requires the use of tractor once every 7-14 days. To control certain pests, multiple applications of organic pesticides might be necessary with associated costs and risks, while similar pest populations could be controlled by fewer chemical pesticide applications. It is very difficult to manage certain plant diseases and arthropod pests through non-chemical means and inadequate control not only leads to crop losses, but can result in their spread to larger areas making their control even more difficult. Many growers prefer a good IPM-based production to an organic production for the ease of operation and profitability. However, they continue to produce organic food to stay in business.
While middle and upper-class consumers may be willing to pay higher prices for organically produced food, many of the low-income groups in developed and underdeveloped countries cannot afford such food. Organic food production can lead to social inequality and a false sense of wellbeing for those can afford. Food security for the growing world population is necessary through optimizing input costs, minimizing wastage, grower adoption of safe and sustainable practices, and consumer confidence in food produced through such practices. IPM addresses all the economic, environmental, and social aspects and provides safe and affordable food to the consumers and profits to producers and sellers, while maintaining environmental health.
- Author: Surendra K. Dara
- Author: Sumanth S. R. Dara
- Author: Suchitra S. Dara
- Author: Ed Lewis
Eggs, nymphs, and adult silverleaf whitefly on zucchini. Photo by Surendra Dara
A study was conducted in the summer of 2017 to evaluate the efficacy of various chemical, botanical, and microbial pesticides against arthropod pests on zucchini. Zucchini plants initially had a high aphid infestation, but populations gradually declined due to natural control by lady beetle activity. However, heavy silverleaf whitefly (Bemisia tabaci) infestations developed by the time the study was initiated. Other pests that were present during the study period were aphids (possibly melon aphids), the western flower thrips (Frankliniella occidentalis), and the pacific spider mite (Tetranychus pacificus).
Pacific spider mite (egg, male, and females), western flower thrips larva, and unknown aphids on zucchini. Photo by Surendra Dara
Methodology
Experiment was conducted using a randomized complete block design with 10 treatments. Each treatment had two 38” wide and 300' long rows of zucchini replicated four times. Treatments included i) untreated control, ii) Sivanto 200 SL (flupyradifurone) 14 fl oz/ac, iii) Sequoia (sulfoxaflor) 2.5 fl oz/ac, iv) Venerate XC (heat-killed bacterium, Burkholderia rinojensis strain A396) 4 qrt/ac, v) PFR-97 20% WDG (entomopathogenic fungus, Isaria fumosorosea Apopka strain 97) 2 lb/ac, vi) I1800AA (undisclosed botanical extract) 10.3 fl oz/ac, vii) I1800A 12.7 fl oz, viii) I1800A 17.1 fl oz, ix) I1800A 20.5 fl oz, and x) VST-00634LC (based on a peptide in spider venom) 25%. A spray volume of 50 gpa for all treatments except for VST-00634LC, which had 25 gpa. Treatments were applied on 28 August and 4 September, 2017 using a tractor-mounted sprayer with three Teeject 8003vs flat spry nozzles that covered the top and both sides of each bed.
Pest populations were counted before the first spray application and 4 days after each application. On each sampling date, one mid-tier leaf was collected from each of the five randomly selected plants within each plot. A 2-square inch disc was cut out from the middle of each leaf and the number of aphids, eggs and nymphs of silverleaf whitefly, larvae of western flower thrips, and eggs and mobile stages of pacific spider mite were counted under a dissecting microscope. Data were analyzed using Statistix software and Tukey's HSD test was used to separate significant means.
Spraying, sampling, and counting
Results
Efficacy varied among different treatments and for different pests.
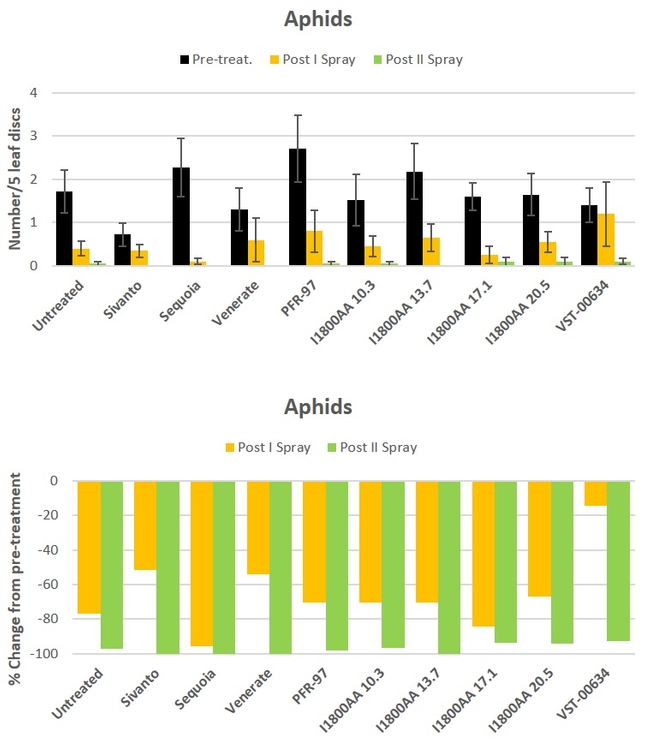
Aphid: There was a general decline in aphid populations during the study period and there was no difference (P > 0.05) among the treatments (Fig. 1).
Fig. 1. Aphid numbers and percent change from pre-treatment counts
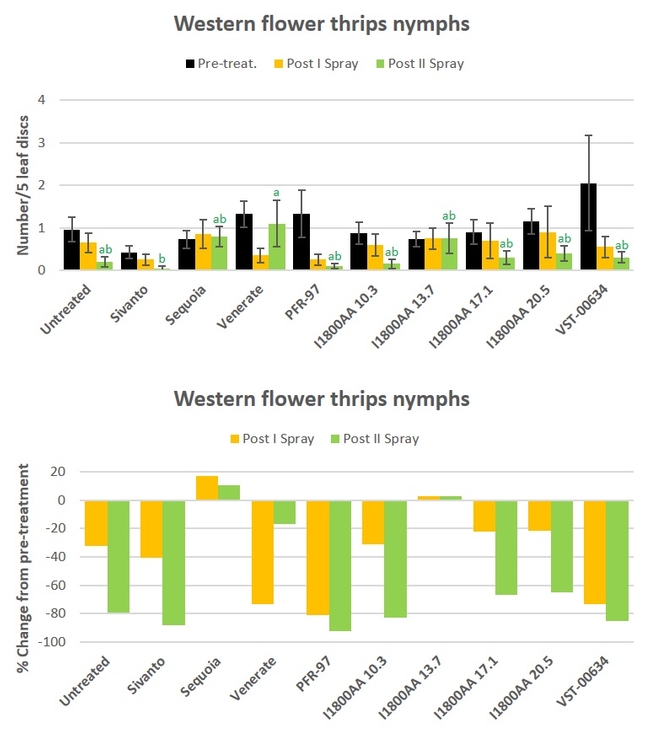
Western flower thrips: Nymphal numbers declined in most of the treatments during the observation period (Fig. 2). However, significant differences (P = 0.0220) only after the second spray application where Sivanto treatment had significantly fewer thrips than Venerate treatment (Fig. 2). There was a 92.5% decline by the end of the study, compared to the pre-treatment counts, from PFR-97 application, followed by 88.1% decline in Sivanto, 85.4% in VST-00634, and 82.9% in I1800AA at 10.3 fl oz.
Fig. 2. Western flower thrips larvae and percent change from pre-treatment counts
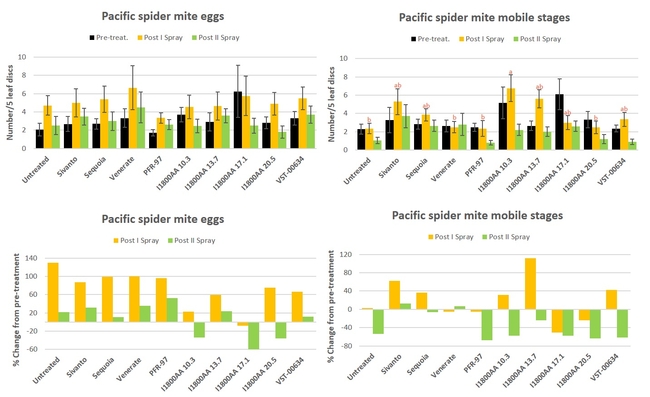
Pacific spider mite: There was an increase in mite eggs in all treatments after the first spray application followed by a decline after the second one without significant differences (P > 0.05) (Fig. 3) Similar trend was also seen in mobile stages in some treatments. Number of mobile stages was significantly different (P = 0.0025) only after the first spray where untreated control, PFR-97, Venerate, and I1800AA at 20.5 fl oz had the lowest. When percent change in egg numbers from the pre-treatment counts, only I1800AA treatments reduced egg numbers after the second spray with a 33.8% decline at 10.3 fl oz rate, 35.7% at 20.5 fl oz, and 60% at 17.1 fl oz. There was also a decline in the mobile stages after the second spray with 54.1% reduction in untreated control to 67.7% in PFR-97 treatment.
Fig. 3. Pacific spider mite egg and mobile stages and percent change from pre-treatment counts
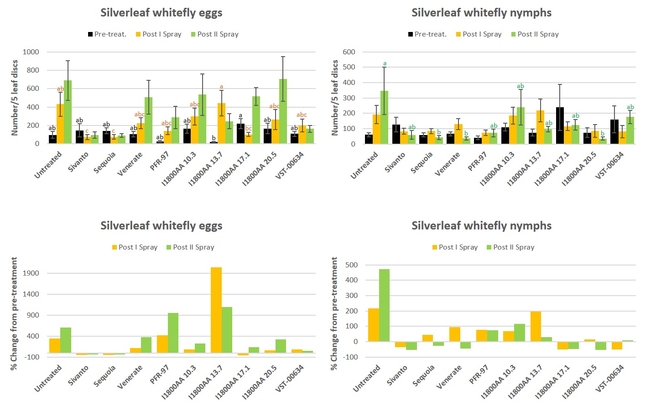
Silverleaf whitefly: There was a general increase in the egg and nymphal stages of whitefly during the study (Fig. 4). Significant differences were seen pre-treatment counts of egg (P = 0.0330) and nymphal stages (P = 0.0011), and after the second spray in nymphal stages (P = 0.0220). Compared to the untreated control, both Sivanto and Sequoia resulted in a significant reduction in egg numbers after the first spray, whereas Sequoia, Venerate, and I1800AA at 20.5 fl oz reduced nymphal stages after the second spray. When the percent change from the pre-treatment counts was compared, only Sivanto and Sequoia reduced whitefly egg numbers after both sprays. There was also a reduction in eggs after the first spray from I1800AA at 17.1 fl oz. However, there was a reduction in nymphal stages after the first spray in Sivanto, I1800AA at 17.1 fl oz, and VST-00634, and after the second spray in Sivanto, Sequoia, Venerate, and I1800AA at 17.1 and 20.5 fl oz.
Fig. 4. Silverleaf whitefly egg and nymphal stages and percent change from pre-treatment counts
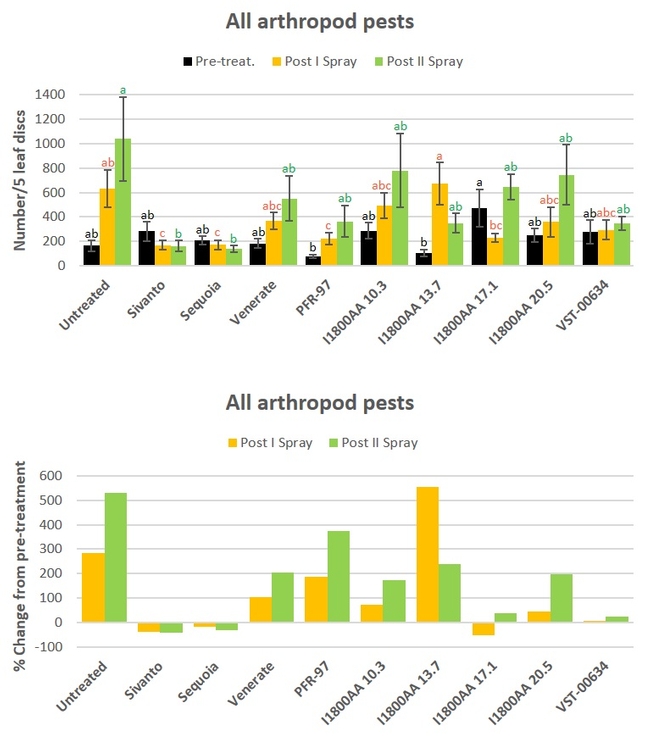
All arthropod pests: When all data were combined for different pests and their life stages, Sivato, Sequia, and PF-97 resulted in a significant (P = 0.0001) decline in pest numbers compared to untreated control after the first spray. Only Sivanto and Sequoia caused such a reduction (P = 0.0048) after the second spray.
Fig. 5. All arthropod pest numbers and percent change from pre-treatment counts
In general, both the chemical pesticides (Sivanto and Sequoia) provided a very good pest control. The efficacy of the botanical extract was moderate to good depending on the pest, life stage, or the application date. Spider venom-based product also provided a good control while microbial products had a moderate impact. Although chemical pesticides appeared to be very efficacious, non-chemical alternatives were also effective. It is important to consider all these options to apply in combinations or rotations to obtain desired pest suppression without posing the risk of insecticide resistance.
Acknowledgements: Thanks for the financial support of Arysta LifeScience, CertisUSA, Dow AgroSciences, and Vestaron, and the technical assistance of Neal Hudson.
- Author: Melissa O'Neal, Marrone Bio Innovations
- Author: Surendra K. Dara
Biopesticide refers to a pesticide which originates from animals, microorganisms, or plants. In addition to preventing yield losses through pest and disease control, biopesticides improve environmental and human health by contributing to the reduction of chemical pesticides as well as by improving the quality of produce (Popp et al., 2012). Additionally, these products have the potential to improve harvest and shipping flexibility, assist with environmental stewardship, and assist growers to achieve sustainability goals. Biopesticides are also important tools in integrated pest management (IPM) programs and reducing the risk of resistance to chemical pesticides (Pretty and Bharucha, 2015), improving worker safety through short restricted entry intervals (Valland, n.d.), conserving natural enemies, and maintaining environmental health (EPA, 2017a).
Biopesticides are inherently less toxic than conventional pesticides. Most affect only the target pest and closely related organisms, in contrast to broad spectrum conventional pesticides that may affect nontarget organisms such as beneficial insects, birds, wildlife, aquatic animals, and mammals. The majority of biopesticides often rapidly decompose, resulting in decreased exposure as well as preventing many pollution problems commonly associated with conventional pesticides. Although relatively safer than chemical pesticides, users or applicators should follow safety guidelines and wear personal protective equipment according to the label directions (EPA, 2017a). It is also important to follow guidelines for spray volume, application rates, droplet size, water pH, compatibility with tank-mix partners, time and frequency of application, and other details to ensure efficacy of the biopesticides (van Zyl et al., 2010; Wang & Liu, 2007; Whitford et al., 2009).
Biopesticides use has been increasing in the recent years. They can be used as standalone treatments or combined or rotated with other pesticides in both organic and conventional production systems. The fact that there are no residues is a huge benefit for exported commodities, as maximum residue limit issues continue to be a challenge in this arena (Berger, 2013).
In expanding upon the role of biopesticides in biocontrol, the topic of resistance management is a key consideration. Pest resistance to conventional chemical pesticides is a significant concern. Scientific research has repeatedly demonstrated that continuous use of the same class of pesticides, especially those reliant on a single mode of action, will result in the emergence of a pest population resistant to those products (Osteen et al., 2012). Populations of insect pests, plant pathogens, nematodes, and weeds all have the ability to develop resistance quickly, even to different types of functionally similar chemistries. This phenomenon is called cross-resistance and is caused by multi-chemistry detoxification mechanisms present in many pest populations (Horowitz and Ishaaya, 2009).
Because of the increasing number of novel, low-impact chemistries available, educators and growers have additional tools to manage resistance within IPM programs (EPA, 2017a). Biopesticides have long been used in combination with synthetic chemistries to provide the basis for excellent control programs that effectively manage resistance. Additionally, they typically have modes of action that are different from synthetic pesticides and do not rely on a single target site for efficacy. Properly used, these products have the potential to extend the effective field life of all products by curtailing the development of resistant pest populations (Horowitz and Ishaaya, 2009).
According to the United States Environmental Protection Agency (EPA), “IPM is an effective and environmentally sensitive approach to pest management that relies on a combination of common-sense practices” (2017b, p. 1). The University of California Statewide Integrated Pest Management Program (UCIPM) (2017) defines the IPM approach as combining prevention, cultural, physical, biological and chemical means to control pests, all the while minimizing economic, public health, beneficial as well as non-target organism, and environmental risks. Biopesticides are noted among the low-risk and most highly effective tools for achieving crop protection in IPM systems. The challenges of farming require that IPM systems actively integrate multiple management approaches to balance optima productivity with sustainability (BPIA, 2017).
Biopesticides should be considered as a component of a holistic total program and used at an appropriate time and pest density. Today, many forward looking IPM professionals are incorporating biopesticides into traditionally conventional pest management strategies (EPA, 2017b). However, education and training are needed to address biopesticide best use practices, the methods of integrating them into IPM programs; as well as instruction to promote an understanding of their unique modes of action (EPA, 2017b). Part of the educational process involves research through fair and realistic field trials that evaluate biopesticides both as standalone treatments as well as in combination and rotation with other options with an objective of improving IPM practices (Abler et al., 1992; Kumar and Singh, 2015). All of these learning experiences are useful in demonstrating the science of biopesticide use and establishing best use practices. A better understanding of biopesticide potential and the mode of action of different active ingredients, increased grant support to promote biopesticide research, and productive grower-industry-researcher collaborations to generate applied research data and design IPM strategies are necessary to make the best use of biopesticides and for environmental sustainability.
References
Abler, D.G., G.P. Rauniyar, and F.M. Goode. 1992. Field trials as an extension technique: The case of Swaziland. NJARE 21(1): 30-35.
Berger, L. 2013. MRL issues and international trade commodity perspectives, pp 3-48. In Proceedings: Idaho Pesticide MRL Workshop, 2 December 2013, Boise, ID. AgBusiness Resources, Visalia, CA.
(BPIA). Biological Products Industry Alliance. 2014. Biopesticides in a program with traditional chemicals offer growers sustainable solutions. http://www.bpia.org/wp-content/uploads/2014/01/grower-final.pdf
(BPIA). Biological Products Industry Alliance. 2017. Benefits of biological products. http://www.bpia.org/benefits-of-biological-products/
(EPA). U.S. Environmental Protection Agency. 2017a. Biopesticides. https://www.epa.gov/pesticides/biopesticides#what
(EPA). U.S. Environmental Protection Agency. 2017b. Integrated pest management principles. https://www.epa.gov/safepestcontrol/integrated-pest-management-ipm-principles
Horowitz, A. and I. Ishaaya. (2009). Biorational control of arthropod pests: Application and resistance management. Springer, New York, NY.
Kumar, S., and A. Singh. 2015. Biopesticides: Present status and the future prospects. J Fertil Pestic 6: e129. doi:10.4172/2471-2728.1000e129
Osteen, C., J. Gottlieb, and U. Vasavada (eds.). 2012. Agricultural Resources and Environmental Indicators. EIB-98, U.S. Department of Agriculture, Economic Research Service, August 2012.
Popp, J., K. Peto, and J. Nagy. 2012. Pesticide productivity and food security: A review. Agron Sustain Dev 33: 243–255. DOI 10.1007/s13593-012-0105-x.
Pretty, J. and Z.P. Bharucha. 2015. Integrated pest management for sustainable intensification of agriculture in Asia and Africa. Insects 6(1): 152–182.
(UCIPM). University of California Statewide Integrated Pest Management Program. 2017. What is integrated pest management (IPM)? http://www2.ipm.ucanr.edu/WhatIsIPM/
Vallad, G.E. n.d. Use of biopesticides for the management of vegetable diseases. University of Florida Gulf Coast Research and Extension Center. http://ipm.ifas.ufl.edu/pdfs/Bio-Pesticides_Slides_IPM_site.pdf
van Zyl, S.A., J. Brink, F.J. Calitz, S. Coertze, and P.J. Fourie. 2009. The use of adjuvants to improve spray deposition and Botrytis cinerea control on Chardonnay grapevine leaves. Crop Prot 29(1): 58-67. https://doi.org/10.1016/j.cropro.2009.08.012
Wang, C.J. and Z.Q. Liu. 2007. Foliar uptake of pesticides: Present status and future challenge. Pest Biochem Phys 87(1): 1-8. https://doi.org/10.1016/j.pestbp.2006.04.004
Whitford, F., D. Penner, B. Johnson, L. Bledsoe, N. Wagoner, et al. 2009. The impact of water quality on pesticide performance. Purdue Extension Publication PPP-86. https://www.extension.purdue.edu/extmedia/ppp/ppp-86.pdf
- Author: Surendra K. Dara
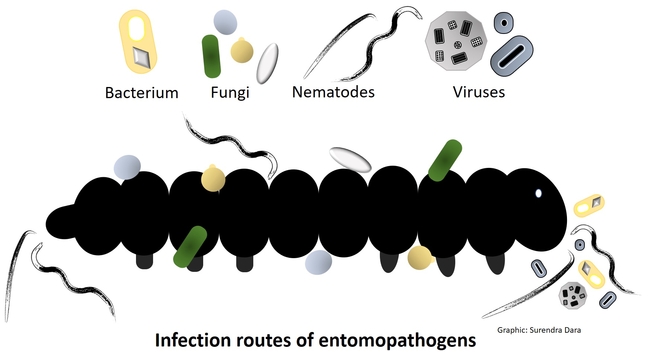
Entomopathogens are microorganisms that are pathogenic to arthropods such as insects, mites, and ticks. Several species of naturally occurring bacteria, fungi, nematodes, and viruses infect a variety of arthropod pests and play an important role in their management. Some entomopathogens are mass-produced in vitro (bacteria, fungi, and nematodes) or in vivo (nematodes and viruses) and sold commercially. In some cases, they are also produced on small scale for non-commercial local use. Using entomopathogens as biopesticides in pest management is called microbial control, which can be a critical part of integrated pest management (IPM) against several pests.
Some entomopathogens have been or are being used in a classical microbial control approach where exotic microorganisms are imported and released for managing invasive pests for long-term control. The release of exotic microorganisms is highly regulated and is done by government agencies only after extensive and rigorous tests. In contrast, commercially available entomopathogens are released through inundative application methods as biopesticides and are commonly used by farmers, government agencies, and homeowners. Understanding the mode of action, ecological adaptations, host range, and dynamics of pathogen-arthropod-plant interactions is essential for successfully utilizing entomopathogen-based biopesticides for pest management in agriculture, horticulture, orchard, landscape, turf grass, and urban environments.
Entomopathogen groups
Important entomopathogen groups and the modes of their infection process are described below.
Bacteria
There are spore-forming bacterial entomopathogens such as Bacillus spp., Paenibacillus spp., and Clostridium spp, and non-spore-forming ones that belong to the genera Pseudomonas, Serratia, Yersinia, Photorhabdus, and Xenorhabdus. Infection occurs when bacteria are ingested by susceptible insect hosts. Pseudomonas, Serratia and Yersinia are not registered in the USA for insect control.Several species of the soilborne bacteria, Bacillus and Paenibacillus are pathogenic to coleopteran, dipteran, and lepidopteran insects. Bacillus thuringiensis subsp. aizawai, Bt subsp. kurstaki, Bt subsp. israelensis, Bt subsp. sphaericus, and Bt subsp. tenebrionis are effectively used for controlling different groups of target insects. For example, Bt subsp. aizawai and Bt subsp. kurstaki are effective against caterpillars, Bt subsp. israelensis and Bt subsp. sphaericus target mosquito larvae, and Bt subsp. tenebrionis is effective against some coleopterans.
When Bt is ingested, alkaline conditions in the insect gut (pH 8-11) activate the toxic protein (delta-endotoxin) that attaches to the receptors sites in the midgut and creates pore in midgut cells. This leads to the loss of osmoregulation, midgut paralysis, and cell lysis. Contents of the gut leak into insect's body cavity (hemocoel) and the blood (hemolymph) leaks into the gut disrupting the pH balance. Bacteria that enter body cavity cause septicemia and eventual death of the host insect. Insects show different kinds of responses to Bt toxins depending on the crystal proteins (delta-endotoxin), receptor sites, production of other toxins (exotoxins), and requirement of spore. The type responses below are based on the susceptibility of caterpillars to Bt toxins.
Type I response – Midgut paralysis occurs within a few minutes after delta-endotoxin is ingested. Symptoms include cessation of feeding, increase in hemolymph pH, vomiting, diarrhea, and sluggishness. General paralysis and septicemia occur in 24-48 hours resulting in the death of the insect. Examples of insects that show Type I response include silkworm, tomato hornworm, and tobacco hornworm.
Type II response – Midgut paralysis occurs within a few minutes after the ingestion of delta-endotoxin, but there will be no general paralysis. Septicemia occurs within 24-72 hours. Examples include inchworms, alfalfa caterpillar, and cabbage butterfly.
Type III response – Midgut paralysis occurs after delta-endotoxin is ingested followed by cessation of feeding. Insect may move actively as there will be no general paralysis. Mortality occurs in 48-96 hours. Higher mortality occurs if spores are ingested. Insect examples include Mediterranean flour moth, corn earworm, gypsy moth, spruce budworm.
Type IV response – Insects are naturally resistant to infection and older instars are less susceptible than the younger ones. Midgut paralysis occurs after delta-endotoxin is ingested followed by cessation of feeding. Insect may move actively as there will be no general paralysis. Mortality occurs in 72-96 or more hours. Higher mortality occurs if spores are ingested. Cutworms and armyworms are examples for this category.
Unlike caterpillars, the response in mosquitoes is different where upon ingestion of Bt subsp. israelensis delta-endotoxin, the mosquito larva is killed within 20-30 min.
While Bt with its toxic proteins is very effective as a biopesticide against several pests, excessive use can lead to resistance development. Corn earworm, diamondback moth, and tobacco budworm are some of the insects that developed resistance to Bt toxins. Genetic engineering allowed genes that express Bt toxins to be inserted into plants such as corn, cotton, eggplant, potato, and soybean and reduced the need to spray pesticides. However, appropriate management strategies are necessary to reduce insect resistant to Bt toxins in transgenic plants.
Paenibacillus popilliae is commonly used against Japanese beetle larvae and known to cause the milky spore disease. Although Serratia is not registered for use in the USA, a species is registered for use against a pasture insect in New Zealand. In the case of Photorhabdus spp. and Xenorhabdus spp., which live in entomopathogenic nematodes symbiotically, bacteria gain entry into the insect host through nematodes. Biopesticides based on heat-killed Chromobacterium subtsugae and Burkholderia rinojensis are reported to have multiple modes of action and target mite and insect pests of different orders.
Fungi
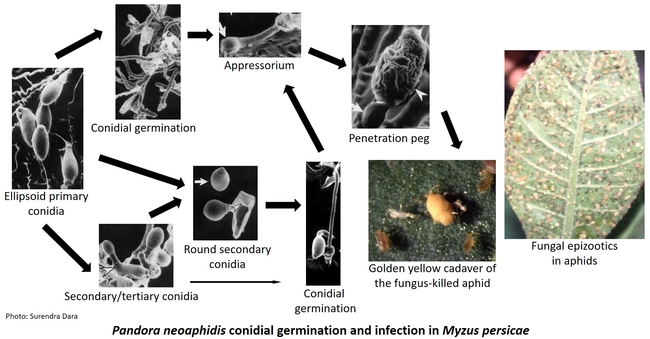
Entomopathogenic fungi typically cause infection when spores come in contact with the arthropod host. Under ideal conditions of moderate temperatures and high relative humidity, fungal spores germinate and breach the insect cuticle through enzymatic degradation and mechanical pressure to gain entry into the insect body. Once inside the body, the fungi multiply, invade the insect tissues, emerge from the dead insect, and produce more spores. Natural epizootics of entomophthoralean fungi such as Entomophaga maimaiga (in gypsy moth), Entomophthora muscae (in flies), Neozygites fresenii (in aphids), N. floridana (in mites), and Pandora neoaphidis (in aphids) are known to cause significant reductions in host populations. Although these fastidious fungi are difficult to culture in artificial media and do not have the potential to be sold as biopesticides they are still important in natural control of some pest species. Hypoclealean fungi such as Beauveria bassiana, Isaria fumosorosea, Hirsutella thompsonii, Lecanicillium lecanii, Metarhizium acridum, M. anisopliae, and M. brunneum, on the other hand, are commercially sold as biopesticides in multiple formulations around the world. Fungal pathogens have a broad host range and are especially suitable for controlling pests that have piercing and sucking mouthparts because spores do not have to be ingested. However, entomopathogenic fungi are also effective against a variety of pests such as wireworms and borers that have chewing mouthparts.
Related to fungi, the spore-forming microsporidium, Paranosema (Nosema) locustae is a pathogen that has been used for controlling locusts, grasshoppers, and some crickets. When P. locustae is ingested, the midgut tissues become infected, followed by infection in the fat body tissues. The disease weakens and eventually kills the orthopteran host within a few weeks.
Various insects killed by different species of entomopathogenic fungi
Nematodes
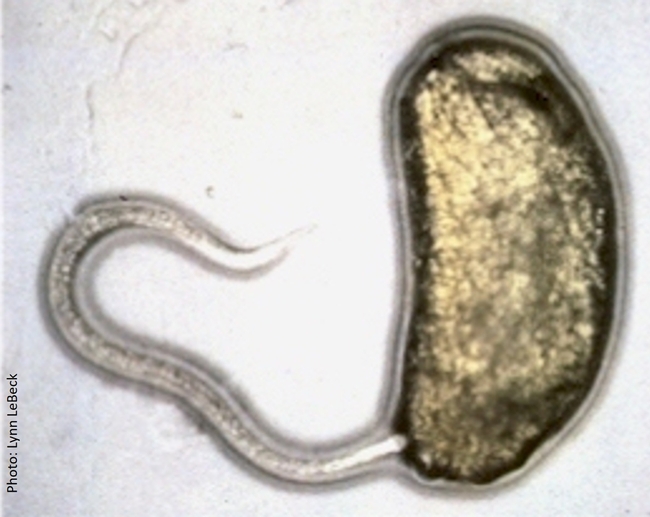
Entomopathogenic nematodes are microscopic, soil-dwelling worms that are parasitic to insects. Several species of Heterorhabditis and Steinernema are available in multiple commercial formulations, primarily for managing soil insect pests. Infective juveniles of entomopathogenic nematodes actively seek out their hosts and enter through natural openings such as the mouth, spiracles, and anus or the intersegmental membrane. Once inside the host body, the nematodes release symbiotic bacteria that kill the host through bacterial septicemia. Heterorhabditis spp. carry Photorhabdus spp. bacteria and Steinernema spp. carry Xenorhabdus spp. bacteria. Phasmarhabditis hermaphrodita is also available for controlling slugs in Europe, but not in the USA.
Infective juvenile of Steinernema carpocapsae entering the first instar larva of a leafminer through its anus.
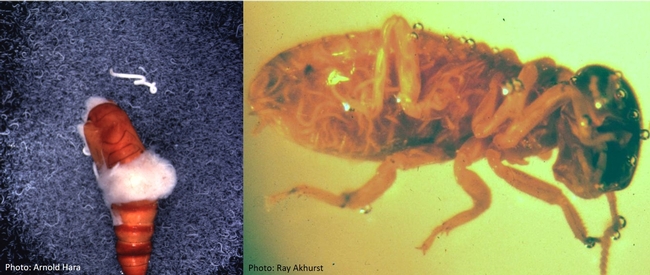
Nematodes in beet armyworm pupa (left) and termite worker (right).
Viruses
Similar to bacteria, entomopathogenic viruses need to be ingested by the insect host and therefore are ideal for controlling pests that have chewing mouthparts. Several lepidopteran pests are important hosts of baculoviruses including nucleopolyhedroviruses (NPV) and granuloviruses (GV). These related viruses have different types of occlusion bodies in which the virus particles (virions) are embedded. Virus particles invade the nucleus of the midgut, fat body or other tissue cells, compromising the integrity of the tissues and liquefying the cadavers. Before death, infected larvae climb higher in the plant canopy, which aids in the dissemination of virus particles from the cadavers to the lower parts of the canopy. This behavior aids in the spread of the virus to cause infection in healthy larvae. Viruses are very host specific and can cause significant reduction of host populations. Examples of some commercially available viruses include Helicoverpa zea single-enveloped nucleopolyhedrovirus (HzSNVP), Spodoptera exigua multi-enveloped nucleopolyhedrovirus (SeMNPV), and Cydia pomonella granulovirus (CpGV).
Most entomopathogens typically take 2-3 days to infect or kill their host except for viruses and P. locustae which take longer. Compared to viruses (highly host specific) and bacteria (moderately host specific), fungi generally have a broader host range and can infect both underground and aboveground pests. Because of the soil-dwelling nature, nematodes are more suitable for managing soil pests or those that have soil inhabiting life stages.
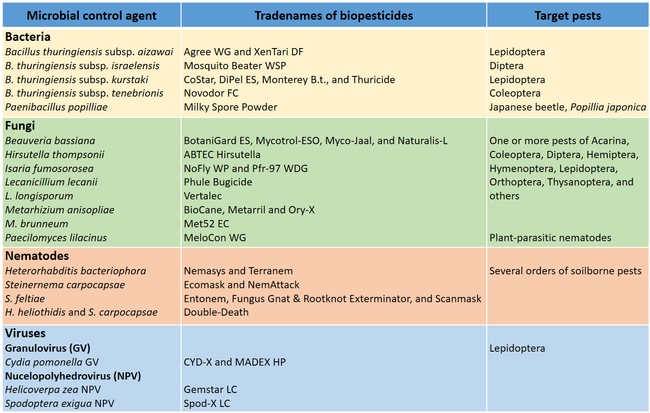
Biopesticides based on various entomopathogenic microorganisms and their target pests
Microbial control and Integrated Pest Management
There are several examples of entomopathogen-based biopesticides that have played a critical role in pest management. Significant reduction in tomato leaf miner, Tuta absoluta, numbers and associated yield loss was achieved by Bt formulations in Spain (Gonzalez-Cabrera et al, 2011). Bt formulations are also recommended for managing a variety of lepidopteran pests on blueberry, grape, and strawberry (Haviland, 2014; Zalom et al, 2014; Bolda and Bettiga, 2014; Varela et al, 2015).
Lecanicellium muscarium-based formulation reducedgreenhouse whitefly (Trialeurodes vaporariorum) populations by 76-96% in Mediterranean greenhouse tomato (Fargues et al, 2005). In other studies, B. bassiana applications resulted in a 93% control of twospotted spider mite (Tetranychus urticae) populations in greenhouse tomato (Chandler et al, 2005) and 60-86% control on different vegetables (Gatarayiha et al, 2010). The combination of B. bassiana and azadirachtin reduced rice root aphid (Rhopalosiphum rufiabdominale) and honeysuckle aphid (Hyadaphis foeniculi) populations by 62% in organic celery in California (Dara, 2015a). Chromobacterium subtsugae and B. rinojensis caused a 29 and 24% reduction, respectively, in the same study. IPM studies in California strawberries also demonstrated the potential of entomopathogenic fungi for managing the western tarnished plant bug (Lygus hesperus) and other insect pests (Dara, 2015b, 2016). Entomopathogenic fungi also have a positive effect on promoting drought tolerance or plant growth as seen in cabbage (Dara et al, 2016) and strawberry (Dara, 2013) and antagonizing plant pathogens (Dara et al, 2017)
Application of SeMNPV was as efficacious as methomyl and permithrin in reducing beet armyworms (S. exigua) in head lettuce in California (Gelernter et al, 1986). Several studies demonstrated PhopGV as an important tool for managing the potato tubermoth (Phthorimaea operculella) (Lacey and Kroschel, 2009).
The entomopathogenic nematode, S. feltiae,reduced raspberry crown borer (Pennisetia marginata) populations by 33-67% (Capinera et al, 1986). For managing the branch and twig borer (Melagus confertus) in California grapes, S. carpocapsae is one of the recommended options (Valera et al, 2015).
Entomopathogens can be important tools in IPM strategies in both organic and conventional production systems. Depending on the crop, pest, and environmental conditions, entomopathogens can be used alone or in combination with chemical, botanical pesticides or other entomopathogens.
Acknowledgements: Thanks to Dr. Harry Kaya for reviewing this article.
References
Bolda, M. P. and L. J. Bettiga. 2015. UC IPM Pest Management Guidelines: Caneberries. UC ANR Pub. 3437.
Capinera, J. L., W. S. Cranshaw, and H. G. Hughes. 1986. Suppression of raspberry crown borer Pennisetia marginata (Harris) (Lepidoptera: Sesiidae) with soil applications of Steinernema feltiae (Rhabditida:Steinernematidae). J. Invertebr. Pathol. 48: 257-258.
Chanlder, D., G. Davidson, and R. J. Jacobson. 2005. Laboratory and glasshouse evaluation of entomopathogenic fungi angainst the two-spotted spider mite, Tetranychus urticae (Acari: Tetranychidae), on tomato, Lycopersicon esculentum. Biocon. Sci. Tech. 15: 37-54.
Dara, S. K. 2013. Entomopathogenic fungus Beauveria bassiana promotes strawberry plant growth and health. UCANR eJournal Strawberries and Vegetables, 30 September, 2013. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11624)
Dara, S. K. 2015a. Reporting the occurrence of rice root aphid and honeysuckle aphid and their management in organic celery. UCANR eJournal Strawberries and Vegetables, 21 August, 2015. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=18740)
Dara, S. K. 2015b. Integrating chemical and non-chemical solutions for managing lygus bug in California strawberries. CAPCA Adviser 18 (1) 40-44.
Dara, S. K. 2016. IPM solutions for insect pests in California strawberries: efficacy of botanical, chemical, mechanical, and microbial options. CAPCA Adviser 19 (2): 40-46.
Dara, S. K., S.S.R. Dara, and S.S. Dara. 2016. First report of entomopathogenic fungi, Beauveria bassiana, Isaria fumosorosea, and Metarhizium brunneum promoting the growth and health of cabbage plants growing under water stress. UCANR eJournal Strawberries and Vegetables, 19 September, 2016. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=22131)
Dara, S.S.R., S. S. Dara, S. K. Dara, and T. Anderson. 2017. Fighting plant pathogenic fungi with entomopathogenic fungi and other biologicals. CAPCA Adviser 20 (1): 40-44.
Fargues, J., N. Smits, M. Rougier, T. Boulard, G. Rdray, J. Lagier, B. Jeannequin, H. Fatnassi, and M. Mermier. 2005. Effect of microclimate heterogeneity and ventilation system on entomopathogenic hyphomycete infectiton of Trialeurodes vaporariorum (Homoptera: Aleyrodidae) in Mediterranean greenhouse tomato. Biological Control 32: 461-472.
Gatarayiha, M. C., M. D. Laing, and M. Ray. 2010. Effects of adjuvant and conidial concentration on the efficacy of Beauveria bassiana for the control of the two-spotted spider mite, Tetranychus urticae. Exp. Appl. Acarol. 50: 217-229.
Gelernter, W. D., N. C. Toscano, K. Kido, and B. A. Federici. 1986. Comparison of a nuclear polyhedrosis virus and chemical insecticides for control of the beet armyworm (Lepidopter: Noctuidae) on head lettuce. J. Econ. Entomol. 79: 714-717.
González-Cabrera, J., J. Mollá, H. Monton, A. Urbaneja. 2011. Efficacy of Bacillus thuringiensis (Berliner) in controlling the tomato borer, Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). BioControl 56: 71–80.
Haviland, D. R. 2014. UC IPM Pest Management Guidelines: Blueberry. UC ANR Pub. 3542.
Lacey, L. A. and J. Kroschel. 2009. Microbial control of the potato tuber moth (Lepidoptera: Gelechiidae). Fruit Veg. Cereal Sci. Biotechnol. 3: 46-54.
Varela, L. G., D. R. Haviland, W. J., Bentley, F. G. Zalom, L. J. Bettiga, R. J. Smith, and K. M. Daane. 2015. UC IPM Pest Management Guidelines: Grape. UC ANR Pub. 3448.
Zalom, F. G., M. P. Bolda, S. K. Dara, and S. Joseph. 2014. UC IPM Pest Management Guidelines: Strawberry. UC ANR Pub. 3468.
- Author: Surendra K. Dara
Seal of IPM - a practical and sustainable crop production system
Arthropod pests or diseases cause a variety of damages to crops. Some by reducing plant vigor resulting in lesser yields and some by causing direct damage to the produce which can be unmarketable due to deformity, unpleasant taste, damaged tissue due to insect feeding, presence of insects and/or frass, decay due to secondary infections, and other factors. It is quite understandable when the produce is not accepted because of the taste or potential health risk. For example, citrus fruit with huanglongbing or citrus greening disease transmitted by Asian citrus psyllid gives a bitter taste to citrus juice. Navel orangeworm larvae bore into almonds and feed on the nut causing complete or partial damage and leave frass and cause fungal infections. Brown marmorated stink bug damage on fruits and vegetables change the texture and taste of the damaged area. Such damage certainly makes the produce unmarketable and applying pesticides or administering other control measures to prevent the damage is warranted.
Brown marmorated stink bug damage to apple (above - Photo by Chris Bergh, Virginia Tech) and navel orangeworm damage to almond (below - Photo by Jack Kelly Clark, UC IPM)
On the other hand, certain damage is only cosmetic with no reported change in taste or quality of the produce. One example would be fruit deformity caused by the lygus bug in strawberries. Strawberry is a high value fruit appreciated for its taste, shape, color, and flavor. Lygus bug feeding on young green berries results in uneven growth and deformity of mature berries. While there is no record of the impact of lygus damage on strawberry fruit quality, millions of pounds of pesticides are applied to control lygus bug or similar pests that cause cosmetic damage in strawberries and other crops.
Cosmetic damage to strawberry by lygus bug (Photo by Surendra Dara)
The preference of consumers for perfectly shaped fruits and vegetables creates a need for intensive pest management practices and results in associated financial and environmental costs. Since chemical pesticides are generally economical and effective tools to manage pests, they are widely used. The overuse of certain effective pesticides causes development of resistance in pest populations. This, in turn, leads to increased use of the same or other pesticides. Excessive use of chemical pesticides can have a harmful effect on beneficial arthropods resulting in secondary pest outbreaks. Organic agriculture is gaining popularity due to environmental and human health concerns from chemical pesticide use. “Organic agriculture produces products using methods that preserve the environment and avoid most synthetic materials, such as pesticides and antibiotics” according to USDA. But organic agriculture is not necessarily the only sustainable solution.
Before agricultural industrialization, there was a better balance between pests and their natural enemies (beneficial arthropods such as predators and parasitoids that attack pests). Once agriculture was industrialized, thousands of acres of monoculture now provide an unlimited supply of food for a variety of pests. When the natural balance is disrupted, natural enemies alone are not sufficient to manage pest populations. This is where an Integrated Pest Management (IPM) strategy plays an important role in bringing a sense of balance into pest management. IPM employs multiple tools that include selecting resistant varieties, modifying planting dates, changing irrigation and nutrient management practices, conserving or releasing natural enemies, applying chemical, botanical, and microbial pesticides, or using mechanical tools. Each of these tools contribute to reducing pest numbers, complement each other, and result in pest management in an environmentally sustainable manner.
Organic agriculture, on the other hand, relies on biopesticides instead of chemical pesticides, which can sometimes be less effective or slow in achieving desired control. For example, an effective chemical pesticide with a specific mode of action could kill pest populations within a few hours of application. However, using a biopesticide based on an insect-pathogenic microorganism like the bacterium Bacillus thuringiensis or the fungus Beauveria bassiana, can take a few days to allow the microorganism to infect and kill the pest. When pest numbers are low, non-chemical solutions may provide required control to minimize damage. However, with heavy pest infestations, chemical pesticides are often needed to provide timely control that prevents further buildup of pest populations and the resulting damage to crops.
Organic agriculture is expensive because of generally higher losses due to pests and higher cost of agronomic and pest management practices. Sometimes, ineffective control of pests on organic farms may result in their spread to neighboring fields and increase the risk of pest damage. Organic agriculture does not mean pesticide-free farming, and biopesticides used on organic farms also require safety guidelines similar to chemical pesticides used on conventional farms. Organic agriculture may require a higher number of pesticide sprays increasing the risk of exposure for workers. In some pest and disease situations in certain crops, organically registered products are not available and yield losses could be higher. Exporting organic produce, in light of exotic and invasive pests spreading to other areas, is also a challenge due to limited options for shipping organically produced pest-free fruits and vegetables.
Using cultural practices to reduce the risk of pest infestations and applying biopesticides when pest populations are low and chemical pesticides when populations are high can be components of an IPM strategy where multiple tools are exploited in a balanced manner. Combining and rotating chemical pesticides with non-chemical alternatives strengthens the effectiveness of IPM by providing desired control without the excessive use of chemicals. Chemical pesticides can be used during early stages of the crop growth while biopesticides can be used closer to harvest.
Considering the challenges and risks associated with organic agriculture and the practicality of IPM-based agriculture, a couple of ideas could be worth pursuing to maintain environmental and human health, reduce harmful chemicals, and ensure food security for the growing world population.
Acceptance of imperfect produce: When consumers are tolerant of imperfectly shaped fruits and vegetables with no health risk from pathogens or arthropod pests, a significant amount of pesticides of all kinds could be avoided. This would translate into saving millions of dollars otherwise spent on pesticides and their application costs, and money earned on selling otherwise unmarketable produce. This may also reduce the disposal of unpicked produce at the grocery stores. When consumers accept imperfect fruits or vegetables, the cost of produce, both to produce and purchase, could come down. I recently came across Imperfect Produce, a company that sells imperfect produce and End Food Waste, an organization that started the Ugly Fruit And Veg Campaign.
IPM: Considering the difficulty in ensuring food security exclusively through non-chemical agriculture for the growing world population (projected to be 9.6 billion by 2050), IPM is an effective, practical, and sustainable tool that uses a balanced approach. While organic agriculture is encouraged and supported, and there are several organizations that certify organic production around the world, IPM hasn't caught the attention of marketers yet. Perhaps a seal of IPM should be considered and promoted in the near future.
Organic certification agencies from around the world. Source organicguidemalaysia.com
Opinions expressed in this article are my own and based on my experience in IPM, microbial control, biological control, and from discussions with several growers and scientists.
http://ucanr.edu/articlefeedback