- (Public Value) UCANR: Promoting economic prosperity in California
- Author: Surendra K. Dara
- Author: Roland C. Bocco
The spotted lanternfly (SLF) [Lycorma delicatula (Hemiptera: Fulgoridae)] is an invasive planthopper, which causes a significant damage to apples, grapes, stone fruit, trees used for timber, and other hosts (Dara et al. 2015). Native to China, SLF was first reported in 2014 in Pennsylvania and has been rapidly spreading in the eastern United States and moving westward. California has 22 cultivated and about 70 wild hosts of SLF and include several high value crops such as apples, cherries, grapes, and plums. The tree-of-heaven, an invasive species, is a favorite host of SLF and is widely distributed in California. SLF is also a nuisance pest with 100s or 1000s of individuals infesting landscape trees and hosts in residential areas. This pest deposits eggs on inanimate objects such as vehicles, furniture, stones, and packages and thus spread to other areas through the movement of these objects. Awareness of the pest and its damage potential, ability of Californians to recognize and report the pest if found, and the knowledge of control practices will help prevent accidental transportation of eggs or other life stages from the infested areas to California and prepare the citizens to take appropriate actions. Outreach efforts have been made in California since 2014 through extension articles, presentations at extension meetings, videos, social media posts, and personal communication (Dara, 2014).
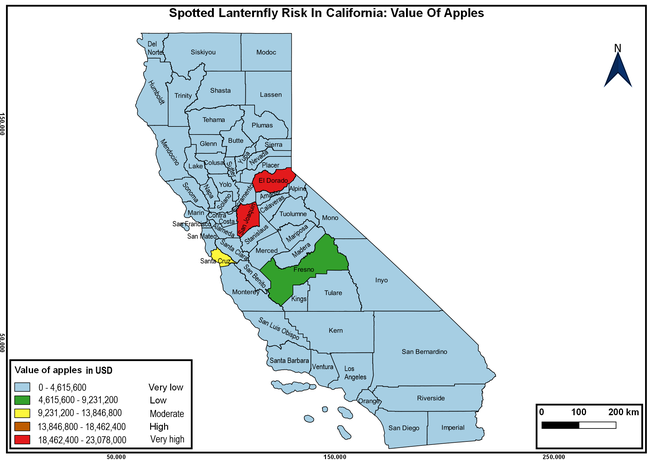
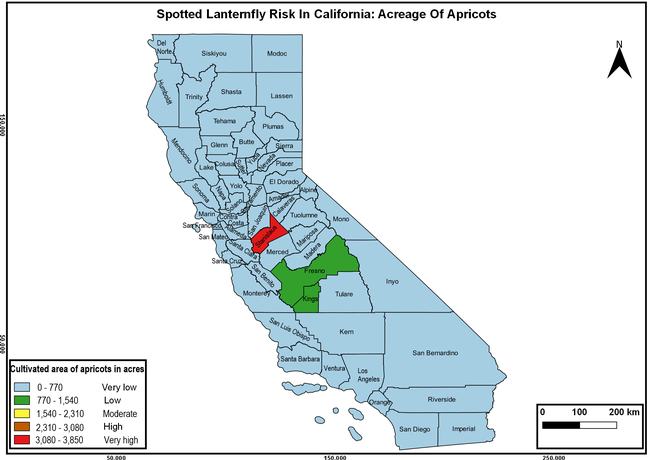
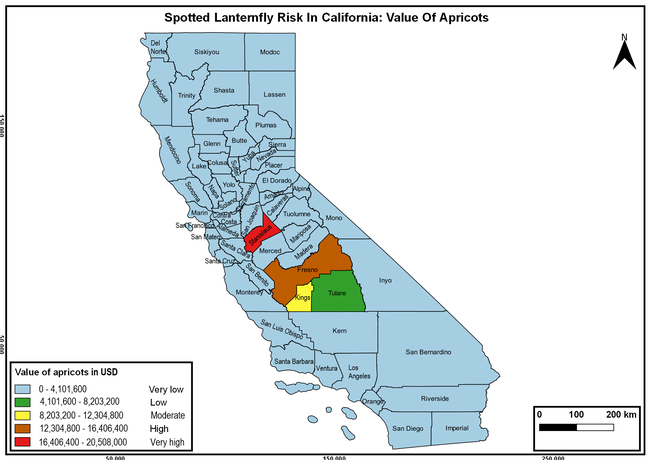
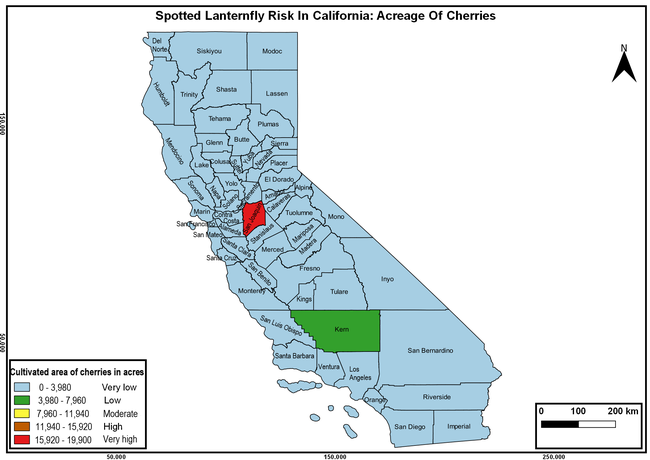
Wakie et al. (2020) modeled the establishment risk of SLF in the United States and around the world and indicated that many coastal regions and the Central Valley of California are among the high-risk areas. Considering the risk to several high-value commodities and the presence of several wild hosts that are distributed all over California, mapping of the risk-prone areas based on the cultivated hosts, their acreage and value in different counties, and the distribution of wild hosts was done to help both growers and other Californians to prepare for potential invasion of SLF.
Methodology
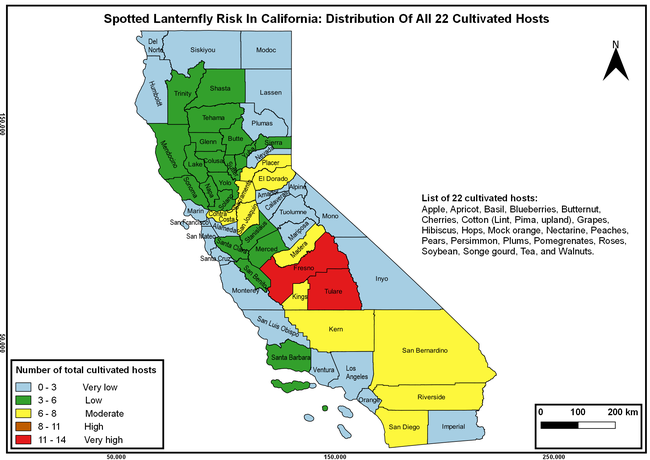
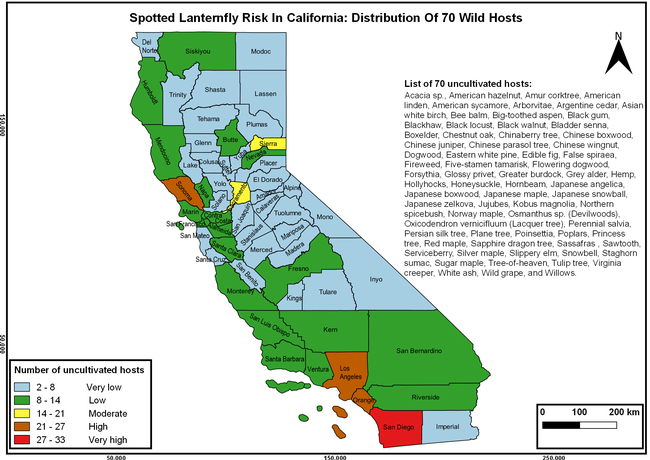
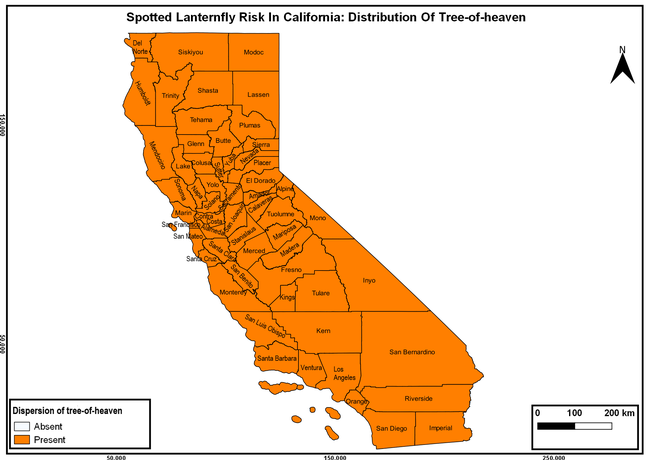
The list of SLF hosts is continuously evolving with host specificity studies in various places. Based on two published resources (Dara et al. 2015; Barringer and Ciafré 2020), 22 cultivated and 70 wild hosts appear to be present in California. Plant species that support some of the feeding life stages or all life stages were included in preparing these lists. The cultivated hosts include apples, apricots, basil, blueberries, butternut, cherries, cotton, grapes, hibiscus, hops, mock orange, nectarine, peaches, pears, persimmon, plums, pomegranates, roses, soybean, sponge gourd, tea, and walnuts; and the wild hosts include Acacia sp., American hazelnut, Amur corktree, American linden, American sycamore, arborvitae, Argentine cedar, Asian white birch, bee balm, big-toothed aspen, black gum, black hawk, black locust, black walnut, Bladder senna, boxelder, chestnut oak, chinaberry tree, Chinese boxwood, Chinese juniper, Chinese parasol tree, Chinese wingnut, devilwoods, dogwood, Eastern white pine, edible fig, false spiraea, fireweed, five-stamen tamarisk, flowering dogwood, Forsythia, Glossy privet, greater burdock, grey alder, hemp, hollyhocks, honeysuckle, hornbeam, Japanese angelica, Japanese boxwood, Japanese maple, Japanese snowball, Japanese zelkova, jujubes, Kobus magnolia, Northern spicebush, Norway maple, lacquer tree, perennial salvia, Persian silk tree, plane tree, Poinsettia, poplars, princess tree, red maple, sapphire dragon tree, sassafras , sawtooth, serviceberry, silver maple, slippery elm, snowbell, staghorn sumac, sugar maple, tree-of-heaven, tulip tree, Virginia creeper, white ash, wild grape, and willows.
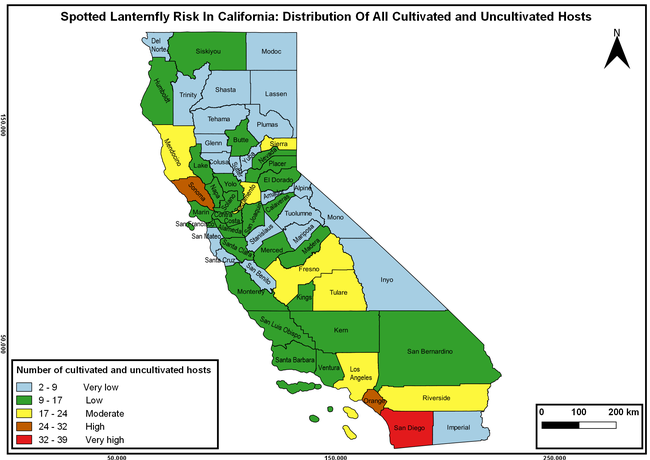
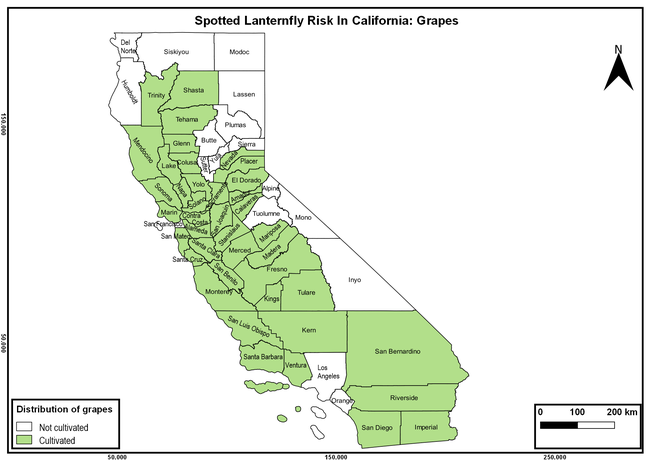
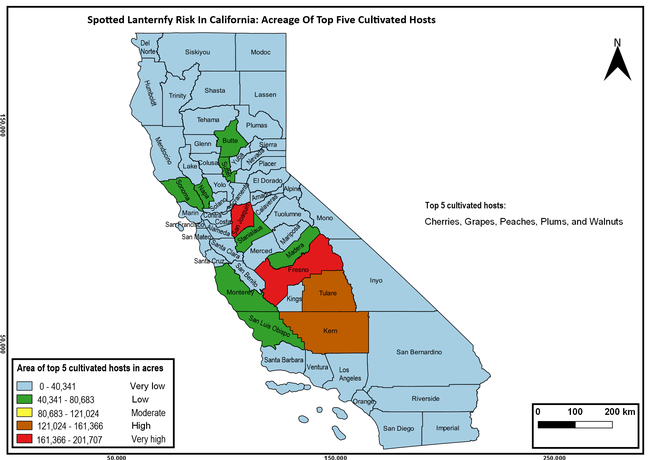
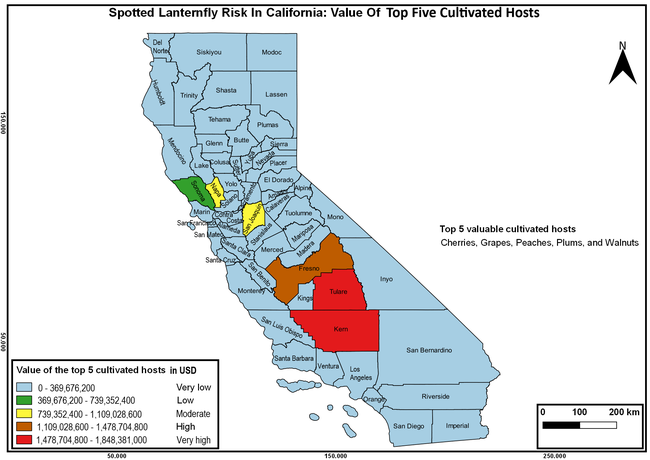
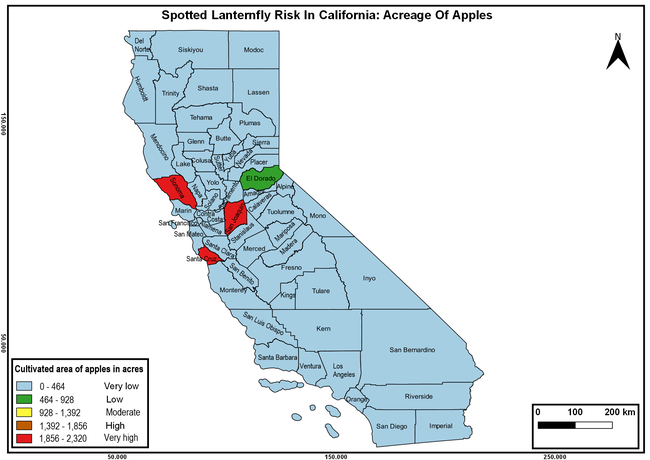
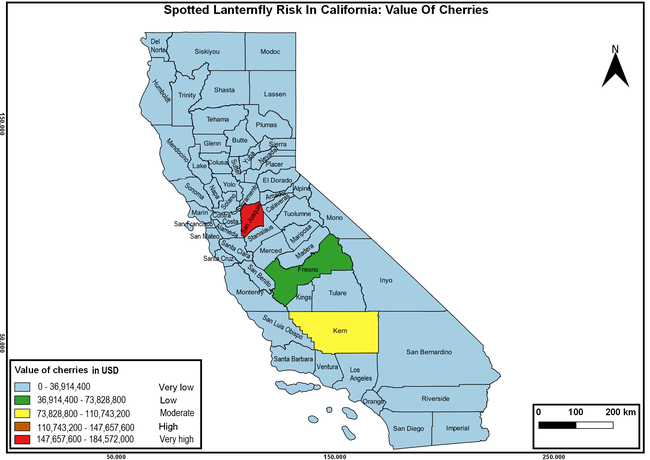
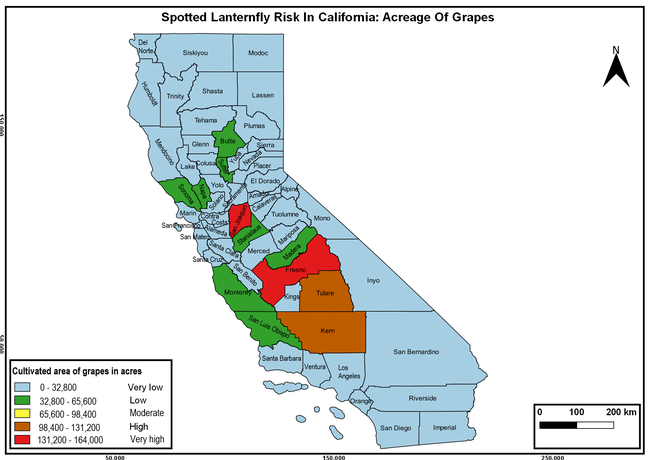
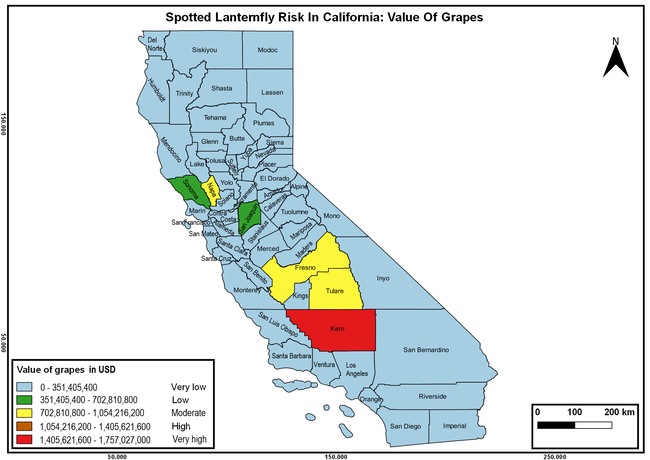
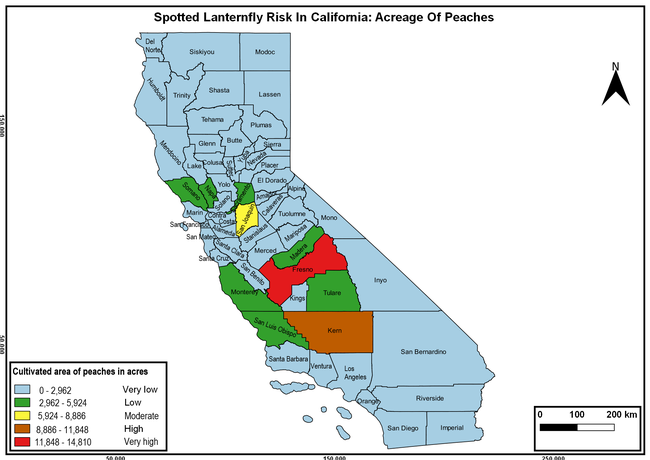
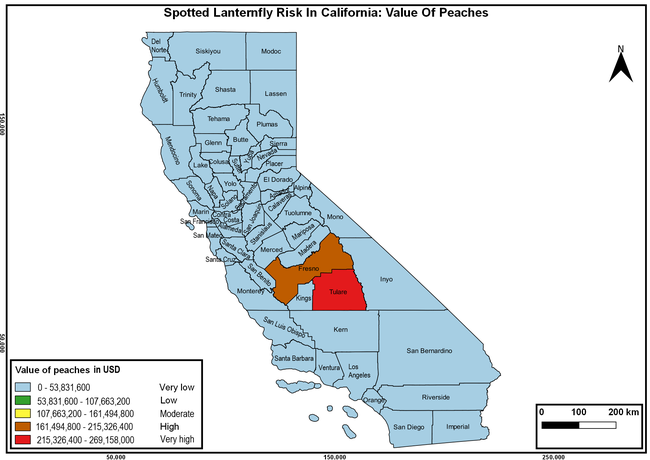
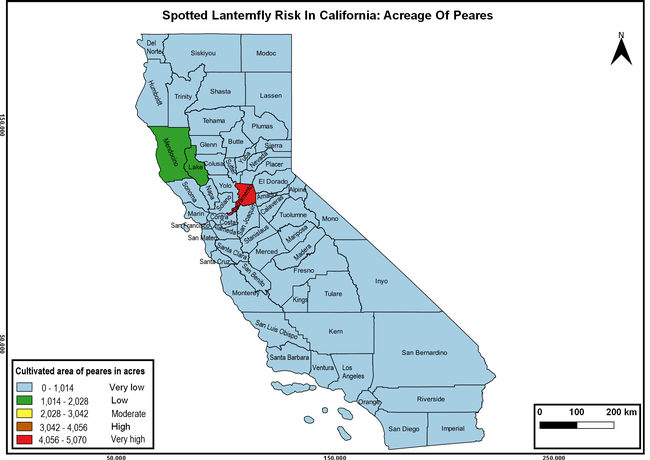
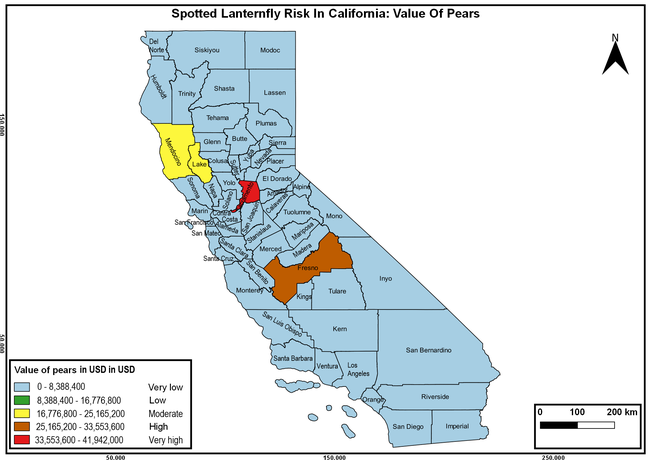
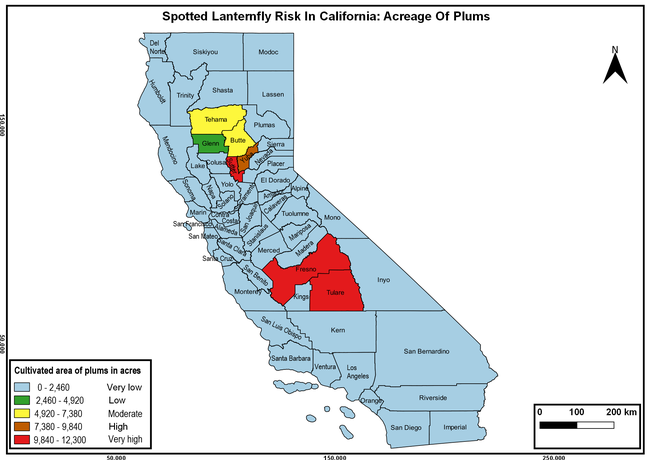
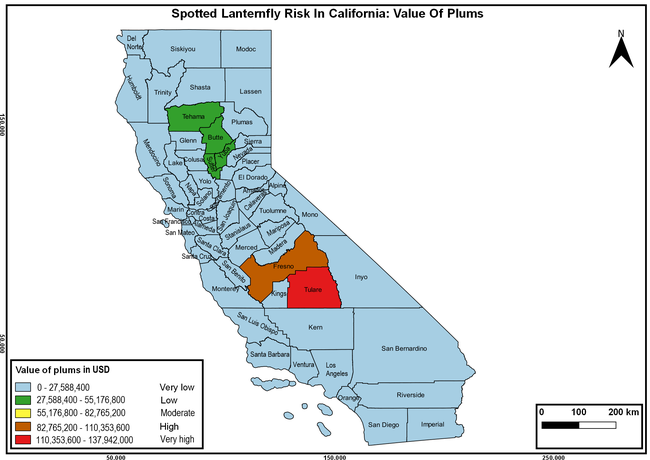
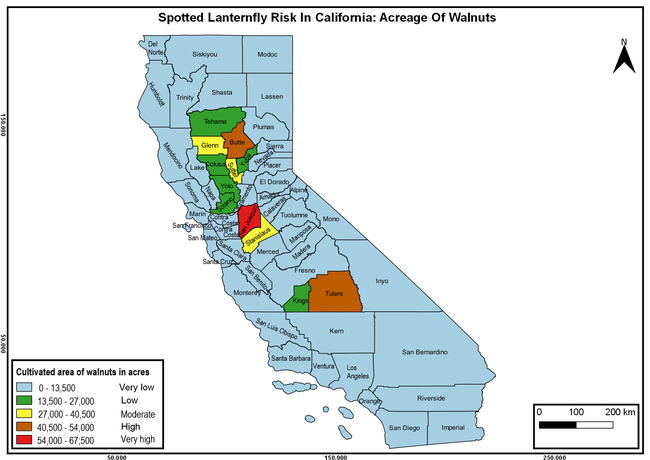
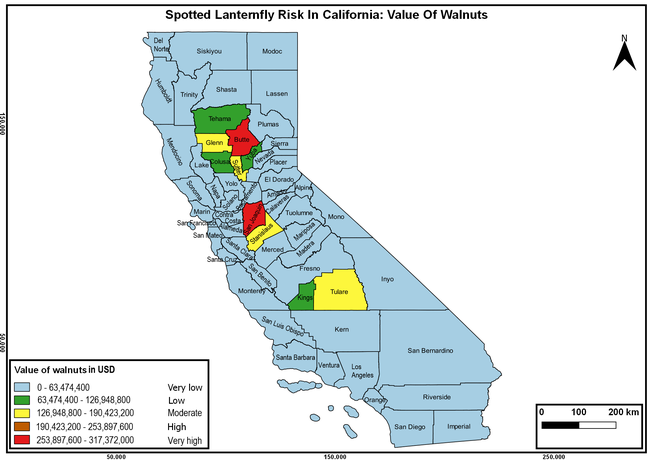
The summary of county crop reports from the California Department of Food and Agriculture (CDFA 2018) was used to determine the value and acreages of the cultivated hosts. To determine the distribution of wild hosts various online resources were used. SLF risk levels were determined as very low, low, moderate, high, and very high for the number of hosts, acreage and value of each cultivated host, and other such parameters within each county. The highest risk value within each parameter was used to determine ‘very high' category and 4/5, 3/5, 2/5, and 1/5 were used for high, moderate, low, and very low categories, respectively. In other words, 0-20% risk was considered very low, 21-40% as low, 41-60% as moderate, 61-80% as high, and 81-100% as very high for each measured parameter. Data were entered into a spreadsheet and maps were generated using QGIS open-source cross-platform geographic information system application.
Risk-prone areas in California
The following maps show areas in California that are prone to SLF risk based on the distribution of cultivated and wild hosts, and the acreage and value of important cultivated crops.
Based on these maps, the entire state of California is at some level of risk. In addition to the commercially produced crops, several backyard or landscape plant species such as roses, grapes, peaches, plums, and others are present throughout the state and can harbor SLF. Such host plants in residential and urban landscapes can serve as SLF sources for commercial crops. The tree-of-heaven is present throughout California and several such uncultivated hosts can serve as sources of undetected infestations. While researchers are working on appropriate biocontrol solutions such as releasing natural enemies, other control options such as synthetic and microbial pesticide applications, sticky traps, removal of egg masses and wild hosts, and other strategies can help manage SLF. In the meantime, Californians will benefit by knowing about this pest and its potential risk to the state. The ability to identify, destroy or capture, and report the pest to county and state departments or University of California Cooperative Extension offices will help prevent or delay SLF invasion and spread in California.
Conclusion
California is at the risk of SLF invasion and spread. Depending on the number of cultivated crops, their acreage, value, and the distribution of wild hosts, the risk level varies in various counties throughout the state. Outreach efforts are helping to alert Californians about SLF and its damage to cultivated crops and nuisance in urban and residential areas.
Additional resources
Refer to https://ucanr.edu/spottedlanternfly for additional information about the pest. If you happen to see this pest in California, please contact your local Agricultural Commissioner, California Department of Food and Agriculture, or UC Cooperative Extension office to report.
Acknowledgments
Thanks to the California Department of Food and Agriculture for funding this study.
References
Barringer, L. and Ciafré, C. M. 2020. Worldwide feeding host plants of spotted lanternfly, with significant additions from North America. Environ. Entomol. 49: 999-1011.
CDFA (California Department of Food and Agriculture). 2018. California County Agricultural Commissioners' Report Crop Year 2016-2017 (https://www.cdfa.ca.gov/statistics/pdfs/2017cropyearcactb00.pdf).
Dara, S. K. 2014. Spotted lanternfly (Lycorma delicatula) is a new invasive pest in the United States. UCANR eJournal Pest News (https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=15861).
Dara, S. K. 2018. An update on the invasive spotted lanternfly, Lycorma delicatula: current distribution, pest detection efforts, and management strategies. UCANR eJournal Pest News (https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=26349).
Dara, S. K., Barringer, L. and Arthurs, S. P. 2015. Lycorma delicatula (Hemiptera: Fulgoridae): a new invasive pest in the United States. J. Integr. Pest Manag. 6: 20.
Wakie, T. T., Nevin, L. G., Yee, W. L. and Lu, Z. 2020. The establishment risk of Lycorma delicatula (Hemiptera: Fulgoridae) in the United States and globally. J. Econ. Entomol. 113: 306-314.
- Author: Surendra K. Dara
The diamondback moth, Plutella xylostella (Lepidoptera: Plutellidae) is an important pest of broccoli, Brussels sprouts, cabbage, cauliflower, collards, kale, and other cruciferous crops. It exclusively feeds on cultivated and weedy crucifers and has a worldwide distribution. Larvae feed on foliage and growing parts of young plants or bore into the heads or flower buds resulting in skeletonization of leaves, stunting of the plants, or failure of head formation in some hosts. In warmer areas, the diamondback moth has up to 12 generations per year. While multiple species of parasitoids and predatory arthropods provide some level of natural control, insecticidal applications are a primary means of diamondback moth management. Although several synthetic and biological pesticides are effective against the diamondback moth, resistance to Bacillus thuringiensis (Ferré et al. 1991), abamectin (Pu et al. 2009), emamectin benzoate, indoxacarb, and spinosad (Zhao et al. 2006), pyrethroids and other pesticides (Leibee and Savage 1992; Endersby et al. 2011) has been well-known from around the world. Excessive use of any kind of pesticide leads to resistance problems (Dara 2020; also see a video presentation) to an individual pesticide or multiple pesticides. Integrated pest management (IPM) strategy encourages the use of various control options both for maintaining pest control efficacy and reducing the risk of resistance development (Dara 2019). Regularly monitoring the pest populations to make treatment decisions, rotating pesticides with different modes of action, exploring the potential of biocontrol agents, and other non-chemical control approaches such as mating disruption with pheromones are some of the IPM strategies for controlling the diamondback moth. While sex pheromones effectively used to manage several lepidopteran pests and are proven to be a critical IPM tool, mating disruption is not fully explored for controlling the diamondback moth. A study was conducted in Brussels sprouts to evaluate the efficacy of a sprayable pheromone against the diamondback moth and to enhance the current IPM strategies.
Methodology
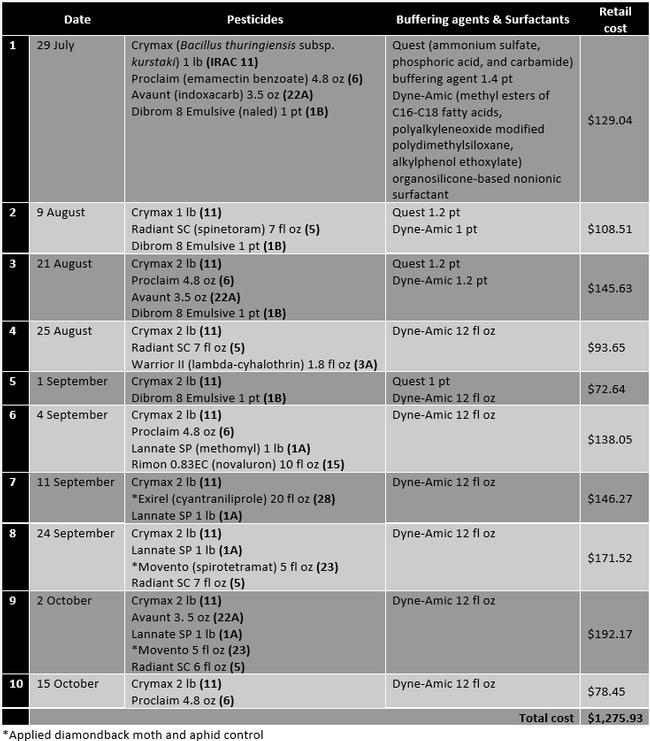
The study was conducted on a 10-acre Brussels sprouts field in Santa Maria. Cultivar Marte was planted in early July 2020 with expected harvesting in mid to late December. A typical diamondback control program includes monitoring diamondback moth populations with the help of sticky traps and lures and applying various combinations of biological and synthetic pesticides at regular intervals. This study evaluated the efficacy of adding CheckMate DBM-F to the grower standard practice of monitoring the diamondback moth populations with traps and lures and applying pesticides. Treatments included i) grower standard pesticide program (Table 1) and ii) grower standard pesticide program with two applications of 3.1 fl oz of CheckMate DBM-F on 9 August and 11 September. Treatment materials were applied by a tractor-mounted sprayer using a 100 gpa spray volume and necessary buffering agents and surfactants. Each treatment was 5 ac and divided into four quadrants representing four replications. In the middle of each quadrant, one Suterra Wing Trap was set up with a Trécé Pherocon Diamondback Moth Lure. Lures were replaced once a month in early September and early October. Sticky liners of the traps were replaced every week to count the number of moths trapped. Traps were placed on 1, 12, and 24 August, 1, 11, 18, and 27 September, and 6 October and the moth counts were taken from respective traps on 8 and 20 August, 1, 11, 18, and 27 September, 6 and 15 October. CheckMate DBM-F was applied at 3.1 fl oz/ac on 9 August and 11 September. The number of larvae and their feeding damage on a scale of 0 to 4 (where 0=no damage, 1=light damage, 2=moderate damage, 3=high damage, 4=extensive/irrecoverable) were recorded from 25 random plants within each replication on 30 August and 6 and 18 October. Data were subjected to analysis of variance using Statistix software and significant means were separated using Tukey's HSD test. The retail value of various pesticides was also obtained to compare the cost of treatments.
Table 1. Pesticides, buffering agents, and surfactants, their active ingredients, rates/ac (along with the IRAC mode of action groups), and retail pricing for those applied in the grower standard diamondback moth control program.
When CheckMate DBM-F [(Z)-11-Hexadecenal (3) , (Z) - 11 - Hexadecen-1-yl Acetate (1)] was applied the first time on 9 August, Dibrom 8 Emulsive was replaced with Warrior II, the buffering agent Quest was not used, and the surfactant Dyne-Amic was replaced with Induce (dimethylpolysiloxane) to avoid potential compatibility issues. The impact of this substitution is expected to be negligible within the scope of this study. The retail cost of 3.1 fl oz CheckMate DBM-F is $45.60. The cost of lures and traps would be about $4-8 per acre for a six-month crop like Brussels sprouts.
Results and Discussion
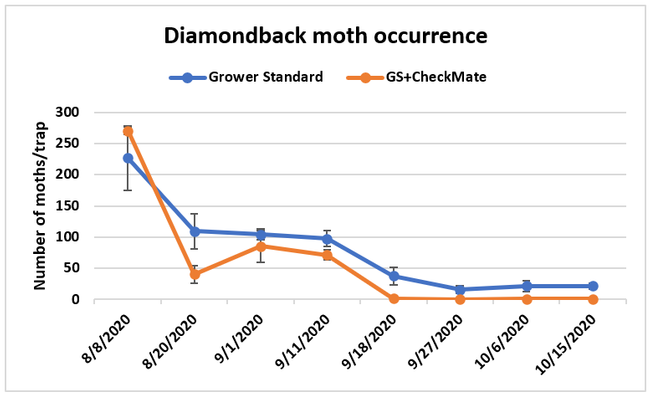
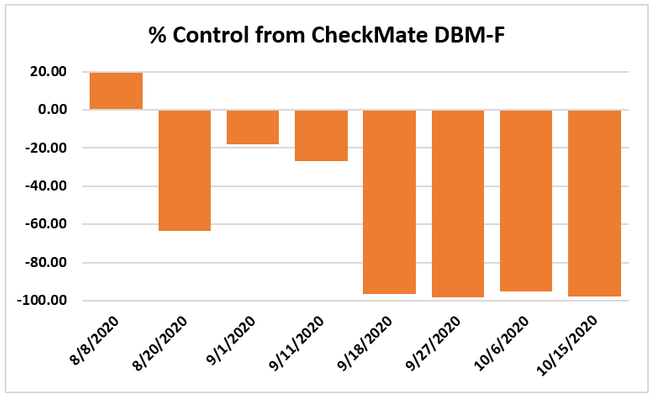
Moth populations: Traps in replication 4 in both treatments on 8 August and replication 1 in the grower standard were missing, probably knocked down by a tractor. The day before CheckMate DBM-F was first applied, the mean number of adult diamondback moths caught were 227 in the grower standard and 271 in the plots that would receive the pheromone application. There was a gradual decline in moth counts during the rest of the observation period in both treatments. However, the decline was higher in the plots that received CheckMate DBM-F. The number of moths per trap were about 19% higher in the pheromone-treated plots compared to the grower standard before the study but were nearly 98% lower by the end of the study. The reduction in moth populations from mating disruption was significant on 18 September (P =0.039) and 15 October (P = 0.006).
Larval populations: The mean number of larvae per 25 plants in a replication was zero on all observation dates except for 0.01 on 30 August in the plots that received CheckMate. Four insecticide applications by the time the study was initiated, and the remaining six applications effectively suppressed larval populations.
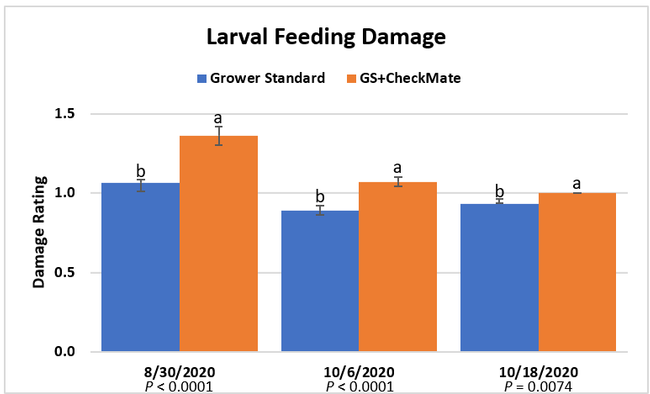
Damage ratings: Larval feeding damage ratings were consistently low (P < 0.0001) in the plants that did not receive CheckMate DBM-F. The damage was limited to the older leaves at the bottom of the plants and must have been from early feeding before the initiation of the study. The lack of larvae and the evidence of new feeding damage also confirm that the crop remained healthy and pest-free.
Since frequent pesticide applications effectively suppressed larval populations and prevented their feeding damage, the effectiveness of mating disruption in reducing yield losses could not be determined in this study. Since larval counts were not made weekly or between pesticide applications, those that were probably present between the pesticide applications could not be determined. Moths found in the traps probably developed from the larvae in the field or could have been those that flew in from other areas. However, lower moth populations in CheckMate DBM-F treatment demonstrated the overall influence of mating disruption and pest suppression.
It is common to make about 10-12 pesticide sprays during the 6-month crop cycle of Brussels sprouts. The cost of each application varied from about $73 to $192 depending on the materials used with an average cost of about $128 per application in this study. The cost of two CheckMate DBM-F applications is $91. If diamondback moth populations could be reduced with mating disruption, it is estimated that 2-3 pesticide applications could be eliminated. That results in $164 to $292 of saving for the pesticide costs and additional savings in the application costs per acre by investing $91 in the mating disruption. Since the diamondback moth can develop resistance to several chemical and natural pesticides, eliminating some applications as a result of mating disruption also contributes to resistance management along with potential negative impact of pesticides on the environment. Compared to other mating disruption strategies, a sprayable formulation compatible with other agricultural inputs is easier and cost-effective to use.
This study demonstrated that mating disruption with CheckMate DBM-F will significantly enhance the current IPM practices by reducing pest populations, contributing to insecticide resistance management, and reducing pest management costs. Additional studies, with fewer pesticide applications that allow larvae to survive and cause some damage, might further help understand the role of mating disruption where pest populations are not managed as effectively as in this field.
Watch a presentation of this study
Acknowledgments: Thanks to the PCA and the grower for their research collaboration, Tamas Zold for his technical assistance in data collection, Ingrid Schumann for market research of pesticide pricing, and Suterra for the financial support.
References
Dara, S. K. 2019. The new integrated pest management paradigm for the modern age. J. Int. Pest Manag. 10: 12.
Dara, S. K. 2020. Arthropod resistance to biopesticides. Organic Farmer 3 (4): 16-19.
Endersby, N. M., K. Viduka, S. W. Baxter, J. Saw, D. G. Heckel, and S. W. McKechnie. 2011. Widespread pyrethroid resistance in Australian diamondback moth, Plutella xylostella (L.), is related to multiple mutations in the para sodium channel gene. Bull. Entomol. Res. 101: 393.
Ferré, J., M. D., Real, J. Van Rie, S. Jansens, and M. Peferoen. 1991. Resistance to the Bacillus thuringiensis bioinsecticide in a field population of Plutella xylostella is due to a change in a midgut membrane receptor. Proc. Nat. Acad. Sci. 88: 5119-5123.
Leibee, G. L. and K. E. Savage. 1992. Evaluation of selected insecticides for control of diamondback moth and cabbage looper in cabbage in Central Florida with observations on insecticide resistance in the diamondback moth. Fla. Entomol. 75: 585-591.
Pu, X., Y. Yang, S. Wu, and Y. Wu. 2009. Characterisation of abamectin resistance in a field-evolved multiresistant population of Plutella xylostella. Pest Manag. Sci. 66: 371-378.
Zhao, J-Z., H. L. Collins, Y-X. Li, R.F.L. Mau, G. D. Thompson, M. Hertlein, J. T. Andaloro, R. Boykin, and A. M. Shelton. 2006. Monitoring of diamondback moth (Lepidoptera: Plutellidae) resistance to spinosad, indoxacarb, and emamectin benzoate. J. Econ. Entomol. 99: 176-181.
- Author: Surendra K. Dara
Strawberry, a high-value specialty crop in California, suffers from several soilborne, fruit, and foliar diseases. Verticillium wilt caused by Verticillium dahliae, Fusarium wilt caused by Fusarium oxysporum f. sp. fragariae, and Macrophomina crown rot or charcoal rot caused by Macrophomina phaseolina are major soilborne diseases that cause significant losses without proper control. Chemical fumigation, crop rotation with broccoli, nutrient and irrigation management to minimize plant stress, and non-chemical soil disinfestation are usual control strategies for these diseases. Botrytis fruit rot or gray mold caused by Botrytis cineaea is a common fruit disease requiring frequent fungicidal applications. Propagules of gray mold fungus survive in the soil and infect flowers and fruits. A study was conducted to evaluate the impact of drip application of various fungicides on improving strawberry health and enhancing fruit yields.
Methodology
This study was conducted in an experimental strawberry field at the Shafter Research Station during 2019-2020. Cultivar San Andreas was planted on 28 October 2019. No pre-plant fertilizer application was made in this non-fumigated field which had Fusarium wilt, Macrophomina crown rot, and Botrytis fruit rot in the previous year's strawberry planting. Each treatment was applied to a 300' long bed with single drip tape in the center and two rows of strawberry plants. Sprinkler irrigation was provided immediately after planting along with drip irrigation, which was provided one or more times weekly as needed for the rest of the experimental period. Each bed was divided into six 30' long plots, representing replications, with an 18' buffer in between. Between 6 November 2019 and 9 May 2020, 1.88 qt of 20-10-0 (a combination of 32-0-0 urea ammonium nitrate and 10-34-0 ammonium phosphate) and 1.32 qt of potassium thiosulfate was applied 20 times at weekly intervals through fertigation. Treatments were applied either as a transplant dip or through the drip system using a Dosatron fertilizer injector (model D14MZ2). The following treatments were evaluated in this study:
i) Untreated control: Neither transplants nor the planted crop was treated with any fungicides.
ii) Abound transplant dip: Transplants were dipped in 7 fl oz of Abound (azoxystrobin) fungicide in 100 gal of water for 4 min immediately prior to planting. Transplant dip in a fungicide is practiced by several growers to protect the crop from fungal diseases.
iii) Rhyme: Applied Rhyme (flutriafol) at 7 fl oz/ac immediately after and 30, 60, and 90 days after planting through the drip system.
iv) Velum Prime with Switch: Applied Velum Prime (fluopyram) at 6.5 fl oz/ac 14 and 28 days after planting followed by Switch 62.5 WG (cyprodinil + fludioxinil) at 14 oz/ac 42 days after planting through the drip system.
v) Rhyme with Switch: Four applications of Rhyme at 7 fl oz/ac were made 14, 28, 56, and 70 days after planting with a single application of Switch 62.5 WG 42 days after planting through the drip system.
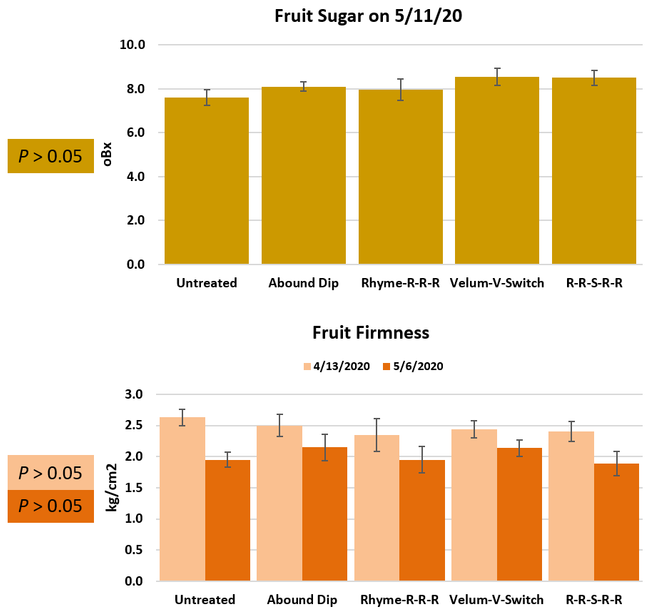
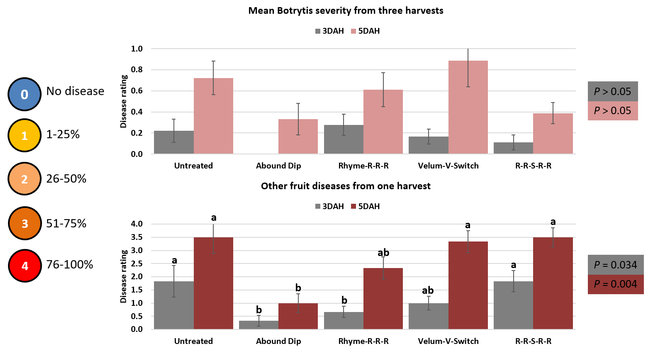
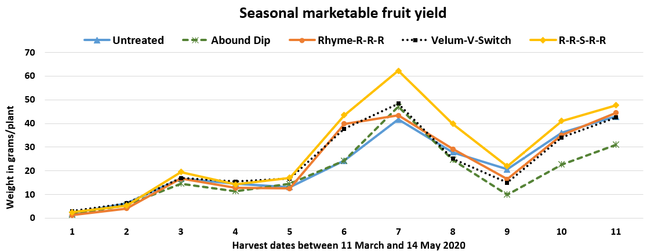
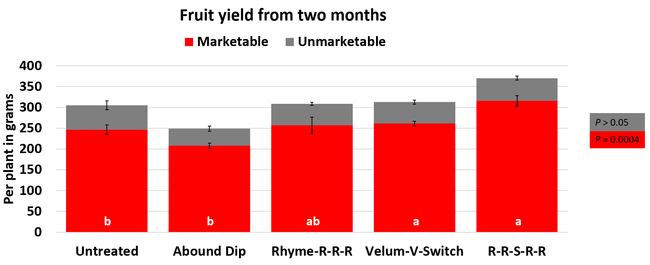
Parameters observed during the study included leaf chlorophyll and leaf nitrogen (with chlorophyll meter) in February and May; fruit sugar (with refractometer) in May; fruit firmness (with penetrometer) in April and May; severity of gray mold (caused by Botrytis cinereae) twice in March and once in May, and other fruit diseases (mucor fruit rot caused by Mucor spp. and Rhizopus fruit rot caused by Rhizopus spp.) once in May 3 and 5 days after harvest (on a scale of 0 to 4 where 0=no infection; 1=1-25%, 2=26-50%, 3=51-75% and 4=76-100% fungal growth); and fruit yield per plant from 11 weekly harvests between 11 March and 14 May 2020. Leaf chlorophyll and nitrogen data for the Abound dip treatment were not collected in February. Data were analyzed using analysis of variance in Statistix software and significant means were separated using the Least Significant Difference test.
Results and Discussion
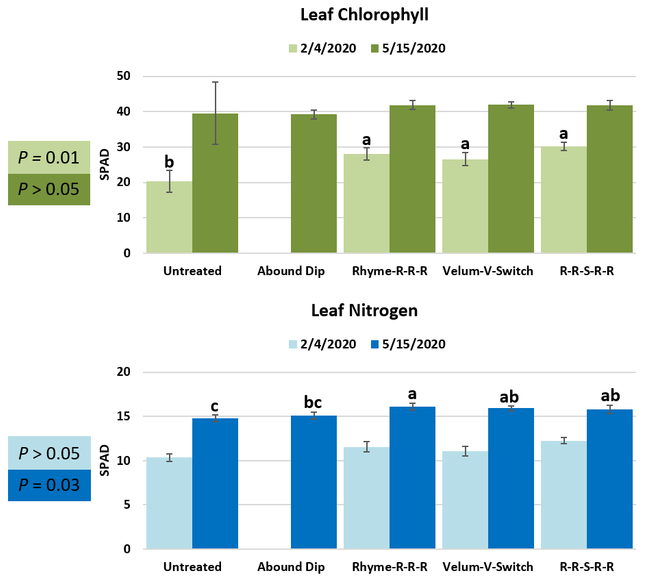
Leaf chlorophyll content was significantly higher in plants that received drip application of fungicides compared to untreated plants in February while leaf nitrogen content was significantly higher in the same treatments during the May observation. There were no differences in fruit sugar or average fruit firmness among the treatments.
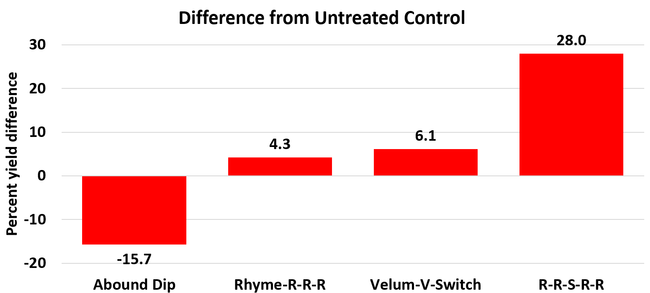
The average gray mold severity from three harvest dates was low and did not statistically differ among the treatments. However, the severity of other diseases was significantly different among various treatments with the lowest rating in Abound transplant dip on both 3 and 5 days after harvest and only 3 days after harvest in plants that received four applications of Rhyme. Unlike the previous year, visible symptoms of the soilborne diseases were not seen during the study period to evaluate the impact of the treatments. However, there were significant differences among treatments for the marketable fruit yield. The highest marketable yield was observed in the treatment that received Rhyme and Switch followed by Velum Prime and Switch and Rhyme alone. The lowest fruit yield was observed in Abound dip treatment. Unmarketable fruit (deformed or diseased) yield was similar among the treatments. Compared to the untreated control, Abound dip resulted in 16% less marketable yield and such a negative impact from transplant dip in fungicides has been seen in other studies (Dara and Peck, 2017 and 2018; Dara, 2020). Marketable fruit yield was 4-28% higher where fungicides were applied to the soil.
Although visible symptoms of soilborne diseases were absent during the study, periodic drip application of the fungicides probably suppressed the fungal inocula and associated stress and might have contributed to increased yields. The direct impact of fungicide treatments on soilborne pathogens was, however, not clear in this study due to the lack of disease symptoms. Considering the cost of chemical fumigation or soil disinfestation and the environmental impact of chemical fumigation, treating the soil with fungicides can be an economical option if they are effective. While this study presents some preliminary data, additional studies in non-fumigated fields in the presence of pathogens are necessary to consider soil fungicide treatment as a control option.
Acknowledgments: Thanks to FMC for funding this study and Marjan Heidarian Dehkordi and Tamas Zold for their technical assistance.
References
Dara, S. K. 2020. Improving strawberry yields with biostimulants and nutrient supplements: a 2019-2020 study. UCANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=43631
Dara, S. K. and D. Peck. 2017. Evaluating beneficial microbe-based products for their impact on strawberry plant growth, health, and fruit yield. UCANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=25122
Dara, S. K. and D. Peck. 2018. Evaluation of additive, soil amendment, and biostimulant products in Santa Maria strawberry. CAPCA Adviser, 21 (5): 44-50.