- (Focus Area) Agriculture
- Author: Surendra K. Dara
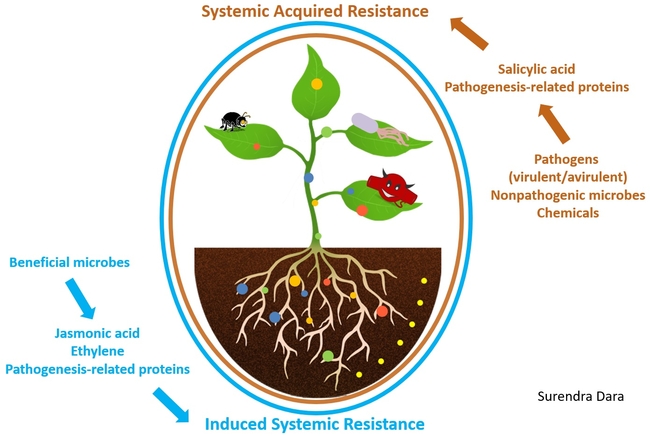
Biostimulants are beneficial microorganisms or substances that can be used in crop production to improve plants' immune responses and their ability to perform well under biotic and abiotic stresses. Biostimulants induce plant resistance to stress factors through systemic acquired resistance or induced systemic resistance. When plants are exposed to virulent and avirulent pathogens, non-pathogenic microorganisms, and some chemicals, the systemic acquired resistance mechanism is activated through the salicylic acid pathway triggering the production of pathogenesis-related proteins. On the other hand, when plants are exposed to beneficial microbes, the induced systemic resistance mechanism is activated through the jasmonic acid and ethylene pathways. The jasmonic acid pathway also leads to pathogenesis-related protein production in plants. In other words, when plants are exposed to pathogens, non-pathogens, or other compounds, various defense genes are activated through two major immune responses, helping plants fight the real infection or prepare them for potential infection. Beneficial microbes and non-microbial biostimulants are like vaccines that prepare plants for potential health problems.
Earlier studies in tomato (Dara and Lewis, 2018; Dara, 2019a) and strawberry (Dara and Peck, 2018; Dara, 2019b) demonstrated varying levels of benefits to crop health and yield improvements from a variety of botanical, microbial, or mineral biostimulants and other supplements. Some of the evaluated products resulted in significant yield improvement in both tomatoes and strawberries compared to the grower standard practices. There are several biostimulant products in the market with a variety of active ingredients, and some also have major plant nutrients such as nitrogen, phosphorus, and potassium. Depending on the crop, growing conditions, potential risk of pests and diseases, and other factors, growers can use one or more of these products. A study was conducted to evaluate the impact of various biostimulants on the yield, quality, and shelf life of strawberries.
Methodology
Strawberry cultivar San Andreas was planted late November 2018 and treatments were administered at the time of planting or soon after, depending on the protocol. Each treatment had a 290' long strawberry bed where 10' of the bed at each end was left out as a buffer. Then, six 30' long plots, each representing a replication, were marked within each bed with an 18' buffer between the plots. Since the test products needed to be applied through the drip system, an entire bed was allocated for each treatment, except for the standard program that had one bed on either side of the experimental block, and plots were marked within each bed for data collection. The following treatment regimens were used in the study:
1. Standard Program (SP): Major nutrients were provided in the form of Urea Ammonium Nitrate Solution 32-0-0, Ammonium Polyphosphate Solution, and Potassium Thiosulfate (KTS 0-0-25). Nitrogen, phosphorus, and potassium were applied before planting in November 2018 at 170, 60, and 130 lb/acre, respectively. From 15 January to 9 May 2019, a total of 26 lb of nitrogen, 13 lb of phosphorus, and 26 lb of potassium were applied through 13 periodic applications.
2. SP + Terramera Program: Formulation labeled as Experimental A (cold-pressed neem 70%) was applied at 1.2% vol/vol immediately after planting. Additional applications were made starting from 2 weeks after planting once every two weeks until the end of February (six times), followed by 13 weekly applications from the beginning of March.
3. SP + Locus Low Rate Program: This program contained Rhizolizer soil amendment (Trichoderma harzianum 1X108 CFU/ml and Bacillus amyloliquefaciens 1X109 CFU/ml) at 3 fl oz/acre, humic acid at 13.5 fl oz/acre, and kelp at 6.8 fl oz/acre. The first application was made within 15 days and at 30 days after planting followed by once in February, March, and April 2019.
4. SP + Locus High Rate Program: This program contained Rhizolizer soil amendment (Trichoderma harzianum 1X108 CFU/ml and Bacillus amyloliquefaciens 1X109 CFU/ml) at 6 fl oz/acre, humic acid at 13.5 fl oz/acre, and kelp at 6.8 fl oz/acre. The first application was made within 15 days and at 30 days after planting followed by once in February, March, and April 2019.
5. SP + BioGro Program: Transplants were treated with Premium Plant BB (Beauveria bassiana 1.1%) by spraying 2 fl oz/acre (1.29 ml in 850 ml of water). About 7 weeks after planting, 30 gpa of Plant-X Rhizo-Pro (botanical extracts), 2 gpa of CHB Premium 21 (humic acid blend), 3 gpa of CHB Premium 6 (3% humic acids), and 5 gpa of NUE Flourish 4-12-0 were applied. Starting from mid-February 2019, 15 gpa of Plant-X Rhizo-Pro, 1 gpa of CHB Premium 21, and 2 gpa of CHB Premium 6 were applied four times every 2 weeks until the end of March. Starting from 5 April 2019, 8 weekly applications of 10 gpa of Plant-X Rhizo-Pro, 1 gpa of CHB Premium 21, 2 gpa of Premium 6, and 4 gpa of NUE Flourish 4-12-0 were made until 26 May 2019.
6. SP + Actagro Program: Structure 7-21-0 at 3 gpa and Liquid Humus 0-0-4 with 22% organic acids at 1 gpa were first applied within 1 week of planting and then three more times every 2 weeks until the end of December 2018. Additional monthly applications were made from the end of January to the end of April 2019.
All the fertilizers and treatment materials were applied through the drip system using the Dosatron (Model D14MZ2) equipment. The following parameters were measured during the experimental period from January to May 2019.
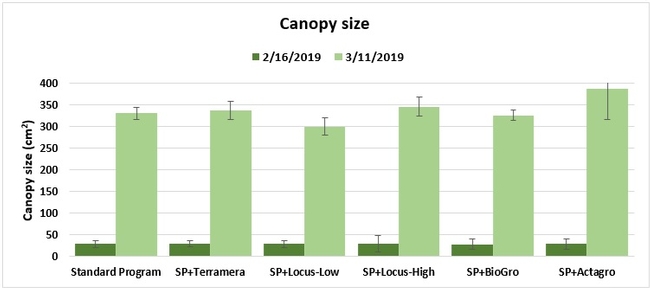
Canopy: The size of the plant canopy was determined on 21 January and again on 17 February 2019 by measuring the spread of the canopy across and along the length of the bed from 16 random plants within each plot, and calculating the area.
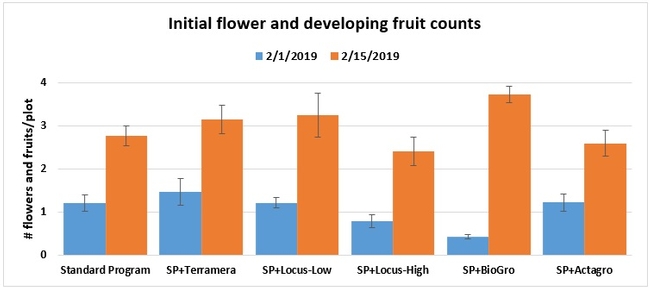
Initial flowering and fruiting: When flowering initiated, the number of flowers and developing fruits was counted from 16 random plants within each plot on 1 and 16 February 2019.
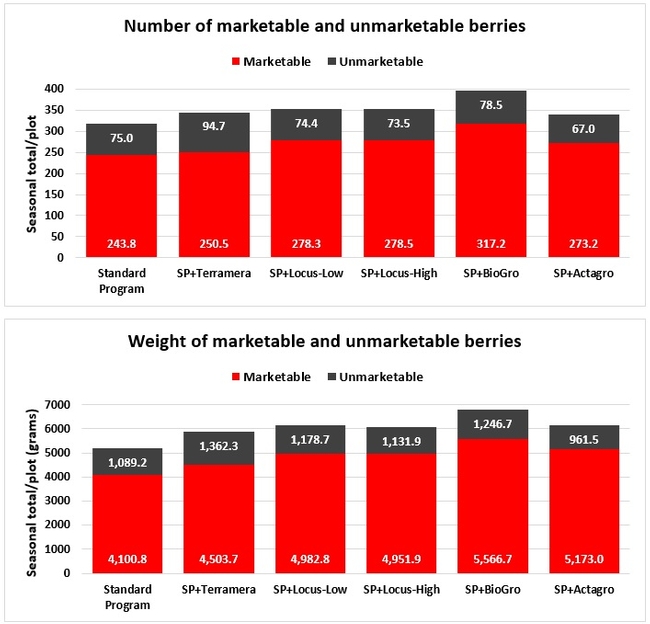
Fruit yield: Fruit was harvested weekly from every plant within each plot from 3 March to 26 May 2019 on 11 dates and the number and weight of the marketable and unmarketable fruit was determined. Due to a technical error, some of the yield data from an additional date (29 March) were lost and excluded from the analysis.
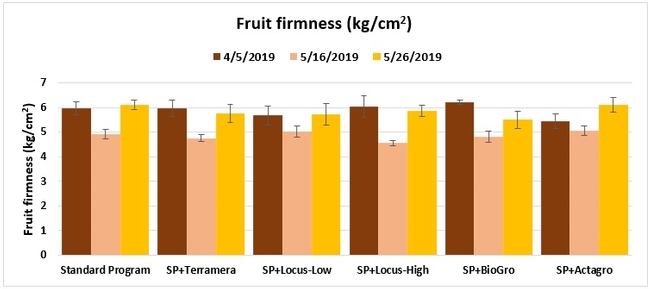
Fruit firmness: The firmness of two marketable fruit from each of five random plants per plot was measured using a penetrometer on 5 April, and 16 and 26 May 2019.
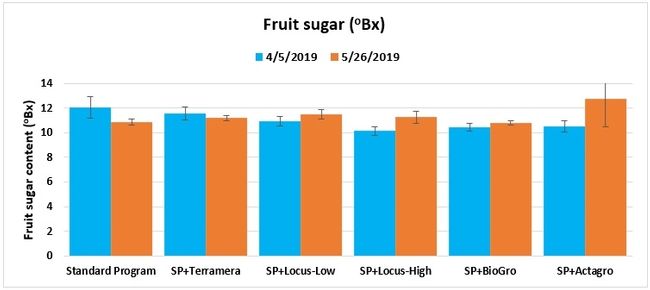
Fruit sugar content: The sugar content from one marketable fruit from each of 10 plants per plot was measured using a refractometer on 5 April and 26 May 2019.
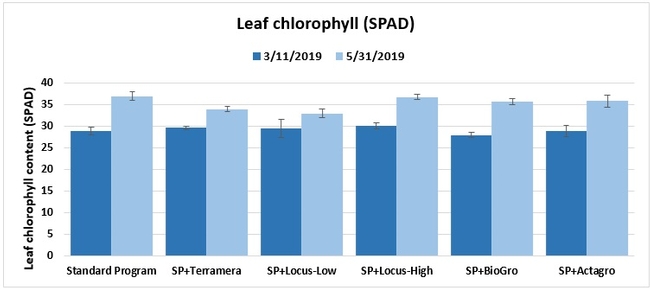
Leaf chlorophyll content: On 11 March and 31 May 2019, the chlorophyll content of one mature leaf from each of five random plants per plot was measured using a chlorophyll meter.
Postharvest disease: Marketable fruit harvested on 21 and 28 April, and 5 and 26 May 2019 was kept at the room temperature in perforated plastic containers (clamshells) and the growth of gray mold (Botrytis cinerea) or Rhizopus fruit rot fungus (Rhizopus spp.) was measured on a scale of 0 to 4 (where 0=no fungus, 1=1-25%, 2=26-50%, 3=51-75%, and 4=76-100% fungal growth) 3 and 5 days after each harvest.
Data were analyzed using analysis of variance in Statistix software and significant means were separated using the Least Significant Difference means separation test.
Results and Discussion
Statistically significant differences among treatments were seen for the seasonal total number of unmarketable berries (P = 0.0172), the initial flower and fruit numbers on 1 February (P < 0.0014), the leaf chlorophyll content on 31 May (P = 0.0144), and the disease rating 3 days after the 28 April harvest (P = 0.0065).
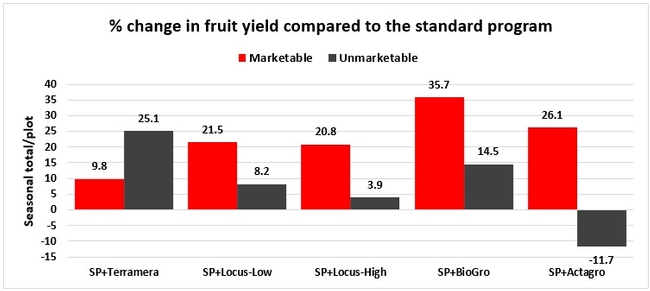
Treatments did not differ (P > 0.05) in any other measured parameters of the plant, fruit quality, or yield. However, the total seasonal fruit yield was 13 to 31% higher and the total marketable fruit yield was 10 to 36% higher in various treatment programs compared to the standard program. The seasonal total of unmarketable fruit yield was also 4 to 25% higher in treatment programs than the standard program except that there were nearly 12% fewer unmarketable berries in the Actagro program compared to the standard program.
While treatments did not statistically differ for many of the measured parameters, numerical differences in marketable fruit yield could be helpful for some understanding of the potential of these biostimulants. Additional studies with larger treatment plots would be useful for generating additional data.
Acknowledgments
Thanks to Dr. Jenita Thinakaran for the assistance at the start of the study, Hamza Khairi for his technical assistance throughout the study, the field staff at the Shafter Research Station for the crop maintenance, NorCal Nursery for the strawberry transplants, and Actagro, BioGro, Locus, and Terramera for their collaboration and financial support
References
Dara, S. K. 2019a. Improving tomato yield with nutrient materials containing microbial and botanical biostimulants. eJournal of Entomology and Biologicals, 6 June 2019 https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=30448
Dara, S. K. 2019b. Evaluating the efficacy of anti-stress supplements on strawberry yield and quality. eJournal of Entomology and Biologicals, 10 August 2019 https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=31044
Dara, S. K. and D. Peck. 2018. Microbial and bioactive soil amendments for improving strawberry crop growth, health, and fruit yields: a 2017-2018 study eJournal of Entomology and Biologicals, 3 August 2018 https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=27891
Dara, S. K. and E. Lewis. 2018. Impact of nutrient and biostimulant materials on tomato crop health and yield. eJournal of Entomology and Biologicals, 9 January 2019 https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=26054
- Author: Surendra K. Dara
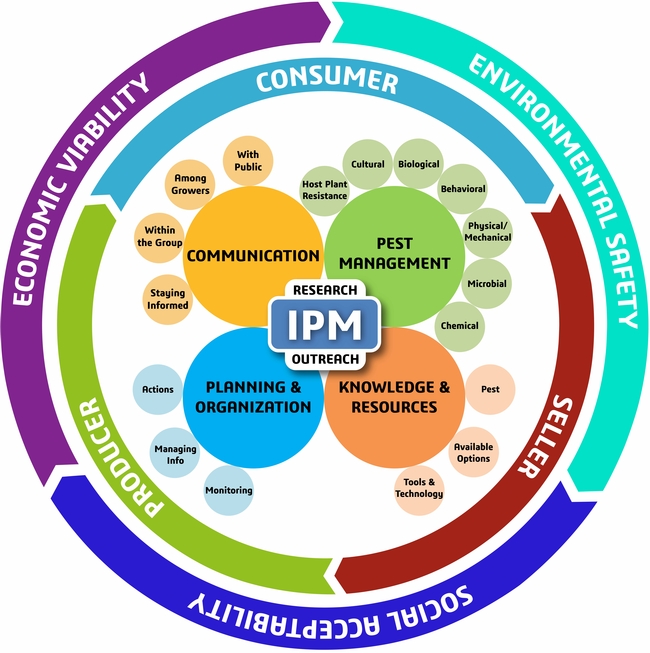
The traditional Integrated Pest Management (IPM) model is focused on maintaining ecological balance in the cropping system with some attention to the economics of pest management related to the yield losses. The new model, recently published in the Journal of Integrated Pest Management, is more comprehensive covering the management, business, and sustainability aspects of pest management and discusses various components within (Dara, 2019). IPM, according to the new model, can be defined as an approach to managing pests in an economically viable, socially acceptable, and environmentally safe manner.
New IPM model from Dara, 2019
Based on the information generated by several studies in California and other reports, here is how the new IPM model can be adapted for producing strawberries sustainably.
1. MANAGEMENT ASPECT
A. Pest Management: The term “pest” includes arthropod pests, diseases, and weeds and the management includes the various practices used to suppress them.
- Select varieties that produce good yields while resisting biotic and abiotic stresses.
- Choosing the right mulch and good irrigation and nutrient management contribute to good plant growth and health. Micro-sprinklers save water and hold pest management benefits.
- Explore the potential of beneficial microbes and biostimulants to improve nutrient and water absorption and to maintain crop health. Inoculate the transplants with biostimulants to induce systemic resistance and periodically apply, especially after fumigation, to improve the beneficial microbial activity in the soil.
- Healthy plants resist pest problems and reduce the need for control options. Plant health can be maintained through good cultural practices (biostimulants, nutrients, irrigation, soil conditioning, etc.).
- Predatory mites effectively control twospotted and Lewis mites, but natural enemy populations may not be sufficient to control the western tarnished plant bug.
- Light traps can be useful for managing lepidopteran pests.
- Tractor-mounted vacuums can be a part of the IPM program for managing the western tarnished plant bug, but their pest control efficiency is not necessarily superior to other strategies and are not without some associated risks. For example, operation of vacuums requires fossil fuels and they are used at a much higher frequency than pesticide applications.
- Use botanical, microbial, and chemical pesticides in combination. Combinations can improve pest control efficacy and rotations reduce the risk of resistance development.
- Rotating strawberries with crops such as broccoli can reduce the severity of certain soilborne diseases.
B. Knowledge and Resources:
- Understand pest biology, vulnerable stages of the pest, and appropriate strategies for each pest, different life stages, season, and budget. For example, relying on natural enemies for the western tarnished plant bug control is not effective and can lead to higher pest damage.
- Accurately identify the issue through visual observation or laboratory diagnosis for proper corrective action.
- Try to explore modern technology to monitor crop health.
C. Planning and Organization:
- Regularly monitor crop health for early detection and prevention of potential pest problems. For example, thorough scouting to determine the level of western tarnished plant bug infestation is very important for making the treatment decision. Deformed fruit is not always an indicator for the treatment threshold as nearly 1/3 of the fruit deformity occurs from environmental and other causes not related to the western tarnished plant bug.
- Look for signs of pesticide resistance and use appropriate strategies to reduce the risk.
- Maintain records of pest occurrence, seasonal trends, strategies that worked, and all relevant information, to build institutional knowledge for future use.
- Take the right action at the right time.
D. Communication:
- Regularly attend extension events and read research updates. Choose or design practices that are ideal for your farm based on the research updates.
- Periodically provide training to all individuals on the farm who directly or indirectly contribute to good agriculture practices.
- Share good management practices with each other for area-wide improvement of crop production and pest management.
- Try to educate the public so that they make better choices when purchasing produce. For example, good IPM practices can be more sustainable than organically approved practices and well-informed consumers can make a choice among conventional, organic, or sustainably produced grains, fruits, and vegetables. Public education can also help to eliminate otherwise good produce that is discarded because of minor imperfections. In strawberry, fruit deformity is caused due to the feeding of the western tarnished plant bug, genetic factors, poor pollination, or very low temperatures. Although most of the deformed strawberries, especially those from insect damage, have equal quality as marketable strawberries, they are discarded because of their shape. If the consumer market can accept deformed strawberries that still have good taste and nutritional quality, it can significantly reduce the wastage and the amount of pesticides sprayed to control the western tarnished plant bug.
2. BUSINESS ASPECT
- A strong IPM program can help growers produce sustainably while ensuring profitability.
- Consumer choices depend on their knowledge of sustainable agriculture. When they understand that produce with an IPM or Sustainably Produced label is safe for human and environmental health, it will have a major impact on food production systems.
3. SUSTAINABILITY ASPECT
- The current interpretation or perception of sustainability does not reflect true sustainability in terms of environmental health, profitability, food security, social equality, and other elements. A good IPM model can address all these issues to ensure farm productivity, food affordability, and environmental safety.
RESEARCH and OUTREACH
- Research and outreach component is the foundation of IPM to identify pest issues, develop appropriate knowledge for their management, and effectively disseminate the related information. Supporting research and outreach efforts of universities and other entities is essential for continuing IPM.
References
In addition to the below references, there are several articles in this eJournal on crop production and protection topics related to strawberry.
- Download “Biology and management of spider mites in strawberry” in English and Spanish at http://ucanr.edu/spidermiteguide or scan the QR code. Information about different species of spider mites and predatory mites is available in this guide.
- Efficacy of botanical, chemical, and microbial pesticides on twospotted spider mite and their impact on predatory mites http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=18553
- Entomopathogenic fungi can endophytically colonize strawberry plants when applied to the soil and negatively impact twospotted spider mite infestations http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=16821
- How to detect resistance to miticides in twospotted spider mite populations and strategies to reduce the resistance development http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=22097
- Comparison between the twospotted spider mite and the Lewis mite http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=5771
- An overview of lygus bug biology, damage, and management in strawberries http://cesantabarbara.ucanr.edu/files/75473.pdf
- Lygus biology, monitoring, and management videos http://ucanr.edu/SDYouTube
- Fruit deformity in strawberry from lygus bug and other factors http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=19630
- Potential of a solar-powered UV light trap as a pest management option in strawberry http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=25307
- IPM tools for controlling western tarnished plant bug in strawberry https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=19641
- Entomopathogens (pathogens of insects, mites, and ticks), their modes of infection, and how they can be used as a powerful tool in IPM http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=24119
- Biopesticides and IPM https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=25912
- Lygus bug and natural enemy populations in organic and conventional strawberries https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=14030
- Microbial and bioactive soil amendments for improving strawberry health and yields (2017-2018 study) https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=27891
- Beneficial microbe-based products for strawberry health and yield (2016-2017 study)
https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=25122
- Beneficial microbes and entomopathogenic fungi for strawberry health and yield (2015-2016 study) https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=22709
- Entomopathogenic fungi antagonizing Macrophomina phaseolina https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=28274
- Entomopathogenic fungi and other biologicals against Fusarium oxysporum
https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=22199
- Micro-sprinklers in strawberry https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=19699
- Author: Surendra K. Dara
Good nutrient management is essential not only for optimal plant growth, but also for maintaining good plant health and the ability of the plant to withstand biotic and abiotic stressors. Strawberry, a $3.2 billion commodity in California, requires good nutrient, water, and health management throughout its lengthy fruit production cycle. In addition to the primary nutrient inputs, certain supplements can be beneficial to the crop. A study was conducted in fall-planted strawberries from 2017 to 2018 using a plant-based anti-stress agent, humates, and sulfur, and a special formulation of NPK as supplements to the standard fertility program to evaluate their impact on strawberry fruit yields and quality.
Methodology
Strawberry cultivar Albion was planted during the second week of December 2017 in 38” wide beds with two rows of plants per bed. This study included the following treatments:
1. Grower standard (GS) program included a total of 6.13 gallons of Urea Ammonium Nitrate Solution 32-0-0, 2.59 gallons of Ammonium Polyphosphate Solution, and 6.95 gallons of Potassium Thiosulfate (KTS 0-0-25) to 0.5 acres of the strawberry field. These fertilizers were applied between 5 January and 18 May 2018 approximately at weekly intervals through the drip irrigation system. Additionally, 1 quart of Nature's Source Organic Plant Food 3-1-1 was applied on 5 and 22 January 2018 and again on 5 February 2018.
2. GS + Bluestim at 3.6 lb/ac in 53 gallons applied as a foliar spray with 0.125% Dyne-Amic once every three weeks for a total of six times. Bluestim is an osmoregulator containing >96% of glycine betaine that is expected to protect plants from abiotic stressors.
3. GS + SKMicrosource Ultrafine powder at 1.4 oz in 4 gallons applied as a foliar spray once a month for a total of three times along with SKMicrosource prill applied at 500 lb/ac at the base of the plant once. Both products contain elemental sulfur, sulfite, and sulfate along with potassium, micronutrients, and rare earth minerals. Additionally, the prill form also has humates. These products are expected to improve plants' natural defenses against biotic stressors like pests and diseases.
4. GS + ISO NPK 3-1-3 at 8 fl oz/ac in 100 gallons once every two weeks for a total of four times. ISO NPK 3-1-3 contains isoprenoid amino complex extracted from a desert shrub guayule (Pathenium argentatum), which is expected to improve nutrient uptake and protect plants from abiotic stressors.
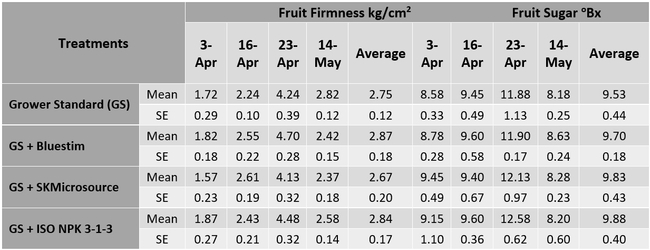
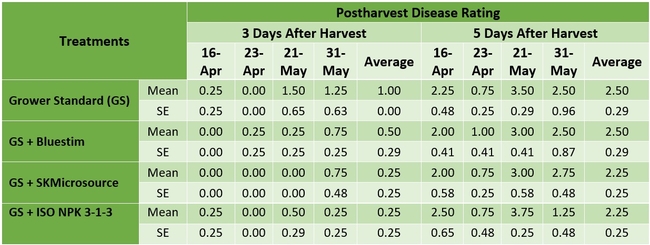
The first application of supplements for treatments 2-4 started on 1 March 2018. Each treatment had a 30' long plot marked on a bed replicated four times in a randomized complete block design. The fruit was harvested one to two times per week between 3 April and 14 June 2018 and the weight of marketable and unmarketable berries was determined for each plot. Using a penetrometer, fruit firmness was measured from four fruits from each plot on 3, 16, and 23 April and 14 May 2018. Sugar content was also measured from two fruits from each plot on those four sampling dates. Postharvest health was measured from the fruit harvested on 16 and 23 April and 21 and 31 May 2018. Fruit was kept in perforated plastic containers (clamshells) at room temperature and the growth of gray mold (Botrytis cinerea) and Rhizopus fruit rot (Rhizopus spp.) was rated 3 and 5 days after harvest on a scale of 0 to 4 (where 0=no disease, 1=1-25% fruit with fungal infection, 2=26-50% infection, 3=51-75%, and 4=76-100%). Data were analyzed using the analysis of variance in Statistix software.
Results
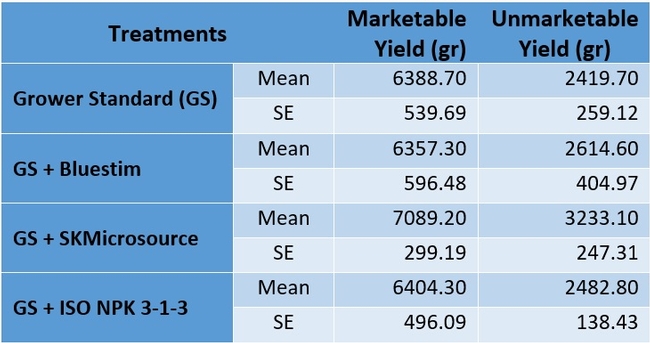
There were no statistically significant (P > 0.05) differences among the treatments in any of the measured parameters. However, the marketable fruit yield was nearly 11% higher in the treatment that received SKMicrosource supplements. While the average sugar content was 9.5 oBx in the grower standard, it varied between 9.7 and 9.8 in other treatments. Similarly, the average disease rating during the postharvest fruit evaluation was 1.00 for the standard at 3 days after harvest, while it varied between 0.25 and 0.50 for the other treatments. Average disease rating at 5 days after harvest was between 2.25 and 2.50 for all treatments.
Table 1. Total marketable and unmarketable fruit yield per plot during the study period
Table 2. Fruit firmness and sugar content on four observation dates and their averages
Table 3. Postharvest fruit disease rating 3 and 5 days after four harvest dates
Discussion
The crop was generally healthy during the study period and there were no signs of any abiotic stresses such as salinity, water stress, and extreme temperature fluctuations, or biotic stresses such as pests or diseases except for uniform weed growth in the furrows and some areas of the beds. Since these supplements are expected to help the plants under stressful conditions, significant differences could not be found, probably due to the lack of unfavorable growth conditions. It also appears from the manufacturer's studies that ISO NPK 3-1-3 performs better at 4 fl oz/acre - half the rate recommended for this study. Additional studies can help further evaluate the potential of these supplements both under normal and stressed conditions and at different application rates and frequencies.
Acknowledgments
Thanks to the technical assistance of Dr. Jenita Thinakaran in carrying out the study, the field staff at the Shafter Research Station for the crop maintenance, the financial support of Biobest and Heart of Nature, and to Beem Biologics for providing the test material.
- Author: Sumanth S. R. Dara
- Author: Suchitra S. Dara
- Author: Surendra K. Dara
Charcoal rot, caused by Macrophomina phaseolina, is one of the important fungal diseases of strawberry in California. Macrophomina phaseolina is a soilborne fungus and has a wide host range, including alfalfa, cabbage, corn, pepper, and potato, some of which are cultivated in the strawberry production areas in California. The fungus infects the vascular system of the plant roots, obstructing the nutrient and water supply and ultimately resulting in stunted growth, wilting, and death of the plant. The fungus survives in the soil and infected plant debris as microsclerotia (resting structures made of hyphal bodies) and can persist for up to three years. Microslerotia germinate and penetrate the root system to initiate infection. Plants are more vulnerable to fungal infection when they are experiencing environmental (extreme weather or drought conditions) and physiological (heavy fruit bearing) stress.
Soil fumigation is the primary management option for addressing charcoal rot in strawberry. Crop rotation with broccoli can also reduce the risk of charcoal rot due to glucosinolates and isothiocyanates in broccoli crop residue that have fungicidal properties. Beneficial microorganisms such as Bacillus spp. and Trichoderma spp. are also considered, especially in organic strawberries, to antagonize M. phaseolina and other soilborne pathogens and provide some protection. The role of beneficial microbes in disease management or improving crop growth and health is gaining popularity in the recent years with the commercial availability of biofungicide, biostimulant, and soil amendment products. In a couple of recent strawberry field studies in Santa Maria, some of the beneficial microbial products improved fruit yield or crop health. These treatments can be administered by inoculating the transplants prior to planting, immediately after planting or periodically applying to the plants and or the soil. Adding beneficial microbes can help improve the soil microbiome especially after chemical or bio-fumigation and anaerobic soil disinfestation.
Similar to the benefits of traditionally used bacteria (e.g., Bacillus spp. and Pseudomonas spp.) and fungi (e.g., Glomus spp. and Trichoderma spp.), studies with entomopathogenic fungi such as Beauveria bassiana, Isaria fumosorosea, and Metarhizium spp. also demonstrated their role in improving water and nutrient absorption or antagonizing plant pathogens. The advantage of entomopathogenic fungi is that they are already used for arthropod pest management in multiple crops, including strawberry; now, there are the additional benefits of promoting crop growth and antagonizing plant pathogens. In light of some promising recent studies exploring these roles, a study was conducted using potted strawberry plants to evaluate the efficacy of two California isolates of Beauveria bassiana and Metarhizium anisopliae s.l. and their application strategies against M. phaseolina.
Methodology
About 6 week old strawberry plants (cultivar Albion) from a strawberry field at the Shafter Research Station were transplanted into 1.6-gallon pots with Miracle-Gro All Purpose Garden Soil (0.09-0.05-0.07 N-P-K) and maintained in an outdoor environment. They were regularly watered, and their health was monitored for about 5 months prior to the commencement of the study. Conidial suspensions of the California isolates of B. bassiana and M. anisopliae s.l. were applied one week before, after, or at the time of applying microsclerotia of M. phaseolina to the potting mix. The following treatments were evaluated in the study:
- Untreated control
- Soil inoculated with M. phaseolina
- Soil inoculated with B. bassiana 1 week prior to M. phaseolina inoculation
- Soil inoculated with M. anisopliae s.l. 1 week prior to M. phaseolina inoculation
- Soil inoculated with B. bassiana at the time of M. phaseolina inoculation
- Soil inoculated with M. anisopliae s.l. at the time of M. phaseolina inoculation
- Soil inoculated with B. bassiana 1 week after to M. phaseolina inoculation
- Soil inoculated with M. anisopliae s.l. 1 week after to M. phaseolina inoculation
Entomopathogenic fungi were applied as 1X1010 viable conidia in 100 ml of 0.01% Dyne-Amic (surfactant) solution distributed around the plant base. To apply M. phaseolina, 5 grams of infested cornmeal-sand inoculum containing 2,500 CFU/gram was added to four 5 cm deep holes around the base of the plant. Each treatment had six pots each planted with a single strawberry plant representing a replication. Treatments were randomly arranged within each replication. The study was repeated once a few days after the initiation of the first experiment.
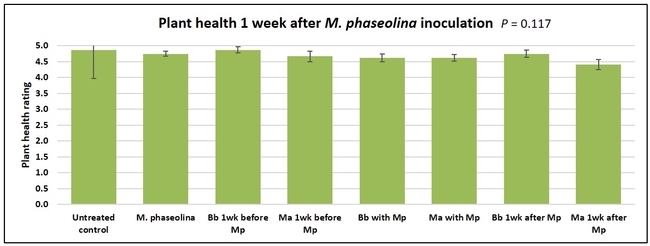
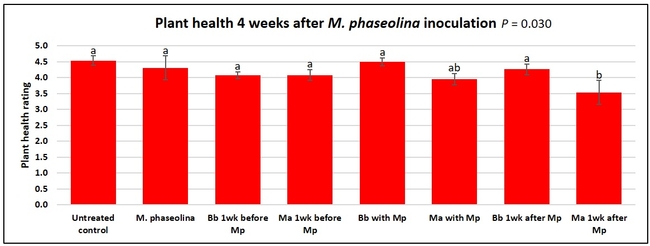
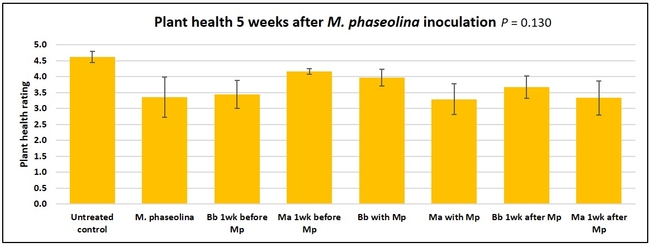
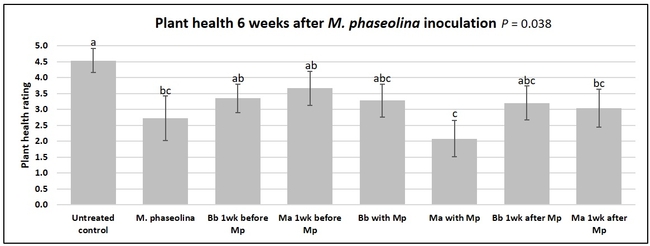
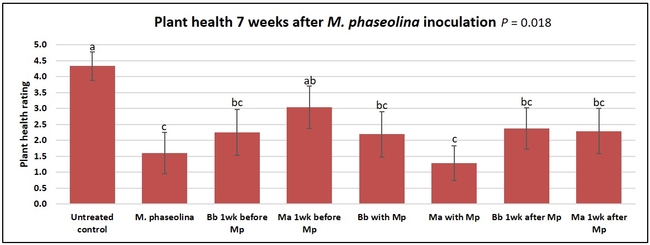
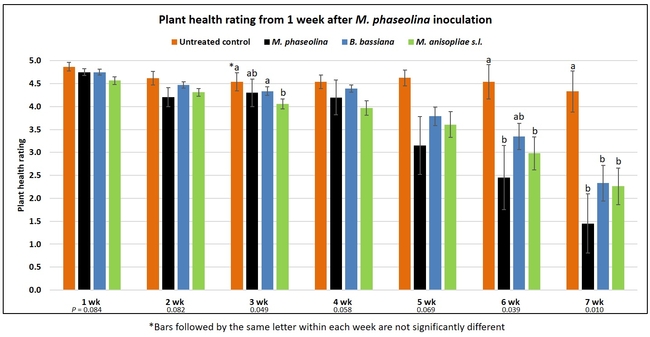
Plant health was monitored starting from the first week after the M. phaseolina inoculation and continued for seven weeks. Plant health was rated on a scale of 0 to 5 where 0=dead and 5=very healthy and the rest of the ratings in between depending on the extent of wilting. Data from both experiments were combined and analyzed by ANOVA using Statistix software and significant means were separated using LSD test. The influence of entomopathogenic fungal treatments applied at different times as well as the combined effect of different applications within each fungus were compared for seven weeks. Ratings for some plants that were scorched from hot summer temperatures and died abruptly were removed from the analyses.
Results
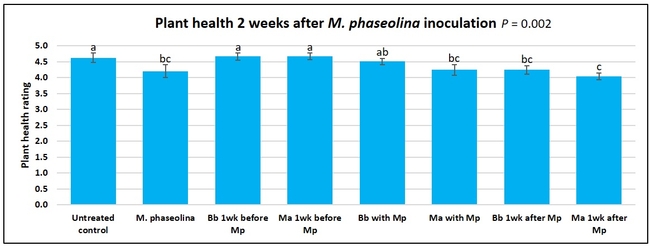
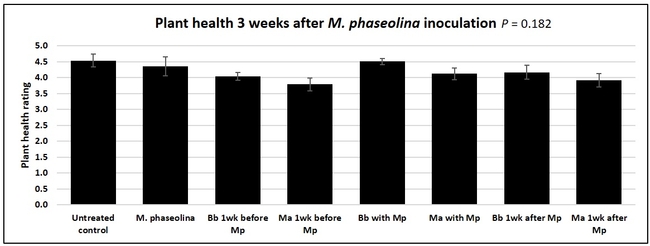
Untreated control plants maintained good health throughout the observation period varying between the rating of 4.3 and 4.9. In general, plant health declined considerably from the 5th week after M. phaseolina inoculation. Plant health appeared to be slightly better in plants treated with entomopathogenic fungi, but there was no statistically significant difference in any except one instance. Plants treated with M. anisopliae one week prior to the application of M. phaseolina had a rating of 3.0 compared to 1.6 rating of plants inoculated with M. phaseolina alone.
When data from different treatments for each entomopathogenic fungus were compared, both B. bassiana and M. anisopliae s.l. appeared to reduce the wilting, but the plant health rating was not significantly different from the M. phaseolina treatment alone.
This is the first report of the impact of entomopathogenic fungi on M. phaseolina with some promise. Additional studies under more uniform environmental conditions and with more treatment options would shed more light on this approach of using entomopathogenic fungi against M. phaseolina. The current study evaluated single application of the entomopathogenic fungi and we plan to conduct additional studies with multiple applications.
Acknowledgements: We thank Dr. Kelly Ivors (previously at Cal Poly San Luis Obispo) for the pathogen inoculum and Dr. Stefan Jaronski, USDA-ARS, Sidney, MT for multiplying the entomopathogenic fungal inocula.
References
Dara, S. K. and D. Peck. 2017. Evaluating beneficial microbe-based products for their impact on strawberry plant growth, health, and fruit yield. UC ANR eJournal Strawberries and Vegetables. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=25122
Dara, S. K. and D. Peck. 2018. Evaluation of additive, soil amendment, and biostimulant products in Santa Maria strawberry. CAPCA Adviser, 21(5): 44-50.
Dara, S. K., S.S.R. Dara, and S. S. Dara. 2017. Impact of entomopathogenic fungi on the growth, development, and health of cabbage growing under water stress. Amer. J. Plant Sci. 8: 1224-1233. http://file.scirp.org/pdf/AJPS_2017051714172937.pdf
Dara, S. K., S. S. Dara, S.S.R. Dara, and T. Anderson. 2016. First report of three entomopathogenic fungi offering protection against the plant pathogen, Fusarium oxysporum f.sp. vasinfectum. UC ANR eJournal Strawberries and Vegetables. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=22199
Koike, S. T., G. T. Browne, and T. R. Gordon. 2013. UC IPM pest management guidelines: Strawberry diseases. UC ANR Publication 3468. http://ipm.ucanr.edu/PMG/r734101511.html
Partridge, D. 2003. Macrophomina phaseolina. PP728 Pathogen Profiles, Department of Plant Pathology, North Carolina State University. https://projects.ncsu.edu/cals/course/pp728/Macrophomina/macrophominia_phaseolinia.HTM
Vasebi, Y., N. Safaie, and A. Alizadeh. 2013. Biological control of soybean charcoal root rot disease using bacterial and fungal antagonists in vitro and greenhouse condition. J. Crop Prot. 2(2): 139-150.
- Author: Surendra K. Dara
- Author: Dave Peck, Manzanita Berry Farms
In a continuous effort to explore the potential of additive, soil amendment, biostimulant, and other products, a new study was conducted in a conventional strawberry field at the Manzanita Berry Farms in Santa Maria. The following treatments were administered at different times, from planting till the end of production season, as requested by the manufacturer.
- Untreated control: Other than the soil incorporated fertilizers during the field preparation, no other nutrient inputs were added during the study.
- Grower standard: Transplants were dipped in Switch 62.5WG (cyprodinil+fludioxonil, at 5 oz/100 gal) before planting and a proprietary nutrient regimen that included administration of a humic acid-based product was followed.
- Innovak Global regimen: Nutrisorb-L (a blend of polyhydroxy carboxylic acids) at 28 fl oz/ac, starting 2 wk after planting and every 3 wk thereafter through drip. Packhard (carboxylic acids with calcium and boron) at 28 fl oz/ac, starting at the first fruit set (early January) and every 2 wk thereafter as a foliar spray.
- TerraVesco regimen: A microbe-rich Vermi-extract (worm extract) at 10% vol/vol as a transplant dip for 3 hours, followed by application through drip at 7.5 gal/ac after planting, and again in December, 2017 and January, 2018.
- Fertum regimen: Transplant dip in 1% vol/vol of Germinal Plus (a product from marine algae), followed by drip applications of Booster (a biostimulant and a natural organic fertilizer made from seaweed) at 0.5 gal/ac in late November and late December, 201; Silicium PK (a biostimulant and a natural organic fertilizer based on silicon enriched with phosphorus, potassium and seaweed extracts) at 0.5 gal/ac late December, 2017 and once a month starting from mid February to early July, 2018; and Foliar (a biostimulant and a natural organic fertilizer from marine algae) at 0.5 gal/ac in mid and late January.
- Shemin Garden regimen: EcoSil (a silica fertilizer) at 800 ml/ac once a month starting from early December, 2017 to May, 2018 through drip, and at 200 ml/ac in early May and June, 2018 as a foliar spray; ComCat (based on a plant extract) at 20 gr/ac and EcoFlora (a consortium of Azotobacter spp., Bacillus spp., Paenibacillus spp., Pseudomonas sp., Trichoderma spp., and Streptomyces spp.) at 12 oz/ac one week after EcoSil through drip until May, 2018 and ComCat at 10 gr/ac and EcoFlora at 12 oz/ac as a foliar spray in May and June, 2018.
- GrowCentia regimen-low: Yeti containing 1% bacterial culture (of Pseudomonas putida, Citrobacter freundii, Comamonas testosterone, and Enterobacter cloacae) and 2% alfalfa extract applied at 0.6 ml/gal through drip for 90 min weekly from the first drip application.
- GrowCentia regimen-high: Yeti at 1 ml/gal through drip for 90 min weekly from the first drip application.
- NanoChem regimen: EX10, a biodegradable fertilizer additive containing thermal polyaspartate at 1 qrt/ac through first drip after planting with follow up applications in early January (first bloom), mid February, and mid May, 2018. The active ingredient binds with cations such as ammonium, calcium, copper, iron, magnesium, manganese, potassium, and zinc and improves their availability for the plant.
- BiOWiSH regimen 1: Formula 1 at 1.33 oz/gal for transplant dip followed by 3.53 oz/ac through drip starting 2 wk after planting and every 4-5 wk thereafter.
- BiOWiSH regimen 2: Formula 1 at 1.33 oz/gal for transplant dip followed by 3.53 oz/ac as a foliar srpay starting 2 wk after planting and every 4-5 wk thereafter.
- BiOWiSH regimen 3: Formula 1 at 1.33 oz/gal for transplant dip followed by 3.53 oz/ac through drip starting 2 wk after planting alternated with a foliar spray every 4-5 wk.
- BiOWiSH regimen 4: Formula 1 at 1.33 oz/gal for transplant dip followed by BiOWiSH Crop 16-40-0, a microbial consortium (Bacillus amyloliquefaciens, B. lichenoformis, B. pumilus, and B. subtilis)at 3.53 oz/ac through drip starting 2 wk after planting and every 4-5 wk thereafter.
Each treatment contained a 165' long 5.7' wide bed and replicated four times in a randomized complete block design. A 15' long plot in the center of the bed was marked and netted for collecting yield and some other parameters that were compared. Strawberry cultivar BG 6-30214 was planted on 7 November, 2017. Other than the untreated control, all other products were administered on top of the grower standard fertility program. However, only the grower standard transplants were dipped in Switch 62.5WG before planting.
Various parameters were measured during the vegetative growth and fruit production periods to evaluate the impact of the treatments on crop growth, health, and yield. Data were analyzed using ANOVA and LSD test was used to separate significant means.

Transplant treatment (above) and drip application (below). Photos by Tamas Zold
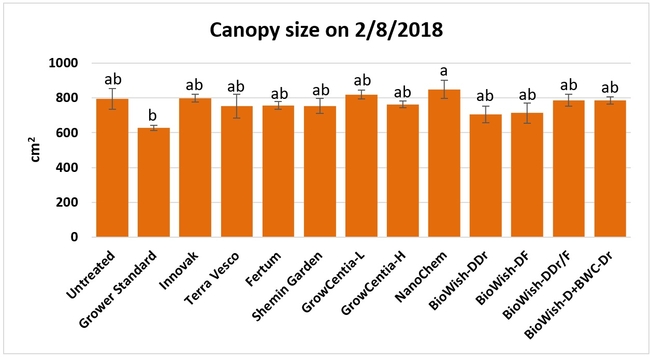
Canopy growth: Canopy growth was observed on 11 December, 2017, 7 and 30 January, and 8 February, 2018 by measuring the size of the canopy along and across the length of the bed from 20 random plants per bed and calculating the area. Canopy size significantly (P = 0.0261) different among the treatments only on the last observation date where plants treated with EX10 and the GrowCentia product at the low concentration were larger than those in the grower standard.
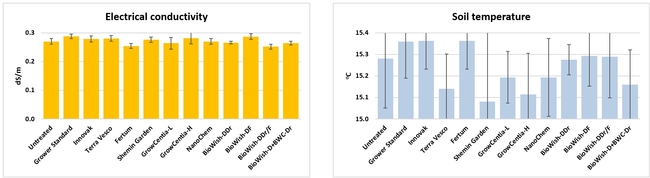
Electrical conductivity and temperature of soil: From two random location on each bed, electrical conductivity (EC in dS/m) and temperature (oC) were measured about 3 inches deep from the surface on 12 and 25 January, 7 February, 19 March, 18 April, and 29 May, 2018. Only soil temperature on 25 January significantly (P = 0.0007) varied among treatments where the difference between the highest (untreated control) and the lowest (Vermi-extract) values was 0.8oC.
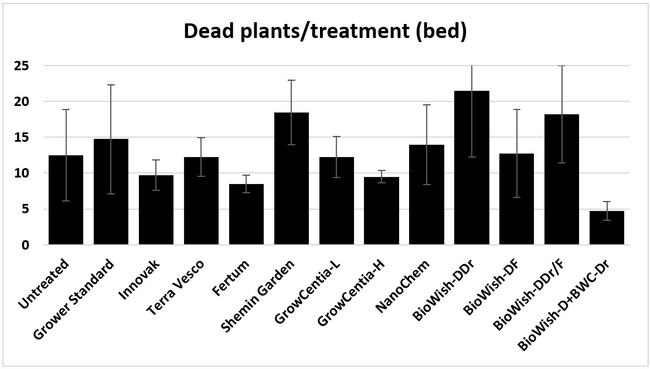
Dead plants: The number of dead plants represents empty spots in the bed due to the death of transplants. There were no obvious signs of disease or a particular stress factor associated with those plants except that they were randomly distributed within each bed and throughout the field. When counted on 18 April, 2018, BiOWiSH regimen 4, Fertum regimen, GrowCentia product at the high rate, and Innovak Global regimen had
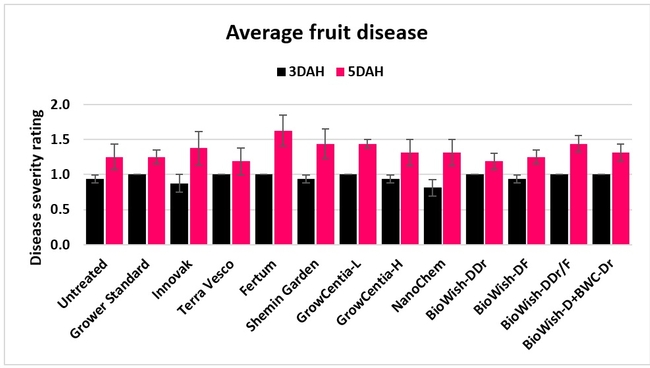
Fruit diseases: Fruit harvested on 12 March, 3 and 13 April, and 17 May, 2018 from each marked plot was incubated at room temperature in dark in plastic containers and the fungal growth was rated 3 and 5 days after harvest (DAH) using a scale of 0 to 4 where 0=no fungal growth, 1=1-25%, 2=26-50%, 3=51-75%, and 4=76-100% fungal growth. Botrytis fruit rot or grey mold was predominant during the first two observation dates and the growth of other fungi (possibly Rhizopus spp.) was also seen during the last two dates. In general, fruit disease occurred at low levels throughout the observation period with
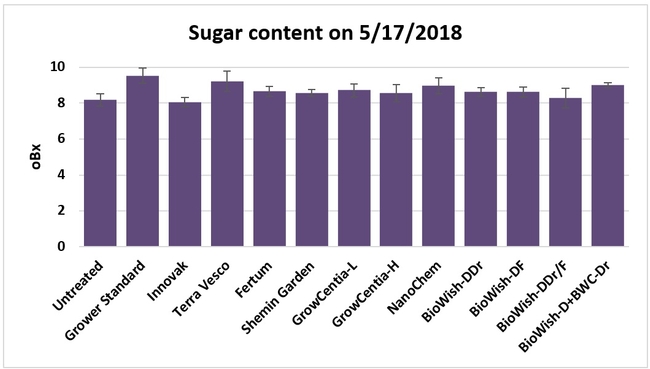
Sugar content in fruit: Sugar content was measured from two harvest-ready berries per bed on 17 May, 2018 using a handheld refractometer. Sugar content varied from 8.06 oBx (Innovak Global regimen) to 9.53 oBx (grower standard).
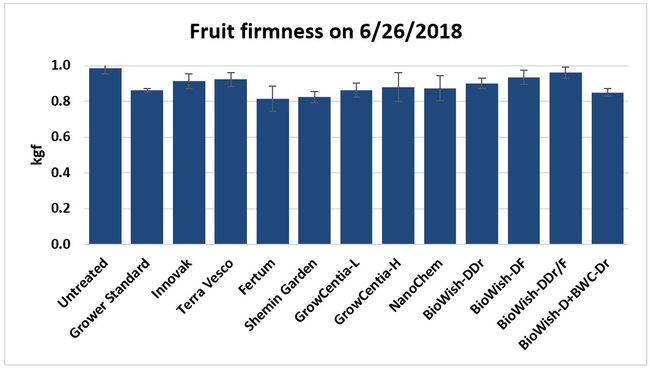
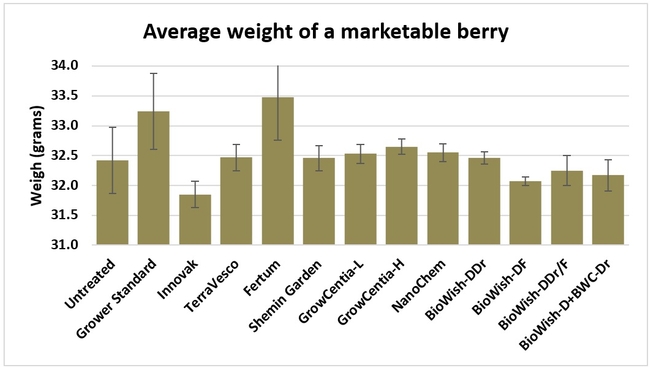
Fruit firmness: Fruit firmness was measured from eight randomly collected harvest-ready berries from each bed on 28 June, 2018. Firmness varied from 0.82 kgf (Fertum and Shemin Garden regimens) to 0.98 kgf (untreated control).
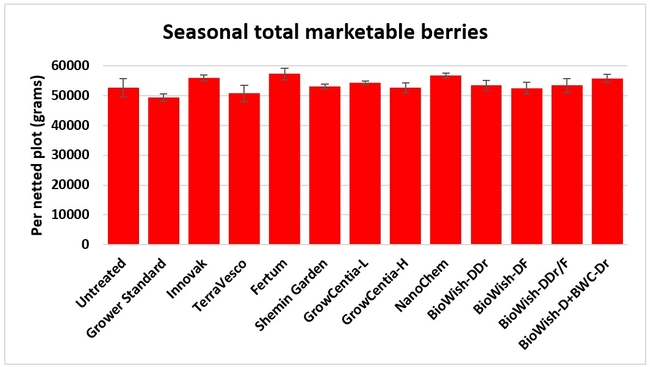
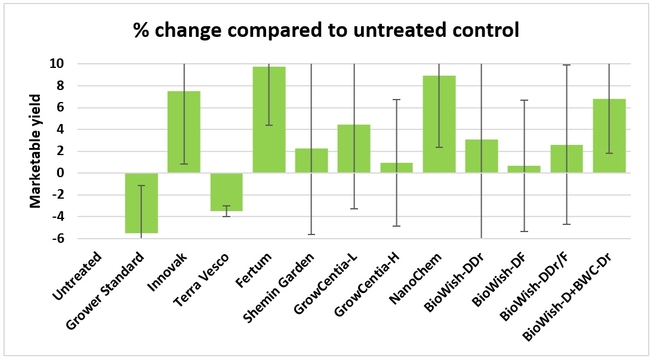
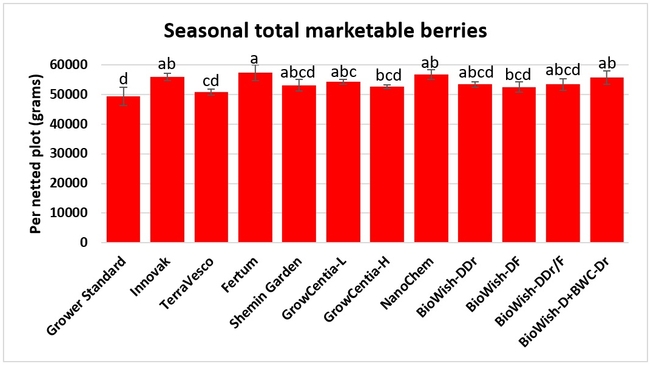
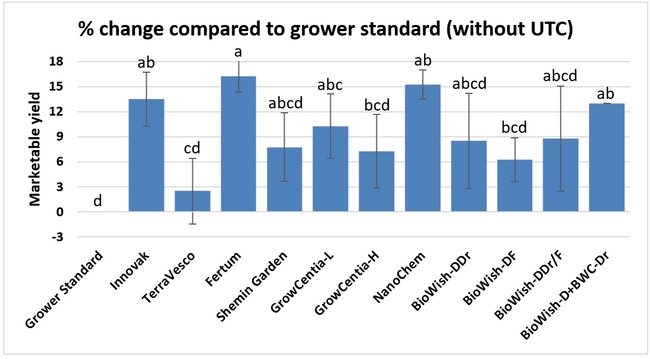
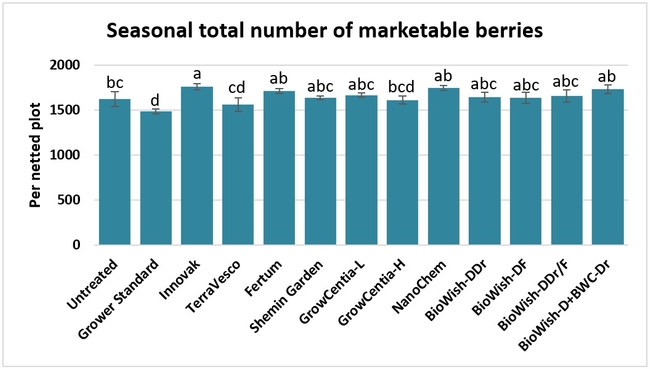
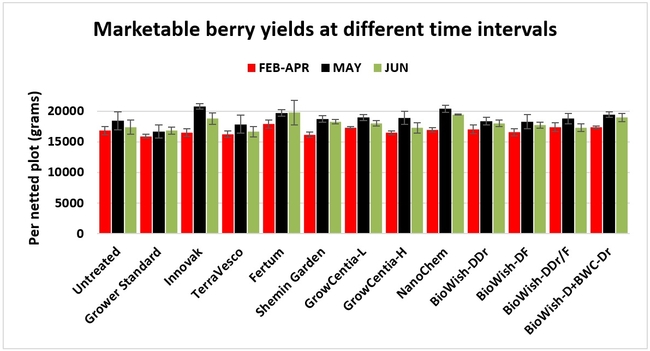
Fruit yield:Strawberries were harvested from 6 February to 22 June, 2018 on 36 dates. When compared to the grower standard, the marketable berry yield was 16.2, 15.1, 13.7, and 13% higher in Fertum regimen, EX10 treatment, Innovak Global regimen, and BiOWiSH regimen 4, respectively. The marketable berry yield was 9.8, 9, 7.5, and 6.8% higher in those respective treatments over the yield from untreated control.
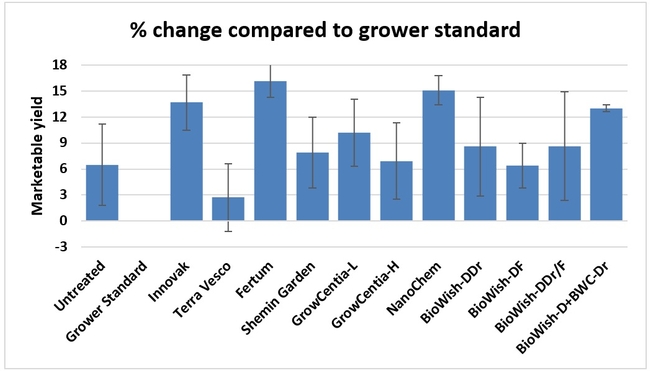
It took 23 harvest dates in three months (from February to April, 2018) to obtain the first third of the total seasonal yield while the remaining two-thirds were obtained from seven harvest dates in May and six dates in June. Marketable fruit yield was higher than the grower standard in all treatments and higher than the untreated control in most treatments.
In general, fruit yields were higher and the pest and disease pressure was lower than usual during the study period. Aleo, a garlic oil based fungicide, at lower label rates was periodically used for disease management and bug vacuums were operated a few times against the western tarnished plant bug as a standard across all treatments.
This study evaluated some treatment regimens as recommended by the collaborating manufacturers and some of them appear to have a potential for use in strawberry production. These results help the manufacturers fine tune their recommendations for achieving better yields through additional studies.
Acknowledgments: We thank the planting and harvest crew at Manzanita Berry Farms for their help with the crop production aspects, Chris Martinez, Tamas Zold, and Maria Murrietta for their technical assistance, Sumanth Dara for statistical analysis, and the support of the industry collaborators who funded the study.