Students, faculty and colleagues are encouraged to take advantage of these opportunities.
Pamela Kan-Rice, Assistant director, news and information outreach, UCANR
Three scholarships are being offered for students studying agriculture.UCANR
Applications and nominations of outstanding students pursuing careers in agriculture will be accepted through May 13, 2024, for UC Agriculture and Natural Resources scholarships and awards.
Students, faculty and colleagues are encouraged to take advantage of these opportunities to honor academic excellence and provide additional support for undergraduate and graduate students.
Bill and Jane Fischer Vegetation Management Scholarship is for students enrolled at ANY accredited California university, with preference given to graduate students. The recipient of the $1,000 (multiple awards possible) will be selected from students who are enrolled in fall 2024 pursuing degrees in vegetation management, weed science or agriculture specializations plant science, soils and plant nutrition, agricultural engineering, agricultural botany, plant pathology, plant protection and pest management, or agricultural economics. Students apply directly.
Howard Walton Clark Prize in Plant Breeding and Soil Building is for students enrolled at UC Berkeley, UC Davis or UC Riverside. The $5,000 (multiple awards possible) will be awarded to a promising student who will be enrolled as a senior in fall 2024 in the College of Agriculture and/or Natural Resources with demonstrated scholastic achievement and talent for independent research with reference to either plant breeding (leading to new/improved crops and new/improved varieties using appropriate tools) or soil building (leading to improving soil quality related to soil productivity and sustainability as a resource). Nomination by faculty member required.
Knowles A. Ryerson Award in Agriculture is for students enrolled at UC Berkeley and UC Davis. $2,500 (minimum one award for each campus) given to an international undergraduate student who will be enrolled in fall 2024 in the College of Agriculture and/or Natural Resources, in any curriculum, preferably after completion of the junior year. The award is based on high scholarship, outstanding character and promise of leadership. Nomination by faculty member required.
More information about the application process can be found on at https://ucanr.edu/anrscholarships.
For questions, contact Andrea Ambrose, UC ANR director of advancement, at apambrose@ucanr.edu.
Source: University of California Division of Agriculture and Natural Resources

- Author: Ben Faber
- Author: Ben Faber
|
The California Department of Food and Agriculture (CDFA) has allocated $5 million of the Healthy Soils Block Grant Program to the California Citrus Quality Council (CCQC) in collaboration with California Farm Bureau (CAFB) and Xerces Society. The California Farm Bureau through its 501(c)(3) science and research nonprofit, the California Bountiful Foundation, will oversee all administrative functions, and financial functions, of this project including reporting and on-farm project verifications. The Xerces Society will provide technical expertise in pollinating plants for hedgerows and other similar management practices.
This grant program supports citrus growers in adopting conservation management practices that improve soil health and sequester carbon. Citrus growers will receive on demand assistance at no cost through the implementation and verification process of their on-farm projects. The program will fund 20-45 on-farm projects over three years, focusing on practices like planting pollinator hedgerows, cover crops, compost application, mulching, whole orchard recycling, and windbreak establishment. Funding for each on-farm project may receive up to $200,000, depending on the project's size and the number of practices implemented.
Enroll Now for the Healthy Soils Block Grant Program
Begin your application by clicking the "Enroll Now" button to submit your online application form.
We can only process applications in English. For Spanish-speaking growers needing assistance with filling out the application in English, please contact Ana Resendiz at aresendiz@ucanr.edu or call (442) 265-7709. View the application in Spanish: here
Got questions? Contact Margaret Honig, Administrative Lead, at (916) 561-5504 or email at mhonig@cfbf.com
Why Citrus Growers?
The citrus sector in California contributes approximately $2 billion annually to the economy and plays a crucial role in job creation and supports agriculture and environmental sustainability efforts. This grant program will help citrus growers in reducing costs associated with conservation management practices, offering insights into effective pest and disease management while protecting pollinators. Citrus growers involved in this program will make meaningful contributions towards pollinator protection, environmental stewardship, climate change resilience, and meet sustainable demands by consumers for citrus production.
|
|

- Author: Ben Faber
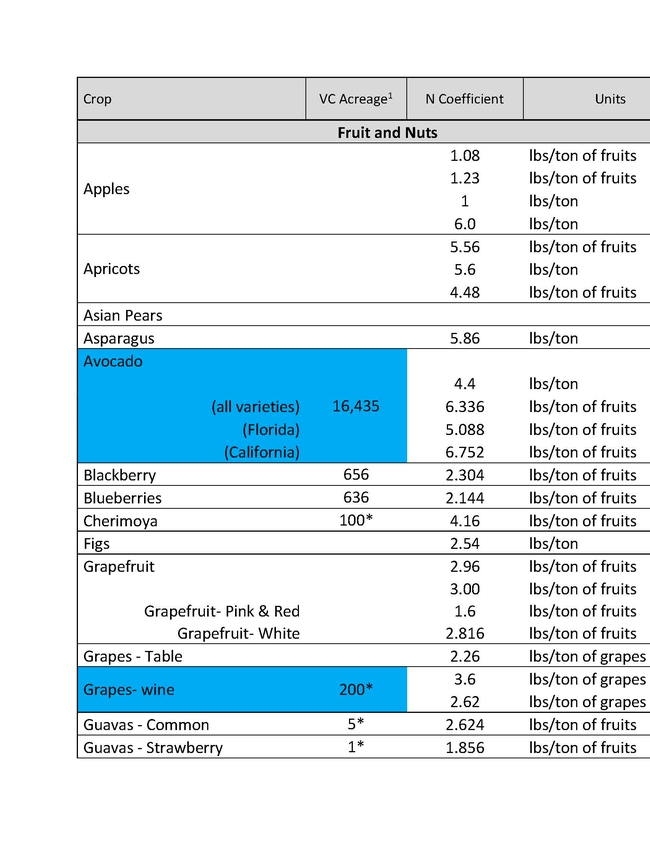
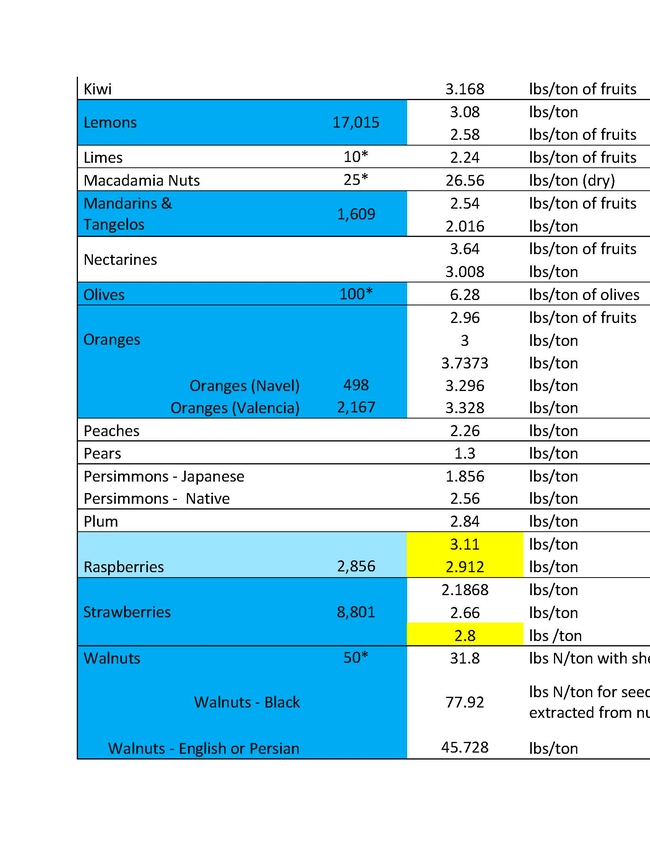
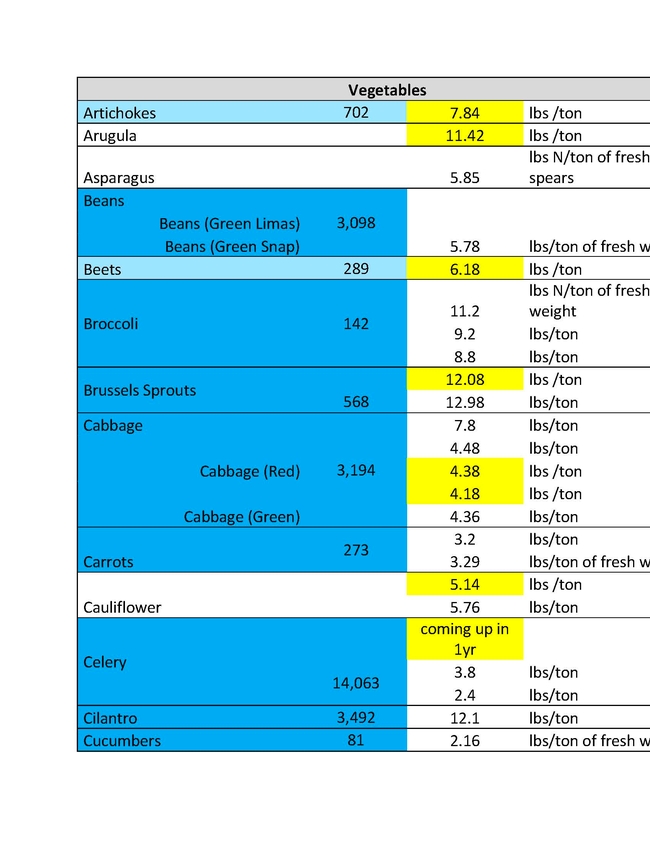
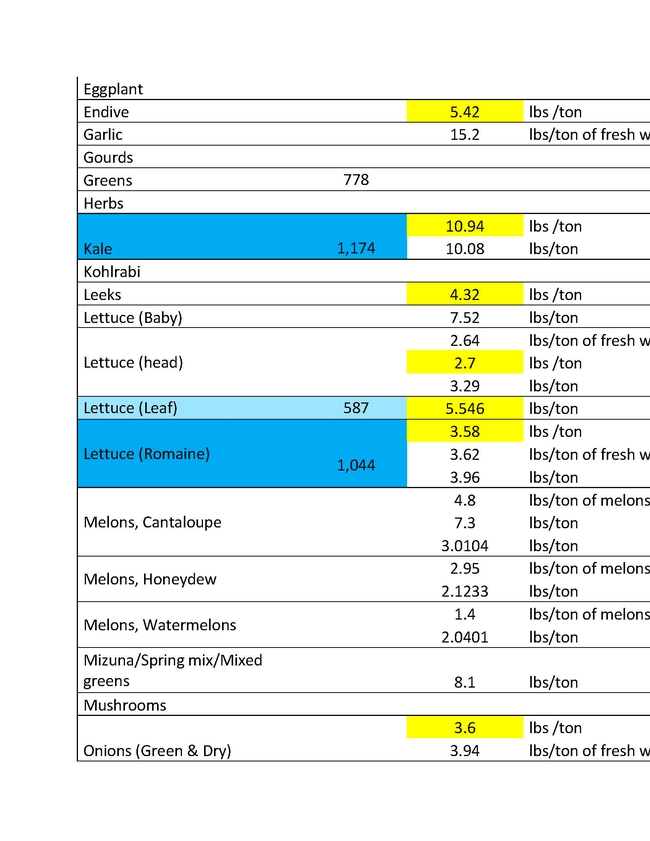
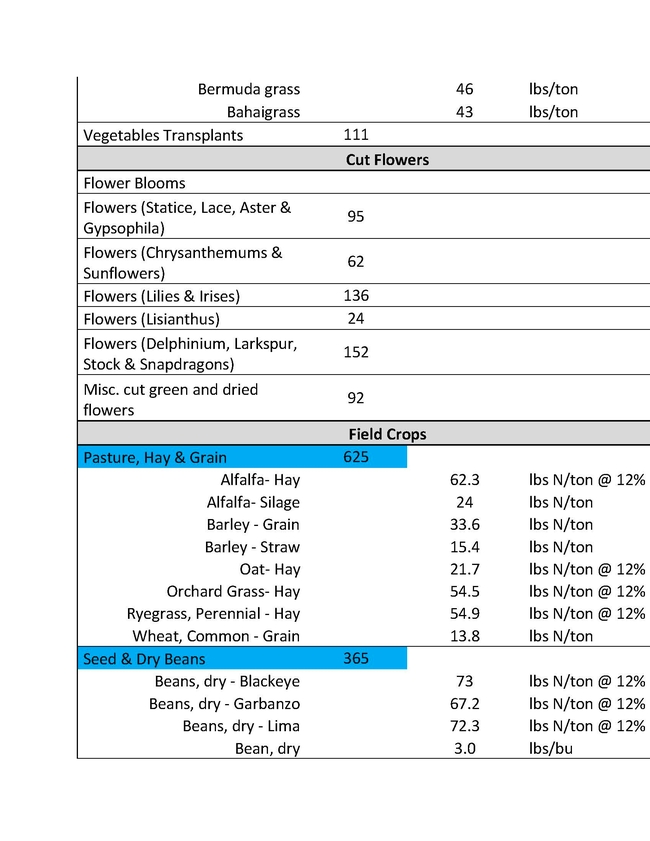
The ratio of N applied to N removed is a key metric in the Central Valley Irrigated Lands Regulatory Program (CVILRP). Growers report applied N and yield to agricultural water quality coalitions. The coalitions in turn convert yield to N removed from fields and report various statistics to the Central Valley Regional Water Quality Control Board. Nitrogen accumulated into perennial plant tissues may also be counted as “removed”. For these calculations, reliable values of N concentrations in the harvested parts and perennial tissues of crops are needed. It's important to know the amounts of N removed by various crops in order to have a better estimate of how to manage N to avoid ground water contamination.
For a report released in 2016, Geissler and team mined the scientific literature for data on N concentrations in harvested crop parts with an emphasis on California data (Geisseler, 2016). For many commodities, a robust dataset of recent samples from California was not available. With financial support from the California Department of Food and Agriculture – Fertilizer Research and Education Program (CDFA- FREP) and the help of the Kings River Watershed Coalition, John Dickey, Ken Miller, and their team at the Southern San Joaquin Valley Management Practices Evaluation Program, a large number of samples were collected and then processed in the author's nutrient management lab at UC Davis. The present report is the second update of the 2016 report, the first being released in March 2021, and includes results for kiwis, lemons, mandarins, oranges, nectarines, and grain sorghum. In addition, recently published data for cotton and N in perennial parts of cherry and citrus trees were included.
This sort of reporting will soon be required in all the Water Quality Control Board Regions. Understanding how much nitrogen to apply is important not only for insuring water quality, but also for tree growth, health and insect and disease management. An expanded list for coastal crops will soon be made available by Richard Smith, Andre Biscaro and Michael Cahn.
You can read the more detailed discussion of the Geissler report HERE.
In the tables presented here "VC Acreage" represents the acreage of a given crop in Ventura County and "N Coefficient" is the amount of that crop removal represented in the different units in the following column. There are different estimates for different crops largely because of varietal differences, soil types and climatic conditions of different growing areas. The values are not exact, but approximations to give growers a better understanding of what a crop would use and therefore how much N to apply.