- Author: Kirsten Pearsons
I have received a handful of calls this season with concerns about “trash bugs,” a catch all term for various soil invertebrates. These soil invertebrates include root maggots, springtails, bulb mites, and symphylans, all which will happily feed on decomposing plant debris, i.e., trash. If given the opportunity, trash bugs will feed on seedlings and transplants, but high pressure can usually be avoided by allowing plant debris to fully breakdown before planting the next crop. A long enough pause between plant debris and seedlings acts as a sort of field-level host-free period, allowing trash bug populations to drop before planting.
Why does it feel like we have more trash bugs this season?
We had an unseasonably cool, wet spring. Under these conditions, crop debris from the fall was breaking down much slower than usual. With the compressed start to the season, folks have been eager to plant into fields as soon as possible, so many were likely planting into fields with higher-than-optimal plant debris.
By mid-April, daily temperatures were starting to get more back to normal, so why are we still dealing with more trash bugs? From talking with a handful of growers and PCAs, the slow start to the season has set back the whole planting schedule. To try and bring things back to schedule as close as possible, folks are still pushing the limit on how quickly they turn around fields for the next crop. A perfect storm for trash bug pressure.
Which trash bug am I dealing with?
A diverse group of pests can cause stunting and stand loss, so accurate pest ID is critical for successful management.
Root Maggots (seedcorn maggots or cabbage maggots, Delia spp.)
Root maggots are the larvae of small grey flies. The adult flies are commonly caught on sticky cards that are deployed for monitoring thrips and aphids.
Seedcorn maggot, Delia platura, seems to be the primary culprit that I have come across that is stunting brassicas. Seedcorn maggots tend to hit fields within a week or two after planting, causing patchy stands and stunted seedlings. Closely related cabbage maggots tend to hit fields a few weeks after planting and can continue to cause damage as plants mature.
To scout for root maggots, pull up stunted plants (see picture #1 below) and check roots for small, yellowish-white maggots (less than ¼ of an inch long). They can be easy to miss when they're all tangled up in the roots (picture #2). If scouting is delayed, you may also find the brown pupa (picture #3). With seedcorn maggots, timing a reactive control (i.e., insecticide application) can be tricky since the damage may go unnoticed until the maggots have already pupated and left the field.
|
From left to right: #1 broccoli stunting caused by root maggot feeding; #2 maggot tangled up in the roots of a broccoli plant; #3 root maggot pupa found next to a stunted broccoli plant. |
Springtails and soil mites
Pest springtails and soil mites are trickier to identify, since non-pest species are common in healthy soils. The one species of springtail that has been identified as an occasional pest of lettuce and brassicas is Protaphorura fimata, while the main pest mites are bulb mites (Rhizoglyphus spp., Tyrophagus spp.). Both P. fimata and bulb mites are small and whiteish while non-pest species are often larger and more colorful.
|
Clockwise from top left; Protaphorura fimata, the springtail species that can be a pest (Photo Credit: Shimat Joseph, previous UC IPM Advisor); non-pest springtails (note that these springtails are more colorful and have long antennae); a predatory soil mite (not a pest); and two bulb mites (Photo Credit: Jack Kelly Clark, UC IPM). |
Symphylans
Symphylans look like small (less than ½ an inch), white centipedes. Since symphylans are fast and mobile, you will be more likely to catch symphylans in action if you dig around stunted plants that are near healthy looking plants. If you readily find symphylans just by digging, the symphylan pressure is probably high enough to cause economic damage.
Lower symphylan densities can be harder to observe, so you may want to try bait-trapping with potato or beet wedges if you suspect symphylans but cannot find any by digging around stunted plants. The bait-trapping method can also be used to scout for springtails.
For more on the identification, scouting, and management of these pests, be sure to reference the UC IPM Pest Management Guidelines:
https://ipm.ucanr.edu/agriculture/cole-crops/
https://ipm.ucanr.edu/agriculture/lettuce/
Related blog posts from past entomology advisors:
Root maggots:
https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=9804
https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=4301
Symphylans:
https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=18819
Springtails:
https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=16769
Bulb Mites:
https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=13271
- Author: Shimat Villanassery Joseph
Springtail (Protaphorura fimata) (Figure 1) is a serious pest of lettuce in the northern part of Salinas Valley of California. The direct seeded young lettuce seedlings in fields with high densities of springtail show retarded or stunted growth and do not emerge in a synchronous pattern (Figure 2). Springtails are reported to feed on soil fungi, decaying plant materials and live roots.
Springtails attack the germinating seeds of the lettuce, but it is not certain if irregular or inconsistent plant stand is due to the persistent feeding by springtail on both germinating and developing seedling stages of lettuce. Springtail continue to occur in the soil beyond 30 days after planting. Knowing the most vulnerable stage(s) of lettuce to springtail feeding will help in determining the best timing for control measure intervention to achieve a uniform lettuce stand.
Lettuce fields are heavily irrigated at least once before and up to three weeks after planting the seeds for uniform seed germination and seedling establishment. However, the behavioral response of springtail to feeding injury on lettuce under high soil moisture condition has not yet been studied in the central coast of California.
Similarly, the temperature has a profound impact on lettuce plant development as well as the growth and activities of springtail. Springtail has been found causing crop losses during February to May in the Salinas Valley and beyond June, springtail related problems are not widely reported. Perhaps slower lettuce seed germination and subsequent development during cooler seasons (January to May) is the pre-disposing factor as seedlings are exposed to springtails for an extended time frame than during the rest of the year. The relationship between temperature and springtail feeding of germinating lettuce seeds has not been investigated.
A study was conducted to determine the effect of germinating stages of lettuce seeds (up to 7 days after planting), soil temperature and moisture to springtail feeding injury.
Germinating seeds or one day old lettuce seedlings were the most vulnerable stage to springtail feeding, resulting in reduction in seedling growth. Thus, it appears that once the roots are established in the soil, lettuce is less susceptible to springtail feeding injury. Because the germinating phase of the plants is more likely to be injured, springtail monitoring activity should start prior to planting the seeds to determine the presence of springtail in the soil. Previous studies showed that, beet or potato slice baits attract springtail if placed in the top layer of the soil; thus, these baits could be used for monitoring springtail activity in the soil. If the soil is not moist, the baits may not capture springtail and springtail activity may go undetected.
When the experiments were conducted with germination phase in the temperatures as low as 41°F, springtail feeding was still evident. This suggests that although the seed germination and seedling development is progressing slowly in the cooler temperatures, springtail can be still active in feeding if there is sufficient moisture in the soil. Also, this suggests that lettuce seedlings might require prolonged protection from springtail with additional insecticide sprays until the seedlings are established in the cooler temperatures especially in spring and early summer (January to May). In the later part of summer and fall, the temperatures are higher than 60°F even at nights, which allows the seeds to germinate and develop quickly and not providing springtail to persistently feed and cause economic damage. In these circumstances, an at-plant application of insecticide is likely to provide adequate springtail control and multiple applications may not be required.
High moisture content in the soil will favor springtail feeding on the germinating lettuce seeds. In the Salinas Valley, before the lettuce seeds are planted, fields are pre-irrigated to aid land preparation and bed shaping. It has been observed that the springtail density increased from the sub-surface of soil when the field was recently irrigated or after a rain event. This cultural practice which maintains high moisture levels for seed germination on the sub-surface profiles of the soil might be favoring the faster buildup of springtail populations. Springtail captures in bait traps were greater immediately after irrigation.
Clearly, this study demonstrates that early lettuce seed development stages are the most vulnerable to springtail feeding injury. Moisture has a profound effect on springtail feeding on germinating lettuce seeds. This study also suggests that springtail can attack the germinating lettuce seeds at all growing temperatures in the Salinas Valley, although the seed germination and subsequent seedling development at cooler temperatures would increase the vulnerability of lettuce seeds to springtail feeding. This information provides insights not only on the timing of protection but the extent of protection under various temperature ranges also in managing springtail in the Salinas Valley. Plants growing the cooler temperature need prolonged protection for springtail if adequate moisture is present in the top soil of the bed. In the warmer temperatures, seed development would occur rather quickly which suggests that prolonged protection against springtail is not necessary. These results warrant the need for more field studies on protecting lettuce seeds from springtail in the cooler temperatures especially during spring and early summer lettuce plantings in the Salinas Valley.
If interested in the details of the study, please read the published article:
http://cemonterey.ucanr.edu/files/253284.pdf
- Author: Shimat Villanassery Joseph
A series of laboratory and field studies were conducted to determine if the insecticides coated lettuce seeds are an option to control key lettuce pests in the Salinas Valley: springtail (Protaphorura fimata; Fig. 1A), leafminers (Liriomyza spp.; Fig. 1B) and western flower thrips (Frankliniella occidentalis; Fig. 1C). In addition, a laboratory test was conducted to determine if “primed” lettuce seeds reduced springtail feeding damage.
Springtails. Springtail (P.fimata) is soil dwelling primitive arthropod primarily attacks germinating lettuce seeds, reducing the plant vigor or death, which cause patchy or area-wide stand loss. Most springtails possess a forked organ (furcula) in the rear-end, which is extended forward and backward to jump; hence, the common name, springtail. However, the springtail species, sampled from lettuce fields causing the stand loss, does not have furcula. This means it cannot jump.
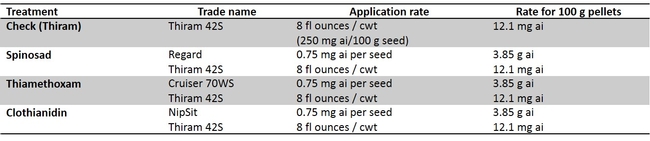
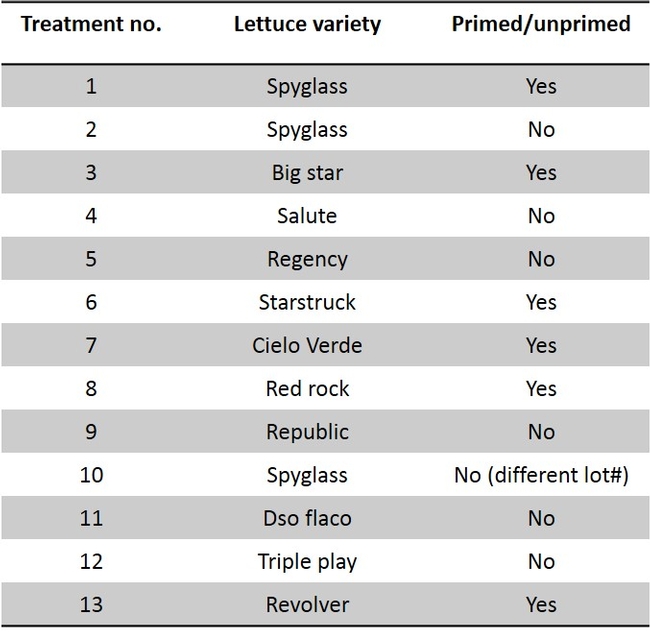
The head lettuce seed ‘Regency' was coated with clothianidin, thiamethoxam, and spinosad (Table 1). The seeds were coated by Dr. Alan Taylor at Cornell University and coating technique mirrored commercial seed coating procedure. Laboratory studies were conducted in containers with springtail (P. fimata) infested soil. The data show that all three insecticides spinosad, clothianidin and thiamethoxam treated seeds significantly reduced the incidence of springtail feeding injury when compared with untreated seeds. Among insecticides, superior performance in efficacy was noted in the following order: clothianidin > thiamethoxam > spinosad (Fig. 2). Two field trials were conducted against springtails using the same seed treatments, however, the springtail pressure was so low that conclusive data were not obtained. Clothianidin (NipSit) in particular, is now registered on head lettuce and could be used for springtail control. This is an important information in that springtails attack the germinating seeds of lettuce especially in the spring time. During spring, we get some rain showers and the wet conditions in the field after planting makes insecticide application along seed line almost impossible. If the insecticide coated seeds are planted, the grower or PCA could avoid at-plant insecticide application which is typically targeted toward springtails. Application of insecticides such as neonicotinoids and pyrethroids along the seed line will protect the germinating seeds from springtail feeding. More field studies will be conducted in the following years to validate these results in the field.
Studies were also conducted to determine if there are any varietal effects exists (Table 2). The much needed attribute for springtail control is faster seed germination so that the springtail would not get sufficient time to feed and cause seed mortality. “Primed” lettuce seeds are used for uniform and a quick germination (cut short 2 to 3 days than “unprimed” seeds). “Primed” and “unprimed” seeds were evaluated to determine if the quick seed development would reduce springtail damage. Data show that germinating “primed” seeds were impacted with springtail feeding affecting their germination and were not different from the “unprimed” seeds when the springtail pressure was moderate to high (Fig. 3). The seeds used for this experiment were from same seed lot (“primed” and “unprimed”) for a lettuce variety. Also, there was no clear variety difference in springtail feeding damage.
Leafminers and western flower thrips. The leafminer eggs are laid within the surface layer of the leaf. The eggs hatch within couple of days and the maggots mine through the surface layer of the leaves. The egg laying and maggot mining creates stippling and mining injuries which make the leaves unmarketable. Although UC recommends few insecticides such as Agri-mek (Abamectin), Trigard (Cyromazine), Aza-direct (Azadirachtin) and Entrust (Spinosad), the management of leafminers are primarily relied upon on Agri-mek applications.
Thrips is another of the major pest of lettuce, and combination of direct feeding injury and viral disease [thrips-transmitted tospoviruses [Impatiens Necrotic Spot Virus (INSV)] can cause significant losses in lettuce production. In addition, because most of the export markets have set higher standards on prevalence of live and dead thrips in the produce, the lettuce industry is constantly battling ways to significantly reduce thrips in the produce targeted for export.
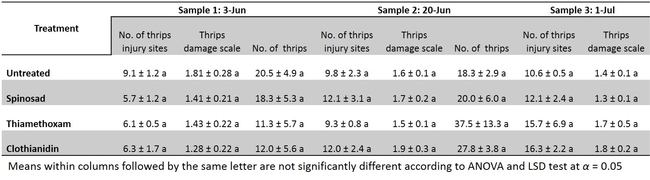
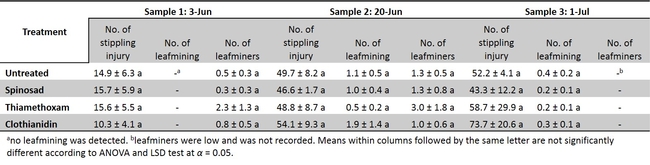
A replicated-field trial was conducted to determine the efficacy of seed coated insecticides (Table 1) on leafminers and western flower thrips incidence and their infestation. The results show that insecticide seed coating may not be an effective option for leafminers and thrips control in head lettuce (Table 3 and 4) under the conditions this experiment was conducted. There was no reduction of leafminer or thrips feeding with insecticide coated seeds compared with untreated control. Further evaluations under varying conditions might be necessary to validate the consistency of these results.

- Author: Shimat Villanassery Joseph
The soil-dwelling springtail (Protaphorura fimata) is a serious pest of lettuce in the Salinas Valley of California. This springtail is ~ 2.5 mm long and white and has no jumping organ (furcula) and eyes. They feed primarily on the radicle of the germinating lettuce and broccoli seeds causing stunted seedling growth, and their high densities at planting is associated with poor lettuce stands. Most springtails are saprophytic feeders considered as beneficial organisms because they aid in the decomposition of decaying plant material, thereby contributing to the cycling of carbon and nitrogen, which in turn improves soil health and structure. These springtails typically known to feed on fungi in the soil.
There is no grower-friendly monitoring method to determine incidence and abundance of springtails in the lettuce and broccoli fields of California's central coast. Garden symphylan another soil-borne arthropod pest that occurs in vegetable fields, is commonly monitored using a potato slice bait. Potato slices are typically deployed on the soil surface, and they attract garden symphylan. The utility of potato slices for captures of springtail has not been investigated. Because potato slices are off-white in color, it might be difficult to quickly quantify lightly-colored arthropods such as springtail and garden symphylan on the potato surface. Beets are dark red in color and may provide background contrast and to help quantify lightly-colored organisms on it.
Recent research suggest that beet and potato attract springtail when placed in lettuce fields and these baits could be used to monitor in commercial field settings. It could only provide the presence or absence information, which might be still important to avoid unnecessary insecticide application with no or low captures on baits. Capture of springtails on the baits suggests that their populations are likely developing in the upper soil profile especially near the root system. Also, the incidence and abundance of springtail is subjected to soil moisture and their captures likely decline sharply as the upper soil layer loses moisture. It is likely that springtail populations multiply as the pre-plant irrigation is initiated. Monitoring the field using baits before or during the initial irrigation may provide an indication of active springtail population in the upper soil profile. Study also show that greater numbers of springtails were collected on beet one day after deployment than extended days of exposure. Perhaps deployment of baits for extended periods of time in soil may cause desiccation and make them less attractive to springtails.
Therefore, beet and potato can attract springtail in the soil and could be used for monitoring springtail.
If you are interested to read further on the published article, click the link below.
http://cemonterey.ucanr.edu/files/230154.pdf

- Author: Shimat Villanassery Joseph
Growers in the Salinas Valley facing an irregular lettuce stand are usually uncertain about what caused the problem and often blame the factors such as poor seed quality, planting error, irregular irrigation timing or distribution, high salt levels in the soil or water, soilborne pathogens of seedlings, bulb mites, and garden symphylan feeding for the losses. Several springtails were collected from the soil associated with lettuce and it is not clear if they were feeding and contributing to the irregular lettuce stand. Often, this springtail is misidentified as garden symphylan.
Recently, I found a large number of a subterranean springtail (Protaphorura fimata) (Fig. 1) in the monitoring potato slice traps deployed in Salinas lettuce fields. I did a series of laboratory and field studies to determine its pest status in lettuce. This springtail (P. fimata) is less than 2.5 mm long, white in color and lacks eyes. Unlike other springtails, this springtail lack a furcula (jumping organ), and when disturbed it does not jump instead curls up. Other similar species of springtails primarily reproduce parthenogenically meaning they reproduce without mating; however, sexual reproduction is also seen on this one (P. fimata). This species (P. fimata) seems to be widely distributed in Europe, but has not been previously reported from the U.S.
Springtails occur in diverse habitats worldwide and are generally considered as beneficial arthropods because they aid in the decomposition of decaying plant material by feeding, thereby contributing to the cycling of carbon and nitrogen which in turn improves soil health and structure. This springtailis primarily known to feed on soil fungi but also feeds on live plant roots. Other springtails in the same family have been associated with feeding damage to germinating sugar beet seeds, sugarcane, poppy seeds and weed seeds (Plantago major). Foliage-feeding springtails (lucerne flea and garden springtail) attack several plants including Lucerne (Medicago sativa), clover (Trifolium sp.), sugar beet (Beta vulgaris) and bean.
The major objectives of the present study were to document the ability of springtail (P. fimata) to injure germinating seeds of lettuce in laboratory and field and characterize the feeding injury of springtail on germinating seeds and seedlings of lettuce.
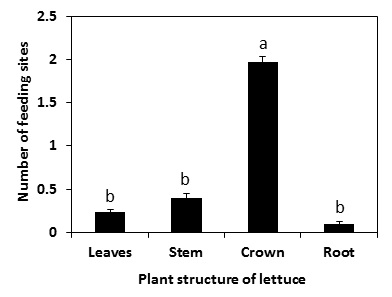
In the laboratory, I conducted experiments in plastic petri dish with and without soil and sprigtails. Then I recorded the ungerminated lettuce seeds due to feeding injury, total number of feeding injury sites, and number of germinated seedlings with distinct feeding injury. I also documented the location (e.g. leaf, stem, plant crown or root) of the feeding injury on the plants. I conducted a field study too. The assumption of the field study was that the repeated use of maximum label rate of selected insecticides at early stages of plant development would suppress springtails and protect the seeds or seedlings from feeding. Two commonly used pyrethroid insecticides were applied (by the grower) three times: 2 days before planting, at planting, and 20 days after planting. Applications were made using a commercial tractor mounted sprayer. Two pyrethroid insecticides used were Mustang (4 fl oz per acre) and Warrior II (1.6 fl oz per acre). Both the Mustang and Warrior II were tank mixed and applied at 2 days before planting and 20 days after planting but only Warrior II was applied at planting to conform to the label. An adjuvant, Widespread Max (2 fl oz per acre) was added with all the applications. I used bait slices to monitor the springtails at weekly intervals. They were placed in the soil at 1.5 inches deep along the seed line and were covered with disposable white plastic bowls. At the end of each 2 days exposure period, beet root slices were removed, placed into plastic bags and transported to the laboratory. In addition, plant samples were collected to assess the plant growth. Please read the full Journal article for details.
Results demonstrate that this springtail (P. fimata) can feed on germinating lettuce seeds or young seedlings, resulting in reduction in lettuce growth (Figure 2).Springtails attacked seeds and young seedlings alike. In the laboratory, springtails directly fed through the seed coat (pericarp) of a few seeds. This is possibly due to the moistening of the pericarp, enabling springtails to feed through the softened coating (Fig. 3a and b). In some instances, springtails fed on the growing radicle of the germinating seeds (Fig. 3c). However, most of the feeding at the seed radicle or elsewhere did not entirely sever it (Fig. 3d), which allowed the seedling to survive but affected the normal development of the plant. Moreover, most of the feeding injury was evident at the crown area rather than on leaf, stem, or root (Fig. 4).
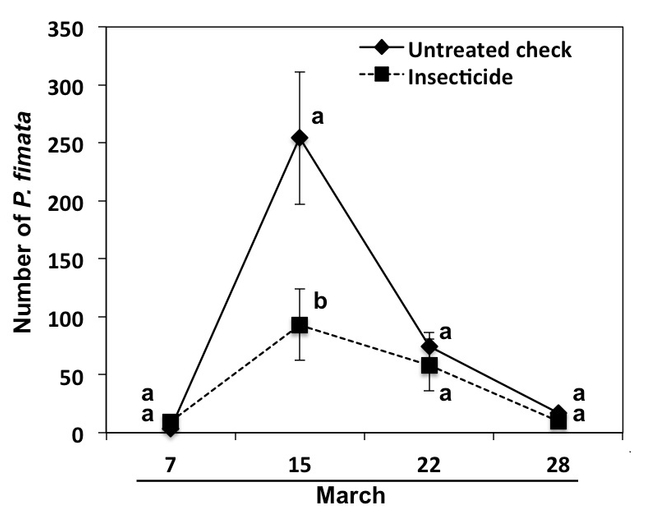
In the Salinas Valley, before the lettuce seeds are planted, fields are watered deeply and irrigations continue for at least three weeks after planting. I observed that the springtaildensity increased when the field was recently irrigated or after a rain event. This cultural practice which maintains high moisture levels for seed germination on the sub-surface profiles of the soil might be favoring faster buildup of springtailpopulations. In the field trial, the springtailcaptures were greater immediately after irrigation in the untreated beds than insecticide treated beds which was reflected in reduced number of springtails and in the untreated beds (Fig. 5).
In conclusion, this study clearly demonstrates that springtail (P. fimata) is an important pest of lettuce and is capable of reducing the crop stand. Incidence of high populations of springtail could be detrimental to germination of seeds in the field (Fig. 6 and 7). Springtails could be effectively suppressed to a large extent with early applications of insecticides directed to the seed line. Monitoring is the key to determine the presence and population size of springtail. Currently, I'm conducting for laboratory and field studies to determine the efficacy and application timing of insecticides.
Again, please use the link below to read the full article.
http://cemonterey.ucanr.edu/files/206762.pdf