- Author: Yu-Chen Wang
2024 Pest Management Meeting
Sponsor: UCCE Monterey County
Date: Thursday, November 14, 2024
Location: Agricultural Center Meeting Room (1432 Abbott St, Salinas CA)
Time: 7:55 am-12:15 pm
Pre-registration is encouraged:
https://surveys.ucanr.edu/survey.cfm?surveynumber=43793
Agenda:
7:55 Introductions
8:00 2024 Plant disease update
Yu-Chen Wang, Plant Pathology Advisor, UC Cooperative Extension, Monterey
8:30 Soil disinfestation with steam for weed and disease control
Steve Fennimore, Extension Vegetable Weed Specialist, UC Davis
9:00 Lettuce breeding for disease resistance
Kelley Richardson, Research Geneticist, USDA-ARS, Salinas
9:30 Pest detection & delimitation 2023-2024
Casey McSwiggin, Deputy Agricultural Commissioner, Monterey County
10:00 Break
10:15 Update on spinach downy mildew and INSV on spinach
Jim Correll, Professor of Plant Pathology, University of Arkansas
10:45 Soilborne pathogens of lettuce and research updates on Fusarium wilt
Alex Putman, Plant Pathology Extension Specialist, UC Riverside
11:15 2024 Update on thrips and INSV
Daniel Hasegawa, Research Entomologist, USDA-ARS, Salinas
11:45 Management of insect pests in lettuce and cole crops - research update
Ian Grettenberger, Extension Entomology Specialist, UC Davis
Addie Abrams, Graduate Student Researcher, UC Davis
4.0 DPR credits have been requested.
For more information, contact the UCCE Office @ 831-759-7350 or email Yu-Chen Wang (yckwang@ucanr.edu )
- Author: Kirsten Ann Pearsons
We are getting to the time of year where lettuce production winds down in the Salinas Valley and ramps up in the desert around Yuma, Arizona. Unlike in the Salinas Valley, the desert has not been hit as hard by Impaciens necrotic spot virus (INSV), the virus that is transmitted to lettuce by Western flower thrips (F. occidentalis). When the virus does show up in the desert, the primary infection can often be traced back to INSV-infected thrips that arrived on vegetable transplants from coastal California (Palumbo, 2022)
These finding stirred up concern in the local ag community – could vegetable transplants also be a significant source of new INSV infections in the Salinas Valley? There are instances where recently transplanted fields start showing INSV symptoms soon after planting (Figure 1), but it is challenging to nail down if they were infected before or after transplanting. After gaining some advice from John Palumbo of the University of Arizona (who has been spearheading the ISNV work in the desert), I set out to sample thrips from local transplant nurseries.
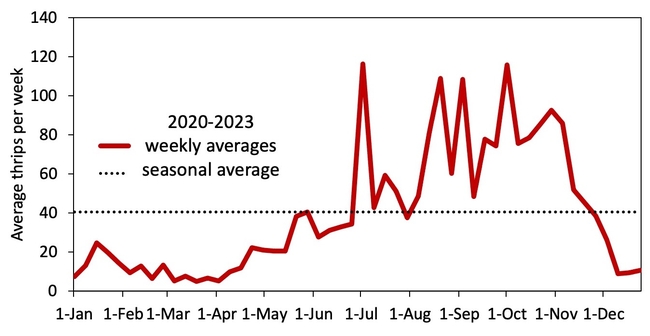
Hypothesis: Vegetable transplants are NOT a major source of INSV-infected thrips in the Salinas Valley. Although this hypothesis may seem contrary to the findings from Yuma, it all comes down to timing of transplant production and background levels of thrips and INSV. As summarized in Figure 2, weekly thrips activity is low in spring and early summer but high in late summer and early spring. [check out an interactive version of these data: Salinas Valley Lettuce Pest Mapping Tool]. This difference in activity somewhat corresponds to when transplants are grown for local use (spring and early summer) versus desert use (late summer and fall). Transplants that are sent to Yuma are grown when thrips populations and INSV pressure has been historically high – putting them at higher risk of carrying INSV+ thrips.

Species confirmation and virus testing: I collaborated with Daniel Hasegawa (USDA-ARS) for species confirmation and virus testing. Daniel and his team used a genetic testing method called multiplex RT-qPCR to determine if the adult and larval thrips were 1) Western flower thrips (F. occidentalis), 2) Onion thrips (Thrips tabaci), or 3) some other thrips species AND if the thrips were carrying 1) INSV or 2) Tomato spotted wilt virus (TSWV, similar to INSV). For small samples (five or fewer thrips), Daniel's team tested each thrips individually. For samples with more than five thrips, they tested half of the thrips as a pooled sample. If this sub-sample tested positive for INSV, a subset of the remaining thrips were tested individually.
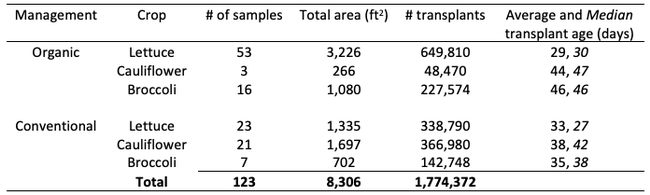
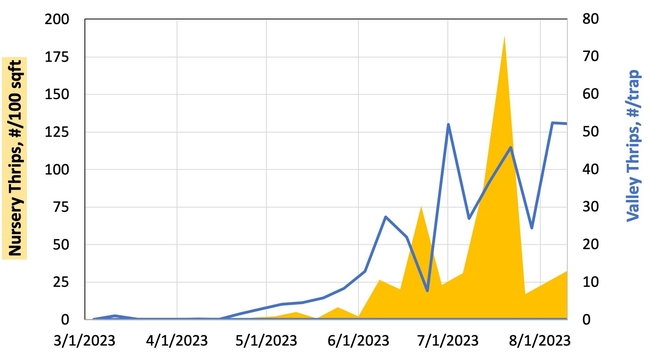
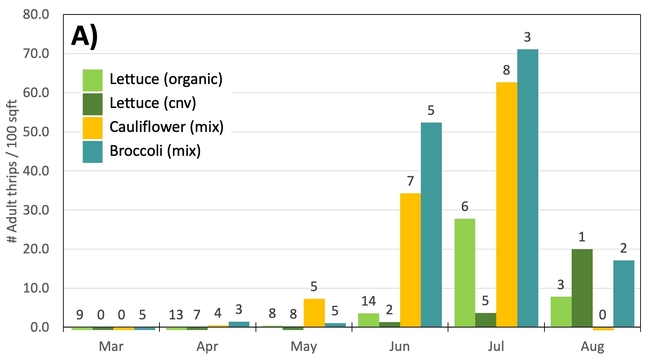
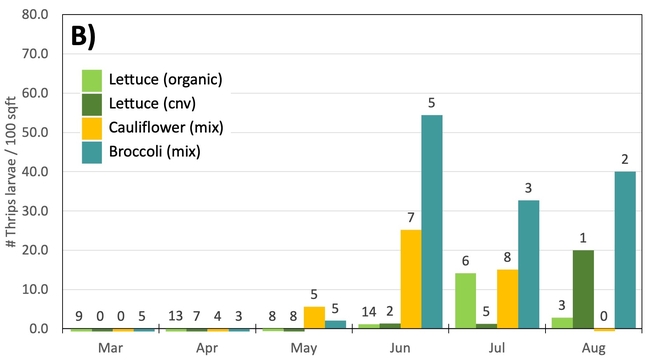
Results: From March through May, we only collected 52 thrips, 50 of which were identified as western flower thrips and none which tested positive for INSV or TSWV. In June, collection shot up almost 10-fold and doubled again in July, somewhat tracking the thrips population increase in the valley (Figure 4). In June and July, thrips densities were consistently higher on brassica transplants compared to lettuce transplants (Figure 5).
Despite collecting over 2,000 individual thrips (1,333 adults and 782 larvae), only two samples, or 1.64% of the tested thrips, tested positive for INSV. The two samples which has positive hits for INSV were collected in June, one off or organic broccoli and one off of conventional cauliflower. None of the thrips collected off of lettuce transplants tested positive for INSV.
Preliminary Conclusions: For the Salinas Valley, the thrips populations on transplants appear to mirror the thrips population in the valley. This is perhaps not too surprising since most transplants are grown on uncovered benches near crop fields. Despite lettuce being a better host for Western flowerthrips (Joseph & Koike, 2021), we consistently collected morethrips frombrassica transplants. We attribute this difference to management practices (e.g., insecticide applications on lettuce transplants) and because brassica transplants tend to stay at nurseries for an additional week.
With this in mind, we may expect that transplanted lettuce fields in the Salinas Valley are more likely to be infected by INSV+ thrips coming in from nearby areas (the prior crop, nearby fields, or weedy areas) rather than thrips from transplant nurseries. Although I would expect to find more INSV+ thrips on vegetable transplants in a year with higher INSV incidence (2023 incidence was considerably lower than prior years), the risk of INSV+ thrips coming in from other sources would increase as well.
For the desert, however, the background risk of INSV is much lower because INSV levels drop off over the summer. In this context, even a handful of INSV+ thrips on transplants pose a proportionally greater risk than in the Salinas Valley. Recognizing this risk to growing regions beyond the Salinas Valley, it is important to continue monitoring INSV levels in transplant nurseries and to work with nurseries to minimize the risk of transporting INSV+ thrips.
References:
- Palumbo, JC, 2022. Thrips and INSV Management in Desert Lettuce. University of Arizona VegIPM Update, Vol 13, No 22, Nov 2, 2022. https://acis.cals.arizona.edu/docs/default-source/agricultural-ipm-documents/vegetable-ipm-updates/2022/thrips-and-insv-management-in-desert-lettuce.pdf?sfvrsn=3088b8b9_2
- Joseph, SV, Koike, ST, 2021. Could Broccoli and Cauliflower Influence the Dispersal Dynamics of Western Flower Thrips (Thysanoptera: Thripidae) to Lettuce in the Salinas Valley of California? Environmental Entomology 50, 995–1005. https://doi.org/10.1093/ee/nvab050
Thanks to Daniel Hasegawa and the entire Hasegawa lab (USDA-ARS, Salinas); John Palumbo (University of Arizona); Kevin Costa, Thomas Costa, and Manuel Aguirre (Headstart Nursery); Francisco Castaneda and Omar Saenz (Growers Transplanting); Lupe Guillen, Maria Alfaro, Alejandro Palma-Carias, and Jim Wilkinson (Dole Fresh Vegetables); Jasmine Rodriguez; Luis Ramirez-Espinoza (CSU-MB); and the California Leafy Greens Research Board.
- Author: Kirsten Pearsons
I have received a handful of calls this season with concerns about “trash bugs,” a catch all term for various soil invertebrates. These soil invertebrates include root maggots, springtails, bulb mites, and symphylans, all which will happily feed on decomposing plant debris, i.e., trash. If given the opportunity, trash bugs will feed on seedlings and transplants, but high pressure can usually be avoided by allowing plant debris to fully breakdown before planting the next crop. A long enough pause between plant debris and seedlings acts as a sort of field-level host-free period, allowing trash bug populations to drop before planting.
Why does it feel like we have more trash bugs this season?
We had an unseasonably cool, wet spring. Under these conditions, crop debris from the fall was breaking down much slower than usual. With the compressed start to the season, folks have been eager to plant into fields as soon as possible, so many were likely planting into fields with higher-than-optimal plant debris.
By mid-April, daily temperatures were starting to get more back to normal, so why are we still dealing with more trash bugs? From talking with a handful of growers and PCAs, the slow start to the season has set back the whole planting schedule. To try and bring things back to schedule as close as possible, folks are still pushing the limit on how quickly they turn around fields for the next crop. A perfect storm for trash bug pressure.
Which trash bug am I dealing with?
A diverse group of pests can cause stunting and stand loss, so accurate pest ID is critical for successful management.
Root Maggots (seedcorn maggots or cabbage maggots, Delia spp.)
Root maggots are the larvae of small grey flies. The adult flies are commonly caught on sticky cards that are deployed for monitoring thrips and aphids.
Seedcorn maggot, Delia platura, seems to be the primary culprit that I have come across that is stunting brassicas. Seedcorn maggots tend to hit fields within a week or two after planting, causing patchy stands and stunted seedlings. Closely related cabbage maggots tend to hit fields a few weeks after planting and can continue to cause damage as plants mature.
To scout for root maggots, pull up stunted plants (see picture #1 below) and check roots for small, yellowish-white maggots (less than ¼ of an inch long). They can be easy to miss when they're all tangled up in the roots (picture #2). If scouting is delayed, you may also find the brown pupa (picture #3). With seedcorn maggots, timing a reactive control (i.e., insecticide application) can be tricky since the damage may go unnoticed until the maggots have already pupated and left the field.
|
From left to right: #1 broccoli stunting caused by root maggot feeding; #2 maggot tangled up in the roots of a broccoli plant; #3 root maggot pupa found next to a stunted broccoli plant. |
Springtails and soil mites
Pest springtails and soil mites are trickier to identify, since non-pest species are common in healthy soils. The one species of springtail that has been identified as an occasional pest of lettuce and brassicas is Protaphorura fimata, while the main pest mites are bulb mites (Rhizoglyphus spp., Tyrophagus spp.). Both P. fimata and bulb mites are small and whiteish while non-pest species are often larger and more colorful.
|
Clockwise from top left; Protaphorura fimata, the springtail species that can be a pest (Photo Credit: Shimat Joseph, previous UC IPM Advisor); non-pest springtails (note that these springtails are more colorful and have long antennae); a predatory soil mite (not a pest); and two bulb mites (Photo Credit: Jack Kelly Clark, UC IPM). |
Symphylans
Symphylans look like small (less than ½ an inch), white centipedes. Since symphylans are fast and mobile, you will be more likely to catch symphylans in action if you dig around stunted plants that are near healthy looking plants. If you readily find symphylans just by digging, the symphylan pressure is probably high enough to cause economic damage.
Lower symphylan densities can be harder to observe, so you may want to try bait-trapping with potato or beet wedges if you suspect symphylans but cannot find any by digging around stunted plants. The bait-trapping method can also be used to scout for springtails.
For more on the identification, scouting, and management of these pests, be sure to reference the UC IPM Pest Management Guidelines:
https://ipm.ucanr.edu/agriculture/cole-crops/
https://ipm.ucanr.edu/agriculture/lettuce/
Related blog posts from past entomology advisors:
Root maggots:
https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=9804
https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=4301
Symphylans:
https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=18819
Springtails:
https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=16769
Bulb Mites:
https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=13271
Richard Smith1, Eric Brennan2, JP Dundore Arias3, Daniel Geisseler4, Peter Henry2, Danyal Kasapligil5, Nicholas LeBlanc2, Karen Lowell6, Jeff Mitchell4, Joji Muramoto7, Radomir Schmidt4, Kate Scow4 and Yu-Chen Wang1
1 – UCCE Monterey; 2 – USDA ARS, Salinas; 3 – CSU, Monterey Bay; 4 – UC Davis; 5 – De La Valley Labs, Fresno; 6 – NRCS, Salinas; 7 – UC Santa Cruz
Lettuce production in the Salinas Valley has suffered unprecedented losses in the last three years due to infection with Impatiens Necrotic Spot Virus (INSV) and co-infection with soilborne pathogens. Pythium wilt (Pythium uncinulatum) has been the primary soilborne disease associated with INSV, but Fusarium wilt and other diseases have also been observed. Given that soilborne pathogens play a significant role in the observed losses and the lack of effective control measures, growers have been asking if there are practices they can employ to improve soil health that may reduce the frequency and intensity of outbreaks of soilborne diseases in their fields. However, it is not clear if the traditional practices used to manage the soil's chemical and physical characteristics that are known to improve soil health could also impact soilborne disease pressure. This article will briefly explore general strategies to manage soilborne diseases (with specific references to Pythium wilt of lettuce), as well as soil health, and their potential interrelationships.
Resistant varieties
For a plant disease to occur, three factors must be present at the same time: a susceptible host, a virulent pathogen, and a conducive environment. Soilborne diseases, just like any plant disease, need those three factors to occur, and therefore, all disease management strategies are aimed at disrupting or weakening the disease triangle. One of the most effective management strategies to control soilborne diseases is the use of tolerant/resistant cultivars. Selection of resistant varieties is a promising strategy for addressing Pythium wilt (https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=55733 ) as well as Fusarium and Verticillium wilts in head and romaine lettuce types (https://calgreens.org/wp-content/uploads/2022/04/monitoring-the-population-of-the-lettuce-fusarium-wilt-pathogen-in-california_2022.pdf and https://calgreens.org/wp-content/uploads/2022/04/verticillium-biology-epidemiology_2022.pdf . There may be limits to the effective use of resistant varieties due to the lack of resistance in desirable lettuce types and to the development of new races of the disease that have overcome current varietal resistance (e.g. Fusarium). As a result, other strategies are also much needed to fight pathogens; here we focus on ways to boost potential benefits of crop rotations and soil biodiversity in fighting soilborne diseases.
Rotations to reduce soilborne disease
Soilborne pathogens use different strategies to persist in the soil such as by 1) Forming chitin-based survival structures ; 2) by saprophytically colonizing plant debris as cellulose-based survival structures; and 3) by colonizing the root tissue of non-susceptible plants .
Crop rotations address the specific survival strategies of a pathogen and may provide specific or general suppression of soilborne diseases. A local example of specific suppression is broccoli's ability to suppress Verticillium wilt in subsequent lettuce crops when grown in rotations. Recent research suggests that broccoli rotations can be enhanced by the addition of crab meal amendment that is rich in chitin compounds to create a "substrate-mediated” microbial community shift which increased fungal antagonists and effectively reduced Verticillium dahlia microsclerotia in the soil by 50 – 78%, thereby reducing crop loss. However, there are only limited examples of rotations in vegetable production that provide specific suppression of soilborne diseases and none that specifically suppress Pythium wilt of lettuce.
Pathogens may be generally suppressed by the rotations with non-susceptible crops because their abundance in the soil usually declines with time. Types of rotational crops used, how frequently they are included in the rotation, and the length of non-host intervals, all influence how much disease suppression can be realized by rotations. Some soilborne pathogens persist in the soil for a very long time and are challenging to effectively reduce with rotations. One such example is the white rot of onions and garlic (Sclerotium cepivorum), whose sclerotia can persist for 20-30 years in the soil. In this case, even in the absence of a plant host, the organism will remain, and future plantings of susceptible crops may be infected. Other pathogens like Fusarium and Pythium, may persist in the soil as saprophytes infecting non-living plant tissue and only express themselves as diseases when a susceptible crop is planted. However, recent research on strawberries has shown that inclusion of rotations with weak hosts can result in a reduction in the amount of Fusarium oxysporum f. sp. fragariae. However, at present, this type of effect has not been shown for F.o. lactucae, which infects lettuce.
As the above examples illustrate, rotations can reduce Verticillium and possibly Fusarium, but other important pathogens, such as Pythium wilt of lettuce, have no field-based observations showing impacts of rotations to date.
What is soil health?
While the phrase ‘soil health' has become commonly used to discuss soil management and stewardship it is important to highlight that the phrase “is a metaphor, not a literal scientific construct” (Janzen et al. 2021). Metaphors like ‘soil health' are used in science to help us understand abstract concepts, because they can make us think about and relate to more familiar things such as our own health. Janzen et al. define soil health as “‘the vitality of a soil in sustaining the socioecological functions of its enfolding land.' And they add that “soil health reflects not the composition of soil per se, rather its capacity to promote the pertinent functions of the land in which it is embedded. This means that the term has little meaning for a soil divorced from its ecosystem, and that properties conferring such health depend on place and time.” So for example, an undisturbed sandy soil in the foot hills of the Salinas valley may be considered healthy if one is considering its ability to support beautiful native flowers, but may not be able to support vegetable production unless it is regularly amended with organic matter and fertilizers. In other words, ‘soil health' depends on who is looking.
Improving Soil Health
Practices to improve soil health for agricultural production mainly revolve around improving carbon management in the soil such as increasing soil organic matter. Sources of carbon to the soil in vegetable production on the Central Coast includes the incorporation of crop residues. For instance, broccoli residue commonly returns 3-4 tons/A of dry biomass to the soil of which 40-42% is carbon. However, given the low C:N ratio of the biomass, mineralization of a large portion of the residue occurs quickly. Inputs of soil carbon can be augmented using cover crops and compost. Cover crops with high C:N ratios are particularly helpful in this regard (https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=55520). In organic production, dry organic fertilizers often contain 30% carbon and, depending on the crops being grown, provide substantial quantities of carbon to the soil. Soil carbon can also be increased by reducing the loss of carbon from the soil by reducing tillage.
Additions of carbon have beneficial impacts on the soil by increasing the diversity, abundance, and activity of soil organisms by providing their carbon food source and facilitating the building of good soil structure that in turn provides good soil organism habitat. These stimulated beneficial organisms may, in turn, outcompete or predate upon pathogens and reduce impacts on crops. In addition, the same soil health practices can reduce abiotic plant stress by improving nutrient availability, reducing compaction, and enhancing drainage. These abiotic stresses can predispose plants to disease, as the defenses of weaker plants are more easily overcome. Reducing abiotic stress can lead to improved crop productivity even with pathogens present.
Growers recognize the benefits of increasing additions of carbon to the soil and have used these practices for many years. However, widespread use of over-wintered cover crop (Oct/Nov to Feb/March) has declined due to planting schedule conflicts; use of compost applications has also declined due to food safety concerns regarding the potential for composts to introduce pathogens of human health impacts. Given the risk of missing spring planting slots with over-wintered cover crops due to wet soils in the spring, some growers are looking at the use of fall-grown cover crops (Aug/Sept to Oct/Nov). This earlier planting window provides an opportunity for growers to grow the cover crops for 50 to 60 days in the fall and incorporate them into the soil before the onset of the rainy season. Fall-grown cover crops can produce 3 - 4 tons biomass/A in this short window which provides a substantial input of carbon to the soil (https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=54986 ). This is just one creative example of how growers can overcome constraints to the use of practices that improve soil health.
Measures of Soil Health
Numerous measures of soil health have been proposed. Examples include evaluating water infiltration rates, measurements of soil enzymes, evaluating the soil microbial biomass and many, many others. Soil health measures may be chemical, biological or physical. The USDA Natural Resource Conservation Service considers several factors when evaluating soil health. Two of the most critical are – is there enough carbon readily available to support abundant and diverse soil organisms and is the soil well aggregated and free of compaction so that the physical environment of the soil favors soil biological activity. Aside from management factors related to contaminants (e.g. excess salts) or disturbance (e.g. tillage), soil health is largely driven by biological activity in the soil.
Soil organic matter is a standard measure in most soil tests and offers insight into whether management is building or depleting soil carbon. But not all carbon is equally available to soil organisms. Carbon may be in chemical structures that are hard for microbes to break down and access as a food source, for example lignin in woody materials, or in forms with simple chemical bonds easily broken, for example sugars. Carbon exudates from living plant roots offer readily available carbon and is a primary driver for maintaining abundant microbial diversity and activity in the rhizosphere. Thus, an important practice in soil health management is to try to avoid long fallow periods when there are no living roots in the soil.
Measurement of Soil Carbon
The most straightforward and practical measure of soil health is measuring total soil carbon. The preferred method to measure soil organic carbon is dry combustion. This method, when corrected for soil carbonates, offers the most reliable and repeatable measure of organic carbon (5%). This test is available from many soil testing labs. Other methods for measuring soil carbon such as Loss on Ignition (LOI) and Walkley-Black Wet Digestion measure soil organic matter and then make an estimate of soil carbon based on this measurement. These methods are less accurate (20%) than the dry combustion method. Good reliable measurements of total soil carbon is the cornerstone method for evaluating soil health.
A portion of the total carbon is “active carbon” that is stored in more simple chemical structures that is available to feed soil biological activity. This form of carbon can be measured by the peroxide oxidizable carbon (POX-C) test. It may offer a more robust insight into how well management provides readily available carbon for soil microbes. For example, a long-term organic systems study with vegetable production in Salinas valley found that POX-C increased with frequent cover cropping and compost inputs (White et al. , 2020). However, this test and many others proposed by the research community are not currently offered by commercial soil labs. This is an indication that the surge of grower interest in soil health is in front of the ability of commercial labs to offer these services. However, commercial labs are responding to demand for soil health measures by growers and certification agencies and this situation may change in the future.
Some soil health assays can be done by the grower: measurements of CO2 respiration in a 24-hour incubation can be done with purchased test kits (e.g. Solvita). This test gives an indirect measure of the activity of soil microbes by measuring how much CO2 is evolved from soil in a 24 hour incubation. In addition, measurements of water stable aggregates which is also an indirect measure of soil health can also be conducted by growers using the SLAKES app on their cell phones. The test measures the stability of soil aggregates against falling apart when wetted with water. For more information go to: ( https://doi.org/10.1002/saj2.20012, and https://www.ars.usda.gov/ARSUserFiles/30501000/SoilAggStabKit.pdf ) .
At this point there are no locally calibrated baseline standards for California growers to compare their soil health test results against. The best approach for a grower is to carry out these tests and watch the trends in the results over time to get a sense if the practices are moving the soil health indicators in a positive direction and whether those changes correlate with positive impacts on crop health and productivity. In almost all cases, it is most helpful to compare results across time and management change in the same field. Inherent soil properties, for example texture, drainage, parent material, etc. may all influence some properties and thus comparisons across fields are less clear cut than those in a single field over time. Given all the research being conducted on soil health, there will undoubtedly be refinements to recommended soil evaluations to assess soil health.
If you choose to send soil samples to a lab out of state, it is important to keep in mind that soil tests are often designed with the local conditions in mind and soils from California my not measure up well when compared with soils from other areas (e.g. lower organic matter levels, higher salts, etc.).
Summary
Frameworks for soil health assessment have been proposed that can help growers identify practical indices that can guide them in assessing the health of their soils. Fundamental to all soil health practices, increasing carbon inputs, is critical to increasing soil microbial activity which may have an impact on suppressing soilborne diseases. Therefore, reliable measurements of soil carbon with the dry combustion test can provide a basic understanding of the impact of management practices on soil health. The impact of soil health practices on soilborne diseases still needs to be examined on a disease-by-disease basis, as undoubtedly each disease will respond differently to beneficial practices. Pythium wilt of lettuce is proving to be an elusive and difficult to understand pathogen; much more work remains to be done to understand biological processes that can effectively suppress it.
The soil health field is an area of active research by scientists collaborating with growers to test the concepts discussed above and to find ways to optimize suppression of soilborne diseases. These crucial collaborations will help us better understand the directions we need to go in to get a better handle on these issues.
For additional reading:
Soil Borne Diseases : Soil Health (ucdavis.edu)

![1699385688184-b7678dfe-37ca-4187-b887-cec08c2a072c 1[1] 1699385688184-b7678dfe-37ca-4187-b887-cec08c2a072c 1[1]](https://ucanr.edu/blogs/SalinasValleyAgriculture/blogfiles/103056.jpg)