- Author: Surendra K. Dara
Botrytis fruit rot or gray mold, caused by Botrytis cinerea, is common fruit disease in California strawberries (Koike et al. 2018). Botrytis cinerea has a wide host range infecting several commercially important crops including blueberry (Saito et al. 2016), grapes (Saito et al., 2019), and tomato (Breeze, 2019). Fungal infection can cause flower or fruit rot. Fruit can be infected directly or through a latent infection in the flowers. Moist and cool conditions favor fungal infections and increased sugar content in the ripening fruit can also contribute to the disease development. Initial symptoms of infection appear as brown lesions and a thick mat of gray conidia is characteristic symptom in the later stages of infection. As chemical fungicides are primarily used for gray mold control, fungicide resistance is a common problem around the world (Panebianco et al., 2015; Liu et al., 2016; Stockwell et al., 2018; Weber and Hahn, 2019). In strawberry, cultural control options such as removing diseased plant material or using cultivars with traits that can reduce gray mold infections may not be practical when the disease is widespread in the field or cultivar choice is made based on other factors. Non-chemical control options are necessary to help reduce the risk of chemical fungicide resistance, prolong the life of available chemical fungicides, achieve desired disease control, and to maintain environmental health. Although there are several botanical and microbial fungicides available for gray mold control, limited information is available on their efficacy in California strawberries. A study was conducted in the spring of 2019 to evaluate the efficacy of several chemical, botanical, and microbial fungicides in certain combinations and rotations to help identify effective options for an integrated disease management strategy.
Methodology
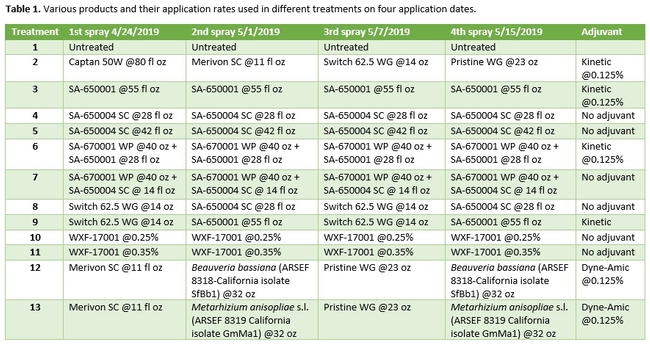
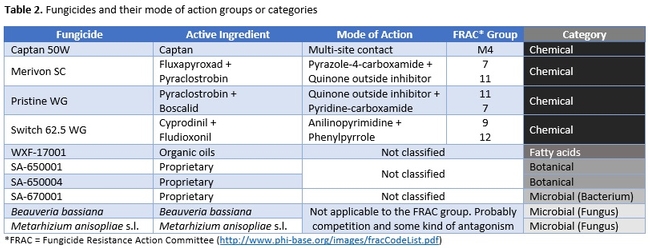
Strawberry cultivar San Andreas was planted late November, 2018 and the study was conducted in April and May, 2019. Each treatment had a 20' long strawberry plot with two rows of plants replicated in a randomized complete block design. Plots were maintained without any fungicidal applications until the study was initiated. Table 1 contains the list of treatments, application rates and dates of application, and Table 2 contains the type of fungicide used and their mode of action. Beauveria bassiana and Metarhizium anisopliae s.l. are California isolates of entomopathogenic fungi, isolated from an insect and a soil sample, respectively. These fungi are pathogenic to a variety of arthropods and some strains are formulated as biopesticides for arthropod control. However, earlier studies in California demonstrated that these fungi are also known to antagonize plant pathogens such as Fusarium oxysporum f.sp. vasinfectum Race 4 (Dara et al., 2016) and Macrophomina phaseolina (Dara et al., 2018) and reduce the disease severity. To further evaluate their efficacy against B. cinerea, these two fungi were also included in this study alternating with two chemical fungicides.
Treatments were applied with a CO2-pressurized backpack sprayer using 66.5 gpa spray volume. Five days before the first spray application and 3 days after each application, all ripe fruit were harvested from each plot and incubated at the room temperature in vented plastic containers. The level of gray mold on fruit from each plot was rated using a 0 to 4 scale (where 0=no disease, 1=1-25% fruit with fungal infection, 2=26-50% infection, 3=51-75%, and 4=76-100%) 3 and 5 days after each harvest (DAH). Due to the rains, fruit could not be harvested after the 3rd spray application for disease rating, but was harvested and discarded after the rains to avoid cross infection for the following week's harvest. Data were analyzed using analysis of variance using Statistix software and significant means were separated using Least Significant Difference separation test.
Results
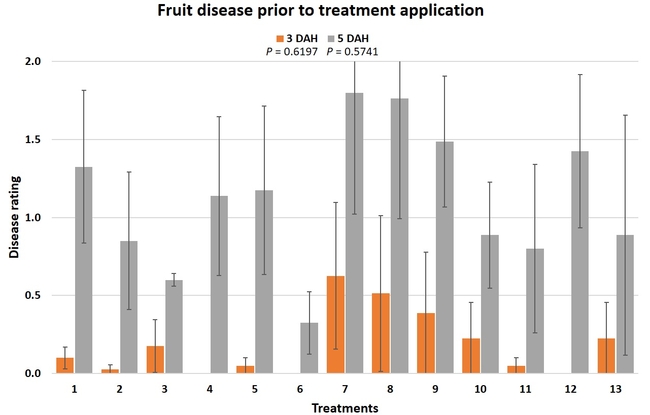
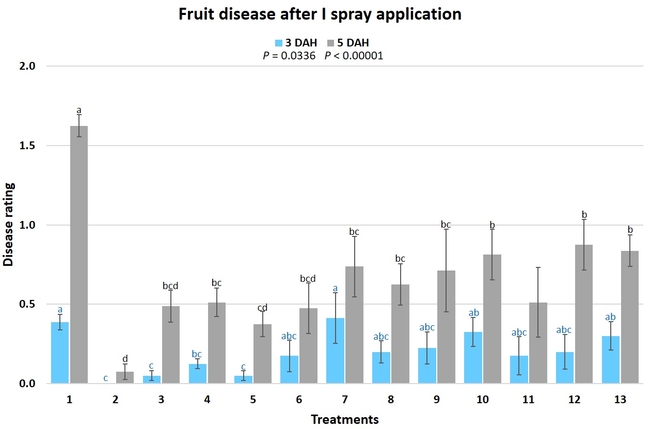
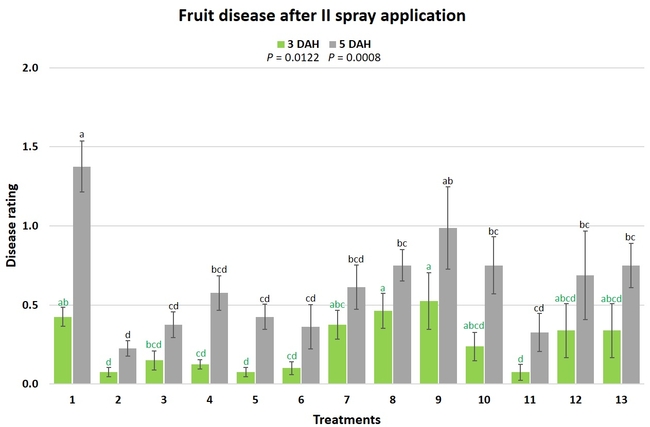
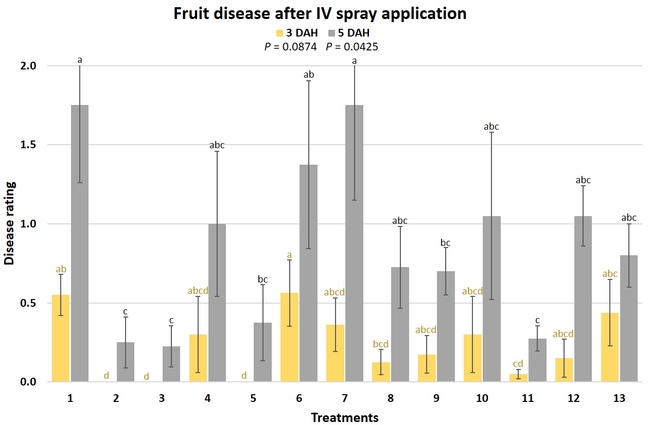
Gray mold occurred at low to moderate levels during the study period. Along with B. cinerea, there were a few instances of minor fungal infections from Rhizopus spp. (Rhizopus fruit rot) and Mucor spp. (Mucor fruit rot). Pre-treatment disease ratings were statistically not significant (P = 0.6197 and 0.5741) 3 and 5 DAH. While the chemical standard treatment with the rotation of Captan, Merivon, Switch, and Pristine (treatment 2) appeared to result in the lowest disease rating throughout the observation period, treatments 3 and 5 after the 1st spray application, treatments 5 and 11 along with 3, 4 and 6 after the 2nd spray application, and treatments 3 and 5 along with 11 after the 4th spray application also had similar disease control at 3 DAH. When disease at 5 DAH was compared, the lowest rating was seen in treatment 2 after the 1st and 2nd spray applications, and treatments 2, 3, and 11 after the 4th application. Several other treatments also provided statistically similar control during these days.
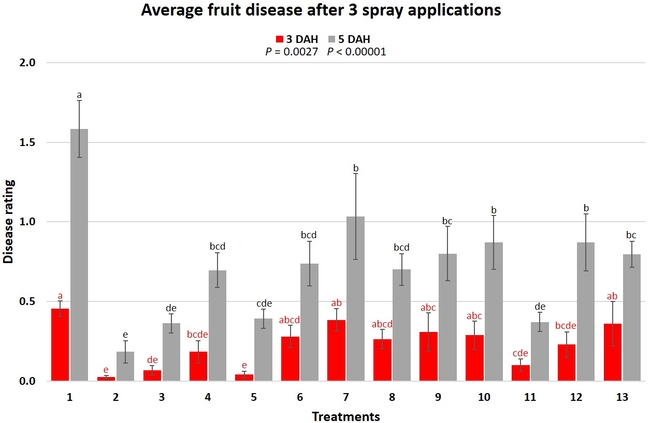
When the average disease rating for the three post-treatment observation events was considered, treatment 2, 3, 5, and 11 had the lowest disease at both 3 and 5 DAH. Treatments 4 and 12 at 3 DAH also had a statistically similar level of disease control to treatment 2.
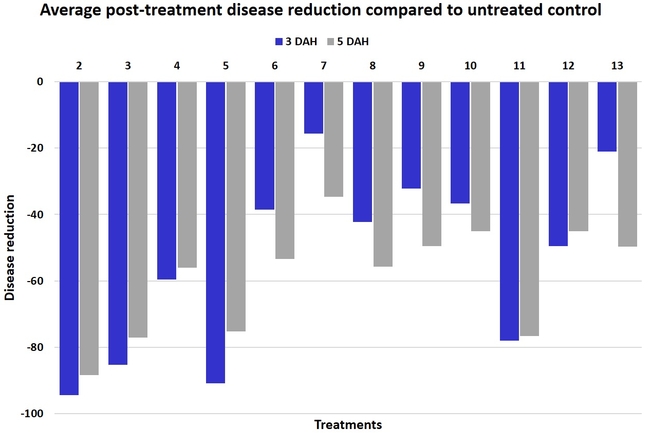
In general, most of the treatments provided moderate to high control compared to the disease in untreated control when the post-treatment averages were considered. Only treatment 7 and 13 had lower control at 3 DAH.
Discussion
This study compared a variety of registered and developmental products along with two entomopathogenic fungi in managing B. cinerea. Considering the fungicide resistance problem in B. cinerea in multiple crops, having multiple non-chemical control options is very important to achieve desirable control with integrated disease management strategies. Since the active ingredients in the botanical and bacterial fungicides used in this study are not public, discuss will be limited on their modes of action and efficacy at this point. Similarly, the active ingredient of WXF-17001 is also not known, however, an earlier study by Calvo-Garrido et al. (2014) demonstrated that a fatty acid-based natural product reduced B. cinerea conidial germination by 54% and disease severity in grapes by 96% compared to untreated control. The product used by Calvo-Garrido et al. (2014) is thought to be fungistatic and reduce the postharvest respiratory activity and ethylene production in fruits.
While chemical fungicides have a specific mode of action, biological and other products act in multiple manners either directly antagonizing the plant pathogen or by triggering the plant defenses. For example, amending the potting medium with biochar resulted in induced systemic resistance in tomato and reduced B. cinerea severity by 50% (Mehari et al., 2015). Luna et al. (2016) also showed that application of β-aminobutyric acid and jasmonic acid promoted seed germination and long-term resistance to B. cinerea in tomato. Burkholderia phytofirmans, beneficial endophytic bacterium, offered protection against B. cinerea in grapes by mobilizing carbon resources (callose deposition), triggering plant immune system (hydrogen peroxide production and priming of defense genese), and through antifungal activity (Miotto-Vilanova et al. 2016). Similarly, entomopathogenic fungi such as B. bassiana are also known to induce systemic resistance against plant pathogens (Griffin et al. 2006). Compared to other options evaluated in the study, entomopathogenic fungi have an advantage of controlling both arthropod pests and diseases, while also having plant growth promoting effect (Dara et al. 2017).
Rotating fungicides with different mode of actions reduces the risk of resistance development and using some combinations will also maintain control efficacy. This study provided the efficacy of multiple control options and their combinations and rotations for B. cinerea. This is also the first study demonstrating the efficacy of entomopathogenic fungi against B. cinerea in strawberry.
Acknowledgements: Thanks to Sipcam Agro and Westbridge for funding the study, technical assistance of Hamza Khairi for data collection, and the field staff at the Shafter Research Station for the crop maintenance.
References
Breeze, E. 2019. 97 Shades of gray: genetic interactions of the gray mold, Botrytis cinerea, with wild and domesticated tomato. The Plant Cell 31: 280-281. https://doi.org/10.1105/tpc.19.00030
Calvo-Garrido, C., A.A.G. Elmer, F. J. Parry, I. Viñas, J. Usall, R. Torres, R.H. Agnew, and N. Teixidó. 2014. Mode of action of a fatty acid-based natural product to control Botrytis cinerea in grapes. J. Appl. Microbiol. 116: 967-979. https://doi.org/10.1111/jam.12430
Dara, S. K., S. S. Dara, S.S.R. Dara, and T. Anderson. 2016. First report of three entomopathogenic fungi offering protection against the plant pathogen, Fusarium oxysporum f.sp. vasinfectum. UC ANR eJournal of Entomology and Biologicals https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=22199
Dara, S. K., S.S.R. Dara, and S. S. Dara. 2017. Impact of entomopathogenic fungi on the growth, development, and health of cabbage growing under water stress. Amer. J. Plant Sci. 8: 1224-1233. https://doi.org/10.4236/ajps.2017.86081
Dara, S.S.R., S. S. Dara, and S. K. Dara. 2018. Preliminary report on the potential of Beauveria bassiana and Metarhizium anisopliae s.l. in antagonizing the charcoal rot causing fungus Macrophomina phaseolina in strawberry. UC ANR eJournal of Entomology and Biologicals https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=28274
Griffin, M. R., B. H. Ownley, W. E. Klingeman, and R. M. Pereira. 2006. Evidence of induced systemic resistance with Beauveria bassiana against Xanthomonas in cotton. Phytopathol. 96.
Koike, S. T., G. T. Browne, T. R. Gordon, and M. P. Bolda. 2018. UC IPM pest management guidelines: strawberry (diseases). UC ANR Publication 3468. https://www2.ipm.ucanr.edu/agriculture/strawberry/Botrytis-Fruit-Rot/
Liu, S., Z. Che, and G. Chen. 2016. Multiple-fungicide resistance to carbendazim, diethofencardb, procymidone, and pyrimethanil in field isolates of Botrytis cinerea from tomato in Henan Province, China. Crop Protection 84: 56-61.
Luna, E., E. Beardon, S. Ravnskov, J. Scholes, and J. Ton. 2016. Optimizing chemically induced resistance in tomato against Botrytis cinerea. Plant Dis. 100: 704-710. https://doi.org/10.1094/PDIS-03-15-0347-RE
Mehari, Z. H., Y. Elad, D. Rav-David, E. R. Graber, and Y. M. Harel. 2015. Induced systemic resistance in tomato (Solanum lycopersicum) against Botrytis cinerea by biochar amendment involves jasmonic acid signaling. Plant and Soil 395: 31-44.
Miotto-Vilanova, L., C. Jacquard, B. Courteaux, L. Wortham, J. Michel, C. Clément, E. A. Barka, and L. Sanchez. 2016. Burkholderia phytofirmans PsJN confers grapevine resistance against Botrytis cinerea via a direct antimicrobial effect combined with a better resource mobilization. Front. Plant Sci. 7: 1236. https://doi.org/10.3389/fpls.2016.01236
Panebianco, A., I. Castello, G. Cirvilleri, G. Perrone, F. Epifani, M. Ferrarra, G. Polizzi, D. R. Walters, and A. Vitale. 2015. Detection of Botrytis cinerea field isolates with multiple fungicide resistance from table grape in Sicily. Crop Protection 77: 65-73.
Saito, S., T. J. Michailides, and C. L. Xiao. 2016. Fungicide resistance profiling in Botrytis cinerea populations from blueberry in California and Washington and their impact on control of gray mold. Plant Dis. 100: 2087-2093. https://doi.org/10.1094/PDIS-02-16-0229-RE
Saito, S., T. J. Michailides, and C. L. Xiao. 2019. Fungicide-resistant phenotypes in Botrytis cinerea populations and their impact on control of gray mold on stored table grapes in California. European J. Plant Pathol. 154: 203-213.
Stockwell, V. O., B. T> Shaffer, L. A. Jones, and J. W. Pscheidt. 2018. Fungicide resistance profiles of Botrytis cinerea isolated from berry crops in Oregon. Abstract for International Congress of Plant Pathology: Plant Health in A Global Economy; 2018 July 29-Aug 3; Boston, MA.
Weber, R.W.S. and M. Hahn. 2019. Grey mould disease of strawberry in northern Germany: causal agents, fungicide resistance and management strategies. Appl. Microbiol. Biotechnol. 103: 1589-1597.
- Author: Sumanth S. R. Dara
- Author: Suchitra S. Dara
- Author: Surendra K. Dara
Charcoal rot, caused by Macrophomina phaseolina, is one of the important fungal diseases of strawberry in California. Macrophomina phaseolina is a soilborne fungus and has a wide host range, including alfalfa, cabbage, corn, pepper, and potato, some of which are cultivated in the strawberry production areas in California. The fungus infects the vascular system of the plant roots, obstructing the nutrient and water supply and ultimately resulting in stunted growth, wilting, and death of the plant. The fungus survives in the soil and infected plant debris as microsclerotia (resting structures made of hyphal bodies) and can persist for up to three years. Microslerotia germinate and penetrate the root system to initiate infection. Plants are more vulnerable to fungal infection when they are experiencing environmental (extreme weather or drought conditions) and physiological (heavy fruit bearing) stress.
Soil fumigation is the primary management option for addressing charcoal rot in strawberry. Crop rotation with broccoli can also reduce the risk of charcoal rot due to glucosinolates and isothiocyanates in broccoli crop residue that have fungicidal properties. Beneficial microorganisms such as Bacillus spp. and Trichoderma spp. are also considered, especially in organic strawberries, to antagonize M. phaseolina and other soilborne pathogens and provide some protection. The role of beneficial microbes in disease management or improving crop growth and health is gaining popularity in the recent years with the commercial availability of biofungicide, biostimulant, and soil amendment products. In a couple of recent strawberry field studies in Santa Maria, some of the beneficial microbial products improved fruit yield or crop health. These treatments can be administered by inoculating the transplants prior to planting, immediately after planting or periodically applying to the plants and or the soil. Adding beneficial microbes can help improve the soil microbiome especially after chemical or bio-fumigation and anaerobic soil disinfestation.
Similar to the benefits of traditionally used bacteria (e.g., Bacillus spp. and Pseudomonas spp.) and fungi (e.g., Glomus spp. and Trichoderma spp.), studies with entomopathogenic fungi such as Beauveria bassiana, Isaria fumosorosea, and Metarhizium spp. also demonstrated their role in improving water and nutrient absorption or antagonizing plant pathogens. The advantage of entomopathogenic fungi is that they are already used for arthropod pest management in multiple crops, including strawberry; now, there are the additional benefits of promoting crop growth and antagonizing plant pathogens. In light of some promising recent studies exploring these roles, a study was conducted using potted strawberry plants to evaluate the efficacy of two California isolates of Beauveria bassiana and Metarhizium anisopliae s.l. and their application strategies against M. phaseolina.
Methodology
About 6 week old strawberry plants (cultivar Albion) from a strawberry field at the Shafter Research Station were transplanted into 1.6-gallon pots with Miracle-Gro All Purpose Garden Soil (0.09-0.05-0.07 N-P-K) and maintained in an outdoor environment. They were regularly watered, and their health was monitored for about 5 months prior to the commencement of the study. Conidial suspensions of the California isolates of B. bassiana and M. anisopliae s.l. were applied one week before, after, or at the time of applying microsclerotia of M. phaseolina to the potting mix. The following treatments were evaluated in the study:
- Untreated control
- Soil inoculated with M. phaseolina
- Soil inoculated with B. bassiana 1 week prior to M. phaseolina inoculation
- Soil inoculated with M. anisopliae s.l. 1 week prior to M. phaseolina inoculation
- Soil inoculated with B. bassiana at the time of M. phaseolina inoculation
- Soil inoculated with M. anisopliae s.l. at the time of M. phaseolina inoculation
- Soil inoculated with B. bassiana 1 week after to M. phaseolina inoculation
- Soil inoculated with M. anisopliae s.l. 1 week after to M. phaseolina inoculation
Entomopathogenic fungi were applied as 1X1010 viable conidia in 100 ml of 0.01% Dyne-Amic (surfactant) solution distributed around the plant base. To apply M. phaseolina, 5 grams of infested cornmeal-sand inoculum containing 2,500 CFU/gram was added to four 5 cm deep holes around the base of the plant. Each treatment had six pots each planted with a single strawberry plant representing a replication. Treatments were randomly arranged within each replication. The study was repeated once a few days after the initiation of the first experiment.
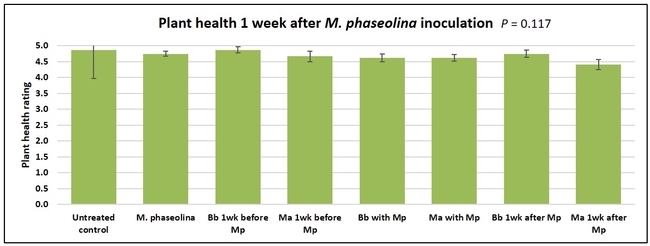
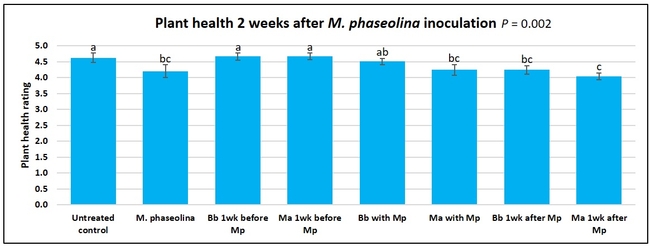
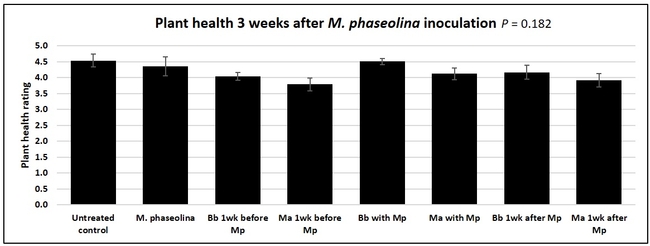
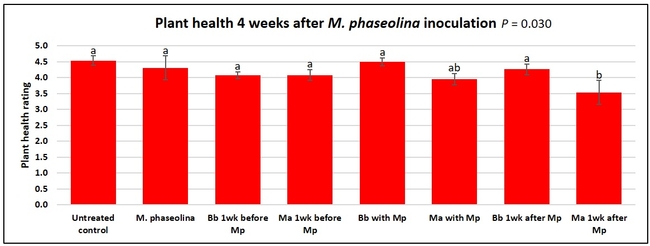
Plant health was monitored starting from the first week after the M. phaseolina inoculation and continued for seven weeks. Plant health was rated on a scale of 0 to 5 where 0=dead and 5=very healthy and the rest of the ratings in between depending on the extent of wilting. Data from both experiments were combined and analyzed by ANOVA using Statistix software and significant means were separated using LSD test. The influence of entomopathogenic fungal treatments applied at different times as well as the combined effect of different applications within each fungus were compared for seven weeks. Ratings for some plants that were scorched from hot summer temperatures and died abruptly were removed from the analyses.
Results
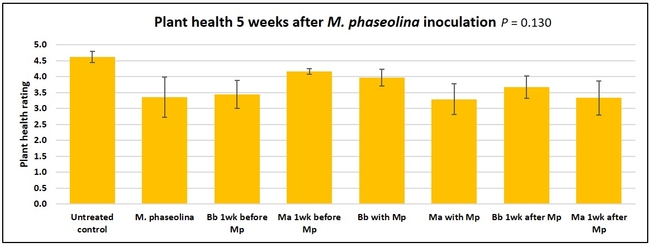
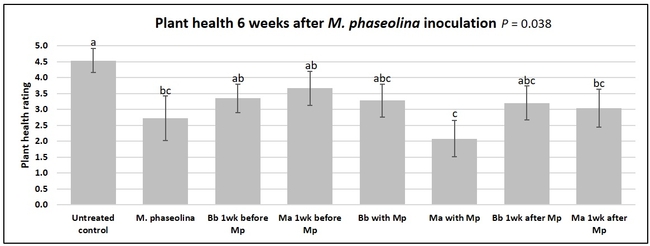
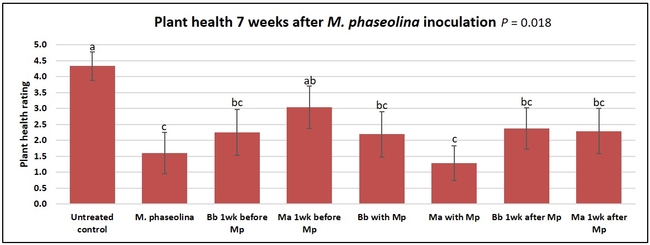
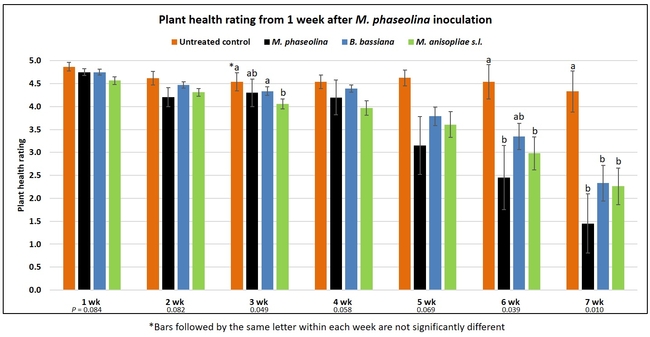
Untreated control plants maintained good health throughout the observation period varying between the rating of 4.3 and 4.9. In general, plant health declined considerably from the 5th week after M. phaseolina inoculation. Plant health appeared to be slightly better in plants treated with entomopathogenic fungi, but there was no statistically significant difference in any except one instance. Plants treated with M. anisopliae one week prior to the application of M. phaseolina had a rating of 3.0 compared to 1.6 rating of plants inoculated with M. phaseolina alone.
When data from different treatments for each entomopathogenic fungus were compared, both B. bassiana and M. anisopliae s.l. appeared to reduce the wilting, but the plant health rating was not significantly different from the M. phaseolina treatment alone.
This is the first report of the impact of entomopathogenic fungi on M. phaseolina with some promise. Additional studies under more uniform environmental conditions and with more treatment options would shed more light on this approach of using entomopathogenic fungi against M. phaseolina. The current study evaluated single application of the entomopathogenic fungi and we plan to conduct additional studies with multiple applications.
Acknowledgements: We thank Dr. Kelly Ivors (previously at Cal Poly San Luis Obispo) for the pathogen inoculum and Dr. Stefan Jaronski, USDA-ARS, Sidney, MT for multiplying the entomopathogenic fungal inocula.
References
Dara, S. K. and D. Peck. 2017. Evaluating beneficial microbe-based products for their impact on strawberry plant growth, health, and fruit yield. UC ANR eJournal Strawberries and Vegetables. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=25122
Dara, S. K. and D. Peck. 2018. Evaluation of additive, soil amendment, and biostimulant products in Santa Maria strawberry. CAPCA Adviser, 21(5): 44-50.
Dara, S. K., S.S.R. Dara, and S. S. Dara. 2017. Impact of entomopathogenic fungi on the growth, development, and health of cabbage growing under water stress. Amer. J. Plant Sci. 8: 1224-1233. http://file.scirp.org/pdf/AJPS_2017051714172937.pdf
Dara, S. K., S. S. Dara, S.S.R. Dara, and T. Anderson. 2016. First report of three entomopathogenic fungi offering protection against the plant pathogen, Fusarium oxysporum f.sp. vasinfectum. UC ANR eJournal Strawberries and Vegetables. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=22199
Koike, S. T., G. T. Browne, and T. R. Gordon. 2013. UC IPM pest management guidelines: Strawberry diseases. UC ANR Publication 3468. http://ipm.ucanr.edu/PMG/r734101511.html
Partridge, D. 2003. Macrophomina phaseolina. PP728 Pathogen Profiles, Department of Plant Pathology, North Carolina State University. https://projects.ncsu.edu/cals/course/pp728/Macrophomina/macrophominia_phaseolinia.HTM
Vasebi, Y., N. Safaie, and A. Alizadeh. 2013. Biological control of soybean charcoal root rot disease using bacterial and fungal antagonists in vitro and greenhouse condition. J. Crop Prot. 2(2): 139-150.
- Author: Surendra K. Dara
Mechanisms of insecticide resistance in insects.
Use of biopesticides or non-chemical pesticides is encouraged as a part of integrated pest management (IPM) for environmental and human safety and to reduce the risk of insecticide resistance. With the increase in biopesticide use in both organic and conventional cropping systems, it is a good time to review the potential of insect resistance to botanical and microbial pesticides.
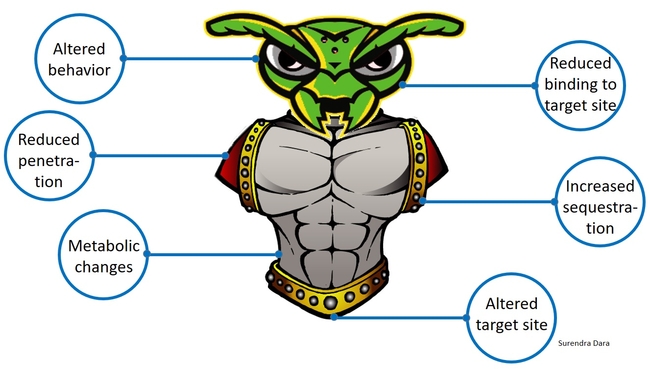
Insects and mites develop resistance to chemical pesticides through genetic, metabolic, or behavioral changes resulting in reduced penetration of toxin, increased sequestration or excretion, reduced binding to the target site, altered target site that prevents binding of the toxin, or reduced exposure to the toxin through modified behavior. When the active ingredient is a toxic molecule and has the mode of action similar to that of a chemical compound, regardless of the plant or microbial origin, arthropods are more likely to develop resistance through one or more of the abovementioned mechanisms. When the mode of action is infection by a microorganism, rather than a toxin, arthropods are less likely to develop resistance. Under natural circumstances, plants, insects, natural enemies, and beneficial or harmful microbes continuously co-evolve and adapt to changing environment. When there is a higher selection pressure, such as indiscriminate use of chemical pesticides, increased mutagenesis can lead to resistance issues. A good understanding of insect resistance to biopesticides will help minimize potential risks and improve their efficient use in IPM.
Resistance to botanical pesticides
Nicotine, an alkaloid from Nicotiana spp., is one of the earlier botanical pesticides known. Although nicotine is not currently used as an insecticide, its synthetic alternatives – neonecotinoids – are commonly used against several pests. Botanical insecticide pyrethrum, extracted from the flowers of Chrysanthemum cinerariaefolium, contains insecticidal pyrethrins (synthetic pyrethrins are referred to as pyrethroids). Although insect resistance to pyrethrum or pyrethroid compounds has been known (Whitehead, 1959; Immaraju et al., 1992; Glenn et al., 1994), they have been effectively used against a number of pests through careful placement in IPM, organic, or conventional management strategies. Additionally, pyrethrin products have been effectively used along with piperonyl butoxide, which acts as a synergist and resistance breaker (Gunning et al. 2015).
Another botanical insecticidal compound, azadirachtin, is a tetranortriterpenoid limonoid from neem (Azadirachta indica) seeds, which acts as an insecticide, antifeedant, repellent and insect growth regulator. While neem oil, which has a lower concentration of azadirachtin, has been used in the United States as a fungicide, acaricide, and insecticide for a long time, several azadirachtin formulations in powder and liquid forms have become popular in recent years and were found effective in managing important pests (Dara 2015a and 2016). Feng and Isman (1995) reported that the green peach aphid, Myzus persicae developed resistance to pure azadirachtin under artificially induced selection pressure after 40 generations, but did not develop resistance to a refined neem seed extract. They suggested that natural blend of azadirachtin compounds in a biopesticide would not exert selection pressure that could lead to resistance. Additionally, Mordue and Nisbet (2000) discussed that azadirachtin can play a role in insecticide resistance management because it reduces the detoxification enzyme production as a protein synthesis inhibitor. Azadirachtin also improved the efficacy of other biopesticides in multiple studies (Trisyono and Whalon, 2000; Dara, 2013 and 2015b).
Insects feeding on plant allelochemicals can develop cross-resistance to insecticides (Després et al., 2007). For example, overproduction of detoxification enzymes such as glutathione S-transferases and monooxygenases in the fall armyworm, Spodoptera frugiperda,when it fed on corn and cowpea, respectively, imparted cross-resistance to various chemical pesticides. It is important to keep this in mind when botanical pesticides are used to detect potential resistance issues.
Resistance to bacterial biopesticides
Bacillus thuringiensis (Bt)is a gram-positive soil bacterium, which contains crystalline toxic protein that is activated upon ingestion by an insect host, binds to the receptor sites in the midgut, and eventually causes insect death. Since the mode of action involves a toxin rather than bacterial infection, several insects developed resistance to Bt pesticides or transgenic crops that contain Bt toxins (Tabashnik et al., 1990; McGaughey and Whalon, 1992; Tabashnik, 1994; Iqbal et al., 1996). However, Bt pesticides are still very popular and used against a variety of lepidopteran (Bt subsp. aizawai and Bt subsp. kurstaki), dipteran (Bt subsp. israelensis and Bt subsp. sphaericus), and coleopteran (Bt subsp. tenebrionis) pests.
Spinosad is a mixture of macrocyclic lactones, spinosyns A and spinosyns D, derived from Saccharopolyspora spinosa, an actinomycete gram-positive bacterium, and is used against dipteran, hymenopteran, lepidopteran, thysanopteran, and other pests. Spinosad products, while naturally derived are registered as chemical pesticides, not as biopesticides. Insect resistance to spinosad later led to the development of spinetoram, which is a mixture of chemically modified spinosyns J and L. Both spinosad and spinetoram are contact and stomach poisons and act on insect nervous system by continuous activation of nicotinic acetylcholine receptors. However, insect resistance to both spinosad (Sayyed et al., 2004; Bielza et al., 2007) and spinetoram (Ahmad and Gull, 2017) has been reported due to extensive use of these pesticides. Cross-resistance between spinosad and some chemical insecticides has also occurred in some insects (Mota-Sanchez et al., 2006; Afzal and Shad, 2017).
Resistance to viral biopesticides
Baculovirus infections in lepidoptera have been known for centuries, especially in silkworms. Currently, there are several commercial formulations of nucleopolyhedroviruses (NPV) and granuloviruses (GV). When virus particles are ingested by the insect host, usually lepidoptera, they invade the nucleii of midgut, fatbody, or other tissue cells and kill the host. Baculoviruses are generally very specific to their host insect species and can be very effective in bringing down the pest populations. However, variations in the susceptibility of certain insect populations and development of resistant to viruses has occurred in several host species (Siegwart et al., 2015). Resistance to different isolates of Cydia pomonella granulovirus (CpGV-M, CpGV-S) in codling moth (Cydia pomonella) populations is well known in Germany and other parts of Europe (Sauer et al., 2017a & b).
Resistance to fungal biopesticides
There are several fungi that infect insects and mites. Fungal infection starts when fungal spores come in contact with an arthropod host. First, they germinate and gain entry into the body by breaching through the cuticle. Fungus later multiplies, invades the host tissues, kills the host, and emerges from the cadaver to produce more spores. Entomophthoralean fungi such as Entomophthora spp., Pandora spp., and Neozygites spp. can be very effective in pest management through natural epizootics, but cannot be cultured in vitro for commercial scale production. Hypocrealean fungi such as Beauveria bassiana, Isarea fumosorosea, Metarhizium brunneum, and Verticillium lecanii,on the other hand, can be mass-produced in vitro and are commercially available. These fungi are comparable to broad-spectrum insecticides and are pathogenic to a variety of soil, foliar, and fruit pests of several major orders. Since botanical, bacterial, and viral biopesticides have insecticidal metabolites, proteins, or viral particles that have specific target sites and mode of action, insects have a higher chance of developing resistance through one or more mechanisms. Although fungi also have insecticidal proteins such as beauvericin in B. bassiana and I. fumosorosea and dextruxin in M. anisopliae and M. brunneum, their mode of action is more through fungal infection and multiplication and arthropods are less prone to developing resistance to entomopathogenic fungi. However, insects can develop resistance to entomopathogenic fungi through increased melanism, phenoloxidase activity, protease inhibitor production, and antimicrobial and antifungal peptide production (Wilson et al., 2001; Zhao et al., 2012; Dubovskiy et al., 2013). It appears that production of detoxification enzymes in insects against fungal infections can also impart resistance to chemical pesticides. Infection of M. anisopliae in the larvae of greater wax moth, Galleria mellonella, increased dexotification enzyme activity and thus resistance to malathion (Serebrov et al., 2006).
These examples show that insects can develop resistance to biopesticides in a manner somewhat similar to chemical pesticides, but due to the typically more complex and multiple modes of action, at a significantly lesser rate depending on the kind of botanical compound or microorganism involved. Resistance to entomopathogenic fungi is less common than with other entomopathogens. Since biopesticide use is not as widespread as chemical pesticides, the risk of resistance development is less for the former. However, excessive use of any single tool has the potential for resistance or other issues and IPM, which uses a variety of management options, is always a good strategy.
Acknowledgements: Thanks to Pam Marrone for reviewing the manuscript.
References
Afzal, M.B.S. and S. A. Shad. 2017. Spinosad resistance in an invasive cotton mealybug, Phenacoccus solenopsis: cross-resistance, stability and relative fitness. J. Asia-Pacific Entomol. 20: 457-462.
Ahmad M. and S. Gull. 2017. Susceptibility of armyworm Spodoptera litura (Lepidoptera: Noctuidae) to novel insecticides in Pakistan. The Can. Entomol. 149: 649-661.
Bielza, P., V. Quinto, E. Fernández, C. Grávalos, and J. Contreras. 2007. Genetics of spinosad resistance in Frankliniella occidentalis (Thysanoptera: Thripidae). J. Econ. Entomol. 100: 916-920.
Dara, S. K. 2013. Strawberry IPM study 2012: managing insect pests with chemical, botanical, and microbial pesticides. UCANR eJournal Strawberries and Vegetables March 13, 2013 (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=9595).
Dara, S. K. 2015a. Root aphids and their management in organic celery. CAPCA Adviser 18(5): 65-70.
Dara, S. K. 2015b. Strawberry IPM study 2015: managing insect pests with chemical, botanical, microbial, and mechanical control options. UCANR eJournal Strawberries and Vegetables November 30, 2015 (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=19641).
Dara, S. K. 2016. Managing strawberry pests with chemical pesticides and non-chemical alternatives. Intl. J. Fruit Sci. https://doi.org/10.1080/15538362.2016.1195311
Després, L., J.-L. David, and C. Gallet. 2007. The evolutionary ecology of insect resistance to plant chemicals. Trends in Ecol. Evol. 22: 298-307.
Dubovskiy, I. M., M.M.A. Whitten, O. N. Yaroslavtseva, C. Greig, V. Y. Kryukov, E. V. Grizanova, K. Mukherjee, A. Vilcinskas, V. V. Glupov, and T. M. Butt. 2013. Can insects develop resistance to insect pathogenic fungi? PloS one 8: e60248.
Feng, R. and M. B. Isman. 1995. Selection for resistance to azadirachtin in the green peach aphid, Myzus persicae. Experientia 51: 831-833.
Glenn, D. C., A. A. Hoffmann, and G. McDonald. 1994. Resistance to pyrethroids in Helicoverpa armigera (Lepidoptera: Noctuidae) from corn: adult resistance, larval resistance, and fitness effects. J. Econ. Entomol. 87: 1165-1171.
Gunning, R., G. Moores, and M. Balfe. 2015. Novel use of pyrethrum to control resistant insect pests on cotton. Acta Hort. 1073: 113-118. https://doi.org/10.17660/ActaHortic.2015.1073.16
Immaraju, J. A., T. D. Paine, J. A. Bethke, K. L. Robb, and J. P Newman. 1992. Western flower thrips (Thysanoptera: Thripidae) resistance to insecticides in coastal California greenhouses. J. Econ. Entomol. 85: 9-14.
Iqbal, K., R.H.J. Vekerk, M. J. Furlong, P. C. Ong, S. A. Rahman, and D. J. Wright. 1996. Evidence for resistance to Bacillus thuringiensis (Bt) subsp. kurstaki HD-1, Bt subsp. aizawai and Abamectin in field populations of Plutella xylostella from Malaysia. Pest Manag. Sci. 48: 89-97.
McGaughey, W. H. and M. E. Whalon. 1992. Managing insect resistance to Bacillus thuringiensis toxins. Science 258: 1451-1455.
Mordue, A. J. and A. J. Nisbet. 2000. Azadirachtin from the neem tree Azadirachta indica: its action against insects. An. Soc. Entomol. Bras. 29: 615-632.
Mota-Sanchez, D., R. M. Hollingworth, E. J. Grafius, and D. D. Moyer. 2006. Resistance and cross-resistance to neonicotinoid insecticides and spinosad in the Colorado potato beetle, Leptinotarsa decemlineata (Say) (Coleoptera: Chrysomelidae). Pest Manag. Sci. 62: 30-37.
Sauer, A. J., E. Fritsch, K. Undorf-Spahn, P. Nguyen, F. Marec, D. G. Heckel, and J. A. Jehle. 2017a. Novel resistance to Cydia pomonella granulovirus (CpGV) in codling moth shows autosomal and dominant inheritance and confers cross-resistance to different CpGV genome groups. PLoS ONE 12(6): e0179157.
Sauer, A. J., S. Schulze-Bopp, E. Fritsch, K. Undorf-Spahn, and J. A. Jehle. 2017b. A third type of resistance of codling moth against Cydia pomonella granulovirus (CpGV) shows a mixture of a Z-linked and autosomal inheritance pattern. Appl. Environ. Microbiol. AEM-01036.
Sayyed, A. H., D. Omar, and D. J. Wright. 2004. Genetics of spinosad resistance in a multi-resistant field-selected population of Plutella xylostella. Pest Manag. Sci. 60: 827-832.
Serebrov, V. V., O. N. Gerber, A. A. Malyarchuk, V. V. Martemyanov, A. A. Alekseev, and V. V. Glupov. Effect of entomopathogenic fungi on detoxification enzyme activity in greater wax moth Galleria mellonella L. (Lepidoptera, Pyralidae) and role of detoxification enzymes in development of insect resistance to entomopathogenic fungi. Anim. Human Physiol. 33: 581-586.
Siegwart, M., B. Graillot, C. B. Lopez, S. Besse, M. Bardin, P. C. Nicot, and M. Lopez-Ferber. 2015. Resistance to bio-insecticides or how to enhance their sustainability: a review. Front. Plant Sci. 6:381. https://doi.org/10.3389/fpls.2015.00381
Tabashnik, B. 1994. Evolution of resistance to Bacillus thuringiensis. Annu. Rev. Entomol. 39: 47-79.
Tabashnik, B. E., N. L. Cushing, N. Finson, and M. W. Johnson. 1990. Field development of resistance to Bacillus thuringiensis in diamondback moth (Lepidoptera: Plutellidae). J. Econ. Entomol. 83: 1671-1676.
Trisyono, A. and M. E. Whalon. 2000. Toxicity of neem applied alone and in combination with Bacillus thuringiensis to Colorado potato beetle (Coleoptera: Chrysomelidae). J. Econ. Entomol. 92: 1281-1288.
Whitehead, G. B. 1959. Pyrethrum resistance conferred by resistance to DDT in the blue tick. Nature 184: 378-379.
Wilson K., S. C. Cotter, A. F. Reeson, and J. K. Pell. 2003. Melanism and disease resistance in insects. Ecology Letters 4: 637-649.
Zhao, P., Z. Dong, J. Duan, G. Wang, L. Wang, Y. Li, Z. Xiang, and Q. Xia. 2012. Genome-wide identification and immune response analysis of serine protease inhibitor genes in the silkworm, Bombyx mori. PloS one 7: e31168.
- Author: Surendra K. Dara
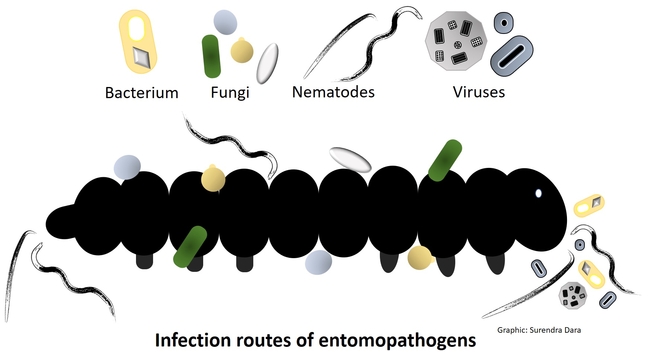
Entomopathogens are microorganisms that are pathogenic to arthropods such as insects, mites, and ticks. Several species of naturally occurring bacteria, fungi, nematodes, and viruses infect a variety of arthropod pests and play an important role in their management. Some entomopathogens are mass-produced in vitro (bacteria, fungi, and nematodes) or in vivo (nematodes and viruses) and sold commercially. In some cases, they are also produced on small scale for non-commercial local use. Using entomopathogens as biopesticides in pest management is called microbial control, which can be a critical part of integrated pest management (IPM) against several pests.
Some entomopathogens have been or are being used in a classical microbial control approach where exotic microorganisms are imported and released for managing invasive pests for long-term control. The release of exotic microorganisms is highly regulated and is done by government agencies only after extensive and rigorous tests. In contrast, commercially available entomopathogens are released through inundative application methods as biopesticides and are commonly used by farmers, government agencies, and homeowners. Understanding the mode of action, ecological adaptations, host range, and dynamics of pathogen-arthropod-plant interactions is essential for successfully utilizing entomopathogen-based biopesticides for pest management in agriculture, horticulture, orchard, landscape, turf grass, and urban environments.
Entomopathogen groups
Important entomopathogen groups and the modes of their infection process are described below.
Bacteria
There are spore-forming bacterial entomopathogens such as Bacillus spp., Paenibacillus spp., and Clostridium spp, and non-spore-forming ones that belong to the genera Pseudomonas, Serratia, Yersinia, Photorhabdus, and Xenorhabdus. Infection occurs when bacteria are ingested by susceptible insect hosts. Pseudomonas, Serratia and Yersinia are not registered in the USA for insect control.Several species of the soilborne bacteria, Bacillus and Paenibacillus are pathogenic to coleopteran, dipteran, and lepidopteran insects. Bacillus thuringiensis subsp. aizawai, Bt subsp. kurstaki, Bt subsp. israelensis, Bt subsp. sphaericus, and Bt subsp. tenebrionis are effectively used for controlling different groups of target insects. For example, Bt subsp. aizawai and Bt subsp. kurstaki are effective against caterpillars, Bt subsp. israelensis and Bt subsp. sphaericus target mosquito larvae, and Bt subsp. tenebrionis is effective against some coleopterans.
When Bt is ingested, alkaline conditions in the insect gut (pH 8-11) activate the toxic protein (delta-endotoxin) that attaches to the receptors sites in the midgut and creates pore in midgut cells. This leads to the loss of osmoregulation, midgut paralysis, and cell lysis. Contents of the gut leak into insect's body cavity (hemocoel) and the blood (hemolymph) leaks into the gut disrupting the pH balance. Bacteria that enter body cavity cause septicemia and eventual death of the host insect. Insects show different kinds of responses to Bt toxins depending on the crystal proteins (delta-endotoxin), receptor sites, production of other toxins (exotoxins), and requirement of spore. The type responses below are based on the susceptibility of caterpillars to Bt toxins.
Type I response – Midgut paralysis occurs within a few minutes after delta-endotoxin is ingested. Symptoms include cessation of feeding, increase in hemolymph pH, vomiting, diarrhea, and sluggishness. General paralysis and septicemia occur in 24-48 hours resulting in the death of the insect. Examples of insects that show Type I response include silkworm, tomato hornworm, and tobacco hornworm.
Type II response – Midgut paralysis occurs within a few minutes after the ingestion of delta-endotoxin, but there will be no general paralysis. Septicemia occurs within 24-72 hours. Examples include inchworms, alfalfa caterpillar, and cabbage butterfly.
Type III response – Midgut paralysis occurs after delta-endotoxin is ingested followed by cessation of feeding. Insect may move actively as there will be no general paralysis. Mortality occurs in 48-96 hours. Higher mortality occurs if spores are ingested. Insect examples include Mediterranean flour moth, corn earworm, gypsy moth, spruce budworm.
Type IV response – Insects are naturally resistant to infection and older instars are less susceptible than the younger ones. Midgut paralysis occurs after delta-endotoxin is ingested followed by cessation of feeding. Insect may move actively as there will be no general paralysis. Mortality occurs in 72-96 or more hours. Higher mortality occurs if spores are ingested. Cutworms and armyworms are examples for this category.
Unlike caterpillars, the response in mosquitoes is different where upon ingestion of Bt subsp. israelensis delta-endotoxin, the mosquito larva is killed within 20-30 min.
While Bt with its toxic proteins is very effective as a biopesticide against several pests, excessive use can lead to resistance development. Corn earworm, diamondback moth, and tobacco budworm are some of the insects that developed resistance to Bt toxins. Genetic engineering allowed genes that express Bt toxins to be inserted into plants such as corn, cotton, eggplant, potato, and soybean and reduced the need to spray pesticides. However, appropriate management strategies are necessary to reduce insect resistant to Bt toxins in transgenic plants.
Paenibacillus popilliae is commonly used against Japanese beetle larvae and known to cause the milky spore disease. Although Serratia is not registered for use in the USA, a species is registered for use against a pasture insect in New Zealand. In the case of Photorhabdus spp. and Xenorhabdus spp., which live in entomopathogenic nematodes symbiotically, bacteria gain entry into the insect host through nematodes. Biopesticides based on heat-killed Chromobacterium subtsugae and Burkholderia rinojensis are reported to have multiple modes of action and target mite and insect pests of different orders.
Fungi
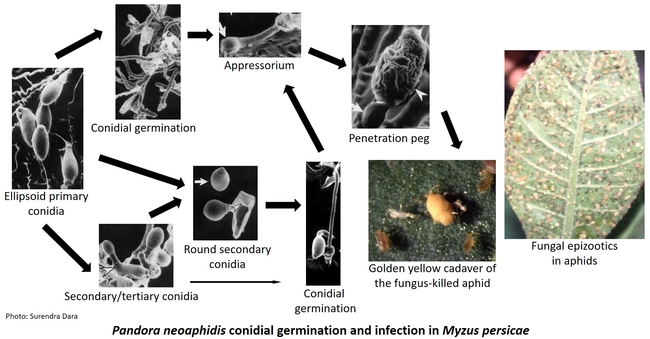
Entomopathogenic fungi typically cause infection when spores come in contact with the arthropod host. Under ideal conditions of moderate temperatures and high relative humidity, fungal spores germinate and breach the insect cuticle through enzymatic degradation and mechanical pressure to gain entry into the insect body. Once inside the body, the fungi multiply, invade the insect tissues, emerge from the dead insect, and produce more spores. Natural epizootics of entomophthoralean fungi such as Entomophaga maimaiga (in gypsy moth), Entomophthora muscae (in flies), Neozygites fresenii (in aphids), N. floridana (in mites), and Pandora neoaphidis (in aphids) are known to cause significant reductions in host populations. Although these fastidious fungi are difficult to culture in artificial media and do not have the potential to be sold as biopesticides they are still important in natural control of some pest species. Hypoclealean fungi such as Beauveria bassiana, Isaria fumosorosea, Hirsutella thompsonii, Lecanicillium lecanii, Metarhizium acridum, M. anisopliae, and M. brunneum, on the other hand, are commercially sold as biopesticides in multiple formulations around the world. Fungal pathogens have a broad host range and are especially suitable for controlling pests that have piercing and sucking mouthparts because spores do not have to be ingested. However, entomopathogenic fungi are also effective against a variety of pests such as wireworms and borers that have chewing mouthparts.
Related to fungi, the spore-forming microsporidium, Paranosema (Nosema) locustae is a pathogen that has been used for controlling locusts, grasshoppers, and some crickets. When P. locustae is ingested, the midgut tissues become infected, followed by infection in the fat body tissues. The disease weakens and eventually kills the orthopteran host within a few weeks.
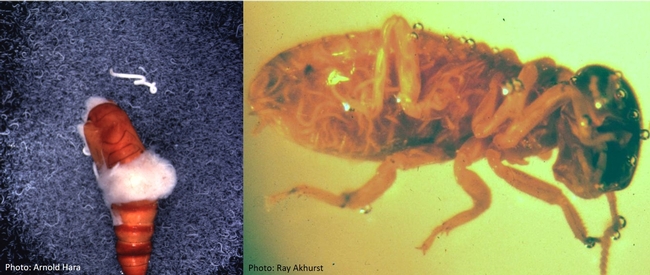
Various insects killed by different species of entomopathogenic fungi
Nematodes
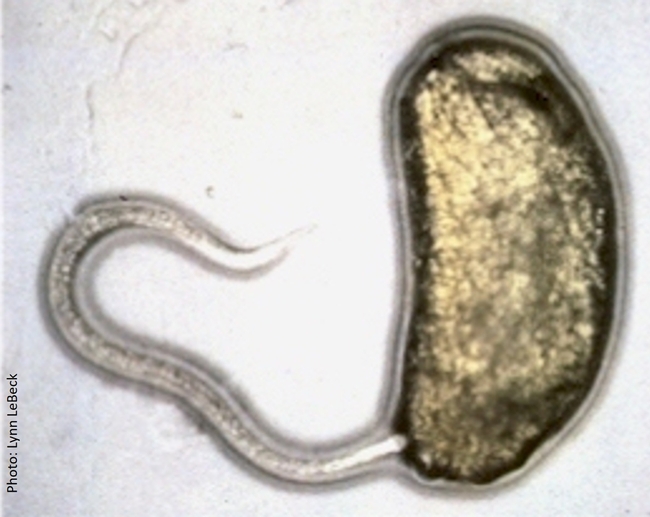
Entomopathogenic nematodes are microscopic, soil-dwelling worms that are parasitic to insects. Several species of Heterorhabditis and Steinernema are available in multiple commercial formulations, primarily for managing soil insect pests. Infective juveniles of entomopathogenic nematodes actively seek out their hosts and enter through natural openings such as the mouth, spiracles, and anus or the intersegmental membrane. Once inside the host body, the nematodes release symbiotic bacteria that kill the host through bacterial septicemia. Heterorhabditis spp. carry Photorhabdus spp. bacteria and Steinernema spp. carry Xenorhabdus spp. bacteria. Phasmarhabditis hermaphrodita is also available for controlling slugs in Europe, but not in the USA.
Infective juvenile of Steinernema carpocapsae entering the first instar larva of a leafminer through its anus.
Nematodes in beet armyworm pupa (left) and termite worker (right).
Viruses
Similar to bacteria, entomopathogenic viruses need to be ingested by the insect host and therefore are ideal for controlling pests that have chewing mouthparts. Several lepidopteran pests are important hosts of baculoviruses including nucleopolyhedroviruses (NPV) and granuloviruses (GV). These related viruses have different types of occlusion bodies in which the virus particles (virions) are embedded. Virus particles invade the nucleus of the midgut, fat body or other tissue cells, compromising the integrity of the tissues and liquefying the cadavers. Before death, infected larvae climb higher in the plant canopy, which aids in the dissemination of virus particles from the cadavers to the lower parts of the canopy. This behavior aids in the spread of the virus to cause infection in healthy larvae. Viruses are very host specific and can cause significant reduction of host populations. Examples of some commercially available viruses include Helicoverpa zea single-enveloped nucleopolyhedrovirus (HzSNVP), Spodoptera exigua multi-enveloped nucleopolyhedrovirus (SeMNPV), and Cydia pomonella granulovirus (CpGV).
Most entomopathogens typically take 2-3 days to infect or kill their host except for viruses and P. locustae which take longer. Compared to viruses (highly host specific) and bacteria (moderately host specific), fungi generally have a broader host range and can infect both underground and aboveground pests. Because of the soil-dwelling nature, nematodes are more suitable for managing soil pests or those that have soil inhabiting life stages.
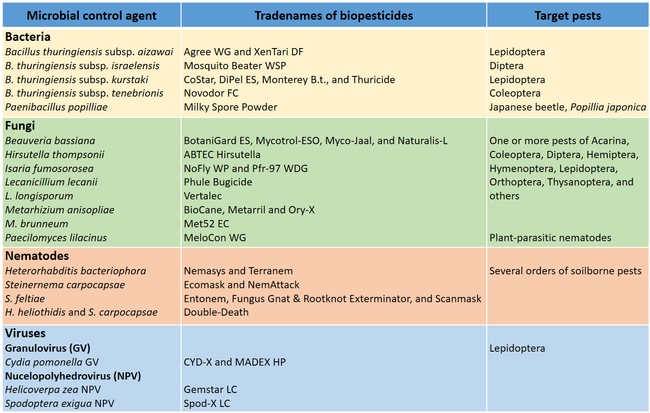
Biopesticides based on various entomopathogenic microorganisms and their target pests
Microbial control and Integrated Pest Management
There are several examples of entomopathogen-based biopesticides that have played a critical role in pest management. Significant reduction in tomato leaf miner, Tuta absoluta, numbers and associated yield loss was achieved by Bt formulations in Spain (Gonzalez-Cabrera et al, 2011). Bt formulations are also recommended for managing a variety of lepidopteran pests on blueberry, grape, and strawberry (Haviland, 2014; Zalom et al, 2014; Bolda and Bettiga, 2014; Varela et al, 2015).
Lecanicellium muscarium-based formulation reducedgreenhouse whitefly (Trialeurodes vaporariorum) populations by 76-96% in Mediterranean greenhouse tomato (Fargues et al, 2005). In other studies, B. bassiana applications resulted in a 93% control of twospotted spider mite (Tetranychus urticae) populations in greenhouse tomato (Chandler et al, 2005) and 60-86% control on different vegetables (Gatarayiha et al, 2010). The combination of B. bassiana and azadirachtin reduced rice root aphid (Rhopalosiphum rufiabdominale) and honeysuckle aphid (Hyadaphis foeniculi) populations by 62% in organic celery in California (Dara, 2015a). Chromobacterium subtsugae and B. rinojensis caused a 29 and 24% reduction, respectively, in the same study. IPM studies in California strawberries also demonstrated the potential of entomopathogenic fungi for managing the western tarnished plant bug (Lygus hesperus) and other insect pests (Dara, 2015b, 2016). Entomopathogenic fungi also have a positive effect on promoting drought tolerance or plant growth as seen in cabbage (Dara et al, 2016) and strawberry (Dara, 2013) and antagonizing plant pathogens (Dara et al, 2017)
Application of SeMNPV was as efficacious as methomyl and permithrin in reducing beet armyworms (S. exigua) in head lettuce in California (Gelernter et al, 1986). Several studies demonstrated PhopGV as an important tool for managing the potato tubermoth (Phthorimaea operculella) (Lacey and Kroschel, 2009).
The entomopathogenic nematode, S. feltiae,reduced raspberry crown borer (Pennisetia marginata) populations by 33-67% (Capinera et al, 1986). For managing the branch and twig borer (Melagus confertus) in California grapes, S. carpocapsae is one of the recommended options (Valera et al, 2015).
Entomopathogens can be important tools in IPM strategies in both organic and conventional production systems. Depending on the crop, pest, and environmental conditions, entomopathogens can be used alone or in combination with chemical, botanical pesticides or other entomopathogens.
Acknowledgements: Thanks to Dr. Harry Kaya for reviewing this article.
References
Bolda, M. P. and L. J. Bettiga. 2015. UC IPM Pest Management Guidelines: Caneberries. UC ANR Pub. 3437.
Capinera, J. L., W. S. Cranshaw, and H. G. Hughes. 1986. Suppression of raspberry crown borer Pennisetia marginata (Harris) (Lepidoptera: Sesiidae) with soil applications of Steinernema feltiae (Rhabditida:Steinernematidae). J. Invertebr. Pathol. 48: 257-258.
Chanlder, D., G. Davidson, and R. J. Jacobson. 2005. Laboratory and glasshouse evaluation of entomopathogenic fungi angainst the two-spotted spider mite, Tetranychus urticae (Acari: Tetranychidae), on tomato, Lycopersicon esculentum. Biocon. Sci. Tech. 15: 37-54.
Dara, S. K. 2013. Entomopathogenic fungus Beauveria bassiana promotes strawberry plant growth and health. UCANR eJournal Strawberries and Vegetables, 30 September, 2013. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11624)
Dara, S. K. 2015a. Reporting the occurrence of rice root aphid and honeysuckle aphid and their management in organic celery. UCANR eJournal Strawberries and Vegetables, 21 August, 2015. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=18740)
Dara, S. K. 2015b. Integrating chemical and non-chemical solutions for managing lygus bug in California strawberries. CAPCA Adviser 18 (1) 40-44.
Dara, S. K. 2016. IPM solutions for insect pests in California strawberries: efficacy of botanical, chemical, mechanical, and microbial options. CAPCA Adviser 19 (2): 40-46.
Dara, S. K., S.S.R. Dara, and S.S. Dara. 2016. First report of entomopathogenic fungi, Beauveria bassiana, Isaria fumosorosea, and Metarhizium brunneum promoting the growth and health of cabbage plants growing under water stress. UCANR eJournal Strawberries and Vegetables, 19 September, 2016. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=22131)
Dara, S.S.R., S. S. Dara, S. K. Dara, and T. Anderson. 2017. Fighting plant pathogenic fungi with entomopathogenic fungi and other biologicals. CAPCA Adviser 20 (1): 40-44.
Fargues, J., N. Smits, M. Rougier, T. Boulard, G. Rdray, J. Lagier, B. Jeannequin, H. Fatnassi, and M. Mermier. 2005. Effect of microclimate heterogeneity and ventilation system on entomopathogenic hyphomycete infectiton of Trialeurodes vaporariorum (Homoptera: Aleyrodidae) in Mediterranean greenhouse tomato. Biological Control 32: 461-472.
Gatarayiha, M. C., M. D. Laing, and M. Ray. 2010. Effects of adjuvant and conidial concentration on the efficacy of Beauveria bassiana for the control of the two-spotted spider mite, Tetranychus urticae. Exp. Appl. Acarol. 50: 217-229.
Gelernter, W. D., N. C. Toscano, K. Kido, and B. A. Federici. 1986. Comparison of a nuclear polyhedrosis virus and chemical insecticides for control of the beet armyworm (Lepidopter: Noctuidae) on head lettuce. J. Econ. Entomol. 79: 714-717.
González-Cabrera, J., J. Mollá, H. Monton, A. Urbaneja. 2011. Efficacy of Bacillus thuringiensis (Berliner) in controlling the tomato borer, Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). BioControl 56: 71–80.
Haviland, D. R. 2014. UC IPM Pest Management Guidelines: Blueberry. UC ANR Pub. 3542.
Lacey, L. A. and J. Kroschel. 2009. Microbial control of the potato tuber moth (Lepidoptera: Gelechiidae). Fruit Veg. Cereal Sci. Biotechnol. 3: 46-54.
Varela, L. G., D. R. Haviland, W. J., Bentley, F. G. Zalom, L. J. Bettiga, R. J. Smith, and K. M. Daane. 2015. UC IPM Pest Management Guidelines: Grape. UC ANR Pub. 3448.
Zalom, F. G., M. P. Bolda, S. K. Dara, and S. Joseph. 2014. UC IPM Pest Management Guidelines: Strawberry. UC ANR Pub. 3468.
- Author: Surendra K. Dara
- Author: David Peck, Manzanita Berry Farms
Six-month old strawberry field.
Under the soil is a complex and dynamic world of moisture, pH, salinity, nutrients, microorganisms, and plant roots along with pests, pathogens, weeds and more. A good balance of essential nutrients, moisture, and beneficial microorganisms provides optimal plant growth and yield. These factors also influence natural plant defenses and help withstand stress caused by biotic and abiotic factors.
Several beneficial microbe-based products are commercially available to promote plant growth and improve health, yield potential and quality. Some of them improve nutrient and water absorption while others provide protection against plant pathogens or improve plant defense mechanism. In addition to the macronutrients such as nitrogen, phosphorus, and potassium, several micronutrients are critical for optimal growth and yield potential. Some of the micronutrient products are also useful in promoting beneficial microbes. Understanding the plant-microbe-nutrient interactions and how different products help crop production are helpful for making appropriate decisions.
Mycorrhizae (fungi of roots) establish a symbiotic relationship with plants and serve as an extended network of the root system. They facilitate improved uptake of moisture and nutrients resulting in better plant growth and yield (Amerian and Stewart, 2001; Wu and Zou, 2009; Bolandnazar et al., 2007; Nedorost et al., 2014). Mycorrhizae can also help absorb certain nutrients more efficiently than plants can and make them more readily available for the plant. With increased moisture and nutrient absorption, plants can become more drought-tolerant. Mycorrhizae also help plants to withstand saline conditions and protect from plant pathogens. A healthy root system can fight soil diseases and weed invasion. Additionally, mycorrhizae increase organic matter content and improve soil structure.
Considering an increasing need for fumigation alternatives to address soilborne pathogens in strawberry, mycorrhizae and other beneficial microbes could be potential tools in maintaining plant health. Additionally, recent studies suggest that entomopathogenic fungi such as Beauveria bassiana, Metarhizium brunneum, and Isaria fumosorosea form mycorrhiza-like and endophytic relationships with various species of plants and could help with plant growth and health (Behie and Bidochka, 2014; Dara et al., 2016). These fungi are currently used for pest management, but their interaction with plants is a new area of research. Understanding this interaction will potentially expand the use of the biopesticides based on these fungi for improving plant growth and health. A study was conducted at Manzanita Berry Farms, Santa Maria in fall-planted strawberry crop during the 2014-2015 production season to evaluate the impact of beneficial microbes on strawberry growth, health, mite infestations, powdery mildew, botrytis fruit rot, and yield.
Methodology:
List of treatments, their application rates and frequencies:
- Untreated control: Received no supplemental treatments other than standard grower practices.
- HealthySoil: NPK (0.1-0.1-0.1).
- BotaniGard ES: Entomopathogenic fungus Beauveria bassiana strain GHA. Rate - 1 qrt in 50 gal for a 30 min transplant dip and 1 qrt/ac every 15 days until January and once a month thereafter until April, 2015.
- Met52: Entomopathogenic fungus Metarhizium brunneum strain F52. Rate – 16 fl oz in 50 gal for a 30 min transplant dip and 16 fl oz/ac every 15 days until January and once a month thereafter until April, 2015.
- NoFly: Entomopathogenic fungus Isaria fumosorosea strain FE9901. Rate – 11.55 oz in 50 gal for a 30 min transplant dip and 11.55 oz/ac every 15 days until January and once a month thereafter until April, 2015.
- Actinovate AG: Beneficial soilborne bacterium Streptomyces lydicus WYEC 108. Rate – 6 oz in 50 gal for a 30 mintransplant dip and 6 oz/ac every month.
- TerraClean 5.0: Hydrogen dioxide and peroxyacetic acid. Rate – 1:256 dilution for a 1 min root dip followed by 2 gal/ac 10 days after planting and then 2 and 1 gal/ac alternated every 15 days until April, 2015.
- TerraGrow: Humic acids, amino acids, sea kelp, glucose based carriers, bacteria – Bacillus licheniformis, B. subtilis, B. pumilus, B. amyloliquefaciens, and B. magaterium, and mycorrhizae – Trichoderma harzianum and T. reesei. Rate – 1.13 g in 10 gal for a 1 min root dip followed by 1.5 lb/ac 10 days after planting and once every month until April, 2015.
- TerraCelan and TerraGrow: Same as individual treatments at the time of planting, but TerraClean at 2 gal/ac and TerraGrow at 1.5 lb/ac 10 days after planting followed by monthly treatments until April, 2015.
- O-MEGA: NPK (0.2-1.0-0.5), bacteria – Azotobacter chroococcum, Azospirillum lipoferum, Lactobacillus acidophilus, Pseudomonas fluorescens, Cellulomonas cellulans and the fungus Aspergillus niger. Rate – 20 ml in 1 gal sprinkled on transplants 30 min before planting followed by 1 qrt/ac every week rest of the season.
Strawberry transplants (variety BG-6.3024) were treated at the time of planting on 6 November, 2014 and treatments are also administered periodically through the drip irrigation system following the abovementioned schedule. Each treatment had two 330' long beds each with four rows of plants. Treatments were randomly arranged in two blocks and two sampling plots (20' long) were established within each bed in a block. The impact of the treatments on plant growth (canopy size), health, spider mite populations, botrytis and powdery mildew severity, and yield were monitored periodically. Plant growth was determined by measuring the canopy size. Plant health was rated on a scale of 0 to 5 where 0=dead, 1=weak, 2=moderate low, 3=moderate high, 4=good, and 5=very good. Powdery mildew severity was determined by observing leaf samples under microscope and rating the severity on a scale of 0 to 4 where 0=no infection, 1=1-25%, 2=26-50%, 3=51-75%, and 4=76-100% of leaf area with powdery mildew. Twenty plants or leaf samples per plot were used for these observations. To monitor botrytis fruit rot, a box of fruits from each plot were held at room temperature and disease was rated 3 and 5 days after harvest on a scale of 0 to 4 where 0=no infection, 1=1-25%, 2=26-50%, 3=51-75%, and 4=76-100% of fruit with botrytis. Yield data were also collected from the plots throughout the production season using grower's harvesting schedule. Mite counts were also taken periodically.
Data were analyzed using analysis of variance and significant means were separated using Tukey's HSD means separation test.
Treating the transplants with different treatment materials and planting in respective beds
Newly transplanted experimental plots.
Chris Martinez (center, front row) and rest of the field crew at Manzanita Berry Farms
Results:
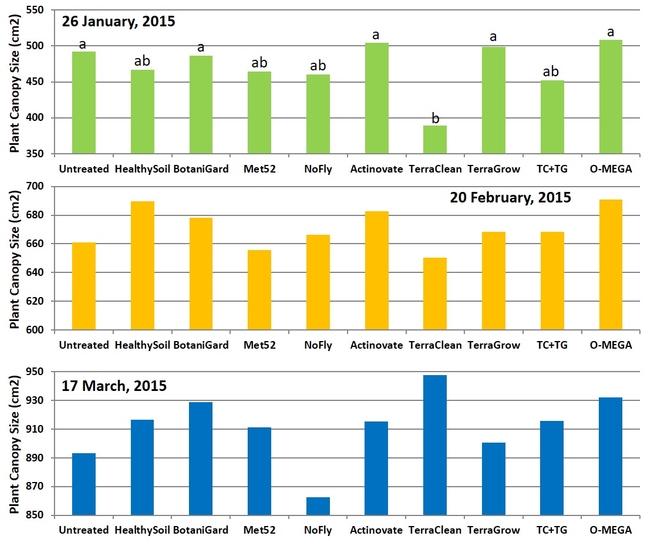
Canopy size: Significant differences (P = 0.002) among treatments were seen only on the first observation date on 26 January, 2015 where TerraClean-treated plants were smaller than some of the treatments. There were no significant differences (P > 0.05) in treatments on the following observations in February and March, however TerraClean-treated plants recovered and plants were larger in some of the treatments.
Size of the plant canopy on three observation dates.
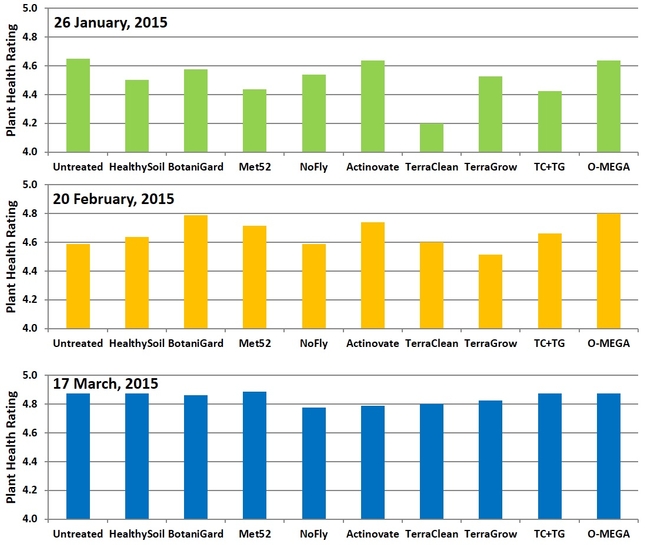
Plant health: Treatments did not have a significant (P > 0.05) impact on plant health. Health ratings varied from 4.2 for TerraClean to 4.6 for untreated, BotaniGard, Actinovate, and O-Mega treatments in January. In February, TerraGrow-treated plants had 4.5 rating and BotaniGard and O-Mega treatments had 4.8. March ratings varied between 4.8 and 4.9 in all the treatments. As there were no soilborne diseases during the study period, the impact of the treatments could not be determined, which was the main objective of the study.
Plant health ratings on three observation dates.
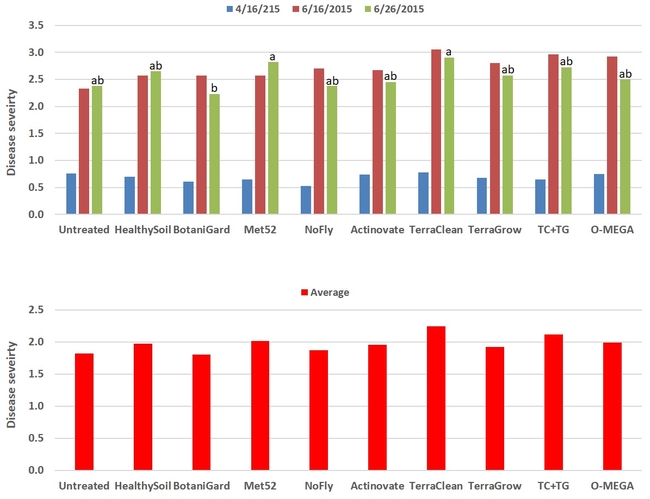
Powdery mildew: Disease severity did not differ among treatments (P > 0.05) on 16 April and 16 June, but significant (P = 0.008) differences were observed on 26 June where BotaniGard-treated plants had the lowest. When data were compared for the three observation dates, severity rating varied from 1.8 for BotaniGard to 2.24 for TerraClean.
Powdery mildew severity on individual observation dates (top) and combined for three observations (bottom)
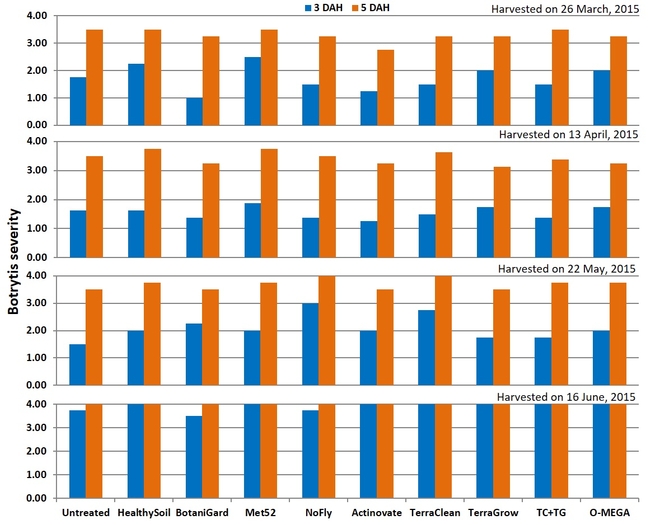
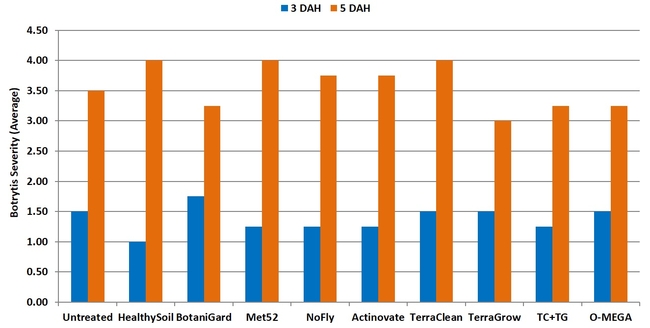
Botrytis fruit rot: There were no significant (P > 0.05) differences among treatments on any of the four observation dates or when data were combined for all observations. In general, fruit rot was less severe 3 days after harvest than 5 days after during the first three observation dates. When data were combined for the observation dates, HealthySoil treatment had a rating of 1 followed by Met52, NoFly, Actinovate, and TerraClean+TerraGrow with a 1.3 rating for 3 days after harvest.
Severity of botrytis fruit rot 3 and 5 days after harvest on individual observation dates (above) and when data were combined (below).
Spider mites: Mite populations were very low in all the plots during observation period and data were not included.
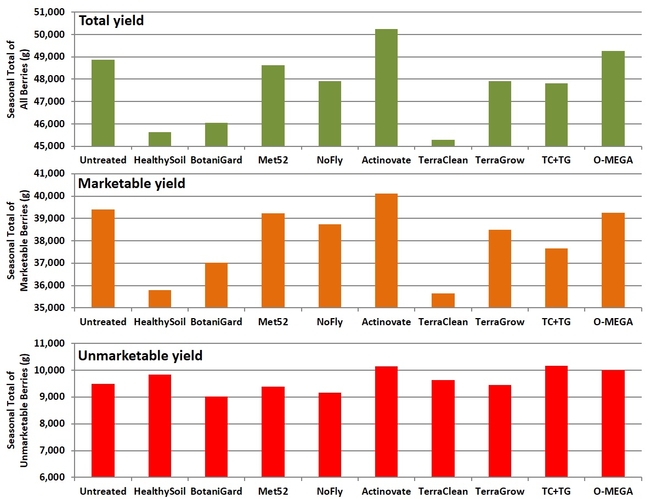
Fruit yield: While the seasonal yield of total, marketable, or unmarketable berries was not significantly (P > 0.05) different for any of the treatments marketable yields had a wider range than unmarketable yields among treatments. The lowest marketable fruit yield was seen in TerraClean (35.6 kg or 79.4 lb) and HealthySoil (35.8 kg or 79.8 lb) while the highest yield was seen in Actinovate (40.1 kg or 89.4 lb) followed by untreated control (39.4 kg or 87.9 lb), O-Mega (39.3 kg or 87.6 lb), Met52 (39.2 kg or 87.4 lb), and NoFly (38.7 kg or 86.3 lb) treatments.
Seasonal yields of total, marketable, and unmarketable strawberries per plot.
This is the first field study evaluating the impact of three popular entomopathogenic fungi along with multiple beneficial microbes on strawberry plant growth, foliar and fruit diseases, and yield. While differences among treatments were not pronounced, it appeared that some had a positive impact on some of the parameters measured. It is interesting to note that yields were higher (although not statistically significant) than the grower standard, HealthySoil. Compared to the grower standard, marketable yield was higher in many other treatments. Since an untreated situation is not common in a commercial field, using beneficial microbes can be useful. Although previous field studies evaluated the impact of with the entomopathogenic fungus B. bassiana in strawberries (Dara, 2013; Dara, 2016), a positive impact on plant growth or yield by I. fumosorosea and M. brunneum in commercial strawberries has never been reported earlier.
Additional studies with different application rates would be useful to understand how beneficial microbes could be exploited more.
Acknowledgments: Thanks to Dave Peck, Manzanita Berry Farms for the collaboration and industry partners for the financial support. Thanks to Chris Martinez and rest of the field crew at Manzanita Berry Farms and Fritz Light and Tamas Zold for the technical assistance.
http://ucanr.edu/articlefeedback
References
Amerian, M.R., and W.S. Stewart. 2001. Effect of two species of arbuscular mycorrhizal fungi on growth, assimilation and leaf water relations in maize (Zea mays). Aspects of Appl. Biol. 63: 1-6.
Behie, S.W., and M.J. Bidochka. 2014. Nutrient transfer in plant-fungal symbioses. Trends in Plant Sci. 19: 734-740.
Bolandnazar, S., N. Aliasgarzad, M.R. Neishabury, and N. Chaparzadeh. 2007. Mycorrhizal colonization improves onion (Allium cepa L.) yield and water use efficiency under water deficit condition. Sci. Horticulturae 114: 11-15.
Dara, S. K. 2013. Entomopathogenic fungus Beauveria bassiana promotes strawberry plant growth and health. UCANR eJournal Strawberries and Vegetables, 30 September, 2013. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11624)
Dara, S. K. 2016. First field study evaluating the impact of the entomopathogenic fungus Beauveria bassiana on strawberry plant growth and yield. UCANR eJournal Strawberrries and Vegetables, 7 November, 2016. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=22546)
Dara, S. K., S.S.R. Dara, and S. S. Dara. 2016. First report of entomopathogenic fungi, Beauveria bassiana, Isaria fumosorosea, and Metarhizium brunneum promoting the growth and health of cabbage plants growing under water stress. UCANR eJournal Strawberries and Vegetables, 16 September, 2016.(http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=22131)
Nedorost, L., J. Vojtiskova, and R. Pokluda. 2014. Influence of watering regime and mycorrhizal inoculation on growth and nutrient uptake of pepper (Capsicum annuum L.). VII International symposium on irrigation of horticultural crops, Braun P., M. Stoll, and J. Zinkernagel (eds). Acta Horticulturae 1038:559-564.
Wu, Q.S., and Y. Zou. 2009. Mycorrhizal influence on nutrient uptake of citrus exposed to drought stress. Philippine Agri. Scientist 92: 33-38.