- Author: Steven T. Koike
- Author: Mark Bolda
Finally. Under the guidance of USDA-ARS and other researchers, and with the help of the California Strawberry Commission and other supporters, the UC Cooperative Extension diagnostic laboratory in Salinas finally has joined the 21st century regarding molecular diagnostic technologies. A DNA-based test is now being implemented to detect and confirm important soilborne diseases of coastal crops. This method can complete the diagnostic process in a few hours, compared to the conventional culture method, which usually takes several days.
The Need. Rapid, accurate, and dependable diagnosis of plant diseases is an essential part of agriculture production. Without knowing the precise agent responsible for the disease, growers, pest control advisers, and other field personnel are hampered when making disease management decisions. Soilborne diseases are particularly challenging to identify. In general, the appearances of the various root rots, crown rots, plant collapses, and vascular wilts all look very similar, making it virtually impossible to diagnose these based on symptoms alone. The UC Cooperative Extension laboratory has some very accurate culture methods to grow out and detect all of these soilborne pathogens. However, such culture techniques are relatively slow and take from three to seven days to complete. For our fast growing coastal crops a more rapid means of disease diagnosis would well serve the growers.
The Method. The new method goes by the name “RPA” which stands for recombinase polymerase amplification. The RPA method, like many other technologies such as the polymerase chain reaction (PCR), is based on DNA components (called primers and probe) that specifically recognize the corresponding DNA found in the target pathogen. However, all PCR methods depend on extracting and purifying the pathogen DNA; getting the DNA out is costly (one needs to buy expensive equipment to analyze the sample) and it requires more time to complete the elaborate procedures. RPA is perfectly suited to an extension laboratory, like the one in Salinas, because DNA purification is not needed as the test uses ground up, crude plant extracts as a source of unpurified DNA (see photos below).
Pathogens Detected. Presently the UC Cooperative Extension laboratory offers RPA diagnostic tests for two pathogens. Phytophthora causes root and crown rots on strawberry, raspberry, and a number of vegetable crops and ornamental/forest plants. Our Phytophthora tests are either genus-specific (it will detect Phytophthora infecting many types of plants) or species-specific for the two pathogens that mostly attack strawberry (P. cactorum and P. fragariae) (Table 1). Our other RPA test detects the specific type of Macrophomina phaseolina that causes severe crown rot and collapse of strawberry. Because this Macrophomina RPA is designed for the strawberry pathogen and not for Macrophomina that infects other crops, this is called a genotype-specific test (Table 1). This summer we anticipate adding tests for the Verticillium wilt pathogen (V. dahliae that infects many crops) and the Fusarium wilt pathogen of strawberry (Fusarium oxysporum f. sp. fragariae). All tests analyze infected plant material only. However, it is hoped that our continuing research will adapt RPA or more sophisticated molecular techniques for detecting these pathogens in soil.
Table 1. RPA tests currently available at the UC Cooperative Extension laboratory.
|
Category of test |
Pathogen |
Comments |
|
Genus-specific |
Phytophthora |
Detects this pathogen in all plants, such as strawberry, raspberry, pepper, asparagus, tomato, avocado, ornamentals, forest species (sudden oak death pathogen) |
|
Species-specific |
Phytophthora cactorum |
Primarily for strawberry but this pathogen can be found on ornamentals such as coffee berry |
|
Species-specific |
Phytophthora fragariae |
For strawberry |
|
Genotype-specific |
Macrophomina phaseolina |
For Macrophomina that infects strawberry |
The Future. Because this RPA method is similar in design to PCR, and PCR is used to develop the DNA tools (primers) to detect any sort of organism, the RPA platform is open ended. It should be possible to readily develop RPA tests for any organism that has DNA, such as other pathogenic fungi and bacteria, nematodes, and even perhaps specialized sub-groups of organisms such as insect biotypes.
Acknowledgements. This advance in rapid, accurate disease diagnostics was developed and implemented by the following research and extension team: Mark Bolda (UC Extension, Santa Cruz Co.), Alyssa Burkhardt (USDA-ARS, Salinas), Oleg Daugovish (UC Extension, Ventura Co.), Steven Koike (UC Extension, Monterey Co.), Frank Martin (USDA-ARS, Salinas), Stacy Mauzey (UC Extension, Monterey Co.), Tim Miles (CSU Monterey Bay), and Cayla Tsuchida (UC Extension, Monterey Co.). This research was supported by the California Strawberry Commission, California Avocado Commission CSU Agricultural Research Institute (grant 5219101A), and the USDA-California Department of Food and Agriculture Specialty Crop Block Grant Program (grants SCB12051 and SCB14052 awarded to F. Martin).
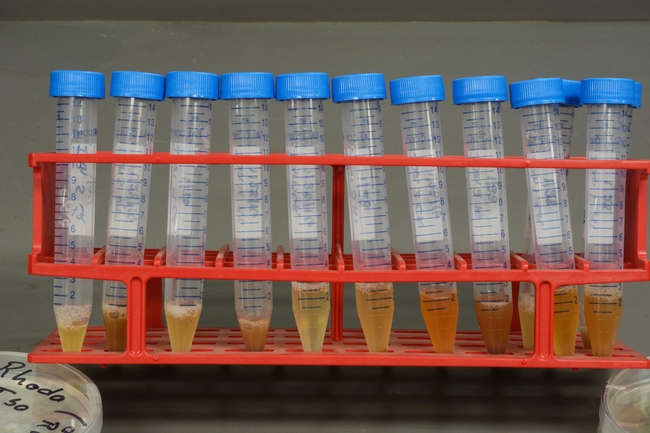
Photo 1: Strawberry samples are examined for symptomatic tissues that indicate possible disease.
Photo 2: Pieces of root, crown, or petiole are macerated and ground up in sample bags containing buffer solutions.
Photo 3: The resulting mixtures are strained and placed into tubes.
Photo 4: Small volumes of the strawberry extracts are loaded into strip tubes.
Photo 5: Loaded strip tubes are placed into the RPA isothermal detection device.
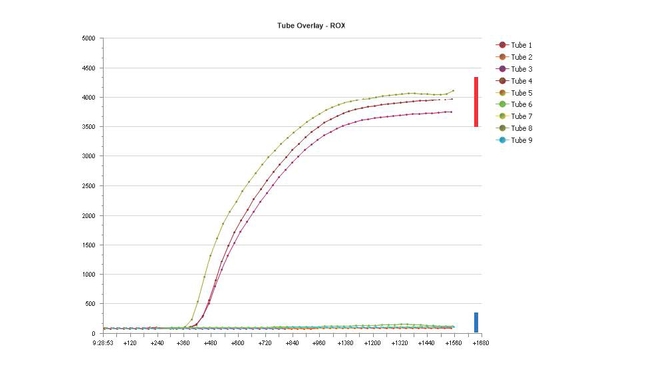
Photo 6: In the computer generated results, samples positive (red bar) for Phytophthora root and crown rot will appear as curved lines moving upwards; negative samples (blue bar) will appear as straight lines mostly remaining flat.
- Author: Richard Smith
- Author: Timothy K Hartz
- Author: Daniel Geisseler
- Author: Patricia Love
Organic production in Monterey County was valued at $335 million in 2015, which was 7% of total agricultural value. Organic agriculture has a prominent role in Monterey County agriculture, but many aspects of organic production are still poorly understood and are in need of more research. The transformations of nitrogen (N) that occur in soil, and organic amendments and fertilizers that ultimately produce plant available forms of nitrogen (nitrate and ammonium, a.k.a. mineral nitrogen) are only understood in a general way. In 2016 we received funding from the Fertilizer Research and Education Program (FREP) which is funded by a mil tax on fertilizer sales, including organic fertilizers, to evaluate nitrogen mineralization (release of mineral nitrogen from soil organic matter) from soils on organic farms and from organic fertilizers. Evaluations included determination of the mineralization of soil organic matter over the course of the cropping cycle of baby vegetables and full-term romaine. In addition, evaluations were conducted of mineralization of organic fertilizers in production fields as well as laboratory incubations carried out at UC, Davis.
Evaluations of soil organic matter
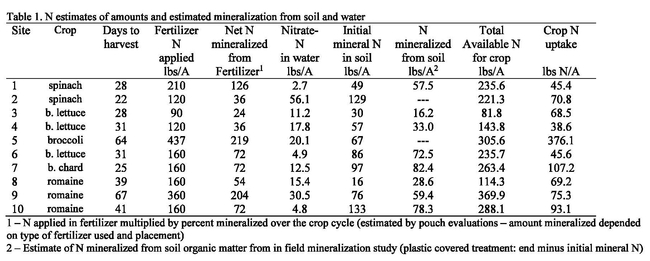
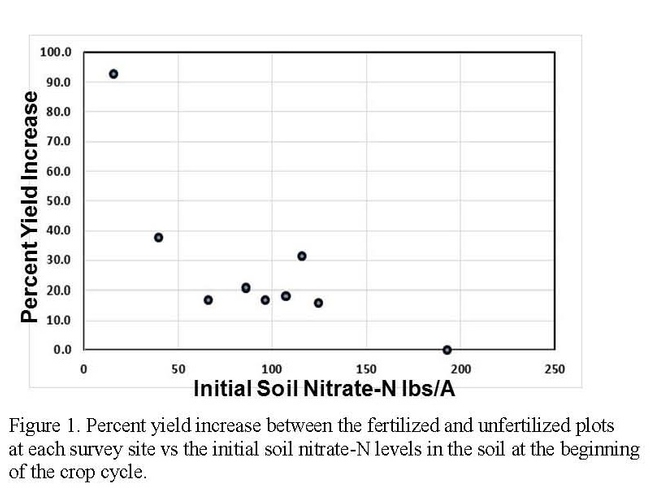
Ten N mineralization trials were conducted in the Salinas Valley with cooperating growers in commercial production fields in diverse locations and with a spectrum of soil types. Field evaluations consisted of establishing replicated unfertilized areas in each field. Within each unfertilized plot, we had areas with and without plants, as well as an area covered with plastic mulch; these areas allowed us to estimate the amount of N mineralized from soil organic matter without the confounding factors of crop uptake and leaching. Unfertilized areas were compared with adjacent fertilized plots. The amount of N mineralized in field evaluations ranged from 0.6 to 3.3 lbs N/A/day; more N mineralization generally occurred in soils with greater quantities of total N in the soil. Nitrate-N at the beginning of the cropping cycle was a useful indication of the amount of N available for crop growth during the crop cycle (Figure 1). Not surprisingly, the sites with low levels of soil nitrate at the start of the crop cycle had greater responses to applied fertilizer. Initial soil nitrate levels would be a great tool for organic growers to utilize to help guide fertilizer decisions, however from these data it is clear that it is not precise enough to fully guide fertilizer decisions except for when levels are extremely high or extremely low. The fields that had initial soil nitrate-N levels of 75-125 lbs N/A had 15-30% greater yield with applied nitrogen fertilizer, even though N uptake by crops was modest for all crops except broccoli. One reason measurements of initial residual soil nitrate levels may not be as good of a predictor of crop response to N might be due to inefficiencies in irrigation management which could result in movement of N beyond the root zone. Water used to germinate the crop or set transplants often exceeds the ability of the soil to retain it, resulting in nitrate leaching at the beginning of the crop cycle. Measuring residual soil nitrate after the germination water applications could improve the estimates of available residual soil nitrate. However, the fast crop cycle of the baby vegetables, necessitates making fertilizer decisions early in the crop cycle.
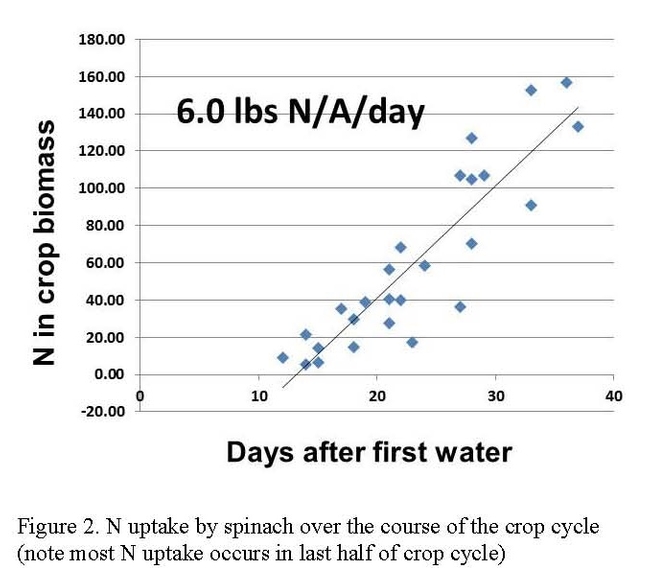
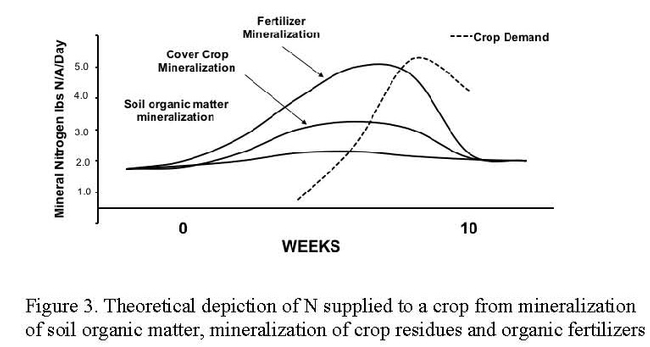
The nitrate levels in the irrigation water in the surveyed sites was low to moderate and did not contribute large amounts of N to the crop budget (Table 1). Mineralization of N from the soil organic matter over the course of the crop cycle was also low to moderate in the surveyed sites. All the crops surveyed in these evaluations have rapid N uptake patterns in the last half of the crop cycle. It is at this time when robust quantities of N are needed to supply the crop with the amounts of N needed to maximize yields (Figures 2 and 3). The N release pattern of organic fertilizers is another factor that makes N management of organic crops difficult.
Evaluations of organic fertilizers
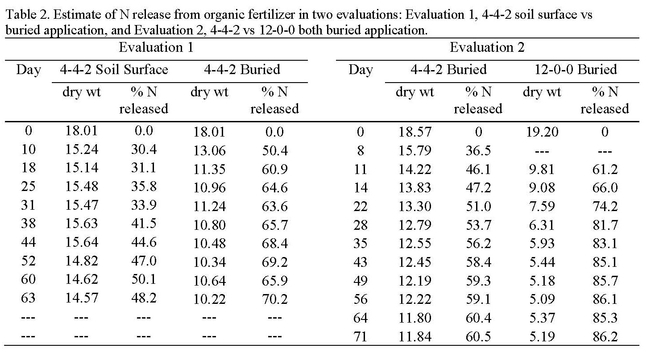
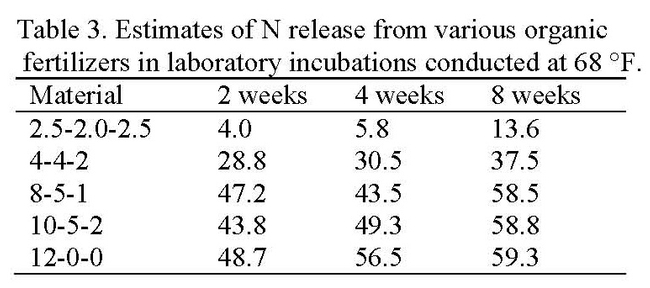
Organic fertilizers used in the production of cool season vegetables are either incorporated into the soil or applied to the top of the beds and sprinkled in. Based on these application methods, field evaluations of organic fertilizer were made by placing organic fertilizers in pouches (See photos below) and applying them to the soil in two ways: 1) buried 3 inches deep to simulate incorporated applications and 2) by placing the pouches on the soil surface to simulate surface (a.k.a. drop-on-top) applications. Pouch evaluations were conducted in commercial production fields and placed in the field at the beginning of the crop cycle. Three to four pouches were collected weekly and the contents were dried, weighed and analyzed for N, phosphorus (P) and potassium (K); the rate of disappearance of nutrients from the pouches estimated the rate of plant-availability. Organic fertilizers used were 4-4-2 (chicken, meat and bone meal) and 12-0-0 (feather meal). Evaluation 1 was a placement evaluation of 4-4-2 (buried vs surface applied). In this trial, 4-4-2 that was buried in the soil released mineral N more quickly and released more N than surface applications, 48.2 and 70.2%, respectively (Table 2). Evaluation 2 was a comparison of 4-4-2 and 12-0-0, both buried in the soil at the beginning of the crop cycle of a broccolini field that was in the ground for 71 days. 12-0-0 released N more quickly and released more mineral N than 4-4-2, 86.2 and 60.5%, respectively.
These evaluations show a rapid release of N from the organic fertilizer over the first 2 to 3 weeks after application. After that time, the rate of release of N is very slow. Burying the material increased the rate of N release; fertilizer with a higher N concentration showed faster N release. It is possible that the pouch evaluations overestimate the N release rate because of movement of small particles of fertilizer that filter out of the pouch; this idea is supported by observations in a laboratory evaluation conducted at Davis that showed about a 20-30% lower rate of N release of 4-4-2 and 12-0-0 over the same period of time (Table 3).
The amount of N applied as fertilizer ranged from 1.2 to 4.8 times crop N uptake (Table 1). However, discounting the amount of N that actually mineralizes from the organic fertilizer, organic fertilizer applications in this survey ranged from 0.4 to 2.7 times crop N uptake.
Soil P values at each survey site were relatively modest for the Salinas Valley vegetable ground, except for one that was located on an old dairy. Bicarbonate extractable P values ranged from 10 to 57 ppm with a mean of 37 ppm (excluding the old dairy site with a bicarbonate P value of 112 ppm). The moderate P values occurred in spite of the common usage of 4-4-2 which has a ratio of 1:1 of N:P2O5. The form of P in 4-4-2 fertilizer comes mostly from bone meal, which is not soluble at soil pH greater than 7.0; all survey sites had pH above 7.0, meaning that bone meal is a highly inefficient P source in this production system. In fertilizer pouch Evaluation 1, phosphorus release was 17% after 63 days in both surface and buried applications. Based on this survey, it appears that soil bicarbonate extractable P levels do not rise rapidly due to the use of 4-4-2. By contrast 82 to 92% of the K contained in 4-4-2 was rapidly released to the soil after 63 days in Evaluation 1.
Summary
Organic fertilization of fast-maturing cool season vegetables is complicated by a number of factors:
- The difficulty of utilizing residual soil nitrate monitoring, given the potential for nitrate leaching with these heavily irrigated, shallowly rooted crops
- Uncertain rates of N release from organic fertilizers due to the variable effects of temperature, fertilizer placement and fertilizer N concentration
- Difficulty synchronizing N release from soil and organic fertilizers with the high crop N demand over the last half of the crop cycle
These studies provided some evidence that measurements of residual soil nitrate can provide a useful tool for planning fertilizer programs in some situations. However, even soils with robust amounts of residual soil nitrate-N (100 – 125 lbs N/A) may still benefit from fertilizer in spite of mineralization of N from soil organic matter and nitrate in the irrigation water, and clearly more study is needed to better understand this issue. The release of N by organic fertilizer is affected by placement, N concentration of the material and temperature and this study provide some measurements of those effects. The fate of N not released from organic fertilizer after the crop cycle is not well understood, but presumably it becomes available to future crops, mineralizing at a slow, steady rate similar to soil organic matter. The high usage of 4-4-2 which is a mix of chicken manure blended with meat and bone meal on larger-scale organic farms brings the benefits of adding significant amounts of organic matter to ranches that may not have extensive cover crop or compost programs. However, use of 2-3,000 lbs 4-4-2 per crop 2-3 times per season adds 2-4.5 tons of carbon-rich organic matter to the soil. Measurements of P release from 4-4-2 indicate that the 1:1 ratio of N:P2O5 does not appear to be resulting in high levels of bicarbonate extractable P buildup in the soils surveyed in this study.


- Author: Richard Smith
- Author: Michael D Cahn, Ph.D.
- Author: Timothy K Hartz
- Author: Patricia Love
Efficient management of nitrogen (N) in the cool season vegetable production areas of the Central Coast has a number of challenges. Many of the vegetable crops, such as leafy greens, are shallow rooted, fast maturing with a high N-demand, and produced in multiple rotations per year. The high N demand by these crops is often during the last half of the crop cycle. For instance, lettuce takes up about 70% of the total N requirement during the last 30 days of a 60-65 day crop cycle, and spinach takes up a similar percentage in the last 15 days of a 30 day crop cycle. These vegetables also have strict quality standards for size, weight and deep green color of the product which can result in greater use of N fertilizer. Crop residues incorporated after harvest can contribute significant amounts of N back into the soil, and increase the risk of nitrate leaching losses during the establishment irrigations of a subsequent crop, or during significant rain events. High land rents restrict the ability of producers to effectively rotate to cover crops that could take up excess residual soil nitrate from the soil.
In response to these challenges, growers are implementing practices such as monitoring residual soil nitrate levels and accounting for them in fertilizer decisions, and have become more careful in managing irrigations to minimize nitrate leaching losses. Less attention has been paid to the impact of rotations on nitrogen use efficiency (NUE). Deep rooted cover crops and vegetable crops grown in rotation with shallow rooted vegetables can sequester nitrate. Careful management of nitrogen inputs and use of rotational crops can provide a means of maintaining N in the rooting zone of the crops, reducing losses to groundwater and improving NUE.
Rotational crops function by capturing residual N in the soil and incorporating it into their crop tissue. By capturing N in the soil profile that might otherwise be lost by leaching beyond the rootzone they mitigate losses of N from prior rotations.
Background on Nitrogen Management
Nitrate (NO3-) is a negatively charged molecule and does not adhere to the negative charges on clay or organic matter. Rather, nitrate resides in the water held in soil pores and can be easily carried downward following an irrigation or rain event. This fact underscores the need for careful irrigation management to effectively manage N in vegetable production systems.
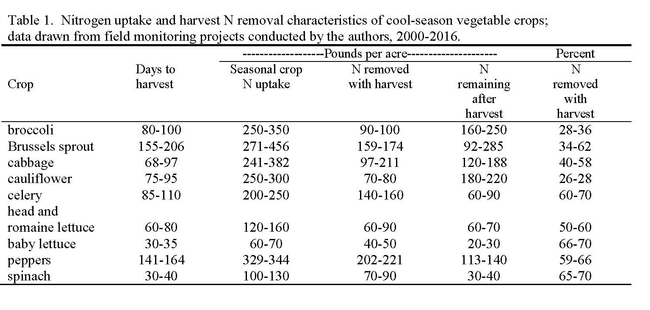
After a winter of normal to high rainfall, soil nitrate levels are typically low at the beginning of the crop cycle in the spring due to leaching during winter fallow. However, as the season progresses, soil nitrate levels build up from unused fertilizer, mineralization of soil organic matter (including prior crop residues) and nitrate from irrigation water. The amount of nitrogen in vegetable crop residues following harvest ranges from 20 to >250 lbs N/acre (Table 1). Nitrogen mineralization from crop residues is initially rapid, with faster mineralization occurring in residues with higher N content. For instance in a trial that we conducted, residue containing 5% N mineralized 63% of its N content in 4 weeks, compared to 43% for residue containing 3.8% N. Following the rapid breakdown phase, crop residues break down at a slower rate that can last over a period of months or longer.
Mineral nitrogen is also released from decomposition of soil organic matter and the quantity made available depends on the percent organic matter in the soil. Soil organic matter contains about 5-6% N. In the summer when soil temperatures are above 70 °F, the quantity of N mineralized typically ranges from 0.5 to 1.5 lbs N/acre/day. During the winter when soil temperatures range between 50 and 60 °F, mineralization from soil organic matter slows, but does not entirely stop.
Nitrate in irrigation water is another source of N. Many irrigation wells in the Salinas Valley contain nitrate-N in the range of 10-30 ppm NO3-N (representing 2.3-6.9 lb N/acre inch of water) and some wells may contain as much as 60-70 ppm NO3-N (13.8-16.1 lbs N/acre inch of water). Several years of field trials have demonstrated that the N in irrigation water contributes to crop growth similar to fertilizer N.
Growers are under increasing regulation to improve NUE. Strategies to improve NUE include moving away from program based fertilizer programs to field by field decision based strategies that utilize measurements of residual soil nitrate. Soil nitrate measurements can be made several ways, but the nitrate quick test provides a reliable method to quickly assess levels of residual soil nitrate in the soil prior to a fertilization event http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=4406. If testing indicates that sufficient residual soil nitrate is present, it is possible to reduce or skip a fertilizer application. In this way, it is possible to take full advantage of residual soil nitrate, reduce the quantity of fertilizer applied to the crop and maintain optimal yields (For a more detailed explanation go to: http://cemonterey.ucanr.edu/files/259776.pdf ).
In spite of best efforts at efficiently managing N in agricultural systems, inefficiencies occur, and some nitrate can leach beyond the crop root zone. Rotating with a crop that takes up and utilizes excess nitrate from the soil would help increase NUE. Winter cover crops can capture a significant portion of excess residual nitrogen during the winter fallow period. Studies in the Salinas Valley showed that cover crops reduced nitrate leaching by 65-75% (Wyland et al 1996; Heinrich et al 2014). Cover crops can accumulate 100-200 lbs N/acre and the residue begins to mineralize N upon incorporation into the soil in the spring. Unfortunately, the use of cover crops in the Salinas Valley is limited by their impact on planting schedules, and as a result, probably no more than 5% of the vegetable acreage is cover cropped each year.
N Scavenging by deep rooted vegetable crops
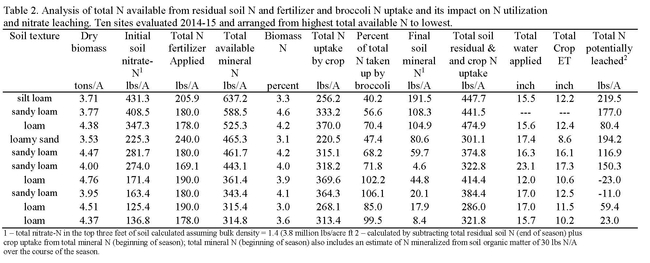
Ten trials were conducted from 2014-15 evaluating the impact of broccoli grown after a prior crop of lettuce on nitrate leaching. Quantities of residual soil nitrate in the top three feet of soil at the beginning of the broccoli crop cycle ranged from 134 to 431 lbs N/acre (Table 2). The quantity of fertilizer N applied to broccoli crop ranged from 169 to 240 lbs N/acre, while the crop took up 220 to 370 lbs N/acre in the above ground biomass. In 9 out of the 10 sites, broccoli took up more N than was applied as fertilizer, and effectively scavenged residual soil nitrate from the soil. By maturity, broccoli took up 40 to 106% of applied fertilizer N and initial residual soil nitrate-N in the top 3 feet of soil. In calculating the N budget for the crop cycle, we estimated that nitrate leaching losses ranged from 0 to 219 lbs N/acre.
The main indicator of whether broccoli could reduce the risk of nitrate leaching was the level of soil nitrate at the beginning of the crop cycle. When initial soil nitrate levels in the top three feet of soil were <200 lbs N/acre, broccoli was more effective at reducing the risk of nitrate leaching. This emphasizes the need to finish the prior lettuce crop with low to moderate amounts of residual soil nitrate. This can be achieved by utilizing the best management practices described above.
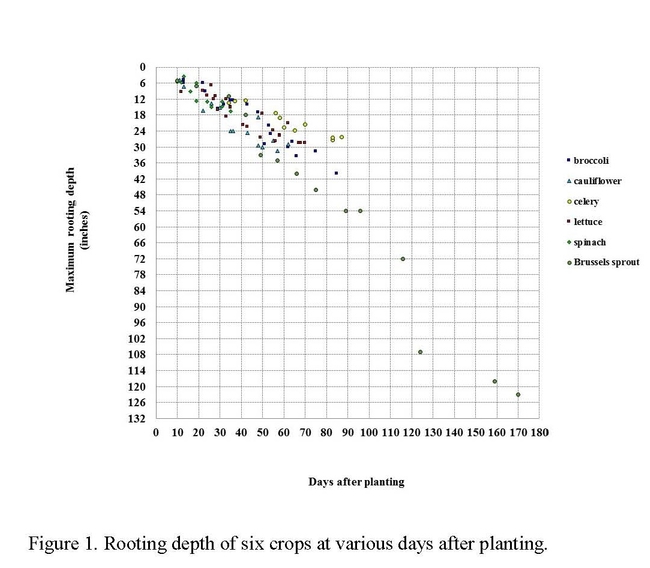
Broccoli roots extend down to 40 inches by the end of the crop cycle (Figure 1). Sufficient root density occurs in the second foot of soil by 50 days after planting such that the crop can effectively utilize nitrate at that depth in the soil profile. Broccoli often uses up most of the N in the top foot of soil after mid-cycle and relies on N in the second foot to reach maturity.
The above-ground biomass of dry broccoli contained from 3.0 to 4.6% N. The high concentration of N in broccoli biomass in conjunction with relatively high dry matter content (4.0 tons dry matter/acre) results in the large amount of N in the biomass.
Broccoli is not the only crop with the ability to scavenge residual soil nitrate; nitrate scavenging has also been observed in Brussels sprouts, cabbage, cauliflower, carrots, celery, peppers and processing tomatoes. What these crops have in common is a relatively long production season that allows roots to grow to a substantial depth, and modest seasonal N fertilization rates compared to their N uptake capacity. Faster, more shallowly-rooted crops (i.e. spinach and other baby greens) have limited scavenging ability, and in fact usually receive substantially more fertilizer N than they take up.
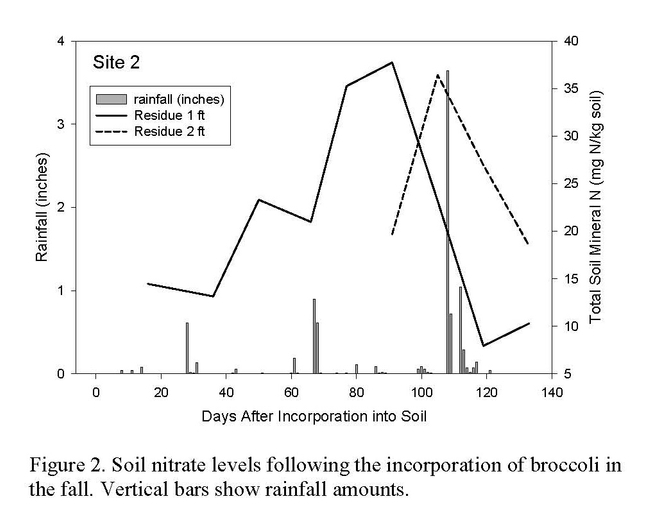
Planting deep rooted cash crops can serve as a best management practice in intensive vegetable cropping systems because their roots can intercept nitrate that would otherwise be lost to leaching, and put it to productive use for crop production. In order to be successful, the scavenged N contained in the crop must be removed from the site in the harvested product or successfully cycled to the next crop. Lower amounts of N leave the field in the harvested product of broccoli and cauliflower than other scavenging crops such as cabbage and peppers (Table 1). The amount of N in crop residue after harvest can be substantial in cole crops. If a cole crop is incorporated into the soil prior to a subsequent crop such as lettuce, the N mineralized from the crop residue can be taken up by the lettuce with careful crop management. If the broccoli crop is incorporated into the soil prior to the winter fallow, the N mineralized from the crop residue can be lost by leaching in winter rains (Figure 2).
In summary, these studies showed that broccoli and other deep rooted vegetable crops can take up more N than is routinely applied to them. Broccoli can take up nitrate deeper in the soil profile than other crops such as spinach and lettuce, which are more shallowly rooted. The scavenged N is put to productive use and kept from immediately loss through leaching. If used effectively, scavenging of N by deep rooted crops can improve NUE and delay and reduce N leaching in cool season vegetable production systems.
- Author: Shimat Villanassery Joseph
Join UC ANR Cooperative Extension for a special Heteroptera insect group Workshop in Monterey, CA. This half-day workshop will feature presentations on topics including insect resistant management, biological control, market regulation and more! After, join us for lunch at the Monterey Boat Harbor and guided tour of Dole Fruit Company and local grower fields.
When: April 20, 2017
Where: Embassy Suites Monterey Bay-Seaside, Seaside, California
3 Hours of DPR CE hours approved
Registration fee: $30
Pre-registration desired: http://ucanr.edu/survey/survey.cfm?surveynumber=20016
Agenda
|
8:00 |
Welcome |
|
8:10 |
Insect Resistance Management – Jesse Richardson, Dow AgroSciences |
|
8:45 |
Brown Marmorated Stinkbug – Tracy Leskey, USDA-ARS, Appalachian Fruit Research Station |
|
9:20 |
Dispersal and Migration of Insects in the Landscape – James Hagler, USDA-ARS, Arid-Land Agriculture Research Center |
|
9:55 |
BREAK |
|
10:40 |
Challenges to Biological Control of Heteroptera – Kim Hoelmer, USDA-ARS Beneficial Insects Introduction Research Unit |
|
11:15 |
Challenges to Pesticide Registration in a Global Market – Luis Gomez, Dow AgroSciences |
|
11:50 |
ADJOURN |
|
|
TOUR - Dole Fruit Company and Local Grower Fields, with lunch at the Monterey Boat Harbor |
|
12:10 |
Tour departs – from Hotel, returns at 5 PM |
For more information visit: http://ucanr.edu/sites/2017bugsymposium/ or contact: Shimat Joseph 831 229 8985
- Author: Richard Smith
- Author: Steven Fennimore
Spinach is susceptible to weed pressure because it is produced on high-density 80-inch wide beds with 18 to 42 seedlines. There is no opportunity to cultivate the bed top so all weed control is accomplished by managing weeds in prior rotations, cultural practices, chemical weed control or hand weeding. Clipped spinach is mechanically harvested and must be kept as weed free as possible to reduce hand weeding costs. In the recent UC publication, Sample costs to produce and harvest organic spinach (Tourte et al, 2015 http://coststudies.ucdavis.edu/current/ ) hand weeding costs averaged $440 per acre. However, weeding costs can easily exceed $1,000 per acre in weedy fields, and if cost get to high, growers may decide to disc the field. A key factor that affects weeding pressure in spinach fields is the prior crop. Long-season crops such as peppers, annual artichokes and leeks tend to allow more weed seed set and can increase weeding costs in a subsequent crop like spinach.
RoNeet and Dual Magnum are registered preemergent herbicides for use on spinach. RoNeet is the most commonly used preemergent herbicide on spinach; it is volatile and must be quickly incorporated into the soil for best efficacy. Post planting, preemergent applications are typically incorporated with sprinkler irrigation, but it can also be mechanically incorporated prior to planting. It has a 48 hour reentry interval following application. RoNeet controls problematic weeds such as malva, purslane, lambsquarters and pigweed.
Dual Magnum provides control of weeds such as burning nettle. One difficulty with the use of this material is that it has a 50 day preharvest interval (PHI). This PHI often necessitates making applications prior to planting clipped spinach which matures in 25-35 days during the summer production season.
Over the past three years we have evaluated Spin-Aid for use in clipped spinach. Spin-Aid is registered for use on processing spinach and has a 21 day preharvest interval (PHI); this use pattern fits the production practices for freezer spinach. However, washing and bagging spinach is also considered processing and would allow its use for clipped spinach. During the summer months, clipped spinach matures in 25-30 days which makes complying with the 21 day preharvest interval difficult. The first opportunity to apply Spin-Aid typically occurs following the germination water at about 8-10 days after planting which makes complying with the 21-day PHI difficult in many situations. Spin-Aid has various warnings on the label regarding phytotoxicity or potential delay of development under some conditions; this is a concern for a rapidly developing crop like clipped spinach. Research conducted a number of years ago indicated that phytotoxicity by phenmedipham was made worse under warm temperatures. As a result, the label stipulates that temperatures >75 °F can cause crop injury. Research carried out by Ran Lati indicated that high light conditions reduced the tolerance of phenmedipham more than temperature in the coastal production district. Given this observation we evaluated the impact of nighttime vs daytime applications of low rates of Spin-Aid (0.5, 1.0 and 2.0 pints/A) for use on clipped spinach.
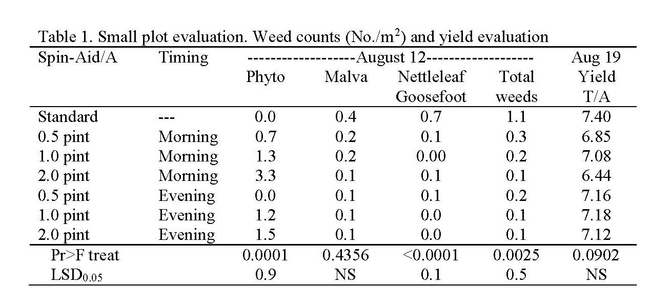
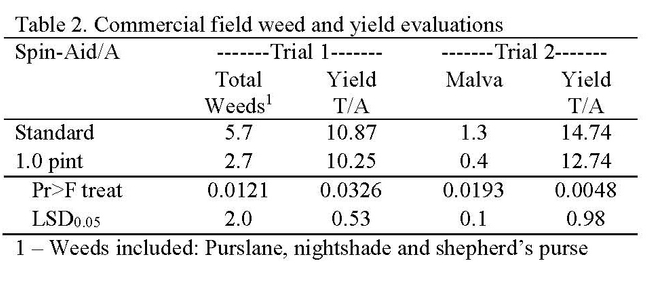
Research consisted of making morning (high light) and evening (low light) applications of Spin-Aid nine days after seeding when the 1st true leaves were <1/4 inch in size. We observed less phytotoxicity from evening than morning applications (Table 1). Damage to spinach was most significant in morning applications of 2 pint/A Spin-Aid and consisted of necrosis on the tips of some cotyledons (Photo 1); these symptoms were not seen in the 2.0 pint/A evening applications (Photo 2). The 0.5 and 1.0 pint/A rates had acceptable levels of phytotoxicity regardless of application timing. Spin-Aid did not significantly reduce the number of malva plants but stunted them (Photo 3) making them less problematic during harvest as is the case of untreated malva (Photo 4). Nettleleaf goosefoot was susceptible to Spin-Aid and all rates significantly reduced the population of this weed. Commercial-scale trials were carried out in fields that had high populations of weeds. At one site Spin-Aid reduced the number of purslane, shepherd's purse and hairy nightshade by half (Table 2). The overall yield of the standard treatment was 11.007 tons/A and the 1.0 pint/A Spin-Aid treatment was 10.061 tons/A indicating that there may be some yield reduction with the use of Spin-Aid.
The results from these and other trials look promising, but modifications to the current label will be necessary and we are currently working with the manufacturer, Engage Agro, to see if that will be possible. If successful, this will provide a powerful tool to reduce weed pressure in fresh market spinach fields.