- Author: Richard Smith
- Author: Timothy K Hartz
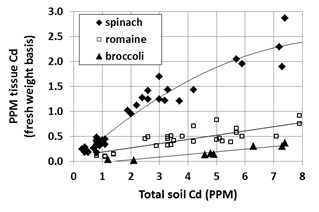
For several years the leafy greens industry has been engaged with regulatory agencies regarding the issue of cadmium (Cd) concentration in fresh produce; Cd, a heavy metal element, poses human health risks if ingested in significant quantity. The 2016 production season marks the first time that California spinach growers may be required to alter their production practices to minimize the amount of Cd taken up by this crop. Spinach is the focus of concern because it takes up much more Cd than other common vegetable crops (Fig. 1).
The graph makes clear that soil Cd content is a major factor determining crop Cd uptake. We have a general understanding about where the highest Cd soils are located, but flooding events over centuries have moved and mixed soil sediments. Therefore, soil testing is a critical step in evaluating the field-specific risk. The soil test procedure for which we have the most data is total soil Cd measured by nitric acid/hydrogen peroxide extraction. Soil samples should be taken from the top foot, which is the effective rooting depth of spinach.
In fig. 1 it is clear that individual fields differ substantially from the general trend line relating total soil Cd to tissue Cd because other factors also have influence. Those factors include:
- Variety – preliminary field evaluations suggests that tissue Cd concentration may differ among varieties by up to 25%.
- Soil pH – Cd bioavailability declines with increasing soil pH.
- Irrigation water chloride (Cl) concentration – increasing Cl concentration makes Cd more bioavailable.
- Soil zinc (Zn) level – Zn and Cd compete for plant uptake, so soil with higher Zn availability tend to have less Cd uptake.
Individual spinach shippers may impose soil Cd limits on their growers, or require remediation practices to be deployed if fields above a certain soil Cd level are used; given the uncertainties in the relationship between spinach Cd concentration to total soil Cd, different shippers may set different rules for their growers to follow.
Where growers are required to use remediation practices to reduce spinach Cd uptake, our research suggests that high-rate Zn application is the most practical approach. Zn and Cd are closely related ions, and plants are not able to distinguish well between these ions. Increasing the ratio of plant-available soil Zn to Cd suppresses Cd uptake. However, to significantly decrease Cd uptake much higher levels of Zn application are needed than would typically be used to remedy a soil Zn deficiency. Across numerous field and pot trials we have observed that applying 25-50 PPM elemental Zn on a soil dry weight basis (equivalent to approximately 100-200 lb elemental Zn per acre foot of soil) suppressed crop Cd uptake by roughly 30-60%. However, the effectiveness of Zn application was affected by the following factors:
Zinc form:
- Zinc sulfate and zinc chelate were more effective in reducing Cd uptake than zinc oxide. Zinc sulfate was more economical than zinc chelate.
- Granular forms of zinc sulfate were less effective in reducing Cd uptake than powdered forms, or zinc solutions. We presume that Zn from the granules did not disperse thoroughly through the soil.
Zinc incorporation:
- To be maximally effective, Zn must be distributed throughout the primary rooting zone of spinach (the top 8-12 inches of soil). Zn distributed only in the top few inches of soil was only marginally effective.
- Disking incorporates Zn to a depth of approximately 6 inches and is therefore more effective than mulching, which incorporates Zn only to about 3 inches. Where practical, incorporation of Zn even deeper than 6 inches would be ideal.
Zinc foliar applications:
- Foliar applications of zinc sulfate or zinc chelate were not effective at reducing Cd uptake in spinach.
Zinc longevity:
- When Zn is applied to soil it slowly becomes less plant-available over time, as chemical compounds of low solubility are formed. However, high-rate Zn application will have measurable effects over several years at least. In a field trial, in the first crop following application of 100 pounds of elemental Zn (280 lbs of zinc sulfate) per acre we observed a 40% reduction in Cd uptake by spinach; after two years the Zn-treated area still showed a 15% reduction in crop Cd uptake. In pot trials, when 80 PPM elemental Zn was added to high-Cd soils, the reduction in Cd uptake of four successive spinach crops was 66%, 66%, 51% and 49%. Based on these observations, it is clear that Zn applications can be effective across years, but additional Zn application may be needed to maintain maximum efficacy over the long term.
Other potential remediation practices include raising soil pH, or applying organic amendments. These practices are aimed at making soil Cd less plant-available. Increasing pH does this by precipitating Cd salts, while organic amendments like compost may tie up Cd on its complex cation exchange sites. Our preliminary data suggest that both practices can have a positive effect, but the effects are less substantial than those we observed with high-rate Zn application.
In summary, regulatory pressure is forcing the spinach industry to adopt practices to limit spinach Cd concentration before we have developed a comprehensive understanding of how best to do that. Based on our current data we believe that fields with total soil Cd below 1 PPM present relatively low risk, while those above 2 PPM will be more difficult to remediate reliably. Remediation practices can be effective in fields between 1-2 PPM total soil Cd, but results in individual fields will vary, based on the complicating factors we have outlined.
- Author: Shimat Villanassery Joseph
Recently (mid April), a large number of grasshoppers has been found attacking broccoli and spinach crops in the southern parts of Salinas Valley (San Lucas, San Ardo etc). It is believed that these grasshopper populations were migrating from the dry grassland to leafy greens and Brassica crops. The feeding damage was typically found along the edge of the fields adjacent to the hills. Also, blister beetles (Epicauta spp.) were seen along with grasshoppers. Blister beetles were mostly feeding on the weed plants surrounding the fields. The feeding damage may not be critical but these insects may contaminate the harvested produce with dead or alive bodies and their excrement. The blister beetle adult is about an inch long, and blackish blue in color. Blister beetle has a distinct look that make them easy to identify from other beetles. The width of the neck is shorter than width of the head. The outer pair of fore wing is flimsy and not held tight on the abdomen.
Ecologically, both grasshopper and blister beetle interact with each other. The larval stages of blister beetle are predaceous and often prey on the eggs of grasshopper. Grasshopper lays eggs in a pod (120 eggs maximum) below the soil. The eggs hatch to first instar nymphs which molt through six nymphal stages before develop into adults. The nymphal stages are wingless but the late nymphal stages have wing pads (incomplete wings). Nymphal stages move only by jumping whereas adults can fly and jump. Most of the grasshoppers found in the fields are nymphs and they are found in aggregation feeding on the broccoli plants. It seems like they like broccoli than spinach. Grasshoppers are migratory in nature meaning they feed on what they find on their way and as the food resource depletes, they move on to another green patch.
Because of the greater size and high mobility, it is relatively difficult to kill these grasshoppers using insecticides. In the Salinas Valley, the wee and morning hours are cooler and grasshoppers are less active and remain in the ground waiting for the day to warms up. These early day time might be the best time for insecticide application but still, they might move away when the sprayer equipment approach them. Pyrethroid insecticides and acephate are effective but multiple applications might be warranted because grasshoppers are continuously moving in from the nearby dry grassland. Please check the label instructions before selecting the insecticide for grasshopper control. Management of grasshopper using organic insecticides will be a greater challenge than synthetic insecticides. Organically approved insecticides such as pyrethrum (Pyganic) might be the option. Because grasshoppers feed on plant material, spinosad (Entrust) might be effective too. Azadirachtin (Neem products) may provide some repellency. However, success is not guaranteed with any of these insecticides. There is a protozoan, Nosema locustae -- commercially available as a bait under the brand name Semaspore. The bran bait is mixed with spores and the spores enter their body while feeding. This bait is slow acting and success may vary. Physical barrier such as fencing between field and grassland might help but a spray on the barrier might check them from progressively moving into the field. Smaller production can use row covers to prevent their access to the crops. Managing grasshoppers in the grassland with approved insecticides might slow down the migration.



- Author: Jim Correll
- Author: Steven T. Koike
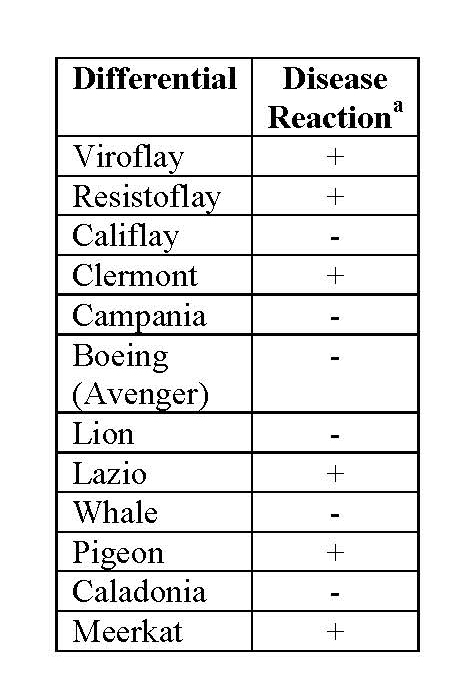
A new race of the downy mildew pathogen (Peronospora farinosa f. sp. spinaciae = P. effusa) on spinach was first identified in March of 2015 in Salinas, CA, U.S. This race was able to overcome most of the race 1-15 varieties evaluated. The isolate, originally designated UA201519B, was characterized based on disease development on a standard set of differential varieties. Subsequently, isolates with the same reaction pattern on the differential set have been found in numerous locations. After careful evaluation of the significance of this development to the spinach industry, the International Working Group on Peronospora (IWGP) has denominated isolate UA201519B race Pfs: 16.
Race Pfs: 16 is able to infect the differentials Viroflay, Resistoflay, Clermont, Lazio, Pigeon, and Meerkat, but not able to infect Califlay, Campania, Boeing (Avenger), Lion, Whale, and Caladonia.
The IWGP is continuously monitoring the appearance of strains of the pathogen that deviate in virulence from the known races. In this way the IWGP aims to promote a consistent and clear communication between public and private entities, such as the seed industry, growers, scientists, and other interested parties about all resistance-breaking races that are persistent enough to survive over several years, occur in a wide area, and cause a significant economic impact.
The IWGP is located in The Netherlands and is administered by Plantum NL. The IWGP consists of spinach seed company representatives (Pop Vriend, Monsanto, RijkZwaan, Bayer, Takii, Sakata, Bejo, Enza, Syngenta, and Advanseed) and Naktuinbouw, and is supported by research centers at the University of Arkansas and the University of California Cooperative Extension (Monterey County) in the U.S. Researchers all over the world are invited to join the IWGP initiative and use the common host differential set to identify new isolates.
For more information on this subject you can contact Jim Correll (jcorrell@uark.edu), Steve Koike (stkoike@ucdavis.edu), Diederik Smilde (d.smilde@naktuinbouw.nl), or the IWGP chairperson Jan de Visser (JandeVisser@popvriendseeds.nl).
Disease reactions of race 16 (UA201519B) observed on spinach differentials by the IWGP compiled February 2016.
aDisease reactions observed in controlled inoculation tests
by the participants of the IWGP. “+” indicates
susceptible; “-“ indicates resistant disease reaction.
- Author: Michael D Cahn, Ph.D.
CropManage 2.0 Workshop: Hands-on training
Monterey County Agricultural Center Conference Room
1432 Abbott St, Salinas CA 93901
Thursday, March 31 2016
(8:30 am – 12 pm)
We will offer a hands-on training to learn in depth about the features of CropManage 2.0, a free online decision support tool for water and nutrient management of coastal crops. CropManage was recently revised with a new user interface and additional capabilities, and now supports iceberg and romaine lettuce, spinach, celery, broccoli, cauliflower, cabbage, and strawberries.
Considering that the drought may continue into a 5th year, and water quality regulations continue to be linked to nutrient management, efficiently using water and nitrogen fertilizer is a high priority for Central Coast growers. CropManage can be a useful tool for providing quick decision support on water and nutrient management on a field-by-field basis. This training will provide an opportunity to learn how to use CropManage for improving the efficiency of your farming operations or for adding value to your consulting services.
The workshop will take you step-by-step on how to navigate and use CropManage for assisting with fertilizer and water management decisions and record keeping. Wi-Fi internet access is available at our conference room so please bring a laptop or tablet computer so that you can follow along as we tour through the features of the software. There should be sufficient time to answer questions as we cover the following topics:
Agenda:
8:30 – 9:00 Registration and Refreshments
9:00 – 9:30 Introduction and update on CropManage 2.0
9:30-10:15 Getting started with CropManage
10:15 (Break)
10:30 – 11:15 Strategies for using CropManage for decision support and record keeping
11:15- 11:45 Advanced features and interfacing sensors with CropManage
11:45-12:00 Discussion of new features or changes needed
Certified Crop Adviser CEU hours (1 hr soil and water management, 1 hr nutrient management, 0.5 hrs professional development)
To keep the group size manageable so that we can provide individual help, we would like to limit the workshop to 30 participants. If you have attended previous workshops and or feel proficient in using the on-line tool, then you are welcome to just attend the second half of the workshop (10:30-12 pm). Whether or not you plan to attend the entire or part of the workshop, please RSVP in advance by sending an email to larriaga@ucanr.edu with the subject heading “CropManage workshop” and let us know the number of participants in your group. We will email you a confirmation. Alternatively, you can contact our office at 831-759-7370. Thank you, and I hope to see you soon.
Michael Cahn
Please feel free to contact us if you need special accommodations
- Author: Richard Smith
- Author: Michael D Cahn, Ph.D.
- Author: Timothy K Hartz
- Author: Patricia Love
- View More...
Summary: The ability of broccoli to scavenge N from depths below 2 feet was evaluated in 2014 and 2015. Broccoli roots extend down to three feet in the soil by the end of the crop cycle. Broccoli is capable of scavenging nitrogen from the 2nd and 3rd foot in the soil, but adequate nitrogen is required to help develop the crop at an early stage for broccoli to achieve maximum yield. Most scavenging of nitrate deeper in the soil profile occurs in the final third of the crop cycle. In a survey of 10 broccoli fields following a prior lettuce crop broccoli took up >80% of the residual + fertilizer N in 4 of these fields. If residual soil nitrate levels are not too high, broccoli can take up a high percent of the N mineralized from the residue of the previous lettuce crop and reduce levels of soil nitrate that is at risk of leaching. Broccoli residue also mineralizes rapidly and releases large quantities of nitrate that could provide for the N needs of a subsequent crop.
Background:Summer-grown broccoli and other cole crops such as cauliflower and cabbage have been shown to routinely take up more nitrogen than is typically applied as fertilizer. This indicates that these crops scavenge excess nitrogen from the soil profile. They are deep rooted and utilize soil nitrate in the second and even the third foot of soil. The phenomenon of scavenging is not unique to the cole crops, as we have observed scavenging in some fields of peppers, celery and lettuce. The question is, however: Where does the residual soil nitrate that these crops scavenge come from?
Residual levels of soil nitrates comes from several sources: mineralization of soil organic matter and prior crop residues, unused N from fertilizers applied to prior crops and nitrate in irrigation water. In warm summer soils, nitrate is the main form of mineral N in the soil and typically ranges from 5 to 30 ppm nitrate-N. Soil ammonium-N levels in the soil in the summer are much less and typically range from 1 to 3 ppm. The nitrate molecule has a negative charge and is not adsorbed onto soil colloids, but rather resides in moisture held in soil pores. As such, nitrate is at risk to be transported below the rooting zone of crops with excess water from irrigation or rainfall. It is therefore best to measure soil nitrate immediately prior to making fertilizer decisions. For this reason, the nitrate quick test is the most common method of measuring soil nitrates in our cool season vegetable area because it allows growers to get the results of soil nitrate levels quickly in order to make fertilizer application decisions.
Residual soil nitrate levels must be high enough to effectively supply a fast growing cool season vegetable. A sufficiency threshold of 20 ppm nitrate-N was first proposed for cool season vegetable crops by Tim Hartz, which if measured in the first foot of soil, is equivalent to 76 lbs N/acre. This quantity of nitrogen is sufficient to supply the immediate needs of the vegetable crop for two to three weeks.
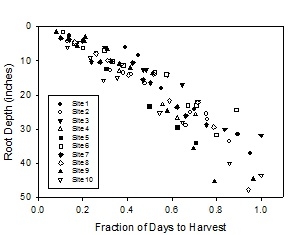
Nitrogen Scavenging Studies: Uptake of N from various depths in the profile. In 2014-15 we evaluated fertilization strategies of broccoli and its ability to scavenge residual soil nitrate from the soil following a lettuce crop. Broccoli roots were found to reach three feet deep in the soil by the end of the cropping cycle (Figure 1). To evaluate the ability of broccoli to utilize nitrogen from deeper in the soil profile, we conducted a trial at the USDA Spence Research Station in which we applied nitrogen using drip tape placed on the surface, and at depths of 12, 18 and 24 inches using equipment for injecting drip tape below the soil surface. The soil profile was low in residual soil nitrate at the beginning of the trial and the same quantity of N (150 lbs N/A) was applied to all the treatments. Fertilizer treatments were applied 27, 38 and 48 days after the first germination water.
The yield was lower in the treatments where the fertilizer was applied at 12 & 18 inch deep in the soil and was particularly low where it was applied 24 inches deep (data not shown). Because it takes time for broccoli roots to grow down through the soil profile, it appears from these results that the early growth suffered from lack of nitrogen, since the root system was not adequately developed to take up the fertilizer from deeper in the soil profile. These results suggest that broccoli needs an adequate supply of N in the upper foot during the early stages of development, even when an ample N may be available deeper in the soil.
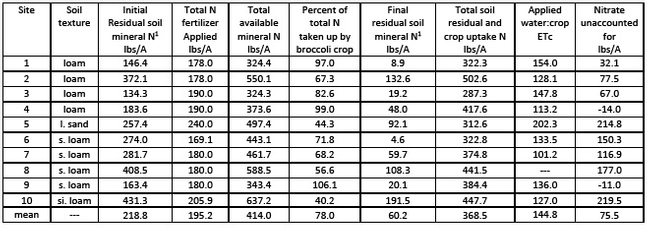
Uptake of residual soil N by broccoli: We also studied the ability of broccoli to take up residual soil nitrate left over from the prior lettuce crop. We surveyed a total of 10 fields over two years, evaluating the amount of residual soil nitrate at the beginning of the broccoli cropping cycle. Nitrogen was measured in the top three feet to account for the total amount of nitrogen accessible to the crop. The levels of soil nitrate in the top three feet of soil at the beginning of the broccoli cropping cycle ranged from 134 to 431 lbs N/A (Table 1). This wide variation in the range may have to do with fertilizer practices in the prior lettuce crop and N mineralized from crop residues. Fertilizer applications to the broccoli crops ranged from 169 to 240 lbs N/A, and the ratio of applied fertilizer N to crop N uptake ranged from 0.48 to 1.09.
The percent of initial mineral N taken up by the broccoli crop ranged from 40 to 106%, with four out of the ten sites having N uptake by the broccoli crop of greater than 82% uptake of initial residual soil nitrate + fertilizer N. The amount of residual soil nitrate left in the top three feet following the broccoli crop ranged from 9 to 192 lbs N/A. The results from this sampling of broccoli fields show that, under the right conditions, broccoli can take up a large percentage of the residual soil nitrogen left by a previous lettuce crop. However, if residual soil nitrate levels are too high, broccoli will not be able to absorb a large percent of the initial load.
In Table 1, the last column shows the levels of nitrate unaccounted-for. This estimate probably reflects leached nitrate. In all sites with unaccounted nitrate levels of >100 lbs N/A, the total amount of mineral N available for the crop (initial residual nitrate + fertilizer) was >440 lbs N/A. The ratio of crop water applied to crop ETc ranged from 1.0 to 2.1, but sites with the highest potential leaching did not necessarily correlate with the more efficient irrigation ratios.
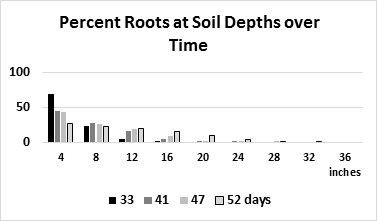
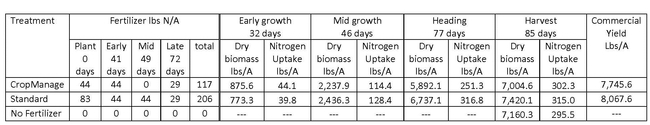
On-farm evaluation of N uptake from 2nd foot of soil: We conducted an on-farm evaluation to see if 2nd-foot soil samples could be used to guide fertilizer application decisions. This trial was conducted to refine the algorithms used in CropManage (CM) to guide water and fertilizer decisions for broccoli. Table 2 shows the quantities of fertilizer applied to the CM and standard treatments. The quantities of fertilizer at each application date were based on soil nitrate quick tests. In total, 56% less fertilizer was applied to the CM treatment. However, the CM treatment also had 4% lower commercial yield than the standard treatment. At the first fertilization, we reduced the fertilizer application in the CM treatment by 44 lbs N/A because of adequate levels of residual soil nitrate in the first foot of soil; subsequent to this application, the soil nitrate levels between CM and the standard treatment did not differ, and we do not think that reducing the N application in this fertilizer event affected the yield of broccoli. On the next application date, at 49 days after planting, 44 lbs N/A were applied in the standard treatment, but no N was applied to the CM treatment because the soil nitrate levels in the 2nd foot were at 25 ppm nitrate-N. We suspect that the observed yield reduction in the CM treatment may have been because the level of nitrate in the upper foot was too low and the root system was not yet sufficiently developed to take up the N from the 2nd foot. In 2015, we observed that 28% of the roots were in the 2nd foot of soil at 52 days after planting (Figure 2), and this level of root activity may not be adequate to utilize deep soil nitrate to support rapid top growth of the crop.
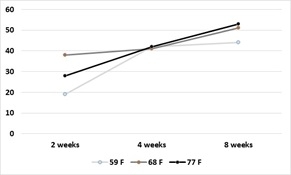
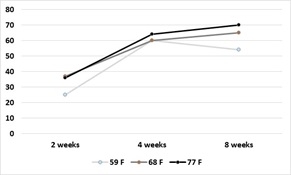
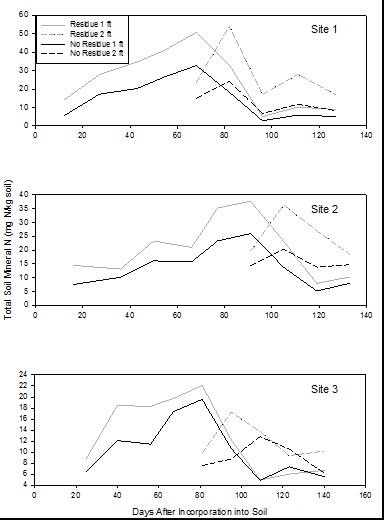
Broccoli residue as a source of N: At harvest, 25-30% of broccoli biomass leaves the field as the harvested product. Given that summer-grown broccoli takes up >330 lbs N/acre, a substantial quantity of nitrogen remains in the field after harvest. For broccoli residue with high N content (5% N), more than 60% of the N is mineralized 4 weeks after incorporation into the soil. Broccoli residues with lower N content (3.75% N) will release about 40% of the N by 4 weeks after incorporation into the soil. (Figures 3a and 3b). Figure 4 shows the results of N mineralization of broccoli residues in the field. Soil nitrate levels were evaluated in plots with and without broccoli residue. Soil nitrate-N levels peaked between 22 to 50 ppm. After the peaks, the levels declined due to rain events that leached the nitrate deeper in the soil profile (Figure 5). Soil nitrate concentrations peaked in the 2nd foot of soil after the decline in concentration in the 1st foot. Later, nitrate was leached from the 2nd foot to deeper in the soil profile. The levels of nitrate mineralized from broccoli residues are substantial and can potentially substitute for a substantial amount of the fertilizer N of a subsequent crop if it is not leached due to a major rain or irrigation event.
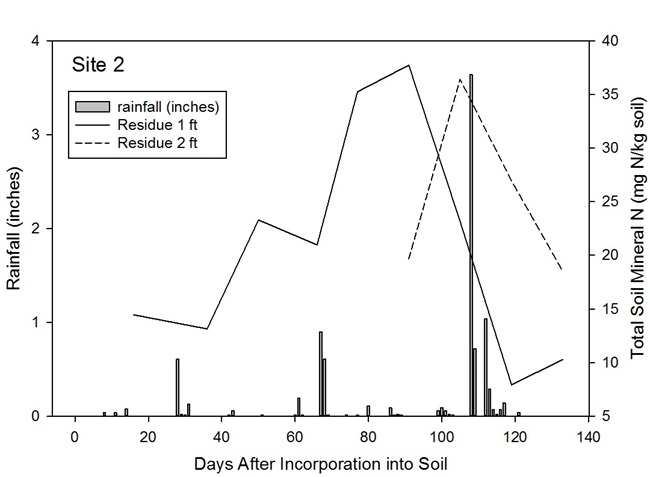
Table 1. Analysis of total N available from residual soil N and fertilizer, broccoli N uptake and its impact on N utilization and nitrate leaching
Table 2. Applied fertilizer and biomass of CropManage vs standard and unfertilized treatments.