- Author: Shimat Villanassery Joseph
Growers in the Salinas Valley facing an irregular lettuce stand are usually uncertain about what caused the problem and often blame the factors such as poor seed quality, planting error, irregular irrigation timing or distribution, high salt levels in the soil or water, soilborne pathogens of seedlings, bulb mites, and garden symphylan feeding for the losses. Several springtails were collected from the soil associated with lettuce and it is not clear if they were feeding and contributing to the irregular lettuce stand. Often, this springtail is misidentified as garden symphylan.
Recently, I found a large number of a subterranean springtail (Protaphorura fimata) (Fig. 1) in the monitoring potato slice traps deployed in Salinas lettuce fields. I did a series of laboratory and field studies to determine its pest status in lettuce. This springtail (P. fimata) is less than 2.5 mm long, white in color and lacks eyes. Unlike other springtails, this springtail lack a furcula (jumping organ), and when disturbed it does not jump instead curls up. Other similar species of springtails primarily reproduce parthenogenically meaning they reproduce without mating; however, sexual reproduction is also seen on this one (P. fimata). This species (P. fimata) seems to be widely distributed in Europe, but has not been previously reported from the U.S.
Springtails occur in diverse habitats worldwide and are generally considered as beneficial arthropods because they aid in the decomposition of decaying plant material by feeding, thereby contributing to the cycling of carbon and nitrogen which in turn improves soil health and structure. This springtailis primarily known to feed on soil fungi but also feeds on live plant roots. Other springtails in the same family have been associated with feeding damage to germinating sugar beet seeds, sugarcane, poppy seeds and weed seeds (Plantago major). Foliage-feeding springtails (lucerne flea and garden springtail) attack several plants including Lucerne (Medicago sativa), clover (Trifolium sp.), sugar beet (Beta vulgaris) and bean.
The major objectives of the present study were to document the ability of springtail (P. fimata) to injure germinating seeds of lettuce in laboratory and field and characterize the feeding injury of springtail on germinating seeds and seedlings of lettuce.
In the laboratory, I conducted experiments in plastic petri dish with and without soil and sprigtails. Then I recorded the ungerminated lettuce seeds due to feeding injury, total number of feeding injury sites, and number of germinated seedlings with distinct feeding injury. I also documented the location (e.g. leaf, stem, plant crown or root) of the feeding injury on the plants. I conducted a field study too. The assumption of the field study was that the repeated use of maximum label rate of selected insecticides at early stages of plant development would suppress springtails and protect the seeds or seedlings from feeding. Two commonly used pyrethroid insecticides were applied (by the grower) three times: 2 days before planting, at planting, and 20 days after planting. Applications were made using a commercial tractor mounted sprayer. Two pyrethroid insecticides used were Mustang (4 fl oz per acre) and Warrior II (1.6 fl oz per acre). Both the Mustang and Warrior II were tank mixed and applied at 2 days before planting and 20 days after planting but only Warrior II was applied at planting to conform to the label. An adjuvant, Widespread Max (2 fl oz per acre) was added with all the applications. I used bait slices to monitor the springtails at weekly intervals. They were placed in the soil at 1.5 inches deep along the seed line and were covered with disposable white plastic bowls. At the end of each 2 days exposure period, beet root slices were removed, placed into plastic bags and transported to the laboratory. In addition, plant samples were collected to assess the plant growth. Please read the full Journal article for details.
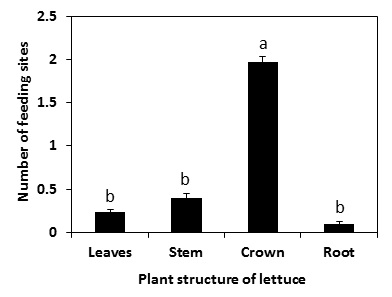
Results demonstrate that this springtail (P. fimata) can feed on germinating lettuce seeds or young seedlings, resulting in reduction in lettuce growth (Figure 2).Springtails attacked seeds and young seedlings alike. In the laboratory, springtails directly fed through the seed coat (pericarp) of a few seeds. This is possibly due to the moistening of the pericarp, enabling springtails to feed through the softened coating (Fig. 3a and b). In some instances, springtails fed on the growing radicle of the germinating seeds (Fig. 3c). However, most of the feeding at the seed radicle or elsewhere did not entirely sever it (Fig. 3d), which allowed the seedling to survive but affected the normal development of the plant. Moreover, most of the feeding injury was evident at the crown area rather than on leaf, stem, or root (Fig. 4).
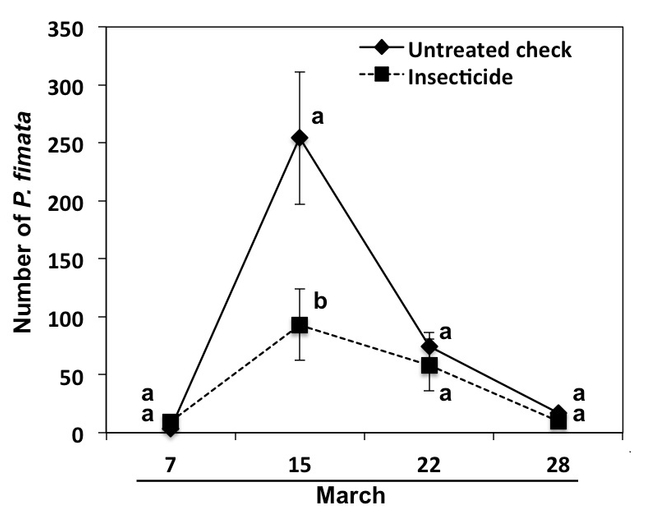
In the Salinas Valley, before the lettuce seeds are planted, fields are watered deeply and irrigations continue for at least three weeks after planting. I observed that the springtaildensity increased when the field was recently irrigated or after a rain event. This cultural practice which maintains high moisture levels for seed germination on the sub-surface profiles of the soil might be favoring faster buildup of springtailpopulations. In the field trial, the springtailcaptures were greater immediately after irrigation in the untreated beds than insecticide treated beds which was reflected in reduced number of springtails and in the untreated beds (Fig. 5).
In conclusion, this study clearly demonstrates that springtail (P. fimata) is an important pest of lettuce and is capable of reducing the crop stand. Incidence of high populations of springtail could be detrimental to germination of seeds in the field (Fig. 6 and 7). Springtails could be effectively suppressed to a large extent with early applications of insecticides directed to the seed line. Monitoring is the key to determine the presence and population size of springtail. Currently, I'm conducting for laboratory and field studies to determine the efficacy and application timing of insecticides.
Again, please use the link below to read the full article.
http://cemonterey.ucanr.edu/files/206762.pdf


- Author: Richard Smith
- Author: Elizabeth Mosqueda
- Author: Anil Shrestha
- Author: Fabian Galvan
- View More...
Summary:The use of automated thinners in the Salinas Valley has increased dramatically over the past two years. There are now four companies that manufacture automated thinners that use a spray system that removes the unwanted lettuce plants as well as weeds. This technology arrived just as the Salinas Valley was experiencing a labor shortage; many growers have adopted this technology or are in the process of seriously examining its usefulness to their operations. An evaluation of automated thinners was conducted in seven commercial lettuce fields in the Salinas Valley in the summer of 2014. The comparison was made with standard hand weeding. Overall, automated thinners were faster and more precise than hand thinning. However, the automated thinner left 7 times more doubles/A than hand thinning and it took more hand labor to remove the doubles in the double removal/weeding operation that occurs about 10 days following thinning; in spite of this issue, the total time to thin and remove doubles/weed in the automated thinner treatment took about 4.3 hours/A less time than hand thinning. A modest increase in yield was observed in the six fields evaluated, but the variability from field to field was quite high.
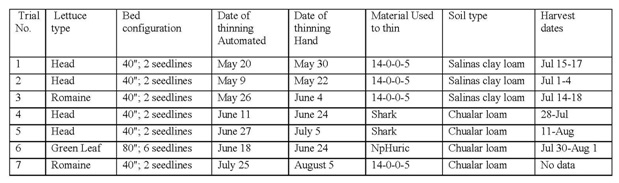
Methods:An evaluation of automated thinners was conducted during the summer of 2014. Trials were conducted with cooperating growers on seven commercial lettuce fields in the Salinas Valley, CA. The bed configuration and lettuce type are shown in Table 1. Each field was split in half with one side thinned by hand (standard practice) and the other side thinned with an automated thinner. The automated thinners used in these studies were: 1) Foothill Packing, 2) Blue River and 3) Agmechtronix. Thinning time was evaluated by recording the time it took to thin a designated area; this information was converted to hours per acre. In each field, four to six replicate areas two 40-inch beds wide by 90 feet were established in each treatment; these areas were used to make stand and weed count evaluations prior to and following the thinning operation. Following the thinning operation, the numbers of doubles (two closely spaced lettuce plants; see photo 1) were counted, and the distance between plants was measured to determine mean plant spacing and spacing distribution. Approximately 7-14 days following the thinning operation, a hand crew passed through the field to remove doubles and weeds; the time to conduct this double/weeding operation was measured and converted to hours per acre. Commercial and small plot yield measurements were made in six of the seven fields evaluated. Immediately prior to commercial harvest, 24 head from each evaluation area were cut and weighed to determine mean head weight. Plants were subsampled and sent to the UC Davis Analytical Laboratory for total N analysis to determine if the fertilizer used to thin the lettuce in some fields had an effect on the nitrogen nutrition of the crop. In addition, the numbers of heads infected with lettuce head drop (Sclerotinia minor) were counted. Commercial yield was measured in each treatment by obtaining box counts and sizes from the harvest crew. All commercial yields were converted to boxes per acre. The number of unharvested heads was counted in the evaluation areas following harvest. A side trial was conducted to evaluate the impact of size unthinned plants on the final plant size. In three of the fields, 20 heads of small and large plants were marked and plant diameter was measured three-four times during the crop cycle; final plant weight of these plants was measured at harvest.
 Photo 1. Chemically thinned lettuce |
 Photo 2. Double plants left by automated thinner |
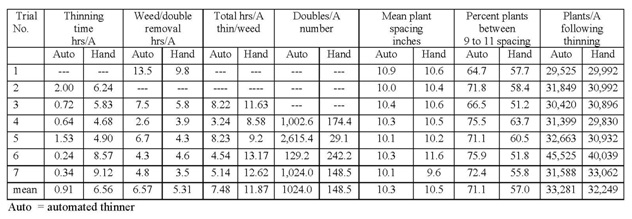
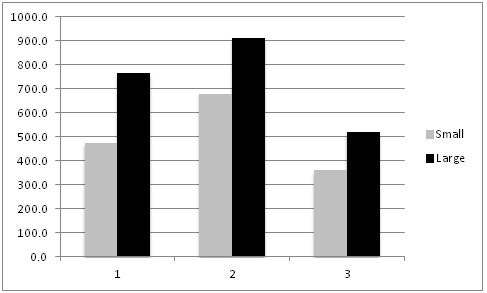
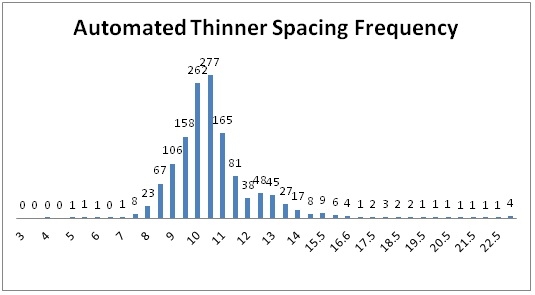
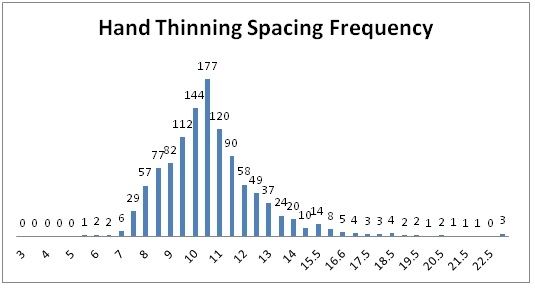
Results: On average automated thinners took 0.91 hours/A (range 0.24 to 2.0 hours/A) and hand thinning took 6.56 hours/A (range 4.68 to 9.12 hours/A) (Table 2). However, the double removal/weeding operation 7-14 days following thinning took 6.57 hours/A in the automated thinner treatment and 5.31 hours/A in the hand thinned treatment; the increase in time for double removal and weeding in the automated thinner area was due to the greater number of doubles in the automated thinner treatment (1,024 double/A) than hand thinning (149 doubles/A). The total time for thinning and double removal/weeding operations was 7.48 hours/A in the automated thinner treatment and 11.87 hours/A in the hand thinned treatment. The desired spacing for all fields in the study was 10.0 inches. The mean plant spacing in the automated thinner treatment was 10.3 inches (range 10.0 to 10.9 inches) and 10.5 inches in the hand thinned treatment (range 9.6 to 11.6 inches). The percent of plants that were between 9 and 11 inch spacing in the automated thinner and hand thinned treatments was 71.1 and 57.0, respectively. Figures 1 and 2 show the spacing distribution for the automated thinner and hand thinned fields, respectively. These graphs clearly show that the automated thinner left a greater percentage of plants closer to the desired spacing of 10 inches than hand thinning.
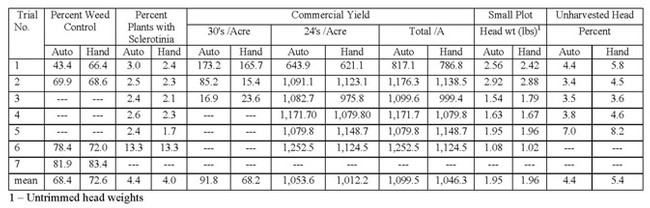
On average over six fields evaluated for yield, the automated thinner had an increase in yield of 53 boxes/A (Table 3). Examining the range of yields in the fields evaluated, it can be seen that there was a great deal of variability among fields and the yield was not always higher in fields thinned by the automated thinner. On average there were more 30's in the automated thinner treatment, but data on the number of 30's in only available from three fields which is a particularly small sample size to draw a firm conclusion. In small plot evaluations, we did not measure a difference in the mean plant weight between treatments. Overall the automated thinner had more plants/A than the hand thinned treatment, but again, the data is quite variable. There were no differences in the percent plants with lettuce head drop or in the percent weed control during the thinning operation.
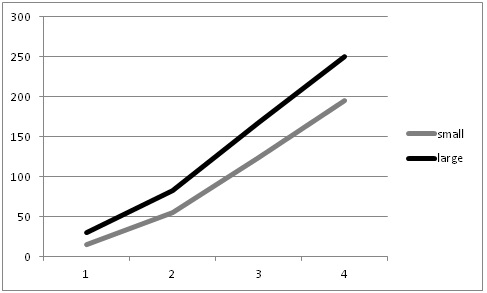
We did not measure the mean plant size left by the two thinning techniques, however, we noticed at one field that the automated thinner treatment may have left a greater proportion of smaller plants. We did a follow-up study to examine the fate of these smaller lettuce plants. Unthinned smaller plants remain smaller through the growth cycle and are smaller at harvest (Figures 3 & 4). This finding indicates that leaving larger plants at thinning (a standard practice used in hand thinning) may help improve yield, but it may sacrifice accuracy. This finding indicates an important detail that may need further research by the automated thinners to help improve the modest yield advantage that we measured in this study.
Table 1. Background information on thinning operations and fields
Table 2. Details on thinning time and spacing
Table 3. Details on pest issues and harvest
- Author: Richard Smith
- Author: Michael D Cahn
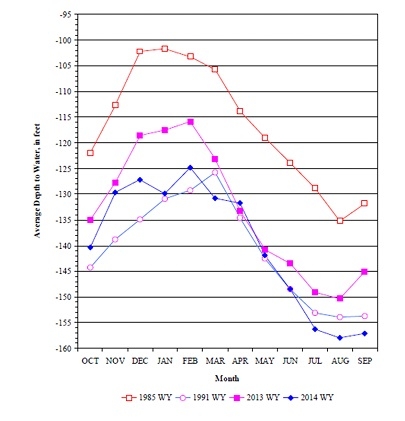
Ground water levels normally fluctuate in the Salinas Valley. Water levels in the aquifer decrease during the spring and summer when crops are being irrigated and recover in the fall and winter when pumping decreases and winter rains begin (Figure 1). However, the prolonged drought has decreased groundwater depths in the Salinas Valley to historically low levels. Because of the limited supply of stored water in the Nacimiento and San Antonio reservoirs this year, groundwater recharge from the Salinas River was minimal this summer and fall. Consequently, ground water levels in areas far from the river, such as the eastside of the valley, are not recovering as rapidly as in past seasons.
As winter approaches it may be worthwhile to think about ways to maximize infiltration of rain from storm events. Ideally storms would be spread apart by several weeks to maximize infiltration, but often several storms hit the central coast within a 2 to 3 week period which contributes to flooding of roadways, erosion, and thousands of acre-feet of water flowing out to the ocean. Infiltrating as much of this rainfall as possible into your fields would maximize salt control and recharge to the aquifer beneath your fields, and minimize water quality impacts downstream.
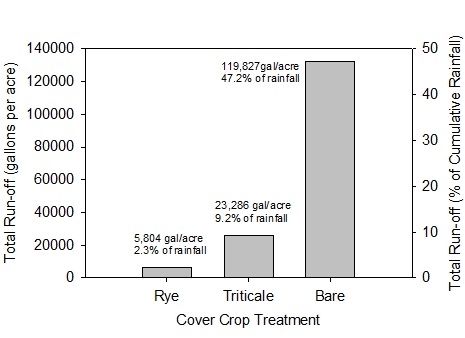
Cover crops can improve the infiltration of water into the soil by protecting the soil surface and eliminating sealing of the soil by crusting caused by the impact of raindrops on the soil surface. In as study that we conducted from 2009 to 2011 we observed greatly improved water infiltration into the soil with the use of low residue cover crops (Figure 1). In the bare-fallow plots, 47.2% of the rainfall ran off of the field; in the rye cover crop treatment only 2.3% of the rainfall ran off. The difference of water infiltrated by the cover crop and bare treatment equaled 1/3 of an acre-foot. In addition, the infiltrated rain water has a very low salt content so it is very effective in leaching accumulated salts near the soil surface.
One concern of using cover crops on the vegetable fields in the Salinas Valley is that they will use a portion of the moisture stored in the soil during the winter. Another concern is that the cover crops can be difficult to incorporate in the soil in early spring and may delay planting. The cover crop system used in this study was not a typical full-term cover crop, but rather was a low-residue cover crop that was killed 55-60 days after planting using an herbicide. They are planted in mid to late November when evapotranspiration is low, and only allowed to produce 0.5-0.75 ton of dry biomass per acre before they are killed. Under ideal conditions the cover crop residue breaks down over the winter and does not become an issue for preparing the peaked beds for planting (see photos 1&2) and YouTube: https://www.youtube.com/watch?v=k0oVVJ_BA7s
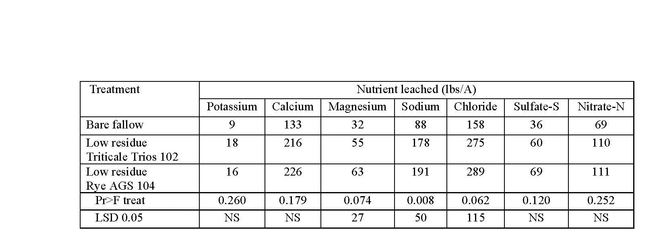
Even dead, the residues on the soil surface are effective in increasing infiltration during rain events. The increase in water infiltration in our trials allowed for greater leaching of salts from the root zone (Table 1).
Full-term cover crops may also allow for greater infiltration of water into the soil, but because the cover crops are growing over a longer period of time over the winter, they lose water due to evapotranspiration. If the winter is particularly wet the water loss from the crop may be of no consequence, but in a drier year, the cover crop may leave the soil drier and not allow for a net gain of water to the soil. For instance in a trial conducted from 2010-11, we observed 5.5 inches of water percolated into the soil in the bare fallow treatment, but only 3.0 inches in the full-season cover crop treatment. The difference in the two treatments was due to evapotranspiration by the cover crop.
There are undoubtedly compelling reasons to use a full-season cover crop and we are not advising against their use. However, given the serious nature of the drought, we are making the argument that low residue cover crops may provide a practice to infiltrate a greater quantity of water into the soil and help contribute to groundwater recharge, especially on the eastside of the valley where recharge from the river plays a smaller role in recharging the ground water in this area. The eastside of the Salinas Valley on the sloping Chualar loam soils is a large area and if a greater proportion of this area used low residue cover crops, a greater proportion of the rainfall could infiltrate into the soil thereby contributing to groundwater recharge.
- Author: Shimat Villanassery Joseph
Bagrada bug, Bagrada hilaris (Fig. 1) continues to be a major pest of brassica crops in the Salinas Valley and Hollister causing severe crop losses for both organic and conventional growers alike. Organic growers are struggling because there are very limited options at disposal to suppress the bagrada bug populations in the field. Conventional growers on the other hand are relying heavily on pyrethroid and neonicotinoid insecticides and are going with more number of applications than normal during early stages pf crop development (Cotyledon to four true leaves stage). This tactic (multiple applications) benefit the young seedlings as insecticide residues protect the plants from bagrada bug feeding especially, on the growing point or apical meristematic tissue. Feeding injury to meristematic tissue would cause “blind” head (no head) and multiple shoots on heading brassica crops such as broccoli, cauliflower or cabbage. With couple of insecticide applications at early stages of brassica crop, conventional growers are facing 5-30% loss from bagrada bug feeding injury. Some growers finding noticeably high mortality of cotyledons forcing them to skip thinning operation to maintain a decent crop stand.
Bagrada bug is taking a huge toll on leafy brassica crops such as Chinese cabbage (Pak Choi or Bak Choi), Arugula, Mizuna, Totsoi and Kale. These crops become unmarketable from direct feeding injury on the leaves (Fig. 2). These crops are like “magnets” for bagrada bugs. Bagrada bugs can precisely detect the seeds planted in the soil and most of the seedlings emerge with bagrada bug feeding injury. Sometimes, severe feeding at young stages cause plants die upon emergence (Fig. 3).
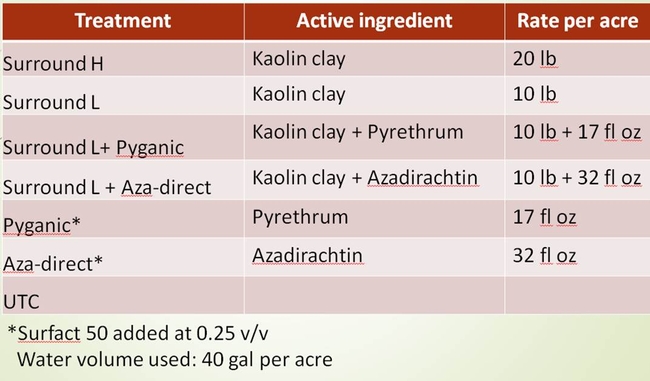
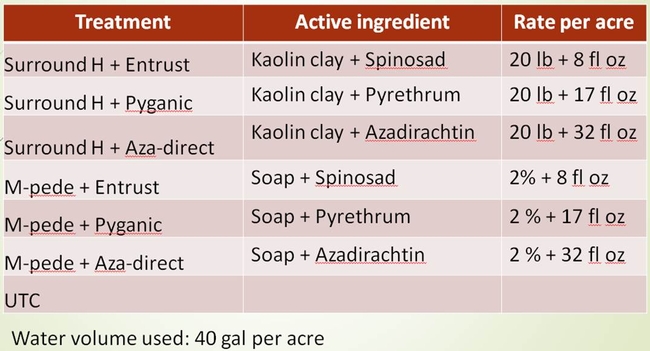
Two field trials were conducted in Hollister seeking organic insecticide options for bagrada bug management. The insecticide products chosen were Surround (Kaolin clay), Pyganic, Aza-direct, Entrust and M-pede. Surfact 50 was added when the Pyganic and Aza-direct were used alone. These organically approved insecticides were combined with other insecticides based on certain assumptions. For example, when insecticide Surround is sprayed, it forms a thin particle film on the leaf surface without affecting light transmission or photosynthesis. As bagrada bugs crawl on the Surround treated surface, the particles could stick to their body possibly cause irritation and force them to crawl off from the treated surface. This phenomenon if occurs, it could reduce the incidence of bagrada bug feeding injury. Moreover, it is possible that combining insecticides such as Pyganic and Aza-direct with Surround may increase insecticide exposure as bagrada bugs groom to remove the clay particles stuck on their body using their legs or wings.
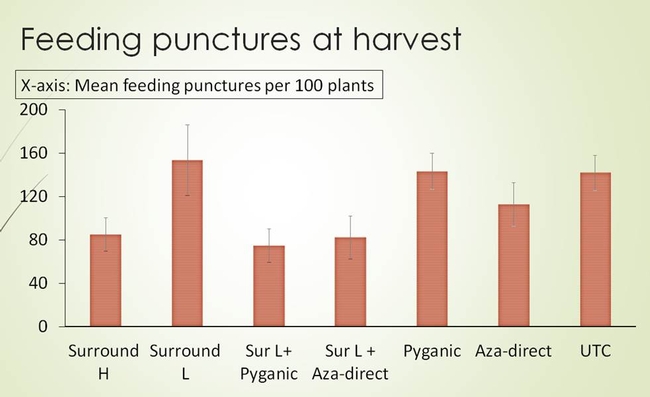
The field where the trials were conducted had enormous bagrada bug pressure. Bagrada bugs were everywhere that one would easily kill “thousands of bugs” just by walking over the beds. All stages of bagrada bug were present in the field at the time of insecticide applications. The applications were targeted to protect the plants from feeding injury and not particularly to kill the bugs. The first trial was conducted in Mizuna field and the second trial was conducted in Arugula field. In both the trials, the products were applied four times at three-day interval until harvest between 7 AM and 9 AM. The water volume used was 40 gal per acre. The products were applied using the pneumatic sprayer or back pack sprayer. The details of the products, active ingredients and the rates used for the trials are shown in the Tables 1 and 2. The design of experiments was Completely Randomized Block Design with four replications (Fig. 4). Plant samples were collected twice a week and were evaluated for bagrada bug feeding injury on the true leaves (Fig. 5).
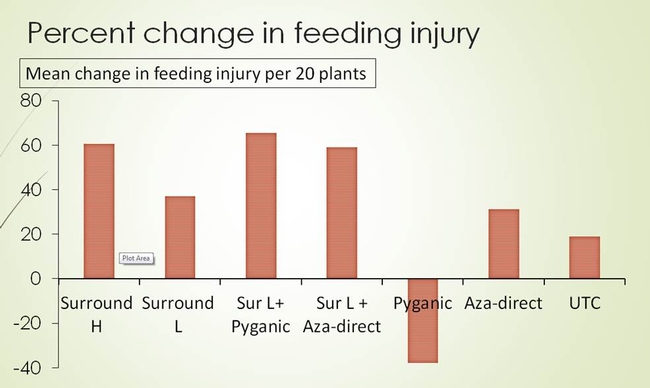
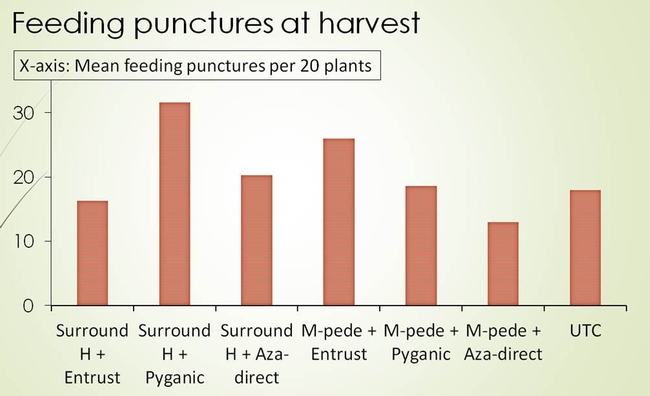
In trial 1, the bagrada bug feeding injury was numerically lower on Mizuna plants that received higher rate of Surround (alone) and Surround combined with Pyganic or Aza-direct than on untreated plants (Fig. 6). When the percentage change in bagrada bug injury on true leaves was calculated (taller the histogram, better the product performance), the plants treated with higher rate of Surround, and Surround combined with Pyganic or Aza-direct had greater reduction of bagrada bug feeding than untreated plants (Fig. 7). In trial 2, none of the treatments showed any indication of suppression when compared with untreated plants (Fig. 8).
Basically, these studies did not provide definite answers to the questions posed or problem but provided some trends. It appears that combining Pyganic or Aza-direct with Surround may have some value rather than applying alone. The Surround rate 20 lb per 40 gal of water is maximum rate for this product. Because Surround could clog the spray tanks, it requires rigorous agitation before application. Also, because Surround easily comes off from the leaf surface with sprinkler irrigation, reapplications are warranted if irrigated at closer intervals especially during the early stages of the crop. The rate of M-pede used in the study was 2% of the water volume. Typically, 2% of M-pede is considered as a high rate and increasing the rate (> 2%) may cause phytotoxicity (burning of leaves).
Then, can we manage bagrada bug?
Few thoughts:
- Perhaps, we should approach this problem differently. Bagrada bug is a landscape scale pest that they could reproduce and build-up to huge populations if the food is available in plenty, and warm and dry conditions persist. So far, we learned that their population build-up starting late July to December in the Salinas Valley and Hollister. The warmer conditions favor rapid reproduction and several overlapping generations of bagrada bug would develop in short period.
- We observed that their population pressure vary across landscapes and is a serious problem where the control options are limited. For example, bagrada bug problem is less severe when the management is aggressive such as conventional fields where growers have effective products that could suppress or knock down their populations at least in the crops grown. Organic growers on the other hand have limited options to fight bagrada bug and its population rapidly grows into uncontrollable size.
- We also noticed that initially these bugs invade the plants in the edge of the field from the surrounding fields or risk zones (e.g. ditches).
- These facts suggest that this is a landscape level problem rather than a field level problem. Bagrada bug management approach probably should include the management of various kinds of hosts that function as reservoir (e.g. brassica weeds) and aid to sustain their populations (brassica crops).
- Cultural management: Avoid planting brassica crops back-to-back pattern or staggered pattern. This would provide opportunity for bagrada bug to utilize the continuous supply of food to reach uncontrollable population size in short period of time. If somehow, the growers could disrupt the ecology of bagrada bug by not growing brassica crops in succession for a period instead rotate with non-brassica hosts, their population might crash and reach to a controllable levels.
- Weed management: Aggressive weed management especially brassicaceous weeds along with tight cultural management would disrupt the food supply and prevent escalation of population size.
- Bagrada bugs have demonstrated the ability to survive on non-brassica hosts especially solanaceous crops such as tomato, potato or pepper but would rarely reach to the levels we are seeing in brassica fields.
For further reading on bagrada bug please click on the following links (UC IPM pest note or blog articles).
http://www.ipm.ucdavis.edu/pestalert/pabagradabug.html
http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=14915
http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11632
http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11591
Please contact me (Shimat Joseph) by email at svjoseph@ucanr.edu or phone at 831-229-8985 if you have any questions or comments.





- Author: Ria Debiase
Nursery workers are our first line of defense in detecting light brown apple moth when growing ornamental plants in commercial nurseries. A new brochure and video can help those in the field distinguish light brown apple moth from several look-alike caterpillars.
Light brown apple moth is currently under a California Department of Food and Agriculture quarantine that regulates the interstate shipment of plants to keep the moth from spreading to new areas. It has been quarantined in various counties throughout coastal California ranging from Mendocino to San Diego.
Correct field identification of the light brown apple moth is the first step in containing the spread of this moth. Unfortunately several other leafroller caterpillars, including the orange tortrix, omnivorous leafroller, avocado leafroller, and apple pandemic moth, look similar to light brown apple moth caterpillars. This makes photo identification tools that can go into the field with workers, like the Field Identification Guide for Light Brown Apple Moth in California Nurseries, a useful resource for nursery workers.
The field guide was created by Steven Tjosvold, Neal Murray, University of California Cooperative Extension; Marc Epstein, Obediah Sage, California Department of Food and Agriculture; and Todd Gilligan, Colorado State University with the Statewide Integrated Pest Management Program (UC IPM).
An exotic and invasive pest from Australia, light brown apple moth has a host range of more than two thousand plants. It is a pest to a wide range of ornamental and agricultural crops, including caneberries, strawberries, citrus, stone fruit, apples, and grapes. The caterpillars eat leaves and buds, leading to weak or disfigured plants. They also can feed directly on fruit, causing the fruit to be unmarketable.
For more information on light brown apple moth and other leafrollers found in nurseries, see the UC Pest Management Guidelines for Floriculture and Nurseries.