- Author: Richard Smith
- Author: Steven T. Koike
Nitrogen fertilizer can have negative impacts on lettuce roots via salt effects and specific ion toxicity from nitrite and ammonium. In the early spring we commonly see ammonium toxicity in coastal lettuce production fields. In the past week, we received numerous samples of lettuce plants with this disorder. The core of the root of affected plants turns reddish brown to black, tissue becomes soft (necrotic), and the root becomes hollow as the plants age (Photo 1). The tops of affected plants suffer from water stress and become stunted; plants often wilt during the day due to the impaired root system (Photo 2). Lettuce plants with damage from ammonium may remain alive but will be stunted and behind in development; if the damage is severe and/or the plants are small, the lettuce can die. A key characteristic of ammonium toxicity is the distribution pattern in the field. Plants affected with this problem will occur randomly scattered throughout the field and will rarely occur in patches or groups.
While ammonium toxicity symptoms may resemble those of some lettuce diseases, in most cases ammonium toxicity is readily differentiated from fungal or bacterial problems of lettuce roots (Table 1). The main disease that may be confused with ammonium toxicity is Fusarium wilt. However, Fusarium wilt is characterized by occurring later in the summer, causing symptoms on both younger and older lettuce plants, occurring in patches and groups of plants, and thus far being found primarily in the southern part of Monterey County.
Table 1. Comparison of various characteristics of ammonium toxicity and soilborne diseases of lettuce
| Characteristic | Ammonium toxicity | Fusarium wilt | Verticillium wilt | Corky root |
| Stunted young plants | Yes | Yes |
No |
Yes, sometimes |
| Root vascular discoloration | Yes: red to brown to black | Yes: usually red to brown | Yes: usually black |
No |
| Root exterior surface | White, healthy | White, healthy | White, healthy | Yellow to brown, cracked |
| Affects mature plants |
No |
Yes | Yes | Yes |
| Field distribution | Random, scattered | In patches, groups | In patches, groups | May be uniform |
| Time of year | Early spring, sometimes June | Summer through fall | Summer through fall | Spring through fall |
The factors that contribute to the buildup of toxic levels of free ammonia and/or ammonium in the soil include fertilizer type, as well as soil type, temperature and moisture content. For instance, urea fertilizers results in the buildup of ammonium resulting from hydrolysis in the presence of the urease enzyme:
| CO(NH2)2 | + | 2H20 | + | H+ | ---------> | 2NH4+ | + | HCO3- |
| Urea | water | acid | urease | ammonium | bicarbonate |
If the pH of the zone where urea hydrolysis is taking place increases to above 8.3 (e.g. in the fertilizer bead), highly toxic amounts of free ammonia can temporarily form. High levels of ammonium in lettuce tissue can also be toxic. In the early spring when soil temperatures are less than 60 °F, nitrification of ammonium to nitrate is slowed and ammonium can build up in the soil following the application of urea or ammonium fertilizers. We have observed soil ammonium levels ranging up to 8 ppm or more in the winter and early spring, whereas in the summer it is rare to see soil ammonium levels exceed 1.0 to 1.5 ppm. Given the sudden onset of a number of fields with ammonium toxicity, it seems clear that weather and/or cool soil temperatures created conditions favorable to the buildup of toxic levels of ammonium in the soil.
Table 2 shows data from a 2001 on-farm evaluation where calcium nitrate was compared with the standard fertilizer program of ammonium nitrate to determine the effect on ammonium toxicity symptoms. In trial 1 we did see a significant reduction in the number of heads per plot with ammonium toxicity; however, there were still some toxicity symptoms even with the use of calcium nitrate, indicating that the form of fertilizer was important, but did not answer all the questions regarding the disorder in these trials.
In coastal California ammonium toxicity is typically an issue that occurs during the months of February and March. As the days lengthen and soil temperatures increase, issue with ammonium toxicity will decrease.
Table 2. Number of heads/plot with toxicity symptoms and levels of ammonium and nitrate in the soil 2 weeks following fertilizer application
| Fertilizer Type |
Heads with toxicity No. |
NH4-N ppm |
NO3-N ppm |
| Trial No. 1 | |||
| Calcium nitrate | 18 a | 0.79 a | 61.1 |
| Ammonium nitrate | 61 b | 3.43 b | 82.6 |
| Trial No. 2 | |||
| Calcium nitrate | 10 | 0.74 | 29.8 |
| Ammonium nitrate | 24 | 0.82 | 35.7 |
- Author: Michael D Cahn
CropManage Workshop: Hands-on training
Monterey County Agricultural Center Conference Room
1432 Abbott St, Salinas CA 93901
Thursday, April 2nd 2015
(8:30 am – 12 pm)
We will offer a hands-on training to learn in depth about the features of CropManage, a free online decision support tool for water and nutrient management of coastal crops. In addition to head and romaine lettuce, CropManage now supports broccoli, cauliflower, cabbage, and strawberries.
Considering that the drought is continuing into a 4th year, and nutrient management continues to be linked to water quality regulations, efficiently using water and nitrogen fertilizer is a high priority for Central Coast growers. CropManage can play an important role in providing quick decision support on water and nutrient management on a field-by-field basis.
This training will provide an opportunity to learn how to use CropManage for improving the efficiency of your farming operations or for adding value to your consulting services. We will provide in depth hands-on training so that you can learn step-by-step how to navigate and use CropManage for assisting with fertilizer and water management decisions and record keeping. Wi-Fi internet access is available at our conference room so please bring a laptop or tablet computer so that you can follow along as we tour through the features of the software. There should be sufficient time to answer questions as we cover the following topics:
Agenda:
8:30 – 9:00 Registration and Refreshments
9:00 – 9:30 Introduction and update on CropManage
9:30-10:15 Getting started with CropManage
10:15 (Break)
10:30 – 11:15 Strategies for using CropManage for decision support and record keeping
11:15- 11:45 Advanced features and interfacing sensors with CropManage
11:45-12:00 Discussion of new features or changes needed.
To keep the group size manageable so that we can provide individual help, we would like to limit the workshop to 30 participants. If you have attended previous workshops and or feel proficient in using the on-line tool, then you are welcome to just attend the second half of the workshop (10:30-12 pm). Whether or not you plan to attend the entire or part of the workshop, please RSVP in advance by sending an email to larriaga@ucdavis.edu or mdcahn@ucdavis.edu with the subject heading “CropManage workshop” and let us know the number of participants in your group. We will email you a confirmation. Thank you, and I hope to see you soon.
Respectfully,
Michael Cahn, Irrigation and Water Resources Advisor
Certified Crop Adviser CEU hours requested (1.5 hrs irrigation management, 1.5 hrs nutrient management)
- Author: Michael D Cahn
If you missed the 2015 Irrigation and Nutrient Meeting or you would like to review the presentations, you can download pdf versions of the powerpoint files from the UCCE Monterey County Website (http://cemonterey.ucanr.edu). The direct link to the presentations is:
http://cemonterey.ucanr.edu/Vegetable_Crops/2015_Irrigation_-_Nutrient_Management_Meeting_
We want to thank all of you who attended for your participation and for the many constructive comments that we received verbally and through the evaluation surveys. Let us know of any topics that you would like us to address in the next irrigation and nutrient management meeting or ways to improve the meeting.
If you are interested to learn more about CropManage for improving irrigation and nutrient management, I plan to host a hands-on training on using this on-line decision support tool on April 2nd. I will send out a formal announcement in the upcoming weeks.
- Author: Richard Smith
Acreage of Brussels sprouts is expanding in Monterey County from the traditional production areas along the immediate coast in Castroville and Moss Landing to as far south as Chualar and Gonzales. Interest by growers in efficiently managing nitrogen (N) applications in Brussels sprout production prompted us to conduct evaluations of mineral nutrition and nitrogen uptake dynamics in 2013 and 2014. In 2013 we conducted a preliminary study of nutrient uptake of a well-managed field. Table 1 shows the results of the total nutrient uptake of Brussels sprouts. The data shows the distribution of the various nutrients in the plant. The highest concentration of N is in the sprout (5.13%) which is almost two times the concentration found in the leaves (2.66%). Phosphorus is also concentrated in the sprouts (0.66%) with a much lower concentration in the leaves (0.24%). Potassium concentration in the sprouts (4.10%) was about the same as in the leaves (3.98%). Potassium had the highest whole-plant concentration (3.85%) than other nutrient. Calcium concentration in Brussels sprout leaves was very high (3.23%), but was much lower in the sprouts (0.36%). The low concentration of calcium in the sprouts is due to the low transpirational flow of water in the inner tissue of the sprout, which may explain why Brussels sprouts can be quite susceptible to tipburn at times (Figure 1). Of the micronutrients, zinc and copper had greater concentrations in the sprouts while iron, manganese and boron were more concentrated in the leaves.
Evaluation of the crop biomass indicates that about 21% of the biomass was in the sprouts, 47% in leaves and 33% in stalks (Table 2). The total dry matter in the crop was 14,862 lbs/A. Potassium uptake by the crop was 593 lbs/A, followed by N uptake 490 lbs/A and calcium 267 lbs/A. The combination of high concentration of nutrient and high crop biomass resulted in nutrient uptake that exceeded values we have observed in other cole crops such as broccoli and cabbage, and may be the highest of any vegetable grown in the Salinas Valley.
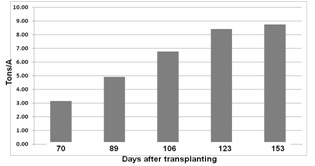
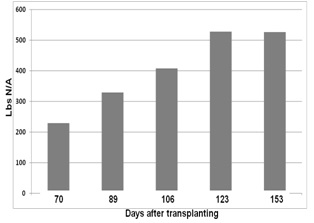
Brussels sprout is a long-season crop which makes nutrient management a challenge; for the majority of the crop cycle, tractors cannot enter the field to make fertilizer applications. The question arises, how can growers supply sufficient N to this high demanding late in the growth cycle? Nitrogen can be injected into the irrigation system, but how much N is needed by this high N demanding crop. In 2014 we worked with a grower who observed strong growth of his Brussels sprouts in spite of low N applications to the field. He still had 4+ months until harvest and had questions about how much N to supply his crop over the remainder of the growing season. One difficulty is that we have limited information on petiole nitrate levels of Brussels sprouts over the course of the growing season. We chose his youngest block for evaluations of N uptake by Brussels sprout. The block was transplanted in mid-June and our evaluations started mid-August. Up to that point the grower had applied 71 lbs N fertilizer/A. We evaluated biomass and biomass N accumulation over the remainder of the growth cycle, as well as nitrate levels in petiole fresh sap and dry tissue. Fresh sap nitrate analyses were made with a Cardy nitrate meter and dry tissue petiole nitrate levels were determined at the UC Davis Analytical Lab. Figure 2 shows the accumulation of dry biomass and Figure 3 shows the biomass N accumulation over the growth cycle. It is interesting to note that the crop had already taken up over 200 lbs N/A by the first evaluation date, 70 days after transplanting. Nitrogen uptake continued at a steady rate of over 5.0 lbs N/A/day until 123 days after transplanting at which time the rate of N uptake slowed. The grower applied a total of 145 lbs N/A in five fertilizer applications and the obvious question is where did the remaining 380 lbs N in the crop came from.
Other sources for nitrogen that could have provided for this crop include:
- Nitrogen in the irrigation water
- Residual soil nitrate
- Nitrate mineralized from soil organic matter and prior crop residues over the crop cycle
- Nitrate deeper in the soil profile that the roots access over the crop cycle
The nitrate levels in the irrigation water were below 2.5 ppm NO3-N which would not contribute a significant amount of N. Soil nitrate down to three feet in the soil amounted to a total of 57 lbs N/A when monitoring began in August, but much of the 200+ pounds of N already in plant biomass at that time may have come from soil residual nitrate. We did not measure the rooting depth of the crop, but given that Brussels sprouts are in the ground for 150+ days, there is a strong possibility that the roots extend rather deep. We have measured broccoli roots down to 3 to 4 feet deep in 90 days (Figure 4). If we assume that Brussels sprout roots grow at the same rate as broccoli roots, they could extend down much deeper (e.g. 4 to 5 feet). This is an areas that needs more research in the coming year. Nitrate mineralization from soil organic matter could have been significant at this site. The soil was a Salinas clay loam which can potentially mineralize 1.0 to 1.5 lbs N/A/day. This amount of N could contribute (100 – 150 lbs N/A) over the crop cycle. It is clear that this crop can efficiently mine the soil for nitrogen and where substantial soil residual N is present, only a modest amount of N fertilizer is required.
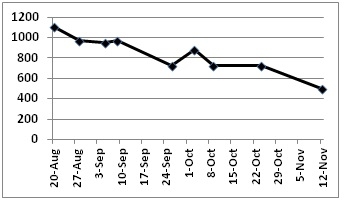
Petiole fresh sap vs dry tissue nitrate values were moderately correlated (R2 = 0.71). Nitrate-N in the fresh sap of the petioles of Brussels sprouts followed a typical trend over the course of the growth cycle (Figure 5). They started at 1100 ppm nitrate-N when the buds on the plant were just beginning to form. They declined to 700 ppm nitrate-N over the course of sprout development and finished the crop cycle at 500 ppm nitrate-N. These values were similar to values that have been observed for broccoli. These preliminary data provide an indication of where Brussels sprout fresh sap petiole levels should be; our goal is to finalize these values to provide a diagnostic tool for growers to use to better manage N Brussels sprouts, a long-season, high-nitrogen demanding crop.

Table 1. 2013. Concentration of plant nutrient in Brussel sprout tissues and whole plant
| Plant tissue |
Nitrogen % |
Phosphorus % |
Potassium % |
Sulfur % |
Calcium % |
Magnesium % |
Boron ppm |
Zinc ppm |
Manganese ppm |
Iron ppm |
Copper ppm |
| Sprouts | 5.13 | 0.66 | 4.10 | 1.35 | 0.36 | 0.18 | 20.8 | 39.2 | 30.0 | 193.1 | 4.3 |
| Leaves | 2.66 | 0.24 | 3.98 | 1.43 | 3.23 | 0.42 | 47.4 | 21.9 | 87.6 | 557.0 | 1.7 |
| Stalks | 2.63 | 0.53 | 3.38 | 0.84 | 0.65 | 0.24 | 19.7 | 22.9 | 19.0 | 177.4 | 3.0 |
| Whole plant | 3.48 | 0.41 | 3.85 | 1.20 | 2.02 | 0.38 | 35.7 | 28.3 | 77.4 | 340.9 | 2.9 |
Table 2. 2013. Plant biomass and nutrient uptake
| Plant tissue |
Fresh weight lbs/A |
Percent of whole plant |
Dry weight lbs/A |
Nitrogen lbs/A |
Phosphorus lbs/A |
Potassium lbs/A |
Sulfur lbs/A |
Calcium lbs/A |
Magnesium lbs/A |
Boron lbs/A |
Zinc lbs/A |
Manganese lbs/A |
Iron lbs/A |
Copper lbs/A |
| Sprouts | 22,272 | 20.1 | 2,827.0 | 153.9 | 19.8 | 122.9 | 40.6 | 10.9 | 5.4 | 0.1 | 0.1 | 0.1 | 0.6 | 0.01 |
| Leaves | 52,026 | 47.0 | 6,393.8 | 179.5 | 16.1 | 269.2 | 97.2 | 218.0 | 28.4 | 0.3 | 0.1 | 0.6 | 3.9 | 0.01 |
| Stalks | 36,315 | 32.8 | 5,641.3 | 157.0 | 31.6 | 201.0 | 50.4 | 38.6 | 14.5 | 0.1 | 0.1 | 0.1 | 1.0 | 0.02 |
| Whole plant | 110,613 | --- | 14,862.1 | 490.3 | 67.4 | 593.2 | 188.2 | 267.4 | 48.3 | 0.5 | 0.4 | 0.8 | 5.5 | 0.04 |
- Author: Richard Smith
- Author: Elizabeth Mosqueda
- Author: Anil Shrestha
- Author: Patricia Love
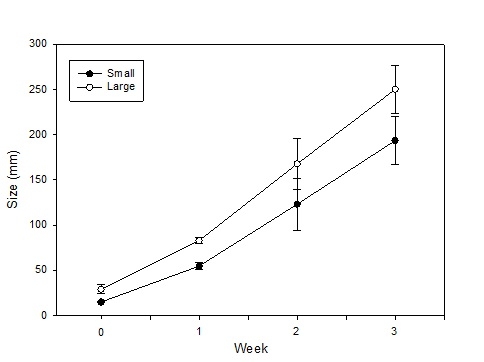
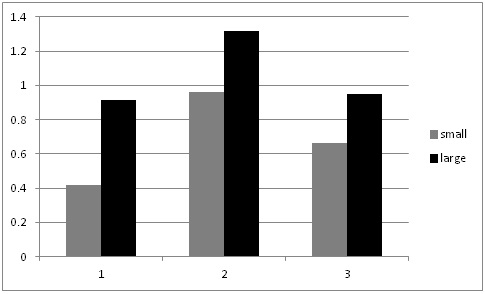
This summer, as we conducted our evaluations of automated thinners we had the opportunity to evaluate the impact on yield of selecting small vs larger lettuce plants at thinning (for more information on this project go to: http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=15932). It stands to reason that selecting larger plants during the thinning process would result in larger plants at harvest and this is a common sense decision that growers and thinning crews routinely make. However, we were not aware of scientific studies that showed the effect of plant size at thinning on crop yield. After thinning by the automated thinner in one field, we noticed that sometimes the automated thinner would select for a smaller plant rather than a larger plant (Figure 1). As a result, we conducted an evaluation of the impact of plant size on final plant yield in three commercial lettuce fields: two head lettuce (Sites 1 & 2) and one green leaf (Site 3). The evaluation was conducted by selecting 20 pairs of ‘small' and ‘larger' lettuce plants; plants were marked so that we could return and evaluate them over the course of the growth cycle. Lettuce plant diameter was measured four times, and at harvest, the heads were stripped to the marketable condition and weighed. Figure 2 shows the trend of plant sizes for three weeks after thinning; the smaller plants never caught up with the larger plants in size during this part of the growth cycle. At harvest, the marketable weight of the smaller plants was significantly less than plants that were larger at thinning (Figure 3).
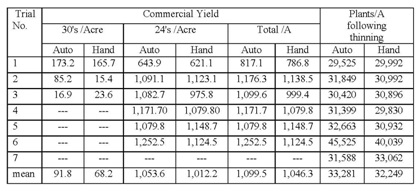
These results underscore the importance of selection for the largest and most vigorous plants at thinning, whether the plants are thinned by hand or by the automated thinners. We noticed that there may have been a tendency by the automated thinners, in their quest for accurate spacing, to select for small plants. In spite of this observation, our data shows that automated thinners are more accurate than hand thinning and that they have higher yields than hand thinning (Table 1). However, there were also more 30's per acre in the automated thinner treatment than in the hand thinning treatment. Automated thinners have the capacity to select for larger sized plants during the thinning process (they do so by evaluating the number of pixels of the plant). This evaluation indicates that that it is important that they do so in order to maximize the size of the selected plants and the ultimate yield of 24 count boxes.
Table 1. Commercial yield evaluation of automated thinner vs hand thinning treatments and total plants per acre.