- Author: Richard Smith
Acreage of Brussels sprouts is expanding in Monterey County from the traditional production areas along the immediate coast in Castroville and Moss Landing to as far south as Chualar and Gonzales. Interest by growers in efficiently managing nitrogen (N) applications in Brussels sprout production prompted us to conduct evaluations of mineral nutrition and nitrogen uptake dynamics in 2013 and 2014. In 2013 we conducted a preliminary study of nutrient uptake of a well-managed field. Table 1 shows the results of the total nutrient uptake of Brussels sprouts. The data shows the distribution of the various nutrients in the plant. The highest concentration of N is in the sprout (5.13%) which is almost two times the concentration found in the leaves (2.66%). Phosphorus is also concentrated in the sprouts (0.66%) with a much lower concentration in the leaves (0.24%). Potassium concentration in the sprouts (4.10%) was about the same as in the leaves (3.98%). Potassium had the highest whole-plant concentration (3.85%) than other nutrient. Calcium concentration in Brussels sprout leaves was very high (3.23%), but was much lower in the sprouts (0.36%). The low concentration of calcium in the sprouts is due to the low transpirational flow of water in the inner tissue of the sprout, which may explain why Brussels sprouts can be quite susceptible to tipburn at times (Figure 1). Of the micronutrients, zinc and copper had greater concentrations in the sprouts while iron, manganese and boron were more concentrated in the leaves.
Evaluation of the crop biomass indicates that about 21% of the biomass was in the sprouts, 47% in leaves and 33% in stalks (Table 2). The total dry matter in the crop was 14,862 lbs/A. Potassium uptake by the crop was 593 lbs/A, followed by N uptake 490 lbs/A and calcium 267 lbs/A. The combination of high concentration of nutrient and high crop biomass resulted in nutrient uptake that exceeded values we have observed in other cole crops such as broccoli and cabbage, and may be the highest of any vegetable grown in the Salinas Valley.
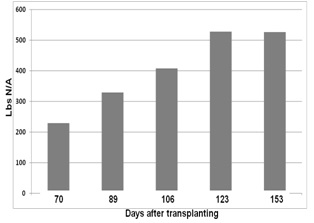
Brussels sprout is a long-season crop which makes nutrient management a challenge; for the majority of the crop cycle, tractors cannot enter the field to make fertilizer applications. The question arises, how can growers supply sufficient N to this high demanding late in the growth cycle? Nitrogen can be injected into the irrigation system, but how much N is needed by this high N demanding crop. In 2014 we worked with a grower who observed strong growth of his Brussels sprouts in spite of low N applications to the field. He still had 4+ months until harvest and had questions about how much N to supply his crop over the remainder of the growing season. One difficulty is that we have limited information on petiole nitrate levels of Brussels sprouts over the course of the growing season. We chose his youngest block for evaluations of N uptake by Brussels sprout. The block was transplanted in mid-June and our evaluations started mid-August. Up to that point the grower had applied 71 lbs N fertilizer/A. We evaluated biomass and biomass N accumulation over the remainder of the growth cycle, as well as nitrate levels in petiole fresh sap and dry tissue. Fresh sap nitrate analyses were made with a Cardy nitrate meter and dry tissue petiole nitrate levels were determined at the UC Davis Analytical Lab. Figure 2 shows the accumulation of dry biomass and Figure 3 shows the biomass N accumulation over the growth cycle. It is interesting to note that the crop had already taken up over 200 lbs N/A by the first evaluation date, 70 days after transplanting. Nitrogen uptake continued at a steady rate of over 5.0 lbs N/A/day until 123 days after transplanting at which time the rate of N uptake slowed. The grower applied a total of 145 lbs N/A in five fertilizer applications and the obvious question is where did the remaining 380 lbs N in the crop came from.
Other sources for nitrogen that could have provided for this crop include:
- Nitrogen in the irrigation water
- Residual soil nitrate
- Nitrate mineralized from soil organic matter and prior crop residues over the crop cycle
- Nitrate deeper in the soil profile that the roots access over the crop cycle
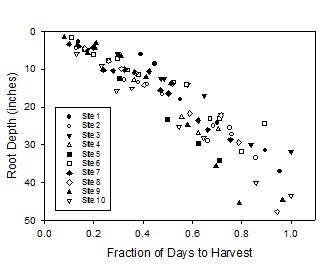
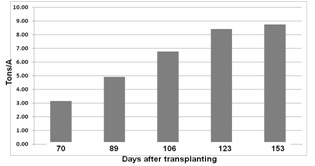
The nitrate levels in the irrigation water were below 2.5 ppm NO3-N which would not contribute a significant amount of N. Soil nitrate down to three feet in the soil amounted to a total of 57 lbs N/A when monitoring began in August, but much of the 200+ pounds of N already in plant biomass at that time may have come from soil residual nitrate. We did not measure the rooting depth of the crop, but given that Brussels sprouts are in the ground for 150+ days, there is a strong possibility that the roots extend rather deep. We have measured broccoli roots down to 3 to 4 feet deep in 90 days (Figure 4). If we assume that Brussels sprout roots grow at the same rate as broccoli roots, they could extend down much deeper (e.g. 4 to 5 feet). This is an areas that needs more research in the coming year. Nitrate mineralization from soil organic matter could have been significant at this site. The soil was a Salinas clay loam which can potentially mineralize 1.0 to 1.5 lbs N/A/day. This amount of N could contribute (100 – 150 lbs N/A) over the crop cycle. It is clear that this crop can efficiently mine the soil for nitrogen and where substantial soil residual N is present, only a modest amount of N fertilizer is required.
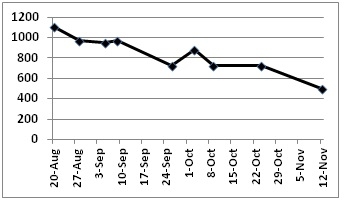
Petiole fresh sap vs dry tissue nitrate values were moderately correlated (R2 = 0.71). Nitrate-N in the fresh sap of the petioles of Brussels sprouts followed a typical trend over the course of the growth cycle (Figure 5). They started at 1100 ppm nitrate-N when the buds on the plant were just beginning to form. They declined to 700 ppm nitrate-N over the course of sprout development and finished the crop cycle at 500 ppm nitrate-N. These values were similar to values that have been observed for broccoli. These preliminary data provide an indication of where Brussels sprout fresh sap petiole levels should be; our goal is to finalize these values to provide a diagnostic tool for growers to use to better manage N Brussels sprouts, a long-season, high-nitrogen demanding crop.

Table 1. 2013. Concentration of plant nutrient in Brussel sprout tissues and whole plant
| Plant tissue |
Nitrogen % |
Phosphorus % |
Potassium % |
Sulfur % |
Calcium % |
Magnesium % |
Boron ppm |
Zinc ppm |
Manganese ppm |
Iron ppm |
Copper ppm |
| Sprouts | 5.13 | 0.66 | 4.10 | 1.35 | 0.36 | 0.18 | 20.8 | 39.2 | 30.0 | 193.1 | 4.3 |
| Leaves | 2.66 | 0.24 | 3.98 | 1.43 | 3.23 | 0.42 | 47.4 | 21.9 | 87.6 | 557.0 | 1.7 |
| Stalks | 2.63 | 0.53 | 3.38 | 0.84 | 0.65 | 0.24 | 19.7 | 22.9 | 19.0 | 177.4 | 3.0 |
| Whole plant | 3.48 | 0.41 | 3.85 | 1.20 | 2.02 | 0.38 | 35.7 | 28.3 | 77.4 | 340.9 | 2.9 |
Table 2. 2013. Plant biomass and nutrient uptake
| Plant tissue |
Fresh weight lbs/A |
Percent of whole plant |
Dry weight lbs/A |
Nitrogen lbs/A |
Phosphorus lbs/A |
Potassium lbs/A |
Sulfur lbs/A |
Calcium lbs/A |
Magnesium lbs/A |
Boron lbs/A |
Zinc lbs/A |
Manganese lbs/A |
Iron lbs/A |
Copper lbs/A |
| Sprouts | 22,272 | 20.1 | 2,827.0 | 153.9 | 19.8 | 122.9 | 40.6 | 10.9 | 5.4 | 0.1 | 0.1 | 0.1 | 0.6 | 0.01 |
| Leaves | 52,026 | 47.0 | 6,393.8 | 179.5 | 16.1 | 269.2 | 97.2 | 218.0 | 28.4 | 0.3 | 0.1 | 0.6 | 3.9 | 0.01 |
| Stalks | 36,315 | 32.8 | 5,641.3 | 157.0 | 31.6 | 201.0 | 50.4 | 38.6 | 14.5 | 0.1 | 0.1 | 0.1 | 1.0 | 0.02 |
| Whole plant | 110,613 | --- | 14,862.1 | 490.3 | 67.4 | 593.2 | 188.2 | 267.4 | 48.3 | 0.5 | 0.4 | 0.8 | 5.5 | 0.04 |
- Author: Richard Smith
- Author: Elizabeth Mosqueda
- Author: Anil Shrestha
- Author: Patricia Love
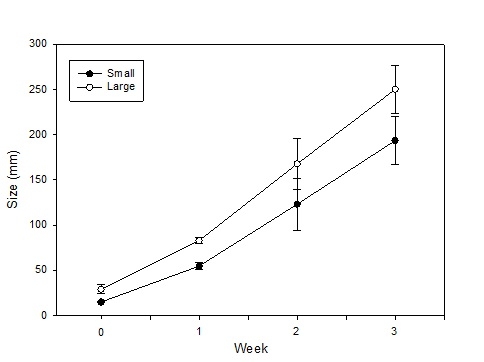
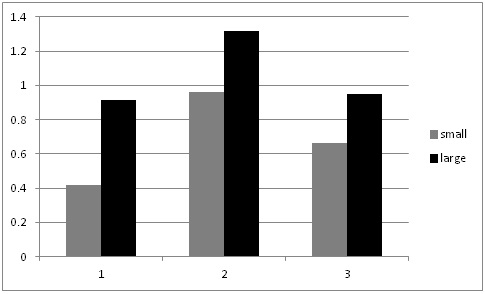
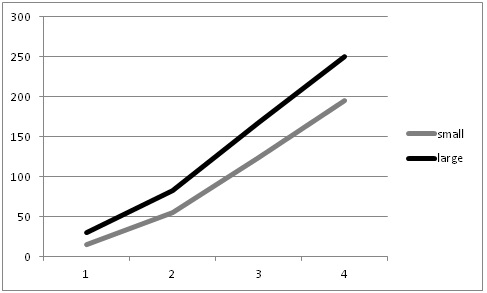
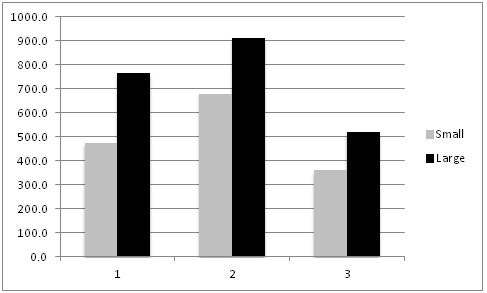
This summer, as we conducted our evaluations of automated thinners we had the opportunity to evaluate the impact on yield of selecting small vs larger lettuce plants at thinning (for more information on this project go to: http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=15932). It stands to reason that selecting larger plants during the thinning process would result in larger plants at harvest and this is a common sense decision that growers and thinning crews routinely make. However, we were not aware of scientific studies that showed the effect of plant size at thinning on crop yield. After thinning by the automated thinner in one field, we noticed that sometimes the automated thinner would select for a smaller plant rather than a larger plant (Figure 1). As a result, we conducted an evaluation of the impact of plant size on final plant yield in three commercial lettuce fields: two head lettuce (Sites 1 & 2) and one green leaf (Site 3). The evaluation was conducted by selecting 20 pairs of ‘small' and ‘larger' lettuce plants; plants were marked so that we could return and evaluate them over the course of the growth cycle. Lettuce plant diameter was measured four times, and at harvest, the heads were stripped to the marketable condition and weighed. Figure 2 shows the trend of plant sizes for three weeks after thinning; the smaller plants never caught up with the larger plants in size during this part of the growth cycle. At harvest, the marketable weight of the smaller plants was significantly less than plants that were larger at thinning (Figure 3).
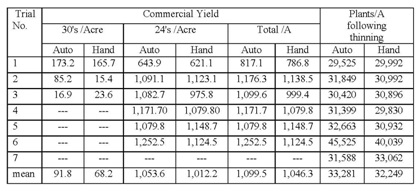
These results underscore the importance of selection for the largest and most vigorous plants at thinning, whether the plants are thinned by hand or by the automated thinners. We noticed that there may have been a tendency by the automated thinners, in their quest for accurate spacing, to select for small plants. In spite of this observation, our data shows that automated thinners are more accurate than hand thinning and that they have higher yields than hand thinning (Table 1). However, there were also more 30's per acre in the automated thinner treatment than in the hand thinning treatment. Automated thinners have the capacity to select for larger sized plants during the thinning process (they do so by evaluating the number of pixels of the plant). This evaluation indicates that that it is important that they do so in order to maximize the size of the selected plants and the ultimate yield of 24 count boxes.
Table 1. Commercial yield evaluation of automated thinner vs hand thinning treatments and total plants per acre.
- Author: Michael D Cahn
Please come to the 2015 Irrigation and Nutrient Management Meeting that will be held at the UC Cooperative Extension Office in the Agricultural Center Conference Room at 1432 Abbott Street, Salinas, CA. This is a free seminar, and will have a lot of good information pertaining to water and nutrient management on the Central Coast. We have a morning session at the conference center, followed with a Pizza lunch and a field demonstration at the USDA ARS Spence research farm on mitigating Chlorpyrifos from run-off.
When: Thursday, February 19
Time: 7:45 a.m. to 1:45 p.m.
7:45 Registration and Refreshments
8:00 N dynamics following anaerobic soil disinfestation
* Joji Muramoto, Project Scientist, UC Santa Cruz
8:30 N dynamics in high density vegetable production
* Richard Smith, Farm Advisor, UCCE Monterey County
9:00 Assessing soil and water salinity and determining leaching requirements
* Michael Cahn, Farm Advisor, UCCE Monterey County
9:30 CDFA nitrogen management tool and mineralization of soil organic matter
* Daniel Giesseler, Soil Specialist, UC Davis, Dept of Land, Air and Water Resources
10:00 Break
10:30 Mineralization of crop residues and impact on N availability
* Tim Hartz, Vegetable Specialist, UC Davis, Dept of Plant Sciences
11:00 Drought effects on groundwater management
* Thomas Harter, Groundwater Specialist, UC Davis, Dept of Land, Air and Water Resources
11:45 Fertilizer value of nitrogen in irrigation water for vegetable production
* Michael Cahn, Farm Advisor, UCCE Monterey County
12:15 Pizza Lunch
Field Day: USDA Spence Research Station, 1572 Old Stage Road
1:15 Demonstration of practices for mitigating Chlorpyrifos in tail water
* Brian Anderson and Bryn Phillips, UCD Environmental Toxicology
* Michael Cahn, UCCE Monterey County
2:15 Conclusion
* CDPR Continuing Education and Certified Crop Advisor Credits have been requested
* For more information call Richard Smith 759-7357 or Michael Cahn 759-7377
- Author: Shimat Villanassery Joseph
Growers in the Salinas Valley facing an irregular lettuce stand are usually uncertain about what caused the problem and often blame the factors such as poor seed quality, planting error, irregular irrigation timing or distribution, high salt levels in the soil or water, soilborne pathogens of seedlings, bulb mites, and garden symphylan feeding for the losses. Several springtails were collected from the soil associated with lettuce and it is not clear if they were feeding and contributing to the irregular lettuce stand. Often, this springtail is misidentified as garden symphylan.
Recently, I found a large number of a subterranean springtail (Protaphorura fimata) (Fig. 1) in the monitoring potato slice traps deployed in Salinas lettuce fields. I did a series of laboratory and field studies to determine its pest status in lettuce. This springtail (P. fimata) is less than 2.5 mm long, white in color and lacks eyes. Unlike other springtails, this springtail lack a furcula (jumping organ), and when disturbed it does not jump instead curls up. Other similar species of springtails primarily reproduce parthenogenically meaning they reproduce without mating; however, sexual reproduction is also seen on this one (P. fimata). This species (P. fimata) seems to be widely distributed in Europe, but has not been previously reported from the U.S.
Springtails occur in diverse habitats worldwide and are generally considered as beneficial arthropods because they aid in the decomposition of decaying plant material by feeding, thereby contributing to the cycling of carbon and nitrogen which in turn improves soil health and structure. This springtailis primarily known to feed on soil fungi but also feeds on live plant roots. Other springtails in the same family have been associated with feeding damage to germinating sugar beet seeds, sugarcane, poppy seeds and weed seeds (Plantago major). Foliage-feeding springtails (lucerne flea and garden springtail) attack several plants including Lucerne (Medicago sativa), clover (Trifolium sp.), sugar beet (Beta vulgaris) and bean.
The major objectives of the present study were to document the ability of springtail (P. fimata) to injure germinating seeds of lettuce in laboratory and field and characterize the feeding injury of springtail on germinating seeds and seedlings of lettuce.
In the laboratory, I conducted experiments in plastic petri dish with and without soil and sprigtails. Then I recorded the ungerminated lettuce seeds due to feeding injury, total number of feeding injury sites, and number of germinated seedlings with distinct feeding injury. I also documented the location (e.g. leaf, stem, plant crown or root) of the feeding injury on the plants. I conducted a field study too. The assumption of the field study was that the repeated use of maximum label rate of selected insecticides at early stages of plant development would suppress springtails and protect the seeds or seedlings from feeding. Two commonly used pyrethroid insecticides were applied (by the grower) three times: 2 days before planting, at planting, and 20 days after planting. Applications were made using a commercial tractor mounted sprayer. Two pyrethroid insecticides used were Mustang (4 fl oz per acre) and Warrior II (1.6 fl oz per acre). Both the Mustang and Warrior II were tank mixed and applied at 2 days before planting and 20 days after planting but only Warrior II was applied at planting to conform to the label. An adjuvant, Widespread Max (2 fl oz per acre) was added with all the applications. I used bait slices to monitor the springtails at weekly intervals. They were placed in the soil at 1.5 inches deep along the seed line and were covered with disposable white plastic bowls. At the end of each 2 days exposure period, beet root slices were removed, placed into plastic bags and transported to the laboratory. In addition, plant samples were collected to assess the plant growth. Please read the full Journal article for details.
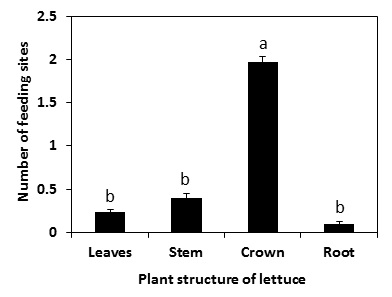
Results demonstrate that this springtail (P. fimata) can feed on germinating lettuce seeds or young seedlings, resulting in reduction in lettuce growth (Figure 2).Springtails attacked seeds and young seedlings alike. In the laboratory, springtails directly fed through the seed coat (pericarp) of a few seeds. This is possibly due to the moistening of the pericarp, enabling springtails to feed through the softened coating (Fig. 3a and b). In some instances, springtails fed on the growing radicle of the germinating seeds (Fig. 3c). However, most of the feeding at the seed radicle or elsewhere did not entirely sever it (Fig. 3d), which allowed the seedling to survive but affected the normal development of the plant. Moreover, most of the feeding injury was evident at the crown area rather than on leaf, stem, or root (Fig. 4).
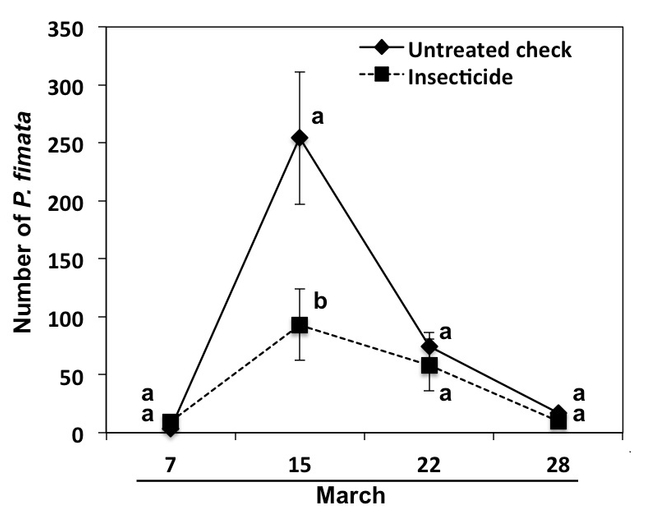
In the Salinas Valley, before the lettuce seeds are planted, fields are watered deeply and irrigations continue for at least three weeks after planting. I observed that the springtaildensity increased when the field was recently irrigated or after a rain event. This cultural practice which maintains high moisture levels for seed germination on the sub-surface profiles of the soil might be favoring faster buildup of springtailpopulations. In the field trial, the springtailcaptures were greater immediately after irrigation in the untreated beds than insecticide treated beds which was reflected in reduced number of springtails and in the untreated beds (Fig. 5).
In conclusion, this study clearly demonstrates that springtail (P. fimata) is an important pest of lettuce and is capable of reducing the crop stand. Incidence of high populations of springtail could be detrimental to germination of seeds in the field (Fig. 6 and 7). Springtails could be effectively suppressed to a large extent with early applications of insecticides directed to the seed line. Monitoring is the key to determine the presence and population size of springtail. Currently, I'm conducting for laboratory and field studies to determine the efficacy and application timing of insecticides.
Again, please use the link below to read the full article.
http://cemonterey.ucanr.edu/files/206762.pdf


- Author: Richard Smith
- Author: Elizabeth Mosqueda
- Author: Anil Shrestha
- Author: Fabian Galvan
- View More...
Summary:The use of automated thinners in the Salinas Valley has increased dramatically over the past two years. There are now four companies that manufacture automated thinners that use a spray system that removes the unwanted lettuce plants as well as weeds. This technology arrived just as the Salinas Valley was experiencing a labor shortage; many growers have adopted this technology or are in the process of seriously examining its usefulness to their operations. An evaluation of automated thinners was conducted in seven commercial lettuce fields in the Salinas Valley in the summer of 2014. The comparison was made with standard hand weeding. Overall, automated thinners were faster and more precise than hand thinning. However, the automated thinner left 7 times more doubles/A than hand thinning and it took more hand labor to remove the doubles in the double removal/weeding operation that occurs about 10 days following thinning; in spite of this issue, the total time to thin and remove doubles/weed in the automated thinner treatment took about 4.3 hours/A less time than hand thinning. A modest increase in yield was observed in the six fields evaluated, but the variability from field to field was quite high.
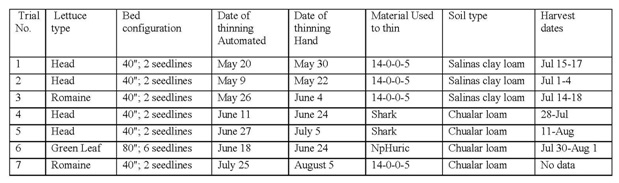
Methods:An evaluation of automated thinners was conducted during the summer of 2014. Trials were conducted with cooperating growers on seven commercial lettuce fields in the Salinas Valley, CA. The bed configuration and lettuce type are shown in Table 1. Each field was split in half with one side thinned by hand (standard practice) and the other side thinned with an automated thinner. The automated thinners used in these studies were: 1) Foothill Packing, 2) Blue River and 3) Agmechtronix. Thinning time was evaluated by recording the time it took to thin a designated area; this information was converted to hours per acre. In each field, four to six replicate areas two 40-inch beds wide by 90 feet were established in each treatment; these areas were used to make stand and weed count evaluations prior to and following the thinning operation. Following the thinning operation, the numbers of doubles (two closely spaced lettuce plants; see photo 1) were counted, and the distance between plants was measured to determine mean plant spacing and spacing distribution. Approximately 7-14 days following the thinning operation, a hand crew passed through the field to remove doubles and weeds; the time to conduct this double/weeding operation was measured and converted to hours per acre. Commercial and small plot yield measurements were made in six of the seven fields evaluated. Immediately prior to commercial harvest, 24 head from each evaluation area were cut and weighed to determine mean head weight. Plants were subsampled and sent to the UC Davis Analytical Laboratory for total N analysis to determine if the fertilizer used to thin the lettuce in some fields had an effect on the nitrogen nutrition of the crop. In addition, the numbers of heads infected with lettuce head drop (Sclerotinia minor) were counted. Commercial yield was measured in each treatment by obtaining box counts and sizes from the harvest crew. All commercial yields were converted to boxes per acre. The number of unharvested heads was counted in the evaluation areas following harvest. A side trial was conducted to evaluate the impact of size unthinned plants on the final plant size. In three of the fields, 20 heads of small and large plants were marked and plant diameter was measured three-four times during the crop cycle; final plant weight of these plants was measured at harvest.
 Photo 1. Chemically thinned lettuce |
 Photo 2. Double plants left by automated thinner |
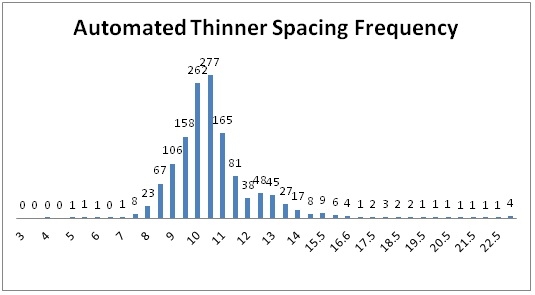
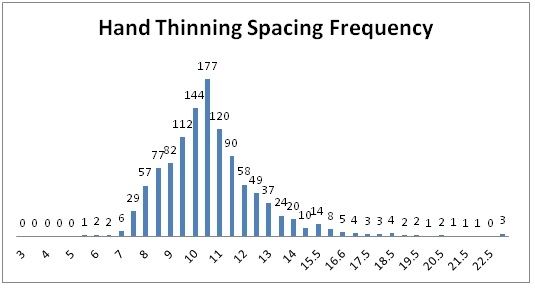
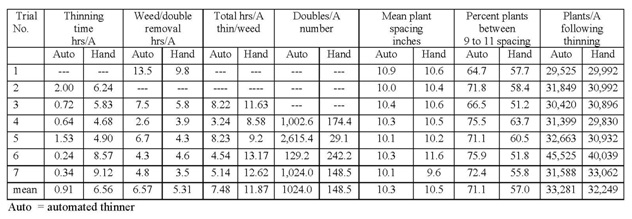
Results: On average automated thinners took 0.91 hours/A (range 0.24 to 2.0 hours/A) and hand thinning took 6.56 hours/A (range 4.68 to 9.12 hours/A) (Table 2). However, the double removal/weeding operation 7-14 days following thinning took 6.57 hours/A in the automated thinner treatment and 5.31 hours/A in the hand thinned treatment; the increase in time for double removal and weeding in the automated thinner area was due to the greater number of doubles in the automated thinner treatment (1,024 double/A) than hand thinning (149 doubles/A). The total time for thinning and double removal/weeding operations was 7.48 hours/A in the automated thinner treatment and 11.87 hours/A in the hand thinned treatment. The desired spacing for all fields in the study was 10.0 inches. The mean plant spacing in the automated thinner treatment was 10.3 inches (range 10.0 to 10.9 inches) and 10.5 inches in the hand thinned treatment (range 9.6 to 11.6 inches). The percent of plants that were between 9 and 11 inch spacing in the automated thinner and hand thinned treatments was 71.1 and 57.0, respectively. Figures 1 and 2 show the spacing distribution for the automated thinner and hand thinned fields, respectively. These graphs clearly show that the automated thinner left a greater percentage of plants closer to the desired spacing of 10 inches than hand thinning.
On average over six fields evaluated for yield, the automated thinner had an increase in yield of 53 boxes/A (Table 3). Examining the range of yields in the fields evaluated, it can be seen that there was a great deal of variability among fields and the yield was not always higher in fields thinned by the automated thinner. On average there were more 30's in the automated thinner treatment, but data on the number of 30's in only available from three fields which is a particularly small sample size to draw a firm conclusion. In small plot evaluations, we did not measure a difference in the mean plant weight between treatments. Overall the automated thinner had more plants/A than the hand thinned treatment, but again, the data is quite variable. There were no differences in the percent plants with lettuce head drop or in the percent weed control during the thinning operation.
We did not measure the mean plant size left by the two thinning techniques, however, we noticed at one field that the automated thinner treatment may have left a greater proportion of smaller plants. We did a follow-up study to examine the fate of these smaller lettuce plants. Unthinned smaller plants remain smaller through the growth cycle and are smaller at harvest (Figures 3 & 4). This finding indicates that leaving larger plants at thinning (a standard practice used in hand thinning) may help improve yield, but it may sacrifice accuracy. This finding indicates an important detail that may need further research by the automated thinners to help improve the modest yield advantage that we measured in this study.
Table 1. Background information on thinning operations and fields
Table 2. Details on thinning time and spacing
Table 3. Details on pest issues and harvest