- Author: Steven T. Koike
University of California Cooperative Extension—Monterey County
2013 Plant Disease Seminar
Tuesday, November 19, 2013
8:00 a.m. to 12:00 noon
**County of Monterey Agricultural Center— Conference Room**
1432 Abbott Street, Salinas, California
8:00 – 8:30 Registration for morning session (no charge).
8:30 – 9:00 2013 plant disease developments in coastal California
Steven Koike. UC Cooperative Extension, Monterey
9:00 – 9:30 Update on Verticillium wilt of lettuce and other crops
Krishna Subbarao. University of California, Davis
9:30 – 10:00 Emerging Produce Safety Issues: Human Parasites and Viruses
Trevor Suslow. University of California, Davis
10:00 – 10:30 Break: Sponsored by CAPCA, Monterey Bay Chapter
10:30 – 11:00 Protecting leafy greens from contamination by filth flies
Alec Gerry. University of California, Riverside
11:00 – 11:30 Grapevine virus diseases: Implications of red blotch virus
Larry Bettiga. UC Cooperative Extension, Monterey
11:30 – 12:00 Spinach downy mildew: research on disease development
Steve Klosterman. USDA-ARS, Salinas
Continuing education credits are requested. Call ahead (at least 24 hrs.) for special needs arrangements; efforts will be made to accommodate full participation. For more information, contact Steven Koike (831-759-7350; 1432 Abbott Street, Salinas, CA 93901) or visit our website at http://cemonterey.ucdavis.edu.
An afternoon session, held in this same conference room, will be hosted by CAPCA, Monterey Bay Chapter.
- Author: Shimat Villanassery Joseph
- Author: Michael Cahn, Irrigation and Water Management Advisor
- Contributor: Thomas Lockhart, Staff Research Associate
- Contributor: Laura Murphy, Staff Research Associate
An important benefit of drip irrigation is the ability to apply fertilizer through the irrigation water, permitting growers to spoon-feed nutrients, such as nitrogen (N), to their crops. By avoiding applications of large amounts of N fertilizer when the crop is small and uptake rates are low, losses of nitrogen by leaching can be minimized. Also, unlike furrow and overhead sprinklers, drip can deliver fertilizer in the zone where roots are most concentrated.
While drip fertigation offers several advantages for managing nitrogen fertilizer during the season, success depends on the management of the drip system and using best practices for fertigation. Drip systems with poor distribution uniformity may likely cause fertilizer to be unevenly distributed within a field. Also, the strategy of injecting fertilizer into a drip system can affect the distribution of fertilizer to the crop. Proper fertigation requires injecting at a steady rate and at a location that provides sufficient mixing of fertilizer with irrigation water. To assure that the fertilizer uniformly distributes within the field after an injection, sufficient irrigation time with clean water is needed so that all of the fertilizer is flushed out of the drip tape before the irrigation ends.
For drip to be economical for vegetable growers on the central coast, most farming operations retrieve drip tape after each crop is harvested and repair and reuse the tape for 8 to 12 crops. Breaks and leaks in the tape are repaired using a splicing machine (Figure 1). Growers have expressed concern that fertigating through their drip systems is not resulting in even applications of N fertilizer after the tape has been reused for multiple crops. Splicing machines often do not fully repair leaks in tape, and emitters tend to plug over time unless the tape was adequately maintained by flushing and chemical treatment.
In response to grower concerns, we evaluated the uniformity of applied water and nitrogen fertilizer for surface placed drip in 11 commercial lettuce fields during the fall of 2012 and during the spring of 2013.
Fig. 1. Splicing machines are used to repair leaks and breaks in drip tape
Procedures
All fields were planted with romaine or iceberg lettuce varieties on 40-inch or 80-inch wide beds. At each site irrigation, pressure, and fertilizer uniformity were evaluated during a single irrigation event. Field sizes ranged from 8 to 20 acres, and the maximum row lengths ranged from 600 to 1340 ft. Drip tape at all field sites was 7/8 inch diameter, medium flow tape (0.34 gpm/100 ft), but varied by manufacturer and age. The location where fertilizer was injected into the irrigation system, and start and end time of the fertigation, as well as the duration of the irrigation, were recorded. Before irrigating, couplers fitted with ¼ gallon per hour pressure compensating emitters that were spliced in to the drip tape at 24 locations within the field, representing the head, tail and middle areas. Water from these emitters was collected into 5 gallon containers during the entire irrigation (Figure 2) and analyzed for NO3-N and NH4-N. The discharge rate of 4 emitters and pressure of the tape was measured near each of the 24 fertilizer sampling locations (total of 96 emitters). Mass (lbs) of N applied at each of the 24 collection locations within a field was estimated by multiplying the measured discharge rate of the drip tape by the irrigation time and by the concentration of N in the collected water. Uniformity of applied water, tape pressure, and fertilizer was calculated by comparing the lowest 25% of measurements to the average of all 24 measurements. In addition to evaluating fertilizer distribution uniformity, we evaluated the time for fertilizer to travel to the furthest distance from the injection point by injecting food dye for a 5 minute period into the irrigation system and monitoring the water for color at the furthest point from the injection location.
Fig. 2. Low flow (1/4 gph) pressure compensating drip emitters were used to collect samples of irrigation water during the entire irrigation cycle.
Results
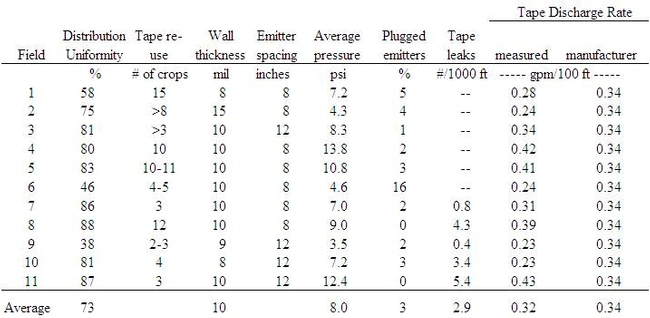
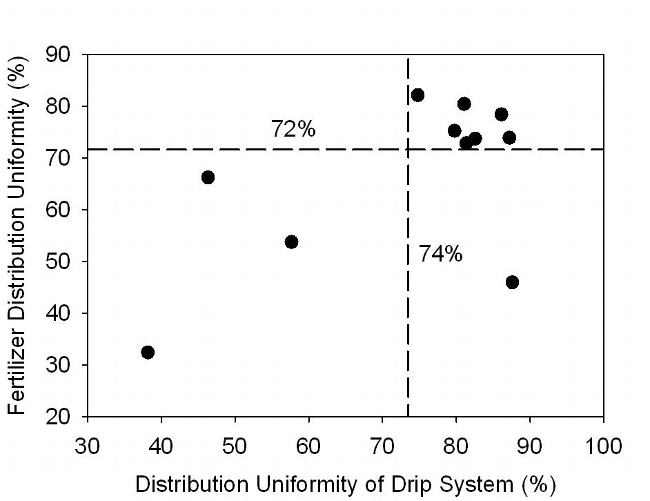
Distribution uniformity of applied water for the 11 fields averaged 73% and ranged from 38% to 88% (Table 1). The industry standard for irrigation uniformity of surface drip is 85%. Fertilizer application uniformity averaged 67% and ranged from 46% to 82%. The distribution uniformity of the drip systems of 7 fields evaluated was greater than 74% (avg = 82%) and fertilizer uniformity was greater than 72% (avg = 77%) (Figure 3).
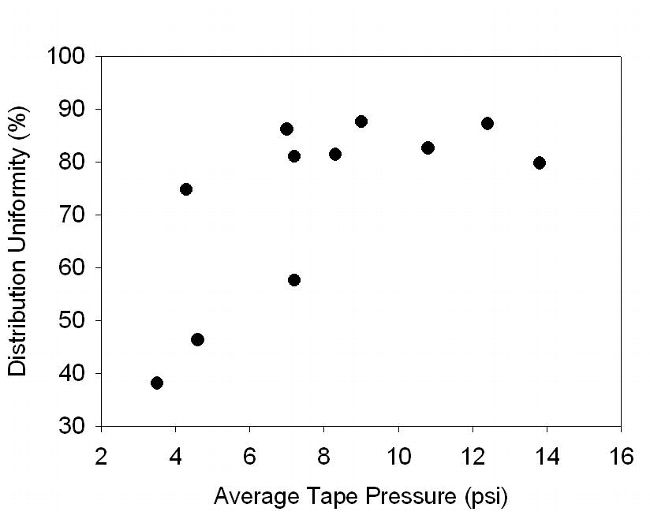
One of the causes for poor distribution uniformity of some drip systems may have been related to pressure. Pressure uniformity averaged 80% and ranged from 43% to 99% (Table 1). Average pressures in the drip tape ranged from 3.5 to 13.8 psi (Table 2). Where the system pressure averaged 4.3 psi, the tape discharge rate was 30% less than the manufacturer's rating. Irrigation distribution uniformity decreased substantially when the average field pressure was less than 5 psi (Figure 4). Additionally, a substantial percentage of emitters of some drip systems were plugged (Table 2) which would reduce irrigation system uniformity. Leaks were evaluated in 5 fields and ranged from 1 to 5 leaks per 1000 ft of tape (Table 2). Significant leaks can potentially reduce drip uniformity by lowering the downstream pressure. Other limitations to good drip uniformity included mixing different types of tape in the same field, fluctuating pressure during the irrigation, and row lengths longer than 800 ft.
Field 8 had a high uniformity of pressure and irrigation distribution but a low fertilizer uniformity. We speculate that the fertilizer which was injected at a “T” connecting the valve in the field with the submain did not have sufficient time to mix with the irrigation water before the flow split into opposite directions. Hence, the average concentration of N on one side of the field was approximately half the concentration measured on the other side of the field. The distribution uniformity of fertilizer on individual sides of the fields was greater than 87%.
With the exception of field 8, fertilizer distribution uniformity was closely related with irrigation system uniformity (Figure 4). Fields with the lowest fertilizer uniformity were operated at the lowest average pressure and/or had the highest level of plugged emitters (Table 2).
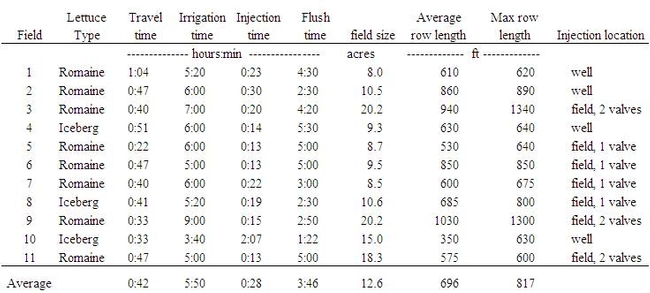
Fertilizer was injected at the well in 4 of the fields and at the submain valve in the other 7 fields (Table 3). Injections were made simultaneously using 2 valves at 3 of the fields. Fertilizer was injected during an average of less than 30 minute period often at the beginning of the irrigation (Table 3). The time required for the fertilizer to travel to the furthest point of the irrigation system averaged 42 minutes but ranged from as short as 22 minutes to as long as 1 hour. Field size, row length, and injection location appeared to affect the travel time of the fertilizer. The average time for flushing the fertilizer was 3.75 hours, which was ample time to allow the fertilizer to completely flush from the system. The irrigation industry recommends that for long irrigations (> 4 hours), fertilizer should be applied in the middle of the irrigation cycle. Only at field 10 was the fertilizer applied during the middle of the irrigation. The long flush time after injecting could potential leach nitrate forms of fertilizer below the root zone of the crop. On average, half of the applied fertilizer N measured in the collection buckets was in the nitrate form.
Conclusions
This survey of commercial lettuce fields demonstrated that N fertilizer applied by drip has an average distribution uniformity of 77% when the injection is properly made and the drip system is operated and maintained to achieve an average distribution uniformity of 82%. The results also showed that N fertilizer applied by drip is frequently distributed to fields unevenly due to poor uniformity of the drip systems, or because proper injection procedures were not followed. Operation procedures observed at these sites would suggest that irrigators may need training to better understand the principles of fertigation so that fertilizer is applied at the highest uniformity possible, and in a manner that will prevent leaching losses of nitrate.
Acknowledgements
We thank the California Leafy Green Research Board for funding this project and the many growers that cooperated with the field trials.
Table 1. Summary of irrigation, fertilizer, and pressure uniformity of drip irrigated lettuce fields.
Table 2. Drip tape characteristics at commercial lettuce sites.
Table 3. Irrigation summary for drip irrigated lettuce fields.
Fig. 3. Relationship between distribution uniformity of retrievable drip systems and fertigation uniformity. Each symbol denotes a commercial lettuce field evaluated during the study.
Fig. 4. Effect of tape pressure on the distribution uniformity of retrievable drip systems. Each symbol denotes a commercial lettuce field.
- Author: Shimat Villanassery Joseph
- Author: Richard Smith
Bagrada bug, Bagrada hilaris is now well established in the southern region of the Salinas Valley. This invasive stink bug, if left unmanaged, could become a serious pest of brassica crops. We studied their occurrence on cover crop, a mustard cover crop blend, broccoli and surrounding weed species, and would like to report few observations.
1. Cover crop, mustard
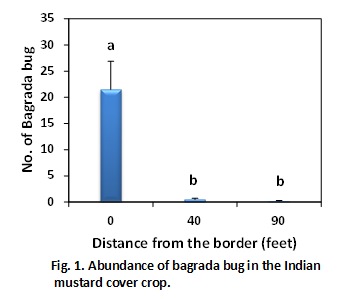
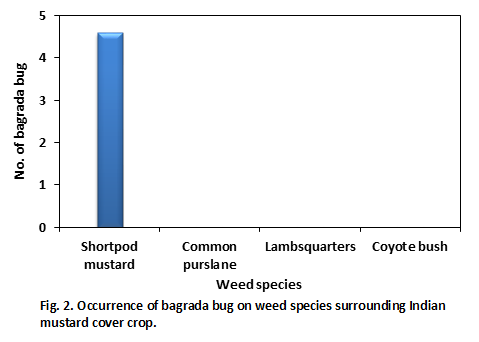
We observed significant populations of bagrada bug on 4-5 week-old mustard cover crop blend (Brassica juncea and Sinapis alba) in San Ardo. Interestingly, the adjacent field was also cover cropped with mustard but was disced few weeks ago. It is likely that the previous adjacent mustard crop might have had a bagrada bugs infestation and they moved to new planting when it was disced. When we walked into mustard stand from the disced field, we noticed that number of bagrada bug numbers declined from the field edge to the interior of the field. To understand better, a border zone (edge of the field), interior zone (90 feet into the field), and intermediate zone (between border and interior zones) were designated. Within each zone, six spots (~ 5.6 feet) were randomly selected and number of bagrada bugs was quantified after spending two minutes per spot. Similarly, two weed species and one native shrub surrounding the mustard field were randomly selected and number of bagrada bugs on them was counted after spending about one minute per plant.
Bagrada bugs tend to be more abundant on the edge of field than interior zones of the field (Fig. 1). It seems that bugs settled on the border plants of the field rather aggressively moving into the field. All life stages were detected. Most of the adults were in the mating position (connected by the rear ends) but very mobile. The adults tend to hide into the soil or under the leaves when we approached the infested plants.
Among the weed species investigated, bagrada bugs were only found on short pod mustard (Fig. 2 and 3). Other weed species investigated were shortpod mustard (Hirschfeldia incana), common purslane (Portulaca oleracea), lambsquarter (Chenopodium album), and the native shrub coyote bush (Baccharis pilularis). It is interesting to note that shortpod mustard plants were senescing, yet we found bagrada bugs on them.
2. Broccoli
The broccoli field was located in San Ardo adjacent to the Salinas River. The riparian plant community along the river contained stands of perennial pepperweed (Lepidium latifolium) as well as other species. Bagrada bug infestation was severe on pepperweed (Figs. 4a, and b).
Bagrada bug feeding injury symptom on broccoli plants was clearly visible on plants along the edge of the field. Feeding injury symptoms on broccoli include leaf distortion, chlorotic patches along the leaf margin and stunting (Fig. 5). Again, feeding symptoms drastically declined and/or plant vigor improved as we walked few steps (~ 10 feet) into the broccoli field from river side. Both nymphs and adults of bagrada bug were active along the edge of the field.
These preliminary observations indicate that some plants in the mustard family are highly attractive to bagrada bug. Shortpod mustard is a common summer-growing species that is commonly found on roadsides, in vineyards and in rangeland. Perennial pepperweed is an invasive plant that is commonly found in the riparian strip along the Salinas River. Both plants provide sufficient food resources for bagrada bug to successfully breed. After the onset of the winter rains, other mustard family weed species such as field mustard (Brassica rapa), black mustard (B. nigra), London rocket (Sisymbrium irio) and wild radish (Raphanus sativus) will begin their growth cycle. These plants are very common along roadsides and in ditches and may also provide over wintering habitat for bagrada bug.
- Author: Shimat Villanassery Joseph
Bagrada bug, Bagrada hilaris is an invasive stink bug and is moving North from the Southern Counties of California, rapidly than anticipated. In the past weeks, I have detected huge populations of these stink bugs on Indian mustard (cover crop), broccoli raab and kale in the King City and San Ardo areas. It is more likely that low populations exist in the other parts of the Salinas Valley.
It is well known that bagrada bug prefers cruciferous hosts (Family: Brassicaceae) including broccoli, cauliflower, cabbage, kale, arugula, collards, and Indian mustard. Major crops, lettuce and spinach are NOT a suitable host for bagrada bug. Also, these bugs can survive on cruciferous weeds such as mustard species (Brassica sp), wild radish, London rocket, short pod mustard and shepherd’s purse, as well as the insectary crop sweet asylum or sunflower. Mustard weeds species are very common in the Salinas Valley along ditches, roadsides and even along the edges of agricultural fields. Mustard cover crops such as white mustard (Sinapsis alba) and Indian mustard (Brassica juncea) in particular, could harbor bagrada bug populations which are not often monitored for pests.
Bagrada bug adult could be easily confused with harlequin bug. Adult of harlequin bug is orange with black and white marks, whereas bagrada bug adult is black with orange and white marks; and adult harlequin bug is about 3 times larger than bagrada bug. Eggs of harlequin bug are white with horizontal, black strips, whereas bagrada bug has no strips but has a “dirty” white appearance.
Damage to brassica crops is varied but could be severe. Severe economic loss has been reported when injury occurred during early developmental stages of the crop such as coldyledon or < 4 leaves stages. Injury on leaves appears initially as small puncher marks, which turn into white patches as leaves expand. In broccoli and cauliflower, the economic injury occurs when the bug feeding kills the apical meristematic tissue of young seedling, which later result in “multiple heads” or sometimes “blind heads” (without a head). Severe feeding also depletes the nutrient reserves of the plant leading to desiccation or wilting.
At this time, monitoring for bugs is the key. Monitoring for bagrada bug during mid-day hours might increase the probability of finding them as the bugs typically hide and stay in the cracks and crevices or on the underside of leaves when the temperature is on the cooler side. Cruciferous weeds in the drains, river bottoms, edges of the field or near residential area increase the risk of establishment. Do not forget to check the brassica cover crops for bugs as well.
Based on the insecticide efficacy studies conducted in University of Arizona, bifenthrin and methomyl were most effective in reducing bagrada bug infestation and injury on broccoli. For organic growers, none of the products were efficacious but pyrethrin and azidirachtin are suggested.
If you detect bagrada bug in Monterey, Santa Cruz and San Benito Counties, please do not hesitate to contact me (Shimat Joseph) at svjoseph@ucdavis.edu or (831) 759-7359.
Further reading:
http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=9023
Video (by Surendra Dara): http://youtu.be/gSj3AZoJIRM