- Author: Shimat Villanassery Joseph
Cabbage maggot (Delia radicum) (Fig. 1) is a serious and destructive pest of brassicas in the Salinas Valley of California. Brassica crops damaged by cabbage maggot are broccoli, cauliflower, cabbage, and Brussels sprouts. Cabbage maggot flies lay eggs in the soil around the base of a plant. Legless, white maggots feed on the taproot and affect plant development. After feeding for about 3 weeks, the maggot pupates in the surrounding soil for 2-4 weeks before emerging into an adult fly. The symptoms of cabbage maggot feeding in the root are yellowing, stunting, and slow growth.
Research showed that infestation by cabbage maggots in direct-seeded broccoli could be severe throughout the growing period, except the first 30 days after seed was planted. Typically, insecticide targeting cabbage maggot is applied immediately after planting seeds and before sprinkler is turned on. Efficacy studies with at-planting application of insecticide did not provide adequate cabbage maggot control. This suggested that insecticide applied at planting might be early relative to cabbage maggot incidence and thus, delaying application might be more effective.
In 2014 and 2015, replicated experiments were done in a commercial planting of baby turnip. The treatments were one chlorpyrifos application at planting and 2 weeks after planting seeds. A tractor-mounted sprayer was used to apply insecticide. Samples were collected and were transported to UCCE entomology laboratory where roots were evaluated for damage by cabbage maggot.
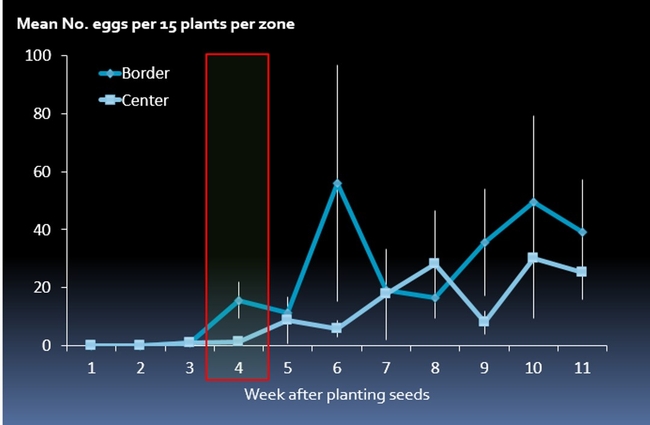
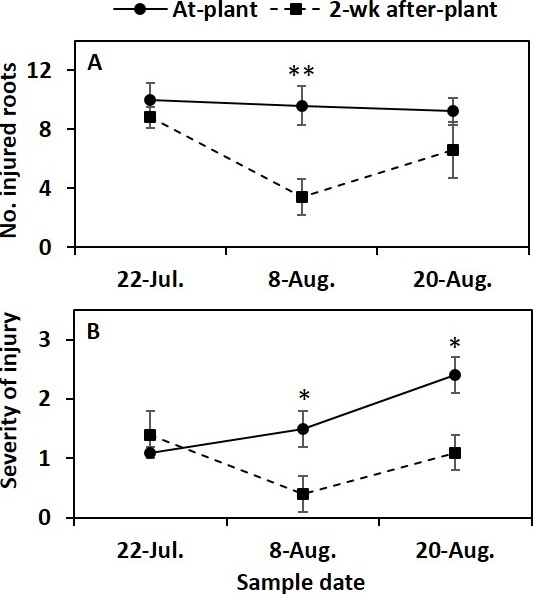
Results suggested that delayed application of effective insecticide suppresses cabbage maggot (Fig. 2). In a previous study, Joseph and Martinez (2014) showed cabbage maggot flies did not lay many eggs at the base of brassica plants until 3 weeks after plant emergence (Fig. 3), despite adult cabbage maggots in the field during early stages of plant development. Also, cabbage maggot infestation tend to be continuous after 3 week stage depending on local pest pressure and crop disturbances (e.g., harvest) in the surrounding fields (Joseph and Martinez 2014).
Delaying insecticide application would increase the likelihood of intercepting cabbage maggot larvae seeking roots. In the Salinas Valley of California, use of organophosphate insecticides including chlorpyrifos is regulated. This stringent regulation is forcing growers to seek alternate insecticides for cabbage maggot control. Previous study showed that clothianidin, thiamethoxam, and spinetoram as well as pyrethroid insecticides such as zeta-cypermethrin, fenpropathrin, bifenthrin, lambda-cyhalothrin, and pyrethrins were effective against cabbage maggot larvae, and efficacy was comparable to chlorpyrifos (Joseph and Zarate 2015). However, alternate insecticides are likely to be less persistent because they break down quickly (e.g., spinetoram) or become immobile in soil under field conditions because they bind to organic matter in contact (e.g., pyrethroid insecticides). Thus, as fewer effective older chemistries (e.g., organophosphate insecticides) are used against cabbage maggot because of use restrictions, delayed application of insecticide might be more critical.
For more details on this study, please read the published paper. http://cemonterey.ucanr.edu/files/248875.pdf
References
Joseph, S. V. 2014. Efficacy of at-planting and basal applications of insecticides on cabbage maggot in seeded-broccoli. Monterey County Crop Report. January/February 2010-2013. http://cemonterey.ucanr.edu/newsletters/i__b_ Monterey_County_Crop_Notes__b___i_50471.pdf
Joseph, S. V.,and J. Martinez. 2014. Incidence of cabbage maggot (Diptera: Anthomyiidae) infestation and plant damage in seeded brassica fields in California's Central Coast. Crop Prot. 62: 72-78.
Joseph, S. V., and J. Zarate. 2015. Comparing efficacy of insecticides against cabbage maggot (Diptera: Anthomyiidae) in the laboratory. Crop Prot. 77: 148-156.

- Author: Ian Grettenberger
- Author: Larry Godfrey
- Author: Richard Smith
- Author: Shimat Villanassery Joseph
Bagrada bug (Bagrada hilaris) is an invasive stink bug that was first observed in the Salinas Valley in October-November 2013. We started monitoring bagrada bug populations in non-crop habitat up and down the Valley starting in January 2015 and have continued to do so since then. We have seen bagrada bug populations beginning to develop on the weeds in spring and summer months. Weeds are clearly a key factor for bagrada bug populations in our region. While cruciferous crops are available year-round in the valley, stands of weeds are typically where populations really build up during early- and mid-summer (Fig 1).
In the Salinas Valley, shortpod mustard (Hirschfeldia incana, or summer mustard; Figs. 2 and 3) and perennial pepperweed (Lepidium latifolium; Figs. 4 and 5) appear to be the two most important weeds for buildup of bagrada bug populations. We have found bugs (in extremely high numbers) on perennial wall rocket (Diplotaxis tenuifolia), a weed commonly found along Hwy 101 between Chualar and Soledad, but this was very late in the year when temperatures were cooling and after the time bagrada bugs are typically problematic. These three weeds are non-native and invasive, just like the bagrada bug.
For our survey, we have been surveying a number of sites that cover the length of the Salinas Valley (Salinas, Chualar, Gonzales, Soledad, Greenfield, King City, San Lucas, San Ardo, and Watsonville). These sites contain a stand of at least one of these weed species, although we focused on pepperweed and/or shortpod mustard at the majority of the sites. Each month, we search for damage and bagrada bugs on the weeds at each site for twenty-minutes or until insects are counted on five plants of each species. When leaves are actively growing and not already damaged and worn out, damage is often easier to detect than insects (Figs. 6 and 7). This is similar to scouting in fields, where checking plants for fresh damage is the recommended method of scouting (Palumbo 2015). Unfortunately, fresh damage can be hard to find on older weeds because old damage obscures fresh damage, leaves are extremely tough, and bugs feed on stems or seed pods, so we primarily rely on detection of actual bugs.
When we surveyed near the end of June this year, we did not find high populations of bagrada bugs or damage, although they were present at some sites. We were left wondering what would happen with bagrada bug populations this year. After our July sample date, how 2016 populations compare with 2015 is still not clear, although we have started finding small populations of bagrada bug, which suggests we won't be having a bagrada bug-free year. If anything, there may be a slight delay in bagrada bug movement into fields, although this is fairly speculative. The spring rainfall this year was definitely greater than in 2015, although what effect of rainfall had on winter and spring bagrada bug populations is not clear. In 2016, we added a number of new sites, so some sites have already been surveyed during one period of population peaks, while others have only been surveyed since the beginning of this year.
At the three sites we surveyed in 2015 with perennial pepperweed, we did not see populations jump until our survey time point in late August. This year, we have not seen populations increase appreciably, although we will have to see what we find at the end of August to compare to 2015. At some of the sites with shortpod mustard that we surveyed in 2015, we had already found a fair number of bagrada bugs by this time last year, including some large populations of nymphs (up to 3 to 5 adults per plant and 18 to 57 nymphs per plant). This year, we haven't seen the same populations yet at those sites.
Next, we have the sites that have only been surveyed in 2016. While the initial sites were chosen based on prior issues with bagrada bugs in nearby fields, the new sites were not necessarily chosen based on the same conditions. We chose these new sites to improve the coverage of our survey. Each site also contains at least one of the three weed species we have focused on (shortpod mustard, perennial pepperweed, or perennial wall-rocket). A few sites were chosen knowing that bagrada bugs were present in 2015. A few of these sites have plenty of weeds available for bagrada bugs, but as of yet, we haven't seen any bugs. Some sites do have developing bagrada bug populations. We found what appears to be growing populations of bagrada bugs on both shortpod mustard and perennial pepperweed at one site near San Ardo on our last survey date (July 25th/26th). In the foothills near Gonzales, we found 1 to 26 bagrada adults and 0 to 22 nymphs on shortpod mustard plants. In addition, we were able to easily find bagrada bugs (0 to 8 adults, 0 to 3 nymphs) on shortpod mustard plants growing along US-101 between Greenfield and King City (Fig. 8). As many of you that have driven this stretch of road may know, there are a lot of weeds there. This means that a handful of bagrada bugs on each plant can quickly add up to large populations when summed over thousands of plants. As the weeds senesce, bagrada bugs will likely concentrate on still-green plants, although this patch can only support the bagrada bugs for so long with plants continuing to senesce.
As we see it, the issue is when these growing populations of bagrada bugs run out of food in the patches of weeds, and then go in search of new food sources. At this point, they can start finding their way into fields of cruciferous crop. This time of year, much of the shortpod mustard in the Valley is starting to set seed and will soon completely dry up if it has not already. In many locations, the vast majority of plants are almost dry. In the same localized area, some shortpod mustard plants will be completely senesced, while others are still green and flowering (see Figs. 9-11). Differences in growing conditions at a fairly small scale can therefore be very important. If all of the plants dry up, bagrada bugs will be forced to disperse, possibly triggering infestations in crop fields. If green plants remain, they may retain bagrada bugs, shifting the risk of infestation. Perennial pepperweed can persist late into the fall, but this is dependent on availability of water, damage by bagrada bugs, and disease pressure (pepperweed is often afflicted by white rust; Koike et al. 2011). The timing and severity of bagrada bug infestations in fields seems to be closely tied to what is happening on the weeds, so a better understanding of population dynamics on weeds will be needed to better predict an influx of bagrada bugs.
The next step will be to figure out how to incorporate bagrada bug populations in weeds into management and scouting plans. It is already common for PCAs to check weeds for bagrada bugs and we believe this to be a useful tactic. At this point, we suggest checking weeds on the edges of fields and any large patches of weeds within ~ 0.5 miles that seem to be likely sources of bagrada bugs. Consider which sides of crop fields often get infested the most or earliest and search for weeds on that side of the field. The dispersal ability of bagrada bugs has not been well characterized, but cases in which fields are colonized even when no cruciferous weeds or only bare ground is nearby show that bagrada bugs can move significant distances. While often seen walking, bagrada bugs readily fly, especially when surface temperatures are above 100° F based on preliminary observations. When perennial pepperweed is green and leaves are growing, new or recent damage should be readily apparent. Damage on “old growth” shortpod mustard is not always readily apparent, so finding bagrada bugs themselves is necessary. Adults are often found feeding on flowers, buds, or seed pods, so these are the best plant structures to scan. We believe bagrada bugs on weeds are mainly a threat to crop fields once the quality of these plants starts to decline, so pay attention to weed phenology and damage severity on weeds from both bagrada bugs and disease.
Management of weeds is another option to limit the population growth potential of bagrada bugs. Sanitation of both weeds and crops has been a recommended cultural management tactic in the pest's Old World range (Palumbo et al. 2016). By removing nearby weeds, you may be able to prevent an economically significant infestations. If weeds are managed, the timing will be important. For shortpod mustard, if it is possible to successfully manage weeds when they are still small, this may prevent build up of bagrada populations from the very beginning. However, resurgence of the weeds may happen with sufficient soil moisture early in the year. A more efficient tactic may be to manage shortpod mustard once it has bolted and is flowering later in the season (~ late April to May). This will help prevent resurgence of weeds but will still intercept bagrada bug populations before they have a chance to build to a significant degree. Managing weeds later and once bagrada bug populations have developed could push insects into crop fields. The timing of weed management could also be tied to the susceptibility of nearby crops, which could help if bagrada bugs are already present on the weeds. Adult bagrada bugs moving into a 40 day-old broccoli field because the weeds they were on were mowed or disced would likely not cause economic damage. However, managing weeds near a newly planted field could make matters worse and create a pest problem. Even if nearby weeds don't harbor bagrada bugs early in the season, they may serve as an intermediate bridge between the crop fields and weeds that are further away. Unfortunately, it is likely impossible to manage weeds far enough out from fields to eliminate the threat of bagrada bugs. The landscape context is important, but exercising control over the entire landscape is not possible. Perennial pepperweed is a trickier weed to deal with given its phenology and ability to rapidly re-sprout. These weed species are not about to disappear from the landscape in the Salinas Valley, so they will continue to play an important role for developing bagrada bug populations.
References
Koike, S. T., M. J. Sullivan, C. Southwick, C. Feng, and J. C. Correll. 2011. Characterization of white rust of perennial pepperweed caused by Albugo candida in California. Plant Disease 95:876.
Palumbo, J. C. 2015. Association between Bagrada hilaris density and feeding damage in broccoli- implications for pest management. Plant Health Progress 16:158–162.
Palumbo, J., T. Perring, J. Millar, and D. A. Reed. 2016. Biology, ecology, and management of an invasive stink bug, Bagrada hilaris, in North America. Annual Review of Entomology 61:453–473.





- Author: Shimat Villanassery Joseph
Cabbage maggot (Delia radicum) is a serious insect pest of Brassica crops such as broccoli and cauliflower in the Central Coast of California. These crops are grown throughout the year; as a result cabbage maggot problems persist year long.Cabbage maggot eggs are primarily laid in the soil around the crown area of the plant. A single female fly can lay 300 eggs under laboratory conditions. The eggs hatch within 2-3 days and the maggots feed on the taproot for up to three weeks and can destroy the root system of the plant. The maggots pupate in the soil surrounding the root system and emerge into flies within 2-4 weeks. Severe cabbage maggot feeding injury to the roots cause yellowing, stunting even plant death.
Control of cabbage maggot on Brassica crops primarily involves the use of soil applied organophosphate insecticides such as chlorpyrifos and diazinon. However, the persistent use of organophosphate insecticides has resulted in high concentrations of the insecticide residues in the water bodies posing risks to non-target organisms and public health through contaminated water. Currently, use of organophosphate insecticides is strictly regulated by California Department of Pesticide Regulation. There is therefore an urgent need to determine the efficacy of alternate insecticides for cabbage maggot control.
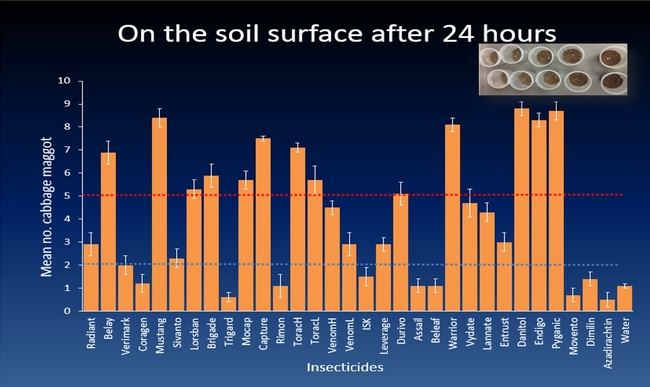
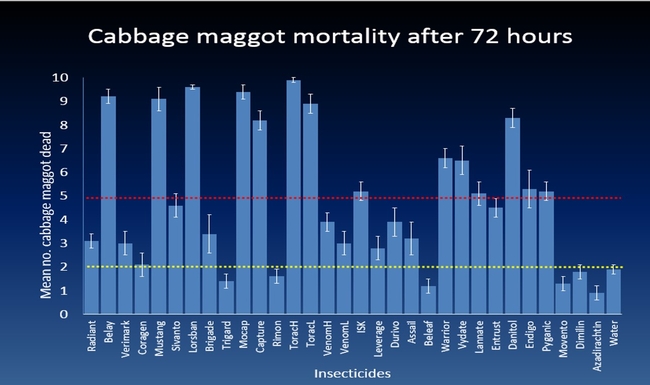
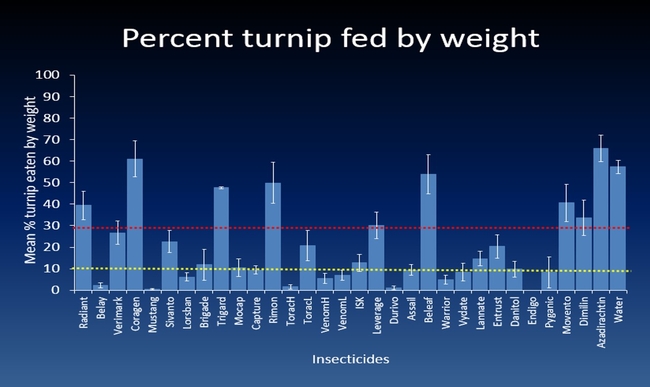
The efficacy of 29 insecticides was determined against cabbage maggot through a laboratory bioassay by exposing field collected maggots to insecticide treated soil immediately after application. Three parameters were used to evaluate efficacy (1) proportion of maggots on the soil surface after 24 h, (2) proportion of change in weight of turnip bait, and (3) dead maggots after 72 h. Based on the assays, 11 insecticides performed better and they were Mustang, Torac, Danitol, Belay, Capture, Warrior II, Lorsban, Mocap, Durivo, Pyganic and Vydate in the order of highest to lowest efficacy. Eight insecticides were selected based on superior efficacy to determine the length of residual activity on cabbage maggot larvae. The persistence of insecticide activity was greater with Capture, Torac and Belay than with other insecticides tested.
The mode of exposure of insecticides in this study was entirely by contact (through skin) and other modes of exposure such as ingestion (through mouth) or through respiratory holes (spiracles) were not investigated. Some of the insecticides tested in the study were insect growth regulators (IGRs) (Dimilin, Rimon, Trigard, and Aza-direct), which normally interfere with the growth and development of the insect and they showed a low efficacy against cabbage maggot larvae. Entrust (spinosad) showed a moderate efficacy possibly because the primary mode of exposure to Entrust is by ingestion. The diamide insecticides (Beleaf, Coragen and Verimark) have systemic activity as they move within the plant and likely away from the site of application. It is possible that the soil applied diamide insecticides are absorbed by the roots and translocated to the above ground plant parts with little effect on the feeding larvae in the tap roots.
This study was conducted under controlled conditions in the laboratory and the results may not be entirely consistent in field conditions. The Brassica fields in the California's Central Coast are profusely sprinkler irrigated up to three weeks after sowing to ensure uniform germination and proper establishment of plants. It is likely that applied insecticides are partially or completely leached out of the root zone area without providing anticipated maggot control. In this study, insecticides were drenched into the cup and none of the applied insecticide solution leached out. Therefore, it is likely that the insecticides were more effective in the laboratory assay than they would be in the field. Certain insecticides such as pyrethroids tend to bind to the soil organic matter. The organic matter in the California's Central Coast soils can be up to 4%, which could reduce the availability of soil applied pyrethroid insecticide to the root zone where cabbage maggot larvae typically colonize. In situations with poor insecticide spray coverage, invading cabbage maggot larvae are possibly exposed to no or sub-lethal doses of the soil applied insecticide and may be able to penetrate the soil and infest the roots. The air temperature in the field at the time of insecticide application may influence the efficacy of the applied insecticide. The efficacy of pyganic decreased as the temperature increased against onion maggot. This suggests that application of pyrethroid insecticides should be avoided during warmer periods of day.
Other field conditions that influence efficacy of insecticides are cabbage maggot incidence and frequency of invading cabbage maggot flies on Brassica crop in the Central Coast of California. The earliest peak of cabbage maggot infestation occur a month after sowing broccoli seeds and infestations can be continuous until harvest. Also, insecticides applied at sowing as a banded spray on the seed lines did not provide adequate cabbage maggot control based on the insecticide efficacy trials conducted in commercial broccoli fields. These findings suggest that delaying the insecticide application by 2-3 weeks after sowing is more likely to maximize maggot control. Because the cabbage maggot infestation can last several weeks, insecticides with extended persistence of efficacy would increase the value for cabbage maggot control. Overall, results show that Capture, Torac and Belay which performed effectively against cabbage maggot for a month after application. This indicates that insecticides used before the first peak of infestation may protect the younger stages of the Brassica plants allowing them to establish and tolerate milder cabbage maggot infestations thereafter.
In conclusion, 11 insecticides with high efficacy were identified for future investigation. Future studies will focus on determining the effects of application timing and delivery methods compatible with cabbage maggot incidence in both directly sown and transplanted Brassica crops in the Central Coast of California.
If you are interested in reading the details of this study, please click the link below to access the published article.





- Author: Shimat Villanassery Joseph
Bagrada bug (Bagrada hilaris) (Figure 1), an invasive stink bug species native to old world countries of southern Africa, Middle East and Asia, was first detected in North America from Los Angeles Co, California in 2008, and is now established in brassica crop production regions of central coast of California.
Bagrada bug prefers cruciferous hosts (Family: Brassicaceae) including broccoli, cauliflower, cabbage, kale, arugula, or collards. Also, bagrada bug could survive on cruciferous weeds such wild radish, London rocket, short pod mustard, and shepherd's purse, as well as the insectary plant, sweet alyssum. Mustard weed species are very common in ditches, roadsides, and along the edges of agricultural fields in the central coast of California. Mustard cover crops such as white mustard and Indian mustard in particular, could harbor bagrada bug populations, which are not often monitored for insect pests.
Damage to brassica crops varies but can be severe. Severe economic loss has been reported when injury occurred during early developmental stages of the crop such as cotyledon or < 4 leaves stages. Injury on leaves appears initially as small puncture marks, which turn into white patches as leaves expand. In broccoli and cauliflower, the economic injury occurs when the bug feeding kills the apical meristematic tissue of young seedling, which later results in “multiple heads” or sometimes “blind head” (without a head). Severe feeding also depletes the nutrient reserves of the plant leading to desiccation or wilting. Other leafy brassica crops such as mizuna mustard or arugula are especially at risk from direct feeding damage of bagrada bug.
At this time, no baseline information on bagrada bug abundance, seasonal activity, or generations has been established in the coastal brassica crop production regions in California. Limited information exists to determine threshold based pest management decisions for bagrada bug. Monitoringfor bagrada bug during mid-day hours might increase the probability of finding them as the bugs typically hide and stay in the cracks and crevices or on the underside of leaves when the temperature is cooler. Strong attraction to specific color could be utilized when developing selective traps for monitoring bagrada bug in the field. Yellow traps are most commonly used for monitoring true bugs such as false chinch bugs, plant bugs, psyllids or stinkbugs. Yellow pyramid traps were used to monitor brown and dusky stink bugs.
This study was conducted to determine the influence of trap color on catches of bagrada bug. In fall 2013, an organically-managed broccoli field in San Ardo, CA was chosen for the study. Planting sweet alyssum is a common practice among organic growers in the Salinas Valley to harbor beneficial insects such as syrphid flies (hover flies) and lady beetles within the cole crop field. Strips of sweet alyssum were planted within the broccoli field. Cross-vane traps were constructed using corrugated plastic sheets and were painted with white, yellow, red, purple, or black paint (Figure 2). All the trap-base containers used in this study were light green colored. Traps were placed within the sweet alyssum plants (Figure 3).Bug captures were recorded for up to 5 weeks.
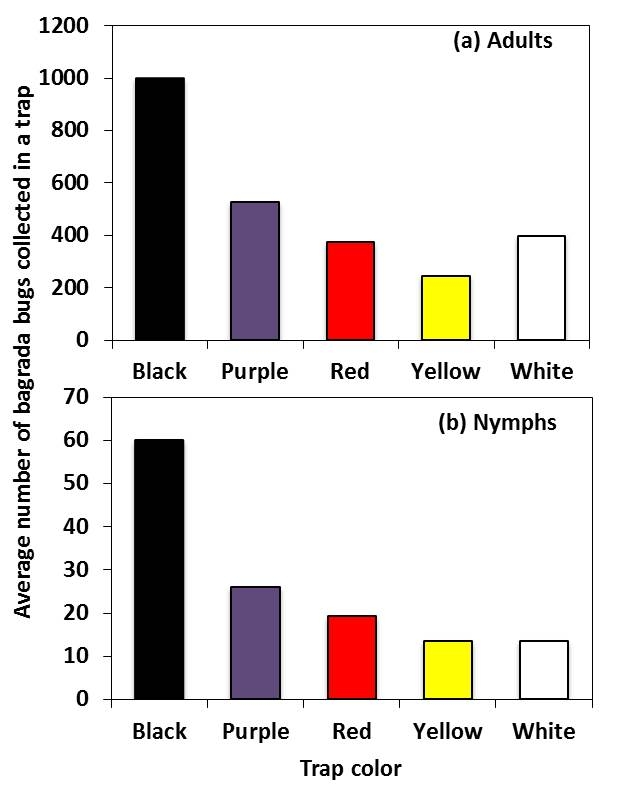
In total, 10,654 bagrada bug were captured and were predominantly adults (95%). Adult bagrada bug captures were influenced by trap color (Figure 4a). Black traps captured almost twice the number of adult bagrada bug than purple traps; however, adult catches were similar among purple, white or red traps. Catches of adults were higher in purple than in yellow traps. Similarly, the numbers of bagrada bug nymphs collected among various colored-traps were higher in black traps than other colored traps (Figure 4b). Future research will investigate the potential use of color preference with plant host odor to develop traps for field monitoring of bagrada bug
For those of you, who are interested to read more on it, please find the published article after clicking the link (below) and feel free to contact me (Shimat Joseph) at svjoseph@ucanr.edu or 831 759 7359.
http://cemonterey.ucanr.edu/files/195965.pdf