- Author: Steven T. Koike
- Author: Mark Bolda
While the recently detected charcoal rot disease (caused by the soilborne fungus Macrophomina phaseolina) was causing collapse of strawberry plants from various parts of California, a second soilborne issue was simultaneously affecting other fields. Fusarium wilt was first confirmed on California strawberry in 2006. Initially found in Ventura County, Fusarium wilt is now present on strawberry in Santa Barbara and Monterey counties. The spread of Fusarium wilt in the state along with the increasing problems with Macrophomina pose long term threats to the strawberry industry which at present does not have satisfactory plant resistance to both of these pathogens and which is facing a changing future without traditional fumigant products.
Symptoms of Fusarium wilt in strawberry consist of wilting of foliage, plant stunting, and drying and death of foliage (Figure 1). When plant crowns are cut open, internal vascular and cortex tissues are dark to orange brown (Figure 2). Disease is often most severe if the infected plant is subject to stresses such as weather extremes, water stress (shortage of water), poor soil conditions, or heavy fruit loads. In locations where the disease has occurred for more than one season, the patches can be quite large and appear to have spread from the initial problem area (Figure 3). Such patterns are consistent with the spread of a soilborne pathogen. It is noteworthy that in these cases we have never isolated other important, well known pathogens such as Colletotrichum, Phytophthora, or Verticillium. However, it is important to note that Fusarium wilt symptoms are virtually identical to those caused by charcoal rot. To complicate matters further, in some fields we have found both Fusarium and Macrophomina infecting the same plant. This overlap of symptoms means that growers and field personnel should have plants tested by a pathology lab in order to confirm which soilborne disease they are encountering.
Fusarium wilt is caused by the fungus Fusarium oxysporum f. sp. fragariae. This pathogen is host specific to strawberry and can only infect this crop. The fungus survives in the soil for long periods by producing resilient, microscopic structures called chlamydospores (Figure 4). The development of Fusarium wilt has also been associated with changes in the practices of pre-plant soil fumigation. The fungus is spread within and between fields mostly by the transport of contaminated soil during soil tillage and preparation operations.
Current management strategies involve the following: (1) Crop rotation. Do not plant strawberry in fields having a known history of the problem and avoid back-to-back strawberry plantings in infested locations. (2) Pre-plant fumigation. This remains a useful tool for managing Fusarium and the other soilborne pests, even though bed-applied fumigants may not provide complete control. (3) Avoid stressing the plants. Stress will hasten the development and increase the severity of symptoms, so use appropriate growing and irrigation practices to reduce stress. Note, however, that even in the absence of stress, infected plants will eventually develop the disease. (4) Sanitation. Growers with Fusarium infested fields need to be concerned with limiting the spread of the fungus from infested to clean fields.
Figure 1. Fusarium wilt results in the collapse and death of strawberry plants.
Figure 2. Internal crown tissue of strawberry infected with Fusarium will show a dark to orange brown discoloration.
Figure 3. Fusarium wilt can affect large portions of a field and cause significant dieback.
Figure 4. Microscopic spherical structures called chlamydospores enable the Fusarium pathogen to survive in the soil. 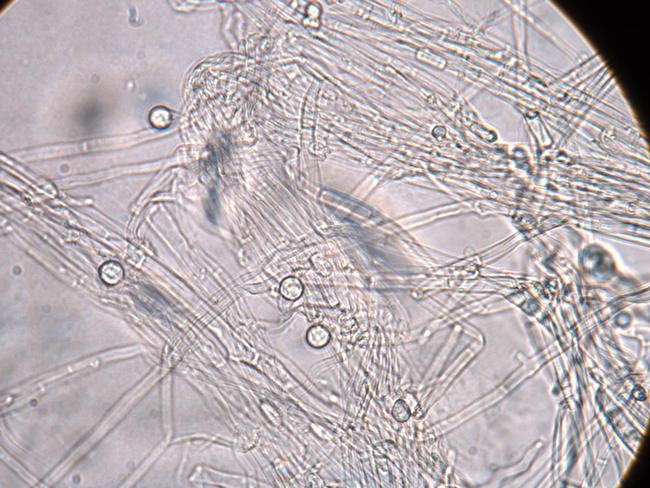
- Author: Steven T. Koike
- Author: Mark Bolda
Beginning at least as early as 2005 and continuing through 2013, collapsing strawberry plants from various parts of California have been associated with the soilborne fungus Macrophomina phaseolina. The disease, called charcoal rot, appears to be the most important current concern for the industry due to its steady increase over this period of time. Each year finds additional new fields infested, and the disease has now been found in all of the major strawberry producing counties in the state. In 2005-2006, charcoal rot was restricted to southern California in Orange and Ventura counties. Most recently this disease has been confirmed in Santa Barbara, Monterey, Santa Cruz, and Santa Clara counties. The spread of Macrophomina to new fields and counties portends that charcoal rot may be a long term threat to the industry which at present does not have satisfactory plant resistance with which to combat the pathogen.
Symptoms of Macrophomina infection in strawberry consist of wilting of foliage, plant stunting, and drying and death of older leaves, with the central youngest leaves often remaining green and alive. Plants can eventually collapse and die (Figure 1). When plant crowns are cut open, internal vascular and cortex tissues are dark to orange brown (Figure 2). Disease is often most severe if the infected plant is subject to stresses such as weather extremes, water stress (shortage of water), poor soil conditions, or heavy fruit loads. In locations where the disease has occurred for more than one season, the patches can be quite large and appear to have spread from the initial problem area (Figure 3). Such patterns are consistent with the spread of a soilborne pathogen. It is noteworthy that in these cases we have never isolated other important, well known pathogens such as Colletotrichum, Phytophthora, or Verticillium. However, it is important to note that another recently described disease, Fusarium wilt, is also occurring in the same regions; symptoms of Fusarium wilt are identical to those caused by charcoal rot.
Macrophomina produces numerous tiny, black, irregularly shaped microsclerotia (Figure 4). These microsclerotia are survival structures that allow the fungus to persist for extended periods in the soil. The fungus is spread within and between fields mostly by the transport of contaminated soil during soil tillage and preparation operations. Spread of Macrophomina in strawberry fields deals with the same issue of field sanitation that concerns growers of many other commodities. Verticillium wilt (lettuce, strawberry, pepper), clubroot (broccoli, cauliflower), Fusarium wilt (lettuce), Fusarium yellows (celery), and lettuce dieback disease (lettuce) are all problems caused by soilborne pathogens that are spread in infested soil.
Current management strategies involve the following: (1) Crop rotation. Do not plant strawberry in fields having a known history of the problem and avoid back-to-back strawberry plantings in infested locations. (2) Pre-plant fumigation. This remains a useful tool for managing Macrophomina and the other soilborne pests, even though bed-applied fumigants may not provide complete control. (3) Avoid stressing the plants. Stress will hasten the development and increase the severity of symptoms, so use appropriate growing and irrigation practices to reduce stress. Note, however, that even in the absence of stress, infected plants will eventually develop the disease. (4) Sanitation. Growers with Macrophomina infested fields need to be concerned with limiting the spread of the fungus from infested to clean fields.
Figure 1. Charcoal rot results in the collapse and death of strawberry plants.
Figure 2. Internal crown tissue of strawberry infected with Macrophomina will show a dark to orange brown discoloration.
Figure 3. Charcoal rot can affect large portions of a field and cause significant dieback.
Figure 4. Tiny, black microsclerotia enable the Macrophomina pathogen to survive in the soil.
- Author: Richard Smith
- Author: Steven T. Koike
In the last year we have been called out to look at lettuce fields showing uneven growth. The fields looked like they were not growing normally due to a cultural practice. The lettuce plants in the field vary in size with normal plants interspersed with stunted plants (Photos 1 and 2). The patterns in the field do not fit with sprinkler patterns or mechanical issues because affected plants are more randomly dispersed. The pattern may fit with soil insect issues, but no soil insects were present in the affected fields. Sometimes stunted plants were more prevalent in areas that might have poor drainage. The randomness of affected plants is similar to some of the nitrogen toxicity issues that we have detected in some fields (http://ucanr.org/blogs/blogcore/postdetail.cfm?postnum=4931). In some cases the taproot of the plant is missing (a typical symptom of nitrogen toxicity), but taproots are not consistently missing and therefore the symptoms do not fit nitrogen toxicity. Upon close examination of the root system, small necrotic sections of the root are present and we were able to confirm that the lettuce was affected by black root rot disease.
Root symptoms of black root rot of lettuce consist of numerous dark brown to black bands and lesions on small feeder and larger secondary roots (Photos 3 and 4). Internal vascular discoloration (as seen with Verticillium wilt) is absent. If severe infections occur early in the crop cycle, the lettuce seedlings will have damaged taproots and experience delayed growth, resulting in stunted, small plants that otherwise may look healthy. The stunting is usually most evident from rosette stage through crop maturity. In advanced cases, the lower leaves of the plant may turn yellow or even brown. In the Salinas Valley, black root rot has been seen to cause stunting and resulting crop loss on both iceberg and romaine cultivars. Black root rot of lettuce has been known for many years in Australia and other regions. The disease was first documented on lettuce in California in 2005. Our diagnostic lab has periodically received black root rot cases since then.
Black root rot is caused by the soilborne fungus Thielaviopsis basicola. The fungus makes two types of spores, with one type being a thick walled spore (Photo 5) that resists weathering and enables the fungus to survive for long periods in the soil. This pathogen is reported to have a broad host range that includes the following vegetable crops: bean, beet, carrot, garlic, lettuce, onion, pea, pepper, tomato. In coastal California we have not found black root rot on most of these crops; black root rot disease has been confirmed only for lettuce and tomato.
Differences in strains or isolates of T. basicola may exist. For example, researchers found that when inoculated onto lettuce and bean, T. basicola isolates from lettuce were very aggressive and caused significant damage. However, the same lettuce isolates caused very little damage to other known hosts. Such research indicates that T. basicola may consist of isolates having some host preferences.
Industry-wide, black root rot of lettuce is a very minor problem that apparently only affects a few fields. However, growers and PCAs should be aware of this disease and notify Farm Advisors if black root rot becomes more common. For other crops, black root rot is managed by a combination of resistant cultivars, crop rotation to non-hosts, sanitation practices so as to not spread contaminated soil/mud to uninfested fields, and application of fungicides to seedlings.

Photo 1. Typical pattern of Thielaviopsis basicola infection in lettuce.

Photo 2. Stunted lettuce affected with black root rot (on left)

Photo 3: Diseased roots of lettuce affected by black root rot.

Photo 4. Close-up of black root rot lesion on fine feeder roots of lettuce.
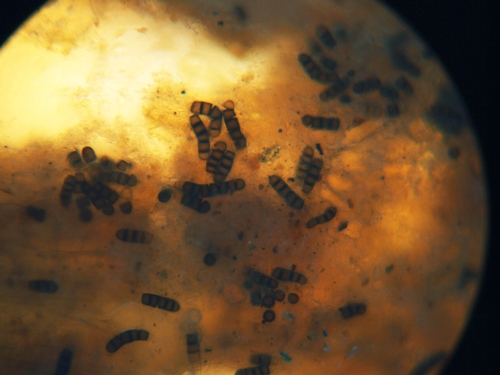
Photo 5: Dark conidia of Thielaviopsis basicola forming on lettuce root.
- Author: Jim Correll, University of Arkansas Steven Koike, University of California Cooperative Extension
Another new race, the 13th, of the downy mildew pathogen (Peronospora farinosa f. sp. spinaciae) of spinach has been found and documented. First identified in January 2010 from spinach in Holtville, California, this race breaks the resistance of several important cultivars. The isolate was initially designated as UA0510C and was characterized with a standard set of differential varieties. Isolates apparently identical to UA0510C have been found in an increasing number of locations throughout California in 2010 and 2011. After careful evaluation of the significance of this development to the spinach industry, the International Working Group on Peronospora (IWGP) has designated this isolate as race Pfs 13. The IWGP is located in The Netherlands and is administered by Plantum NL.
Race Pfs 13 poses a threat to the spinach industry because it is particularly well-adapted to modern hybrids with resistance to races 1-12. The appearance of a new race is not unexpected because hybrids with resistance to races 1-12 have been widely planted over the past few years. Similar developments have taken place when races Pfs 5 (1996), Pfs 6 (1998), Pfs 7 (1999), Pfs 8 and 10 (2004), Pfs 11 (2009), and Pfs 12 (2009) were identified and named. The occurrence of Pfs 13 will clearly encourage the industry to develop and use new spinach cultivars having resistance to races 1-13. A history of the detection of the various spinach downy mildew races is presented in Table 1.
A collaboration of researchers with the IWGP, University of Arkansas (Correll), and University of California (Koike) is monitoring the development of new races of spinach downy mildew on a global scale by collecting and testing suspected new isolates. In this way it is hoped that research findings and conclusions will be agreed upon and better communicated between the seed industry, spinach growers, and other interested parties. For California and Arizona, the Correll-Koike team will continue to receive and test spinach downy mildew samples for growers, pest control advisors, and seed companies. Industry is encouraged to continue to submit downy mildew outbreak samples to Correll-Koike, as such samples facilitate the discovery of additional new races. The Correll-Koike research is made possible by support from the California Leafy Greens Research Board and by active participation by the agricultural industries in California and Arizona.
For more information on this subject you can contact Steven Koike (stkoike@ucdavis.edu), Jim Correll (jcorrell@uark.edu), Diederik Smilde (d.smilde@naktuinbouw.nl), or IWGP chairperson Jan de Visser (JandeVisser@popvriendseeds.nl).
Table 1. Races of spinach downy mildew and year of detection
| Year | Race |
| 1824 | 1 |
| 1958 | 2 |
| 1976 | 3 |
| 1990 | 4 |
| 1996 | 5 |
| 1998 | 6 |
| 1999 | 7 |
| 2004 | 8 |
| ... | (9)* |
| 2004 | 10 |
| 2008 | 11 |
| 2009 | 12 |
| 2010 | 13 |
*One time detection only

Downy mildew of spinach is the most important disease on this crop and results in quality and yield losses.
- Author: Steven T. Koike, Plant Pathology Farm Advisor
Speck on coastal tomato. In summer (May through July) 2011, bacterial speck disease of tomato is commonly found in a number of fields in coastal tomato-growing areas in Santa Clara and Monterey counties. No doubt the periodic and prolonged rains earlier this year are a major factor in this unusual development. Symptoms consist of dark brown to almost black spots on leaves and sometimes stems. Leaf spots can be circular or angular in shape and individual spots are generally smaller than ¼ inch in diameter. Spots are visible from both top and bottom sides of the infected leaf and are sometimes surrounded by a yellow halo. Leaf spots can merge together and result in the death of large areas of the leaf. On occasion the leaf spots may be located along the edges of the leaves and result in elongated lesions. Stems and petioles also can develop dark brown to black spots that are irregular in shape but tend to be slightly elongated along the axis of the stem. If the pathogen is splashed onto the fruit, disease symptoms on green fruit will consist of small (generally less than 1/8 inch in diameter), slightly raised, superficial black lesions or specks. Such specks usually are not surrounded by haloes.
Confusion of symptoms. Symptoms of bacterial speck on tomato foliage can closely resemble symptoms from the other bacterial leaf spot disease of tomato, bacterial spot (caused by Xanthomonas campestris pv. vesicatoria). In addition, Tomato spotted wilt virus may infect tomato and also cause circular to irregularly shaped, dark brown spots on leaves, again resembling bacterial speck. For all three of these problems, precise confirmation of the pathogen will require laboratory testing.
The speck pathogen. Bacterial speck is caused by the pathogenic bacterium Pseudomonas syringae pv. tomato. Strains of this pathogen are thought to be host specific to tomato, and two distinct races (races 0 and 1) have been documented. This pathogen is seedborne. Bacterial speck on the coastal tomato crop is usually quite minor in severity; once rains stop and if overhead sprinklers are not used, the disease is rarely seen in the summer.
Disease development. Primary inoculum can come from infested seed, diseased plant debris in the soil, or infected volunteer tomato plants. Infested seed is a particularly important inoculum source; if infested seed is used to produce transplants, these plants are often grown under greenhouse conditions that encourage disease development and spread. The practice of using overhead sprinkler irrigation (in the greenhouse or in the field) can significantly spread the pathogen. Once diseased transplants are in the field, the pathogen can be spread plant-to-plant via splashing water (rain, sprinkler irrigation), contaminated tools and implements, and worker’s hands. Disease development is favored by high humidity (greater than 80% relativity humidity), free moisture, and relatively cool temperatures. Again, the rains that occurred through June 2011 certainly encouraged the development of this problem. Interestingly, for all the numerous tomato samples tested by the UCCE diagnostic lab in Salinas, all samples were positive for speck only; bacterial spot disease was not detected at all thus far in 2011.
Control. Obtain and plant high quality seed that does not have detectable, economically important levels of P. syringae pv. tomato. Use a hot water seed treatment or treat seed with hydrochloric acid, calcium hypochlorite, or other recommended materials. Seed treatments must be applied carefully because hot water treatments can reduce seed viability and germination. Avoid using overhead sprinkler irrigation in the field. Applying preventative spray applications to transplants still in trays or to field planted crops may be appropriate. Before using any pesticides, check with your local Agricultural Commissioner's Office and consult product labels for current status of product registration, restrictions, and use information.

Photo 1. Oval to rectangular leaf spots caused by bacterial speck disease of tomato.

Photo 2. Angular spots on leaf margin caused by bacterial speck disease of tomato.

Photo 3. Angular spots on leaf margin caused by bacterial speck disease of tomato.

Photo 4. Elongated stem lesions on transplants caused by bacterial speck disease of tomato.

Photo 5. Fruit speck symptom caused by bacterial speck disease of tomato.






