- Posted By: Chris M. Webb
- Written by: W. Thomas Lanini
In recent years, several organic herbicide products have appeared on the market. These include Weed Pharm (20% ace c acid), C Cide (5% citric acid), GreenMatch (55% d limonene), Matratec (50% clove oil), WeedZap (45% clove oil + 45% cinnamon oil), and GreenMatch EX (50% lemongrass oil), among others. These products are all contact type herbicides and will damage any green vegeta on they contact, though they are safe as directed sprays against woody stems and trunks. These herbicides kill weeds that have emerged, but have no residual activity on those emerging subsequently. Additionally, these herbicides can burn back the tops of perennial weeds, but perennial weeds recover quickly.
These products are effective in controlling weeds when the weeds are small and the environmental conditions are op mum. In a recent study, we found that weeds in the cotyledon or first true leaf stage were much easier to control than older weeds (Tables 1 and 2). Broadleaf weeds were also found to be easier to control than grasses, possibly due to the location of the growing point (at or below the soil surface for grasses), or the orientation of the leaves (horizontal for most broadleaf weeds) (Tables 1 and 2).
Organic herbicides only kill contacted tissue; thus, good coverage is essential. In test comparing various spray volumes and product concentrations, we found that high concentrations at low spray volumes (20% concentration in 35 gallons per acre) were less effective than lower concentrations at high spray volumes (10% concentration in 70 gallons per acre). Applying these materials through a green sprayer (only living plants are treated), can reduce the amount of material and the overall cost (http://www.ntechindustries.com/weedseeker-home.html). Adding an organically acceptable adjuvant has resulted in improved control. Among the organic adjuvants tested thus far, Natural wet, Nu Film P, Nu Film 17, and Silwet ECO spreader have performed the best. The Silwet ECO spreader is an organic silicone adjuvant which works very well on most broadleaf weeds, but tends to roll o of grass weeds. The Natural wet, Nu Film 17 and Nu Film P work well for both broad leaf and grass weeds. Although the recommended rates of these adjuvants is 0.25 % v/v, we have found that increasing the adjuvant concentration up to 1% v/v o en leads to improved weed control, possibly due to better coverage. Work continues in this area, as manufacturers continue to develop more organic adjuvants. Because organic herbicides lack residual activity, repeat applications will be needed to control new flushes of weeds.
Temperature and sunlight have both been suggested as factors affecting organic herbicide efficacy. In several field studies, we have observed that organic herbicides work better when temperatures are above 75F. Weed Pharm (acetic acid) is the exception, working well at temperatures as low as 55F. Sunlight has also been suggested as an important factor for effective weed control. Anecdotal reports indicate that control is better in full sunlight. However, in a greenhouse test using shade cloth to block 70% of the light, it was found that weed control with WeedZap improved in shaded conditions (Table 3). The greenhouse temperature was around 80F. It may be that under warm temperatures, sunlight is less of a factor.
Organic herbicides are expensive at this time and may not be affordable for commercial crop producti on. Because these materials lack residual activity, repeat applications will be needed to control perennial weeds or new flushes of weed seedlings. Finally, approval by one's organic certifier should also be checked in advance as use of such alternative herbicides is not cleared by all agencies.
Review tables below...
(Table 1. Broadleaf (pigweed and black nightshade) weed control (% control at 15 days a er treatment), when treated 12, 19, or 26 days after emergence.
|
Weed |
age |
||
|
|
12 Days old |
19 days old |
26 days old |
|
GreenMatch Ex 15% |
89 |
11 |
0 |
|
GreenMatch 15% |
83 |
96 |
17 |
|
Matran 15% |
88 |
28 |
0 |
|
Ace c acid 20% |
61 |
11 |
17 |
|
WeedZap 10% |
100 |
33 |
38 |
|
Untreated |
0 |
0 |
0 |
Table 2. Grass (Barnyardgrass and crabgrass) weed control (% control at 15 days after treatment), when treated 12, 19, or 26 days after emergence.
|
Weed |
age |
||
|
|
12 Days old |
19 days old |
26 days old |
|
GreenMatch Ex 15% |
25 |
19 |
8 |
|
GreenMatch 15% |
42 |
42 |
0 |
|
Matran 15% |
25 |
17 |
0 |
|
Ace c acid 20% |
25 |
0 |
0 |
|
WeedZap 10% |
0 |
11 |
0 |
|
Untreated |
0 |
0 |
0 |
| Table 3. Weed control with WeedZap (10% v/v) in relation to adjuvant, spray volumne and light levels. Plants grown in the greenhouse in either open conditions or under shade cloth, which reduced light by 70%. |
|
||||
|
Pigweed control (%) |
Mustard control (%) |
|
|||
|
|
Sun |
Shade |
Sun |
Shade |
|
|
WeedZap + 0.1%v/v Eco Silwet (10 gpa) |
31.7 |
93.3 |
26.7 |
35.0 |
|
|
WeedZap + 0.5%v/v Eco Silwet (10 gpa) |
31.7 |
48.3 |
43.3 |
71.7 |
|
|
WeedZap + 0.5%v/v Natural Wet (70 gpa) |
26.7 |
94.7 |
26.7 |
30.0 |
|
|
Untreated |
0.0 |
0.0 |
0.0 |
0.0 |
|
|
LSD.05* |
5.7 |
11.5 |
|
|
|
* Values for comparing any two means. Pigweed and mustard were each analyzed separately.

- Posted By: Jeannette E. Warnert
- Written by: Franz Niederholzer, UC Farm Advisor, Sutter/Yuba Counties and Rhonda Smith, UC Farm Advisor, Sonoma Co.
Agricultural spray adjuvants are materials added to the spray tank when loading the sprayer. They include products classified as activator adjuvants and marketed as wetters/spreaders, stickers, humectants, and/or penetrators. Activator adjuvants are marketed to improve the performance of pesticides and foliar fertilizers.
Activator adjuvants can have a place in tree (and vine) crop sprays, but matching the material to the job can be tricky. A bad match can lead to minor or major losses to the grower. Minor losses can result from excess spreading and pesticide runoff from the target plant. Phytotoxicity can cause major damage.
This article describes ingredients and functions of activator adjuvants commonly sprayed on tree and vine crops. Suggestions regarding activator adjuvant selection are offered. Growers must make their own activator adjuvant use decisions based on experience, particular needs, and risk tolerance.
Do I need to add an activator adjuvant?
Read and follow the specific instructions on the label. If the pesticide or foliar fertilizer label indicates the product should be used with certain types or brand of adjuvant(s), that’s what you need to use.
Do I want to add an activator adjuvant?
If the label includes phrases such as "use of an adjuvant may improve results" or “complete coverage is needed for best results” then you may want to look into selecting and using an appropriate activator adjuvant. Before proceeding with use of an activator adjuvant, first look at your existing spray program. Are you already doing the best spray job you can? Good spray coverage begins with proper sprayer calibration and set up. Is your sprayer calibration dialed in for different stages of canopy development?
Optimum sprayer set up – gallons of spray per acre, ground speed, fan output, and nozzle selection/arrangement-- changes from dormant to bloom to early growing season to preharvest sprays. Adjusting your sprayer to best match orchard and vineyard conditions at each general stage in canopy development is the foundation of an effective, efficient spray program. An activator adjuvant will not make up forexcessive tractor speed, poor nozzle arrangement and/or worn nozzles. Your money is best spent first dialing in your sprayer(s) for the whole season, before considering an extra material in the tank (that is not required on the label). If you have your sprayer(s) dialed in for each orchard and stage of growth, now is the time to say “OK, I want to think about a little extra boost to my spray job”.
Which activator adjuvant properties do I want?
First, know the properties of the pesticide you will use. Does it work on the plant surface or inside the plant? This is a key point in selecting adjuvants. Here is a quick review of the main classifications and characteristics of activator adjuvants as they currently appear in the field. Note: Certain products can provide more than one adjuvant property – that can be beneficial in the field. For example, non-ionic surfactants can work as surfactants and penetrators, depending on use rate.
Wetters/spreaders: These materials contain surfactants that decrease the contact angle and increase the spreading of the spray droplet on the target. High rates of wetters/spreaders may also increase penetration of pesticide into the target tissue (leaves or fruit), potentially causing phytotoxicity. Excessive spreading of pesticide spray solution and runoff from the target may result when using a new or higher rate of spreader -- especially when using silicon “super-spreaders”. Test new combinations of spreader material(s) and spray volume before regular use. Spray volume per acre or adjuvant use rate will probably have to be reduced if a labeled rate of adjuvant provides excessive spreading.
To check for excessive spreading, place alength of black plastic sheeting under several trees or vines in a row. Secure the plastic with spikes, wire staples, and/or weights. Spray the new adjuvant and pesticide combination using your current sprayer set up. Reenter the field right after spraying, wearing appropriate PPE, and evaluate coverage. If material is pooling at the lower portion of leaves and/or fruit, excessive spreading is occurring. Check to see if pooling is occurring only in a certain area(s) of the canopy or throughout the canopy. If more spray solution is landing on the black plastic tarp under the trees/vines than between them, then runoff is occurring. [Some ground deposit should be expected from standard airblast sprayer use.]
Compare the results of your adjuvant test with a similar application of your current pesticide/adjuvant combination on another portion of the row. If there is no pooling or runoff with the new adjuvant in the tank, you can use the adjuvant with confidence. A lack of pooling or run off with the new adjuvant also might mean that your old sprayer setup and tank mix didn’t deliver adequate coverage. If the test with the new adjuvant showed pooling on leaves and/or runoff on the ground, you have several choices.
- You can reduce spray volume per acre by replacing some or all nozzles with smaller nozzle sizes on the sprayer in an effort to reduce overspreading. If you saw overspreading on some portions of the canopy, but not others, reduce nozzle size only on the part of the spray boom that targets the over-sprayed part of the canopy. Recheck spray coverage if nozzling changes were made.
- Reduce the adjuvant rate and recheck coverage/spreading.
- You can just go back to your established program without the new adjuvant. What’s the “best” course of action? That depends on your farming operation.Reducing spray volume per acre means more ground covered per full spray tank – a potential time and cost savings. If spraying is done during the heat of the day in hot, dry climate, spray water evaporation is a major issue and it may be best to keep the higher spray volume and reduce the spreader rate or eliminate it entirely. Checking coverage and overspreading allows you to make the best decision possible; avoid damage and, hopefully, save money. All farming operations are different. Make the choice that best fits your farm.
Stickers: These adjuvants can increase the retention time of the pesticide on the leaf and reduce rain wash off. They may limit movement of systemic pesticides into the plant, and are probably most beneficial when used with protectant materials (cover sprays). Do you overhead irrigate? Is there rain on the horizon? If you answer yes to either one of these questions, you may benefit from using a sticker.
Humectants: Under low humidity conditions humectants can help reduce spray droplet evaporation before and after deposition on the plant. This is especially valuable when small droplets and/or materials that must be absorbed into the plant (systemic pesticides, PGRs, nutrients, etc.) are used in the summer under high temperature and low relative humidity conditions.
Penetrators: Frequently used with herbicides, these products include oils (petroleum, vegetable, or modified vegetable oils) and non-ionic surfactants used at higher rates. In crop sprays, penetrators can be used to increase absorption of systemic pesticides (for example, oil with Agri-Mek) as well as translaminar materials. Penetrator adjuvants should be used with caution oravoided entirely with surface active pesticides such as cover sprays or else phyto may result. Finally, some penetrators can increase the rain-fastness of some pesticides.
Which adjuvant material should I select?
Use a product intended for crop spraying. Many activator adjuvants were developed and intended for use with herbicides. Products that are advertised for use with plant growth regulators should have a higher chance of crop safety compared with those that don't. This is still no guarantee of a phyto-free application. Ask for help from your PCA or the adjuvant manufacturer’s sales rep. How much do they know about the particular activator adjuvant in the spray mix you are planning? Can they show you the kind of information on a single product similar to what you can find at: http://www.ast-us.com? (This website is intended as an example, not an endorsement of the web pages it contains including specific adjuvants.)
Will the adjuvant I selected work in the spray I’m planning?
If you choose to use an adjuvant that is not specifically listed on the pesticide or foliar fertilizer label, jar test the planned spray solution first. Use the same spray water source. Include all leaf feeds, other adjuvants, and pesticide(s) that you plan to put in the spray tank. Do this before tank mixing these materials. A lot of time and money rides on effective pesticide application. Do your homework before the spray tank is filled and you will be well on your way to solid results.

- Written by: Craig Kallsen
University of California (UC) researchers and private industry consultants have invested much effort in correlating optimal citrus tree growth, fruit quality and yield to concentrations of necessary plant nutrients in citrus (especially orange) leaf tissue. The grower can remove much of the guesswork of fertilization by adhering to UC recommendations of critical levels of nutrients in the tissues of appropriately sampled leaves. Optimal values for elements important in plant nutrition are presented on a dry-weight basis in Table 1. Adding them in appropriate rates by broadcasting to the soil, fertigating through the irrigation system or spraying them foliarly may correct concentrations of nutrients in the deficient or low range. Compared to the cost of fertilizers, and the loss of fruit yield and quality that can occur as a result of nutrient deficiencies or excesses, leaf tissue analysis is a bargain. At a minimum, the grower should monitor the nitrogen status of the grove through tissue sampling on an annual basis.
Leaves of the spring flush are sampled during the time period from about August 15 through October 15. Pick healthy, undamaged leaves that are 4-6 months old on non-fruiting branches. Select leaves that reflect the average size leaf for the spring flush and do not pick the terminal leaf of a branch. Typically 75 to 100 leaves from a uniform 20- acre block of citrus are sufficient for testing. Generally, the sampler will walk diagonally across the area to be sampled, and randomly pick leaves, one per tree. Leaves should be taken so that the final sample includes roughly the same number of leaves from each of the four quadrants of the tree canopy. Values in Table 1 will not reflect the nutritional status of the orchard if these sampling guidelines are not followed. Typically, citrus is able to store considerable quantities of nutrients in the tree. Sampling leaves from trees more frequently than once a year in the fall is usually unnecessary. A single annual sample in the fall provides ample time for detecting and correcting developing deficiencies.
Table 1. Mineral nutrition standards for leaves from mature orange trees based on dry-weight concentration of elements in 4 to 7 month old spring flush leaves from non-fruiting branch terminals.
|
element |
unit |
deficiency |
low |
optimum |
high |
excess |
|
|
|
|
|
|
|
|
|
N |
% |
2.2 |
2.2-2.4 |
2.5-2.7 |
2.7-2.8 |
3.0 |
|
P |
% |
0.9 |
0.9-0.11 |
0.12-0.16 |
0.17-0.29 |
0.3 |
|
K (Calif.*) |
% |
0.40 |
0.40-0.69 |
0.70-1.09 |
1.1-2.0 |
2.3 |
|
K (Florida*) |
% |
0.7 |
0.7-1.1 |
1.2-1.7 |
1.8-2.3 |
2.4 |
|
Ca |
% |
1.5 |
1.6-2.9 |
3.0-5.5 |
5.6-6.9 |
7.0 |
|
Mg |
% |
0.16 |
0.16-0.25 |
0.26-0.6 |
0.7-1.1 |
1.2 |
|
S |
% |
0.14 |
0.14-0.19 |
0.2-0.3 |
0.4-0.5 |
0.6 |
|
Cl |
% |
? |
? |
<0.03 |
0.4-0.6 |
0.7 |
|
Na |
% |
? |
? |
<0.16 |
0.17-0.24 |
0.25 |
|
B |
ppm |
21 |
21-30 |
31-100 |
101.260 |
260 |
|
Fe |
ppm |
36 |
36-59 |
60-120 |
130-200 |
250? |
|
Mn |
ppm |
16 |
16-24 |
25-200 |
300-500? |
1000 |
|
Zn |
ppm |
16 |
16-24 |
25-100 |
110-200 |
300 |
|
Cu |
ppm |
3.6 |
3.6-4.9 |
5 - 16 |
17-22? |
22 |
*California and Florida recommendations for K are sufficiently different that they are presented separately. The California standards are based on production of table navels and Valencias, and those for Florida were developed primarily for juice oranges like Valencia.
The sampled leaves should be placed in a paper bag, and protected from excessive heat (like in a hot trunk or cab) during the day. If possible, find a laboratory that will wash the leaves as part of their procedure instead of requiring the sampler to do this. Leaf samples can be held in the refrigerator (not the freezer) overnight. Leaves should be taken to the lab for washing and analysis as quickly as is feasible.
Often separate samples are taken within a block if areas exist that appear to have special nutrient problems. The temptation encountered in sampling areas with weak trees is to take the worst looking, most severely chlorotic or necrotic leaves on the tree. Selecting this type of leaf may be counter-productive in that the tree may have already reabsorbed most of the nutrients from these leaves before they were sampled. A leaf-tissue analysis based on leaves like this often results in a report of general starvation, and the true cause of the tree decline if the result of a single nutritional deficiency may not be obvious. Often in weak areas, it is beneficial to sample normal appearing or slightly affected leaves. If the problem is a deficiency, the nutrient will, generally, be deficient in the healthy-looking tissue as well.
Groves of early navels that are not normally treated with copper and lime as a fungicide should include an analysis for copper. Copper deficiency is a real possibility on trees growing in sandy, organic, or calcareous soils. For later harvested varieties, leaves should be sampled before fall fungicidal or nutritional sprays are applied because nutrients adhering to the exterior of leaves will give an inaccurate picture of the actual nutritional status of the tree.
Usually leaf samples taken from trees deficient in nitrogen will overestimate the true quantity of nitrogen storage in the trees. Trees deficient in nitrogen typically rob nitrogen from older leaves to use in the production of new leaves. Frequently, by the time fall leaf samples are collected in nitrogen deficient groves, these spent spring flush leaves have already fallen. Nitrogen deficient trees typically have thin-looking canopies as a result of this physiological response. Since the spring flush leaves are no longer present on the tree in the fall when leaves are sampled, younger leaves are often taken by mistake for analysis. These leaves are higher in nitrogen than the now missing spring flush leaves would have been and provide an inaccurately higher nitrogen status in the grove than actually exists.
Critical levels for leaf-nitrogen for some varieties of citrus, like the grapefruits, pummelos, pummelo x grapefruit hybrids and the mandarins, have not been investigated as well as those for oranges. However, the mineral nutrient requirements of most citrus varieties are probably similar to those for sweet oranges presented in Table 1, except for lemons, where the recommended nitrogen dry-weight percentage is in the range of 2.2- 2.4%.
A complete soil sample in conjunction with the leaf sample can provide valuable information on the native fertility of the soil with respect to some mineral nutrients and information on how best to amend the soil if necessary to improve uptake of fertilizers and improve water infiltration.

- Posted By: Chris M. Webb
- Written by: Ben Faber
Introduction
In numerous publications world-wide, planting hole recommendations for avocado and other subtropical crops are made for large holes from 2 feet by 2 by 2 to as much as a cubic yard. These recommendations also include incorporation of manures or composts comprising 25% by volume with the native soil. I have noted the use of large holes and amendments in several countries, including New Zealand, Guatemala, Brazil, Costa Rica, Mexico and the United States.
The various reasons given for making these large holes are to disrupt any compaction or limiting soil layers and to create a more conducive environment for root growth. In the case of replanting deciduous orchards, McKenry found it to be beneficial in actually replacing the native soil in the hole with pathogen free soil. In many cases, research has shown that holes much larger than the planting ball and using organic amendments can cause problems for many tree species. Improper mixing of the organic amendment can cause anaerobic conditions and settling due to amendment decomposition. Soil that has not been properly firmed in the hole can also lead to plant settling and stems can drop below grade leading to crown rot.
Nonetheless, on the basis of recommendations made in many countries there could be some value in these planting practices, especially in the light of the effect organic matter has on avocado root rot. Numerous studies have shown organic matter suppresses the causal agent of root rot. This study evaluated the effect of hole size and amendments on avocado growth in an ideal environment with excellent soil conditions and in a more harsh one with heavy soil texture and the presence of the root rot pathogen.
Materials and methods
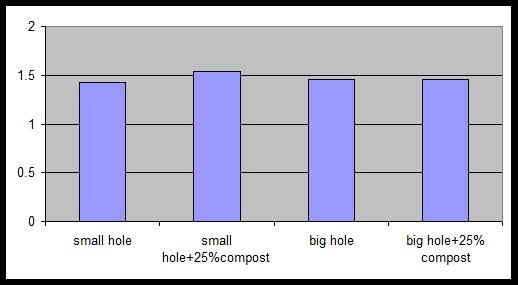
On the north island of New Zealand in the Bay of Plenty, 20 trees each were planted to one of four treatments: a) small holes (12 by 18 inches) without amendment; b) small holes with 25% by volume compost; c) big holes (60 deep by 30 wide by 24 wide inches) without amendment and d) big holes with 25% by volume compost. Big holes were dug with a backhoe, while small holes were dug by shovel. Trees were approximately 2 feet tall at planting. Soil was a deep sandy loam at both sites. Trees were irrigated by drip irrigation. Trees were ‘Hass’ on ‘Zutano’ seedling rootstock. Trees were planted the second week of spring 2000. Tree height, trunk caliper and canopy volume were measured on a monthly basis for eight months and then twice a year for the next year. In Carpinteria, California a similar trial was established using ‘Hass’ on ‘Toro Canyon’ rootstock. Trees were approximately 2 feet tall at planting. The grove had a heavy clay loam soil and a history of root rot. The trees were on drip irrigation. The trees were planted summer 2001 and monitored for 18 months after planting.
Results and discussion
Figures 1and 2 show the results of the different planting treatments at sites in New Zealand on ideal soils and on the heavy soil infected with root rot in California. Only tree height is shown; trunk girth and canopy volume followed similar patterns. From planting onwards, there were no differences in tree growth in any of the treatments at any of the sites. This would lead one to the conclusion that there is no value in and a great expense in making big holes and incorporating amendment. This is especially so in hillside situations where moving equipment and amendments on steep slopes would be very difficult.
The trees at the Carpinteria site, although infested with root rot, all looked good. The addition of organic matter in conjunction with the clonal rootstocks did not apparently provide any greater disease resistance. This is in accordance with work done by John Menge which shows that the greatest benefit derived from mulching are seedling rootstocks. The effect of mulch on disease suppression diminishes with the rootstock’s resistance to root rot.
Figure 1. Tree height (meters) at site 1 in New Zealand 20 months after planting. No differences were found at the 5% level of significance.
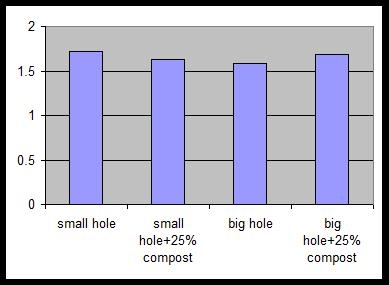
Figure 2. Tree height (meters) in California 18 months after planting. No differences were found at the 5% level of significance.

- Posted By: Chris M. Webb
- Written by: Mary Bianchi
We’d like to challenge you to take the following quiz. Take a minute to place a check mark next to all the practices you regularly employ in your operation. Go ahead – we won’t be collecting them!
Part 1
Yes/ No I know what the nitrogen requirements (lbs actual N/acre/year or /tree/year) are for my crops
Yes/ No I know what the nitrogen levels are in soil amendments I use in my operation (compost, manure, crop residues, etc.)
Yes/ No I have lab analysis of my well/irrigation water.
Yes/ No I monitor tissue levels of nitrogen in my crops to help with fertilizer decisions.
Yes/ No I have put together a nutrient budget that considers all sources of nitrogen for the crops I produce.
Part 2
Yes/ No When I do apply nitrogen, applications are timed according to crop requirements.
Yes/ No I use fertigation to apply nitrogen.
Yes/ No Applications of nitrogen are split into smaller doses to improve efficiency of uptake.
Yes/ No I use cover crops that help manage nitrogen availability.
Yes/ No I manage irrigations to avoid nutrient loss below the rootzone of the crop.
If you marked yes to these as regular activities, you’ve just taken steps in showing how your production decisions can protect water quality. The combined activities noted in Part 1 constitute a Management Practice that protects water quality by developing a nutrient budget to help apply only the appropriate amounts of fertilizer. Activities in Part 2 may alone or in combination constitute Management Practices that help ensure fertilizers are applied efficiently.
Every grower uses ‘management practices’, many of which are meant to generate the best possible product for market. Depending on who you’re talking with, the term ‘management practice’ can be something your Farm Advisor recommends (i.e., pruning to control tree height), your produce buyer suggests (protect avocados in bins from sun scald), or the term can have regulatory connotations.
You’ve all probably heard the term Best Management Practices. Best Management Practice (BMP) is defined in the Federal Clean Water Act of 1987, as “a practice or combination of practices that is determined by a state to be the most effective means of preventing or reducing the amount of pollution generated by nonpoint sources to a level compatible with water quality goals.” The term “best” is subject to interpretation and point of view. In recognition of this, the Coastal Zone Reauthorization Amendment (2000) substituted the terms Management Measures and Management Practices.
How can you tell if any individual activity constitutes a Management Practice that meets the needs of a regulatory program to protect water quality? Ask yourself this question: Can the activity stand alone and result in water quality benefits? Just knowing the nitrogen requirements of your crop doesn’t result in any water quality benefits – developing and using a nitrogen budget for your crop can. A nitrogen budget that takes into account the nutrients applied in amendments, irrigation water, and fertilizers in meeting the requirements of your crop does have the potential to protect water quality from nitrogen pollution from your operation.
Some Management Practices can have water quality benefits as a stand alone activity. Cover crops are recognized as a Management Practice that can help manage both sediment and nutrients to reduce the potential of pollution when used appropriately.
Water quality protection is being asked of all industries in California. You have the opportunity to take credit for all of the activities you already do, like the ones listed above, that protect your local water bodies and/or groundwater from nonpoint source pollution from your operation. Look for additional articles in the coming issues to help you in this effort.
For additional background information on water quality legislation, and nonpoint source pollution from agriculture you can download the following free publications from the University of California’s Farm Water Quality Program:
Water Pollution Control Legislation
Nonpoint Sources of Pollution from Irrigated Agriculture