Some pest problems can be easily handled at home yourself. But if your pest issue is a bit more serious, or you don't have the time or tools to address it yourself, hiring a pest control company might be your best option. Pest management professionals are trained in pest control regulations and methods as well as the principles of integrated pest management (IPM). They can accurately identify your pest and get rid of the problem safely and effectively. While their services may seem costly, the investment can actually save you time and money in the long term.
Before hiring a pest control company, try to do some research on your suspected pest and its management. Consult the UC IPM Pest Notes for help with identification and management to see what control options are available. When you contact a pest control company, prepare yourself to ask about these options and whether they provide IPM services like monitoring, pest exclusion, baiting, trapping, and reduced-risk (less toxic) pesticides.
For detailed steps and questions to ask when hiring a pest control company, consult the newly revised Pest Notes: Hiring a Pest Control Company authored by UC IPM advisors Siavash Taravati, Andrew Sutherland, and UCCE advisor Darren Haver.
- Author: Siavash Taravati
Licensing for perimeter spraying under California's pest control laws is dependent on the situation and site (Table 1). Structural Pest Control Board (SPCB) licensees, Department of Pesticide Regulation (DPR) Qualified Applicator License (QAL) holders, and DPR Qualified Applicator Certificate (QAC) holders are all legally allowed to make perimeter spray applications to control ants and other pests immediately around structures. However, the intent of these applications determines the specific license needed.

SPCB licenses
If the perimeter spraying is performed to prevent the entry of ants or other insects into the structure, then a structural license is required. If the pests are coming from the lawn, then the lawn is considered as a “pest reservoir” and can be treated by the Branch 2 licensee.
DPR licenses
It is important to know what DPR license type allows the applicator to perform perimeter sprays.
- QAL holders with Category A (Residential, Industrial, and Institutional) can do perimeter treatments for ants or other pests that occur indoors and near the structure. QAL Category A licensees, however, cannot provide commercial service to customers (for hire). This license category is most common for governmental employees or institutional employees who perform in-house pest control services as part of their job.
- QAL holders with Category B (Landscape Maintenance) can do perimeter sprays as part of their landscape management efforts. For instance, if landscape shrubs get infested by aphids and tended by ants, then treatment of those plants and perimeter spraying, which incidentally prevents an ant invasion to the structure, is permitted.
- QAC, subcategory Q (Maintenance Gardeners) are also permitted to do perimeter sprays when the application is made as an incidental part of their maintenance gardening work.
It is wise for any pesticide applicator to make sure they are applying pesticides in the right place, at the right time, and for the right reason. Such forethought helps reduce environmental contamination and helps applicators avoid regulatory violations and their consequences.
|
Site/ Situation |
License Required |
Section Cited |
Description of Pest Control Conducted |
|
Adjacent to structure, garden, or orchard pest |
QAL Cat A, Cat B or QAC Subcat Q |
FAC §§ 11701 or 11704 |
Control (for the purpose of eliminating ants, earwigs, garden or non-crop orchard pests, or to assist in the control of honeydew producing insects) even where treatment is applied adjacent to a structure and incidentally prevents invasion of the structure. |
|
Landscape plants & turf |
QAL Cat B or QAC Subcat Q |
FAC §§ 11701 or 11704 |
Any application of a pesticide to existing landscape plants & turf, irrespective of their location in respect to a structure, except for incidental contact of foliage or plants with a pesticide arising from structural pest control activities. Any pesticide application made directly to interiorscapes in business buildings, office complexes, malls, houseplants within households. |
|
Structures, interior or exterior |
Structural |
B&PC § 8550 |
Control of ants, earwigs, cockroaches, silverfish, termites, birds, or rodents invading structures is strictly structural pest control, whether carried on within or outside of the structure. |
|
Structures, in or around |
Structural Branch 2 |
B&PC § 8550 |
Control of pests to humans and their pets, (it would apply to pests other than fleas, for example, mosquitoes) in or around a structure, including treatment of outside areas to control nearby nest or pest reservoir. |
[Originally featured in the Fall 2021 edition of the Green Bulletin Newsletter for structural and landscape pest professionals.]
Disclaimer: Readers should get the most updated regulatory information from federal, state, county, and city resources. Remember, laws change all the time!
Reference: PUE Program Standards Compendium Volume 1: General Administration of the Pesticide Use Enforcement Program – Section 10: Quick Reference Charts. cdpr.ca.gov/docs/enforce/compend/vol_1/entirerep.pdf
- Author: Siavash Taravati
- Posted by: Elaine Lander
Human lice (singular “louse”) are parasitic insects found on people. Adult lice are small (about 1/8 inch (3 mm) long or less) wingless insects that move by crawling. There are three species of lice that exclusively feed on humans: the head louse, the body louse, and the crab or pubic louse.
The head louse (Pediculus humanus capitis) and the body louse (Pediculus humanus humanus) are members of the same species, but the crab (pubic) louse (Pthirus pubis) is from a different insect family. All are pests of public health importance. Treatment is usually prescribed by medical professionals and should be focused on the infested person and their belongings. Use the following descriptions to identify these pests and help your clients seek out proper care from medical professionals.
Crab louse
These lice have a short, round body (not elongated), with hairy tubercles (small, knobby projections of the body wall) on the abdomen. The front legs are shorter and slimmer than the middle and hind legs (Figure 1). Adults and eggs are usually found on pubic hairs but may sometimes be found on other areas of the body such as beards, mustaches, eyelashes, and underneath the arms.
Head louse vs. body louse
Unlike the crab louse, these lice have elongated bodies (Figure 1) and lack tubercles on their abdomen. Head and body lice look almost identical since they are members of the same species. Positive identification requires consideration of specific morphological and behavioral differences (see Table 1). Body lice are known to transmit serious human diseases such as epidemic typhus, trench fever, and louse-borne relapsing fever. Head lice are not known to be an effective vector of diseases even when a pathogen is found in the louse's body.
Human lice elimination involves removal of lice from human body as well as from clothing and living environments. A head lice infestation on a person should be managed by the client. Head lice can be removed with a lice comb or treated with special lice shampoo or lotions containing one or more active ingredients. Human lice have developed resistance to many over-the-counter products containing permethrin or other pyrethroids. Therefore, it is believed that the most effective head lice treatment products are the ones containing ivermectin or spinosad. Body lice can be killed by dry cleaning clothing and bed sheets. Also, vacuuming and treating carpets may improve body lice management especially when the infestation is heavy. Body lice infestations can also be managed by reducing crowding and improving body and clothing hygiene. Head lice infestations, however, are not associated with poor hygiene, at least in developed countries.
For more details about the head louse, including thorough management options and guidance, see the recently revised Pest Notes: Head Lice.
[Article originally published in the Fall 2020 issue of the Green Bulletin.]
- Author: Siavash Taravati
- Posted by: Elaine Lander
Western drywood termites (Incisitermes minor, Figure 1) are an important pest of structural wood in California, causing millions of dollars in damage annually. These termites are very cryptic, hidden in their galleries within wood members (pieces of wood), and only emerge during swarming. As a result, wood damage usually goes unnoticed for a long time.

Control options are generally categorized as either whole-structure treatment (heat treatment and fumigation) or local treatments (insecticide injection into the wood, high-power microwaves, electrocution, and other techniques).
Despite the high efficacy of fumigation, there has been increasing interest by property owners to use local treatments for eradicating drywood termites. This may be due to the high cost and inconvenience of fumigation. To learn more about decision-making associated with fumigation, visit this webpage. Local treatment of drywood termites can be ineffective because of the difficulty in locating active infestation sites within structures.
To address this issue, practitioners and researchers have considered different detection methods using traditional and modern technologies such as borescopes, moisture meters, and heat sensors as well as devices using X-rays, acoustic emission, and low-energy microwaves. Here we provide a review and some technical details on how to operate a specific device using microwave technology for detecting termite movement in structures.
Termatrac
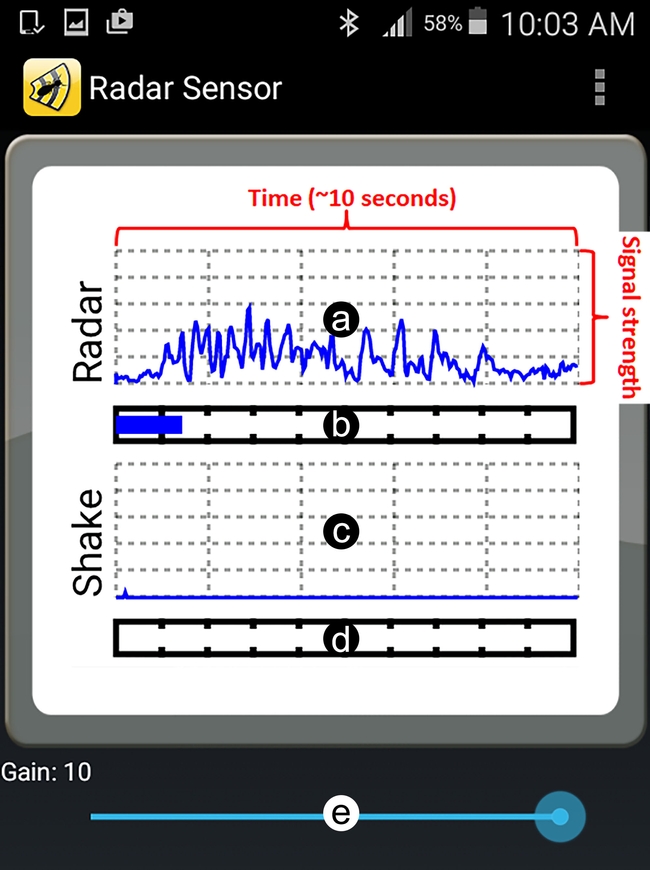
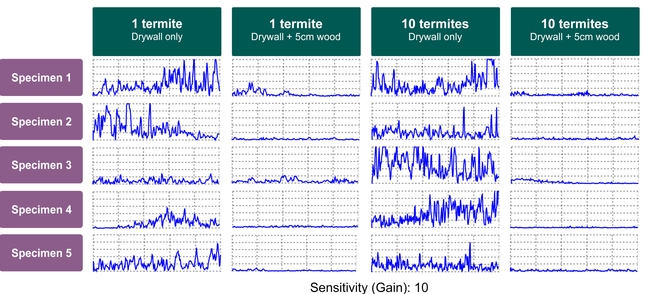
Although such output can be informative, interpreting the results might not always be easy and may also require considerable expertise. First, the output's line graph may represent detection of non-termite objects or the user themselves (body movement or hand shaking while holding the device). Second, the signal intensity varies depending on the depth of termite activity, so Gain settings may need to be adjusted for higher or lower sensitivity. Third, the relationship between termite density (number of termites per unit of area) and signal strength is not easily understood by users (Figure 3). Fourth, termites may not be present or active during inspection and this may lead to a false negative conclusion (concluding “no termites” when they are present) when inspecting an infestation. To address these issues, field and lab research experiments were conducted in California to evaluate the efficacy of the Termatrac device and to help termite inspectors accurately interpret the output signal.

- hand-held with radar surface flush against the inspection surface
- mounted on a tripod with radar surface flush against the inspection surface
- resting on a horizontal surface with radar surface flush against the inspection surface
- with radar at 45° angle to the inspection surface using the back flap or a tripod
Field studies revealed that hand-held uses produce less accurate results than tripod/flap supported uses due to user hand shaking. Also, the device's output showed more noise (Noise is referred to a detected signal in the output that is not coming from drywood termites) from the user's body movement when used at 45° to the inspection surface as compared to flush against the inspection surface.
Recommendations
For optimal readings, Termatrac users should keep the following in mind. Users need to stand still when reading the output or the device will pick up their body movement and produce a false positive signal. This is especially true at high sensitivities.
Users also need to ensure that there are no moving objects (vehicles, plants swaying with the wind, airborne debris such as leaves and dusts, children, or animals (such as pets and birds) on the other side of the inspection surface (a wall for instance) which may create false positive signals. Also, water passing through pipes behind inspection surfaces may produce a strong signal. However, heavy machinery around the experiment sites did not produce any detectable noise despite being very loud.
The device should not be used to inspect unstable surfaces or non-fixed objects (e.g. yard fence) since these situations will increase the chance of false positive signals and inaccurate detection of termites.
To save time and increase accuracy when inspecting standard interior walls, users should first try to locate studs using a stud finder and then use Termatrac on those areas only. Users may also choose to focus on wooden window frames and windowsills since these have been observed to be one of the most commonly drywood termite detected spots in homes.
Findings
Lab studies showed that higher densities of termites may not necessarily produce stronger signal (Figure 3). At the highest sensitivity setting, Termatrac T3i was able to detect a single drywood termite behind 5 cm (2 inch) of wood and 1.3 cm (0.5 inch) of drywall (total thickness of test “wall”: 6.3 cm / 2.5 inch).
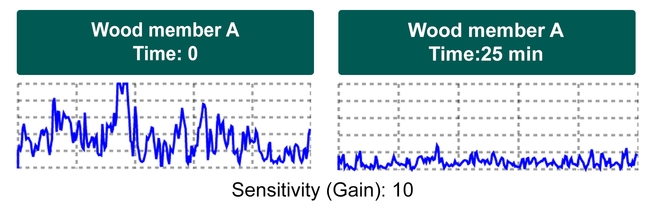
Drywood termites move within their galleries continually and therefore may not be present in all gallery regions at all times. Furthermore, termite activity may change throughout the day depending on temperature and other factors (Figure 5). As a result, if you suspect an active infestation in a wall but are not getting a detectable Termatrac signal, it is worth moving on to other areas and then returning in a few minutes to re-inspect the suspect location.
To conclude, Termatrac can be very useful in some termite detection. Like other termite detection devices, Termatrac has limitations and requires training and experience before a user can efficiently and accurately detect termites. With this said, an experienced Termatrac user can obtain valuable information about termite presence and activity when the infested wood members are in accessible locations.
1Mention of a product does not constitute an endorsement.
[Originally published in the Winter 2020 issue of the Green Bulletin.]
- Author: Siavash Taravati
[From the Fall 2018 issue of UC IPM's Green Bulletin newsletter]
The dark rover ant (Brachymyrmex patagonicus) is an invasive species which is increasingly being noticed in Southern California. It is a nuisance species that invades structures as both workers and winged (alate) individuals.
This article aims to introduce structural and landscape pest management professionals (PMPs) to the dark rover ant and includes several important references for readers to learn what is currently known about its biology, behavior, and control.

Identification
Dark rover ant (DRA) workers are usually 1–2 mm long and are typically reddish-brown to blackish-brown (Figure 1). They have one node on the waist hidden under the gaster (abdomen), with long erect hairs mixed with sparse fine pubescence on the gaster (Figure 2), and nine-segmented antennae (Figure 3). The only other rover ant species reported from California is B. depilis which is rarely seen in the field (1) and can be distinguished from DRA by their yellow to yellowish-brown color (2).

Where are dark rover ants found?
The dark rover ant is native to South America and is found in several countries including Argentina, Paraguay, Brazil, and Venezuela(3).It was first discovered in the United States in 1978 in Louisiana (4).
This ant was found in California for the first time in 2010 in Orange and Riverside Counties (5). The author found this species in Los Angeles County in Pacoima and Sylmar as well as Citrus Community Park (Riverside, CA) and Andulka Park (Corona, CA). Pat Copps of Rollins Inc., reported DRA presence in Bakersfield. This species may already be widely distributed in Southern California and could be dispersing to newer areas.
Biology
DRA nests both indoors and outdoors (6). They forage without making trails but trailing has been observed in the laboratory between the sub-units of a colony (7). High levels of aggression toward individuals of other colonies has been observed, which may prevent the establishment of multiple colonies in a small area (8).
Different authors have reported that this species can coexist with other ant species, most notably Argentine ant (Linepithema humile) and red imported fire ant (Solenopsis invicta), and has even nested alongside the latter (4, 5). In Los Angeles and Riverside counties, the author has seen DRA workers walking near trails of red imported fire ants and Argentine ants without alarming the workers of these other two species.
DRA have been observed feeding on plant nectar in the field. In the lab, a 30% honey solution along with a liquid tuna mixture and caterpillars has been observed to support reproducing colonies (7).
DRA as a Nuisance Pest
Foragers and alates of DRA invade structures and can become nuisances. In Orange County, there were more incidences of DRA infestations in 2018 as compared to 2017. In June and July of 2018, customer calls were made regarding reproductives when clients saw them at bright lights inside. David Taylor from The Bugman Pest and Termite Control reported that clients were often unaware that workers were also present.
According to Pat Copps, in southern California, DRA infestations have been more common in Orange County but are still relatively rare as compared to the pervasive Argentine ant, which accounts for about 90% of customer calls for ant infestations. He also says DRA foragers are usually seen in kitchens, bathrooms, and laundry rooms inside and around grade/wall junctures, stone borders, patios, and tree roots.
How to Manage DRA
Pest management professionals have reported difficulty in managing dark rover ants in other parts of the United States (4). This is believed to be due to the existence of long-lived satellite colonies, which can survive for around three months when separated from the main colony (8). Consequently, a structure treated for DRA may soon be re-invaded by ant individuals from adjacent satellite colonies.
In California, similar difficulties have been reported in controlling this pest. These difficulties have been overcome by some PMPs by using combinations of baits, dusts, and liquid sprays as interior and exterior treatments. Copps reported that very limited success has been achieved when attempting to control this pest outdoors.
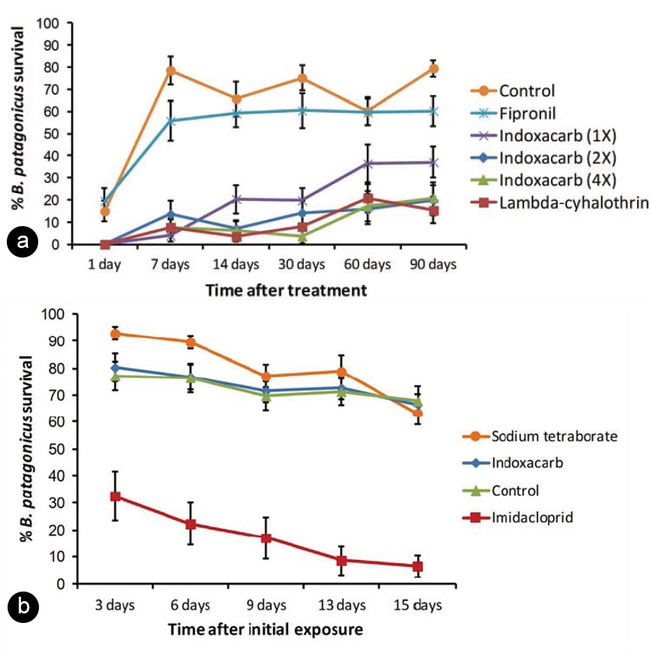
Research by Miguelena and Baker (6) on the efficacy of selected pesticides against DRA showed higher ant mortality when using Arilon (indoxacarb, Syngenta) and Demand CS (lambda-cyhalothrin, Syngenta) liquids as well as Maxforce Quantum ant bait (imidacloprid, Bayer CropScience) when compared to other insecticides used in the study (Figure 4).
Since DRA control failure is very common, it is important for PMPs to consider using products which have been scientifically proven to be effective in controlling DRA. Other products may eventually prove to be effective against DRA, but at this time there has been limited published research on the management of this pest.
References
- Ward, P.S., A synoptic review of the ants of California (Hymenoptera: Formicidae). Zootaxa, 2005. 936(1): p. 1-68.
- McGown, J.A. Ants of the Southeastern United States. 2003.
- Quirán, E.M., J.J. Martínez, and A.O. Bachmann, The neotropical genus Brachymyrmex Mayr, 1868 (Hymenoptera: Formicidae) in Argentina: Redescription of the type species, B. patagonicus Mayr, 1868; B. bruchi Foel, 1912 and B. oculatus Santschi, 1919. Acta zoológica mexicana, 2004. 20(1): p. 273-285.
- MacGown, J.A., J.G. Hill, and M.A. Deyrup, Brachymyrmex patagonicus (Hymenoptera: Formicidae), an emerging pest species in the southeastern United States. Florida Entomologist, 2007. 90(3): p. 457-464.
- Martinez, M.J., et al., New records for the exotic ants Brachymyrmex patagonicus Mayr and Pheidole moerens Wheeler (Hymenoptera: Formicidae) in California. The Pan-Pacific Entomologist, 2011. 87(1): p. 47-50.
- Miguelena, J.G. and P.B. Baker, Evaluation of liquid and bait insecticides against the dark rover ant (Brachymyrmex patagonicus). Insects, 2014. 5(4): p. 832-848.
- Tamayo, D. Dark Rover Ant: Brachymyrmex patagonicus Mayr. 2011.
- Miguelena, J. and P. Baker, Why are rover ants (Brachymyrmex patagonicus) so difficult to control? Graduate Student Poster Session, Entomological Society of America Annual Meeting, San Diego, California, 2010.