- Author: Pershang Hosseini
- Author: Tong Zhen
- Author: Matthew Fatino
- Author: Brad Hanson
- View More...
Broomrapes (Orobanche and Phelipanche spp.) are obligate plant parasites with a broad range of agricultural crop hosts. In non-parasitic plant species, seeds generally initiate germination when exposed to favorable conditions of temperature, humidity, oxygen, and, occasionally, light. However, for obligate parasitic plants like broomrapes, a chemical signal from the host plant is essential. Germination of broomrape can only occur under appropriate soil conditions and when the seed receives a strigolactone chemical signal released from the roots of a suitable host. Strigolactones (SLs) are carotenoid-derived hormones that play a crucial role in various aspects of plant growth and development. Fertilizers can regulate the production of these plant hormones (Xie et al. 2010).
Fertilization can improve soil conditions and lead to reduced initiation of broomrape parasitism (Fernández-Aparicio et al. 2016). Studies have shown that heavy infestations of crenate broomrape (Orobanche crenata Forsk.) on faba beans are linked to lower soil fertility (Trabelsi et al. 2017), and parasitism of Egyptian broomrape on tomato occurs more frequently in low-nutrient conditions (Jain and Foy 1992). The application of fertilizers has been reported to suppress the occurrence of other parasitic plants such as Striga (Jamil et al. 2011) and Egyptian broomrape (Phelipanche aegyptiaca) (Jain and Foy 1992). Fertilizers can reduce parasitism and enhance crop tolerance both directly, through toxic effects, and indirectly by improving soil fertility and plant health.
Direct toxic effect of fertilizers
Nutrient management can enhance both resistance and tolerance to broomrape parasitism in crops at the pre-attachment and post-establishment stages. Increasing the levels of nitrogen (N) and phosphorus (P) in the soil through fertilizer application can reduce the germination and subsequent infestation rates of parasitic weeds (Jamil et al. 2011). Ammonium nitrate combined with potassium phosphate or the use of ammonium phosphate alone proved to be effective in reducing parasitism and promoting the growth of tomato plants compared to potassium sulfate (Jain and Foy 1992). The direct inhibitory effects of nutrients on broomrape seeds can occur during the preconditioning, germination, and seedling elongation stages. Preconditioning Egyptian broomrape seeds in the presence ofammonium salts, such as ammonium sulfate or urea, significantly inhibited their germination; in contrast, nitrate did not have the same inhibitory effect (Jain and Foy 1992). Increasing nitrogen rate (ammonium nitrate) decreased seed germination and radicle length of branched broomrape (Irmaileh 1994). Another experiment showed that nitrogen in the ammonium form resulted in greater inhibition than nitrate, and the inhibition mechanism was actually a reduction in radicle elongation rather than inhibition of germination (Westwood and Foy. 1999).
Down-regulating of Strigolactones (SLs)
Fertilization can protect crops from parasitism by downregulating the synthesis and exudation of strigolactones, which are the most potent germination-inducing factors for root parasites (Fernández-Aparicio et al. 2016). Plants release SLs in different situations, including the establishment of symbiotic relationships between plants and certain soil microorganisms (Besserer et al. 2006; Kapulnik and Koltai 2014) and during stress response (Kapulnik and Koltai 2014). It is likely that plants produce strigolactones as a "cry for help," which broomrape exploits to its advantage. The availability of nutrients, particularly nitrogen, can decrease plant stress and subsequently downregulate the production of strigolactones. Effects of N, P, and K deficiencies on SL production showed that both N and P deficiencies enhanced SL exudation in resistant genotypes of faba bean (Trabelsi et al. 2017) and red clover (Yoneyama et al. 2012), while K deficiency had no effect (Trabelsi et al. 2017). A similar positive effect of low phosphate levels on SL production was also observed in tomato (López-Ráez et al. 2008).
In summary, effective nutrient management is a vital strategy in reducing broomrape parasitism and enhancing crop tolerance. By manipulating soil fertility and nutrient availability, it is possible to directly inhibit broomrape development and indirectly protect crops by downregulating strigolactone production. Appropriate fertilization and other stress-reducing management practices can reduce broomrape parasitism.
In addition to the indirect effects on broomrape, researchers in the Hanson lab are investigating the direct toxic effects of various fertilizers on broomrape seeds during three stages: preconditioning, germination, and post-germination (Figure 1). In future studies, we aim to explore the indirect effects of fertilization on broomrape parasitism, focusing on how nutrient management can influence the production of strigolactones and other related mechanisms. The ultimate goal of this work is to determine if manipulating fertilizer form, timing, or rates could directly inhibit branched broomrape and maximize tomato resilience to broomrape parasitism as part of an integrated management strategy.
 |
 |
 |
| a | b | c |
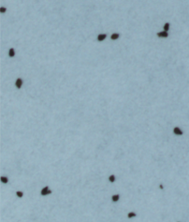
Figure 1: Branched broomrape seeds in different treatment conditions:
a) Germination (elongated radicle) observed in the control group.
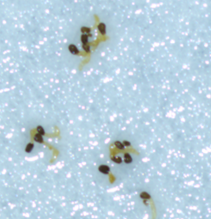
b) No germination was observed when ammonium phosphate was applied at the germination stage.
c) Elongated radicle changed color when ammonium phosphate was applied post-germination.
References
Besserer, A., Puech-Pagès, V., Kiefer, P., Gomez-Roldan, V., Jauneau, A., Roy, S., ... & Séjalon-Delmas, N. (2006). Strigolactones stimulate arbuscular mycorrhizal fungi by activating mitochondria. PLoS Biology, 4(7), e226.
Fernández-Aparicio, M., Reboud, X., & Gibot-Leclerc, S. (2016). Broomrape weeds. Underground mechanisms of parasitism and associated strategies for their control: a review. Frontiers in Plant Science, 7, 171714.
Irmaileh, B. A. (1994). Nitrogen reduces branched broomrape (Orobanche ramosa) seed germination. Weed Science, 42(1), 57-60.
Jain, R., & Foy, C. L. (1992). Nutrient effects on parasitism and germination of Egyptian broomrape (Orobanche aegyptiaca). Weed Technology, 6(2), 269-275.
Jamil, M., Charnikhova, T., Cardoso, C., Jamil, T., Ueno, K., Verstappen, F., ... & Bouwmeester, H. J. (2011). Quantification of the relationship between strigolactones and Striga hermonthica infection in rice under varying levels of nitrogen and phosphorus. Weed Research, 51(4), 373-385.
Kapulnik, Y., & Koltai, H. (2014). Strigolactone involvement in root development, response to abiotic stress, and interactions with the biotic soil environment. Plant Physiology, 166(2), 560-569.
Trabelsi, I., Yoneyama, K., Abbes, Z., Amri, M., Xie, X., Kisugi, T., ... & Kharrat, M. (2017). Characterization of strigolactones produced by Orobanche foetida and Orobanche crenata resistant faba bean (Vicia faba L.) genotypes and effects of phosphorous, nitrogen, and potassium deficiencies on strigolactone production. South African Journal of Botany, 108, 15-22.
Westwood, J. H., & Foy, C. L. (1999). Influence of nitrogen on germination and early development of broomrape (Orobanche spp.). Weed Science, 47(1), 2-7.
Xie, X., Yoneyama, K., & Yoneyama, K. (2010). The strigolactone story. Annual Review of Phytopathology, 48, 93-117.
Yoneyama, K., Xie, X., Kim, H. I., Kisugi, T., Nomura, T., Sekimoto, H., ... & Yoneyama, K. (2012). How do nitrogen and phosphorus deficiencies affect strigolactone production and exudation?. Planta, 235, 1197-1207.
- Author: Bradley Hanson

My interest in California is largely driven by concerns about spreading seeds of the CDFA "A-list" parasitic weeds branched broomrape and related species in processing tomato, a topic with lots of current research. However, the risks of moving new pests around on field equipment and the mitigation strategies are relatively similar whether you're talking about seeds of new weedy pests, new disease pathogens or insect pests, or even some human health pathogens.
In an effort to develop baseline information and guide future research and extension efforts, we are asking for growers and industry participation in a very brief survey (less than 5 minutes) so that we can get some sense of the concerns, practices, and barriers related to equipment sanitation in various crop sectors around the U.S.
Your participation would be greatly appreciated whether you think about this kind of thing regularly, or not at all
The survey can be accessed online using the QR code in the slides below, in the upper corner of this blog post, or at this link:
Sanitation of Agricultural Field Equipment Questionnaire UCD
Thanks!
-Brad


- Posted by: Gale Perez
From the Weed Science Society of America (WSSA) Graduate Student Organization newsletter (Oct. 2023)
* * * * * *
Graduate Student of the Month

What is your long-term goal?
My long-term goal is to land a career in industry or the public sector that enables me to work with growers and stakeholders and help address the issues they face. I have been fortunate to see the positive impacts on growers while working in the industry as well as working with extension professionals of the University of California throughout the state, and would be honored to serve my local stakeholders in either setting.
What drew you to weed science?
My introduction to weed science was through an internship during my undergraduate degree doing applied research for a chemical company. The applied nature of weed science was very appealing to me, as well as the intricacies of managing weeds in the various specialty cropping systems we have in California.
What is your favorite weed and why?
Branched broomrape, and broomrapes in general, are a very unique and challenging species to work with and I really enjoy the dynamic challenges they present. My least favorite weed by far is field bindweed (Convolvulus arvensis.)
What is your favorite thing about the WSSA or your regional society?
My favorite thing about WSSA is its ability to connect scientists from all over the world. Working with a unique weed in a unique cropping system, it is often hard to meet with folks regionally with experience studying or managing branched broomrape in tomatoes. Attending a WSSA conference enabled me to meet with researchers from across the nation and the globe who have experience working with branched broomrape, and our discussions and connections were invaluable to my research and career in weed science.
What university do you attend and what is your research focus?
I am a Ph.D. candidate at the University of California, Davis, and my research focus is on in-season management of the invasive noxious weed, branched broomrape (Phelipanche ramosa). It is a parasitic plant that attaches to a host plant's roots and presents a major threat to the California processing tomato and seed industries.
To contact Matt Fatino, email him at mfatino@ucdavis.edu.
- Author: Emily Dooley
- Posted by: Gale Perez
Hanson and team battle invasive species
At first glance, Orobanche ramosa looks like an interesting blossoming plant, one that could add a unique flair to flower arrangements. But it's a parasitic weed that attaches to roots, sucks out nutrients and is threatening California's $1.5 billion processing tomato industry.
The weed's tiny seeds — smaller than finely ground pepper — can survive in soil for many decades and be carried by wind, water, soil transfers and even footwear. If found attached to crop plants and reported to the state, farmers are required to destroy the field before harvest, taking large losses not covered by crop insurance.
Its resurgence concerns state regulators and industry, which is helping fund multidisciplinary research at the University of California, Davis, on ways to detect, manage and fight the weed.
“Most of the damage occurs before you can see it,” said Brad Hanson, a professor of Cooperative Extension in the Department of Plant Sciences. “There's a lot of ripples to the problem. We could see it spread to other crops and other regions in the state if it's not managed.”
Across three colleges at UC Davis, researchers are working on ways to detect the pest, manage it in the field throughout its life cycle and develop long-term solutions to minimize the threat to California agriculture. The work is happening in labs and the field, using drones, human spotters and new techniques to sniff out volatile organic chemicals that are emitted when the weed is present.
They are also testing ways to sanitize farm equipment to reduce the risk of spreading seeds from contaminated fields to clean ones. And they are testing dozens of other crops to see if they are susceptible or could be used as false hosts to kill off the Orobanche seeds in the soil.
Invasive species alert: A weed resurgence in California agriculture
The California Department of Food and Agriculture and industry had a program from the 1950s through the 1970s to eradicate the weed, which is commonly known as branched broomrape. But the weed showed up again in Yolo County in 2017.

“Believing it to be eradicated, the industry moved on to other challenges,” said Zach Bagley, managing director for the California Tomato Research Institute Inc., or CTRI. “We've been aggressive, with this as our top priority, and we've been putting the funding behind it.”
CTRI's research budget for 2022 and 2023 is nearly $1 million, and half of that money has gone to fund six researchers and their teams at UC Davis and UC Davis Chile, as well as some work at University of Wisconsin.
“Unfortunately, or fortunately, tomatoes are going to have to be the tip on the spear of trying to address this,” Bagley said. “UC Davis has relationships with industry. They have the expertise in the areas we need for this problem.”
Last year, CTRI, Corteva and Hanson's team got state permission for an herbicide treatment that, in trials, has reduced Orobanche emergence fourfold. “That management treatment has been used extensively by growers this year,” Bagley said.
The CTRI funding has helped leverage other money, bolstering the research capability.
“It shows the power of the university,” Hanson said. “It shows the kind of things we can do with the research and extension expertise in a land-grant university. When something like this arises, we can spring into action.”
Severe regulatory triggers impact California tomato farmers
It's hard to know quite how far Orobanche has spread.
Yolo County farmers reported 71 to 403 acres affected between 2017 and 2022, with some years seeing less and others more. No cases have been reported this year, Yolo County Agriculture Commissioner Humberto Izquierdo said.
But Orobanche weeds could be sprouting up elsewhere.
“The issue with this pest is that growers are very reluctant to report it and it's not an easy issue to spot,” Izquierdo said. “The regulatory requirements are very strict. Once it's found, we limit harvesting on that field.”

Izquierdo works with canneries and the CTRI to promote good sanitation policies, so the weeds don't spread. But with a limited number of inspectors, it's hard to get a handle on the extent of the problem without consistent reporting.
“If we don't know where the problem is, we can't deal with it,” he said. “It really takes cooperation from industry to be able to move forward.”
Hanson says it's likely that there are more infested fields in Yolo County than what has been reported, and there's no obvious reason Orobanche could not spread to other tomato-producing regions. But the harsh repercussions for reporting can be a barrier: “Do the right thing, lose hundreds of thousands of dollars,” he said.
The low reporting numbers combined with the hardiness, number and sheer longevity of Orobanche seeds requires more than regulatory action.
“This is not something you're going to sweep under the rug,” said Matt Fatino, a Ph.D. student researcher in Hanson's lab. He has been working on chemical control strategies, including on projects with counterparts in Mediterranean climates, where the weed is more prevalent.
Field research on tomato plant diseases promotes sustainable agriculture
A local grower recognized the issue needed research and allows Hanson, Fatino and other UC Davis scientists access to a 3-acre plot in Woodland where tomatoes are planted in infected soil. There, researchers test out different ways to control the weeds on the ground and others take to the air to scan for evidence of the pest.

A half dozen other weeds also clustered around that one tomato plant, attached at the roots underground by what looked like an undifferentiated mass once unearthed.
In the distance, students and other lab members kicked at the tomato plants, searching for the weeds and placing tiny flags to mark their presence.
In Fatino's experiment, different herbicides of varying concentrations have been applied via irrigation to the rows in an attempt to gauge what may work best controlling the Orobanche. The chemicals must be authorized with the state and have restrictions on use.
Every week, spotters arrive with differently colored flags to mark their latest finds. In past years, 800-900 flags were planted in the 1-acre experiment.
“It's a needle in a haystack project,” Hanson said.
At that same plot, Ph.D. student Mohammadreza Narimani and others from the Digital Agriculture Laboratory, which is run by associate professor of Cooperative Extension Alireza Pourezza, use drones equipped with special cameras and technology to scan the field.
The lab, which is housed in the College of Engineering and College of Agricultural and Environmental Sciences, specializes in using digital technology and data analytics to support sustainable agriculture.
The team has divided the field into quadrants, flagging spots with and without Orobanche weeds. Four drones fly over the field, equipped with Light Detection and Ranging, or LIDAR, scanners, RGB, multispectral and hyperspectral cameras. As the drones fly, real-time images appear on a laptop, filling in the screen square by square with images and data from the camera's sensors to be analyzed later in the lab.

“Different characteristics of plants reflect light in a different way, which results in them appearing in different colors,” Narimani, the drone pilot, said. “We can monitor different levels of nutrients in the plant and identify if there are any signs of broomrape.”
Secure lab research on plant pathology and broomrape weed
Back on campus, the weeds are carefully studied in a secure environment known as the Contained Research Facility, or CRF, which is dedicated to studying invasive plant pests, ranging from pathogens and weeds to nematodes and certain invertebrates like spiders and insects.
The facility is the first of its kind in the west and houses labs, greenhouses and growth chambers. People working in the space must adhere to strict protocols meant to ensure none of the pests escape the space. Waste is sterilized on site and people, plus phones and other personal items brought inside, must shower out to leave the facility.
In a small space set aside for Orobanche, postdoctoral scholar Pershang Hosseini has a handful of projects; Hosseini also is in the Department of Plant Sciences. One of her projects is testing whether certain sanitizing chemicals can kill the seed, removing the threat of Orobanche and other pathogens spreading on farm equipment. Hosseini is working on that project with plant pathologist Cassandra Swett, an associate professor of Cooperative Extension.
Another project involves planting more than two dozen crops to see whether they are susceptible to Orobanche or if they could serve as false hosts, causing the seeds to die out because they germinate but don't find a suitable host to parasitize.
Along those lines, Hosseini is also studying a chemical that is similar to a natural substance in tomato plants that signals Orobanche to germinate when no host exists, essentially causing the seeds to commit suicide.
“No seeds can germinate twice, so this could be a way to directly reduce the amount of seed in the soil seedbank,” Hosseini said.
Innovative plant disease detection: Sniffing out broomrape in tomato plants
Other professors are also using the space for related research, and one project involves smelling volatile organic chemicals.
“We're basically sniffing out diseases or infections or parasites,” said Cristina Davis, professor of mechanical and aerospace engineering.
Davis and her lab director and staff researcher Mitchell McCartney developed a sensor to detect Orobanche by measuring odors emitted from plants affected by the weed.
“The plants tend to off gas an alert signal around themselves to warn other plants,” McCartney said. “There's all this information that's being passed around us in the air, but the human olfactory nerve isn't sensitive enough to pick up on those changes.”
At the Contained Research Facility, tomato plants are enclosed in an airtight space and fresh air is pumped in, while a sponge-like sensor collects what is emitted into the air. Back at their home lab in the College of Engineering, Davis and McCartney superheat the sponges to release volatile organic chemicals.
“It captures a snapshot of odor,” he said.
If a unique chemical signature can be isolated, fields could one day be sniffed for Orobanche weeds, serving as an alert system.
Biological innovations in plant breeding could mean solutions for California farms

“The goal is to figure out if there are very early gene expressions when plant roots are attached by broomrape,” Sinha said. “Early response is where we feel resistance will lie.”
Brady is looking at if the cell types of tomato plant roots could be modified to create a barrier to the weed from attaching.
“It's a combinational approach to attack from all these different points in the life cycle,” Brady said of the UC Davis research. “It's attacking the attacker.”
For Hanson, the research is among the most collaborative that he's been a part of and shows how real-world problems can be addressed when industry, regulators and researchers work together.
“We're making progress on a major threat to California agriculture,” he said, “and it's really rewarding to be a part of the research team.”
Media Resources
- Brad Hanson, UC Davis Department of Plant Sciences, bhanson@ucdavis.edu
- Emily C. Dooley, College of Agricultural and Environmental Sciences, ecdooley@ucdavis.edu
- Amy Quinton, UC Davis News and Media Relations, 530-601-8077, amquinton@ucdavis.edu
Media kit of images for download.
Original source: UC Davis Dept. of Plant Sciences
Emily Dooley is a Communications Specialist with the College of Agricultural and Environmental Sciences at UC Davis.
- Author: Pershang Hosseini
- Author: Alison Colwell
- Author: Bradley Hanson
- Posted by: Gale Perez
The Orobanchaceae family comprises 270 holoparasitic species that cannot photosynthesize. Instead, they rely entirely on the host plant roots for nutrition, and can produce great numbers of minute, dust-like seeds that last for years in the soil. This family contains some of the most serious agricultural parasites in the genera of Phelipanche and Orobanche some of which are present in California. The different species can be distinguished by the degree of branching of their stems and presence/absence of bracteoles at the base of the flower Orobanche species have unbranched stems and no bracteoles, while Phelipanche species feature branched stems with bracteoles. In this paper, we discuss three economically significant broomrape species, and two that are not crop pests.
Phelipanche ramosa (Branched broomrape)
The natural distribution of branched broomrape (Table 1, Figure 2b) encompasses Europe, the Middle East, West Asia, and North Africa. It is established in southern Africa, Chile, and north America, having been reported in IL, TX, WA, OR, DE, WV, VA since early in the last century. Notable new infestations have been documented in Australia, resulting in several million dollars in annual expenses for quarantine and control efforts across 200,000 hectares. Further economic losses accrue from restrictions on the sale of produce or crop seed from the infested areas; these measures are aimed at preventing the spread of the parasite to other agricultural fields (Warren, 2006). Although reported occasionally in California in past decades, a new infestation of branched broomrape has been documented in California in recent years (Osipitan et al., 2021).
The host range of branched broomrape is exceptionally broad, encompassing various crops in the Solanaceae (tomato, eggplant, tobacco, pepper, and potato), Brassicaceae (rapeseed), Cannabaceae (hemp), Fabaceae (chickpea, clovers, groundnut, faba bean, lentil, pea), Apiaceae (carrot, celery, fennel, parsnip) and Asteraceae (lettuce, sunflower, and many ornamental species). Furthermore, branched broomrape can parasitize wild hosts within a range of families, including Chenopodiaceae, Amaranthaceae, Malvaceae, and Rosaceae. Although there have been reports of it affecting onions, it typically does not infest monocots.
There is evidence of lineages of this species having undergone both host specialization and host extension. For instance, one population initially parasitizing tomatoes was found to also parasitize tobacco and lettuce. In southwestern Germany, some farmers switched from cultivating tobacco to parsley, leading to an infestation of branched broomrape in the parsley crop (Kohlschmid et al., 2011). However, a population originating from tobacco failed to parasitize lettuce (Musselman & Parker, 1982), suggesting the host shift requires some selective adjustments in order to succeed.
Economic Importance: Yield losses in tomato and tobacco are commonly reported to be 30–50% (Parker, 2013). Branched broomrape was found to reduce tomato shoot dry weight by 60-70%, while also causing a concurrent 50% decrease in chlorophyll content and photosynthesis levels (Mauromicale et al., 2008) in pot experiments.
Phelipanche aegyptiaca (Egyptian broomrape)
Branched and Egyptian broomrape (Table 1, Figure 2a) are closely related and easily confused for one another. Egyptian broomrape is a more robust, taller plant than a typical branched broomrape plant but the most easily noted difference is the larger size of the flowers at 20–35 mm long (branched broomrape typically no more than 20 mm). The distribution of Egyptian broomrape overlaps with branched broomrape in South Europe, the Mediterranean, and North Africa but extends much further eastwards into South Asia and China.
Egyptian broomrape can attack most of the same crops affected by branched broomrape. It differs in occurring on a wider range of Brassicaceae, especially various mustard species in India. It is also a more important pest on Cucurbitaceae compared to branched broomrape.
Economic Importance: The effects of Egyptian broomrape on the host are the same as branched broomrape, and damage can be similarly severe.
Orobanche minor (small broomrape)
Small broomrape (Table 1, Figure 2c) is typically a one (or few) stalked, unbranched plant up to 70 cm tall and occasionally 1 m. It is very widely distributed, being native throughout most of Europe, Western Asia and Northern Africa, as far south as Ethiopia and Somalia, while it has also been sporadically introduced to Japan, New Zealand, Australia and several countries in North and South America. This species is a federally listed noxious weed in the United States that has infested red clover fields in Washington and Oregon (Mallory-Smith & Colquhoun, 2012; Osterbauer & Rehms, 2002 ).
The host range for small broomrape is very wide, particularly in Fabaceae (clover, alfalfa, vetch). Small broomrape can also parasitize Asteraceae (lettuce, safflower) and Apiaceae (carrot, celery), Solanaceae, and other families.
Economic Importance: The greatest economic damage is to clover, reported in several countries in Europe. This problem has led to the abandonment of the clover seed industry in some of those countries including the UK.
Orobanche hederae (ivy broomrape)
While the majority of Orobanche species exhibit a broad host range for parasitism, some, such as ivy broomrape (Table 1, Figure 2d), exhibit narrow preferences. In this case, the parisite exclusively attacks species within the Hedera genus, commonly known as ivy. Ivy broomrape is a short-lived monocarpic perennial, reaching heights of up to 60 cm. Its distribution area spans across Western and Southern Europe, North Africa, and Asia Minor. It is a close relative of Orobanche minor, and arguably derived from it.
Economic Importance: None known.
Aphyllon Cooperi or Orobanche Cooperi (desert broomrape)
Desert broomrape (Table 1, Figure 2e) is a biennial broomrape that is native to the desert southwest of the US and Mexico. It parasitizes shrubs of Ambrosia and Encelia (Asteraceae). It was segregated from Orobanche based on chromosome number and 5 calyx lobes instead of 4.
Economic Importance: None known. It has been reported occasionally in tomato fields, but does not appear to persist under cultivation of the crop. It has been grown in potted tomatoes by the second author, but it is slow to reach the flowering stage and the phenology unfolds slowly, so the plant is unlikely to set seed during a tomato crop produciton cycle.
References
Kohlschmid E, Mu¨ller-Sto¨ver D, Sauerborn J. (2011). Ausbreitung des parasitischen Unkrauts Phelipanche ramosa in der deutschen Landwirtschaft. Gesunde Pflanzen, 63:69–74.
Mallory-Smith, C., & Colquhoun, J. (2012). Small broomrape (Orobanche minor) in Oregon and the 3 Rs: regulation, research, and reality. Weed science, 60(2), 277-282.
Mauromicale G, Lo Monaco A, Longo AMG. (2008). Effect of branched broomrape (Orobanche ramosa) infection on the growth and photosynthesis of tomato. Weed Science, 56: 574–581
Musselman LJ, Parker C. (1982). Preliminary host ranges of some strains of economically important broomrapes (Orobanche). Economic Botany, 36:270–273.
Osipitan O, Hanson B, Goldwasser Y, Fatino M, Mesgaran M. (2021). The potential threat of branched broomrape for California processing tomato: A review. Calif Agriculture, 75(2):64-73. https://doi.org/10.3733/ca.2021a0012.
Osterbauer, N. K., & Rehms, L. (2002). Detecting single seeds of small broomrape (Orobanche minor) with a polymerase chain reaction. Plant Health Progress, 3(1), 1.
Parker, C. (2013). The parasitic weeds of the Orobanchaceae. In Parasitic Orobanchaceae: parasitic mechanisms and control strategies (pp. 313-344). Berlin, Heidelberg: Springer Berlin Heidelberg.
Warren, P. (2006). The branched broomrape eradication program in Australia. In: Preston C, Watts JH, Crossman ND (eds) 15th Australian Weeds Conference, Adelaide, September 2006, pp 610–613.



