- Author: Kathy Keatley Garvey

It's well deserved!
Scott, a UC Davis distinguished professor who officially retired in 2015, is internationally known for his work on the ecology and epidemiology of dengue, a mosquito-borne viral infection transmitted mainly by Aedes aegypti. He now resides in Luck, Wis., where his wife's family is from.
Only 1 percent of researchers make the global list of Highly Recited Researchers, as announced by Clarivate. The Web of Science Group, the information and technology provider for the global scientific research community, annually honors the 1 percent of scientists whose publications are the most cited in scientific papers.
"Of the world's scientists and social scientists, Highly Cited Researchers truly are one in 1,000," according to the Web of Science website.
Scott is one of 14 researchers from UC Davis--and one of some 6660 worldwide--to achieve the 2021 honor.
Scott's 19 publications listed in the report have been cited a total of 402 times. His most cited publication: “The Current and Future Global Distribution and Population at Risk of Dengue,” published in Nature Microbiology in 2019.
“Being a Highly Cited Researcher means a lot to me because it's an objective measure of the extent to which the scientific community finds helpful the work that my colleagues and I did,” said Scott, who joined the UC Davis Department of Entomology (now the Department of Entomology and Nematology) in 1996. “ I was privileged to work with exceptionally smart, hard-working, and insightful people. We had a lot of fun, but we also took our science seriously. We challenged each other in constructive and collegial ways. We are proud of the results of our efforts.”

Healthy Cites, Healthy People. Scott co-chairs a Lancet Commission that focuses on how prevention of viruses transmitted by Aedes mosquitoes fits into the growing Healthy Cities, Healthy People movement. Lancet Commissions are tasked with identifying the most pressing issues in science, medicine, and global health, with the aim of providing recommendations that change health policy or improve practice. “In this case, we are making the case for Cities without Aedes,” Scott said. "We aim to reduce the burden and threat from Aedes transmitted viruses through improved construction and management of modern urban environments that build Aedes mosquitoes out of cities and towns.”
Scott is a collaborator in a clinical trial designed to demonstrate and quantify the protective efficacy of a spatial repellent to reduce human mosquito transmitted virus infection in Sri Lanka. “This new project,” he said, “builds on a randomized controlled clinical trial that my colleagues I recently completed in Iquitos, Peru, which revealed a significant protective efficacy of a spatial repellent against human infection. Publication of those results a currently under review.”
“In addition to the clinical trial, my colleagues and I are publishing a series of papers from Iquitos that examine the role of people with in-apparent and mild infections in virus transmission. By accounting for factors underlying variation in each person's contribution to transmission we will be able to better determine the type and extent of effort needed to prevent disease.”
Scott's other activities include being a scientific advisor for a clinical trial in Brazil that is testing the public health benefit of Wolbachia for prevention of viruses transmitted by Aedes mosquitoes. “The study will assess the efficacy of releasing Wolbachia-infected Aedes aegypti into the environment in reducing human virus infection compared to standard vector control measures alone,” he said.
Scott serves on three World Health Organization committees, “which I find particularly rewarding because of their potential to improve public health policy and thus global health.”
Among his other activities:
- Chair of a group that is writing a chapter on Dengue Vector Control Guidelines that will be included in the updated version of WHO guidelines for dengue diagnosis, treatment, prevention, and control.
- Co-chair of the Wolbachia Evidence Review Group. “Mosquitoes infected with the endosymbionic bacteria Wolbachia are designed to have reduced capacity to become infected with and transmit a variety of viruses, which is expected to reduce human disease,” Scott explains. “Results from our deliberations will help the World Health Organization to develop guidelines for member States on the application of this exciting new intervention strategy.”
- Member of the World Health Organization Technical Advisory Group on the Global Integrated Arboviruses Initiative. The Arbovirus Initiative focuses on strengthening the coordination, communication, capacity building,research, preparedness, and response needed to mitigate the growing risk of epidemics due to arthropod transmitted viral diseases. Scott describes it as “a collaborative effort between the World Health Emergency Program, the Department of Control of Neglected Tropical Diseases, and the Immunization, Vaccines, and Biological Department at WHO. Members of the Technical Advisory Group have a broad range of expertise (clinical management, diagnostics, epidemiology, vector control, virology, vaccines, and travel medicine) and serve in an advisory capacity to WHO with a focus on essential and strategic guidance on management of disease. Our current focus is finalizing the Global Integrated Arboviruses Initiative, which will be presented to the World Health Assembly for review and approval.”
Challenging and Rewarding. “I am excited about and enjoy all these activities,” the medical entomologist commented. “They are challenging and rewarding extensions of the work I have done for that past 40 years.”
Scott, who holds bachelor and master's degrees from Bowling Green (Ohio) State University, received his doctorate in ecology in 1981 from Pennsylvania State University and did postdoctoral research in epidemiology at Yale University School of Medicine's Arbovirus Research Unit, part of the Department of Epidemiology and Public Health. He served on the faculty of the Department of Entomology, University of Maryland, from 1983 to 1996 before joining the UC Davis entomology faculty as a professor of entomology and director of the Vector-Borne Disease Laboratory. He was acting director of the UC Davis Center for Vector-Borne Research from 1996 to 1999, and director of the UC Davis Arbovirus Research Unit (2001-2003). He was selected vice chair of the Department of Entomology in 2006, serving until 2008.
Highly honored by his peers, Scott won the coveted Harry Hoogstraal Medal from the American Society of Tropical Medicine and Hygiene in 2018. His other honors include fellow of three organizations: American Society of Tropical Medicine and Hygiene (2014), Entomological Society of America (2010), and the American Association for the Advancement of Science (2008). He was named a UC Davis distinguished professor in 2014. In 2015, he won the Charles W. Woodworth Award, the highest honor awarded by the Pacific Branch of the Entomological Society of America.
The methodology that determines the “who's who” of influential researchers draws on the data and analysis performed by bibliometric experts and data scientists at the Institute for Scientific Information at Clarivate. It also uses the tallies to identify the countries and research institutions where these citation elite are based.

- Author: Kathy Keatley Garvey

It's an honor well-deserved.
The scientists rank in the top 1 percent by citations, which represent how often their papers have been cited in other scientific papers.
Or, as the website says: "Recognizing the true pioneers in their fields over the last decade, demonstrated by the production of multiple highly-cited papers that rank in the top 1% by citations for field and year in the Web of Science™. Of the world's scientists and social scientists, Clarivate™ Highly Cited Researchers truly are one in 1,000."
Scott, who joined the UC Davis faculty in 1996, retiring in 2015--but not retiring from science--has published 288 papers to date. His total number of citations: more than 33,500. He is internationally known for his work on the ecology and epidemiology of dengue, a mosquito-borne viral infection transmitted mainly by Aedes aegypti.
“Although I retired from UC Davis in 2015, I have continued to carry out research just as I had previously,” Scott said. “In reality, I retired from UC Davis, but I did not retire from science.”
Scott focuses his research on epidemiology of mosquito-borne diseases, mosquito ecology, evolution of mosquito-pathogen interactions, and evaluation of novel products and strategies for disease control.
“I aim to generate the detailed, difficult to obtain data that are necessary for assessing current recommendations for disease prevention, rigorously testing fundamental assumptions in public health policy, and developing innovative, cost, and operationally effective strategic concepts for prevention of mosquito-borne disease.”
Two Large Grants
Scott has remained active on a variety of fronts. In 2015, he was in the early stages of two large grants (National Institutes of Health Program Project grant “Quantifying Heterogeneities in Dengue Virus Transmission Dynamics” and a sub-award from a Bill and Melinda Gates grant, “Spatial Repellent Products for Control of Vector-Borne Diseases”), which he continued to run and manage through the Department of Entomology and Nematology.

Scott served as the program leader of the Program Project grant, “which required a lot of work because it had a large cast of international collaborators,” he explained. “That project examined the role of people with mild or unapparent infections in dengue virus transmission dynamics, similar to what is currently being seen with COVID. We determined that people who don't get sick at all or don't get sick enough to go to the doctor can contribute as much as 85 percent to forward transmission; that is, new infections.”
The Gates grant was a clinical trial to determine whether and to what extent a chemical that repelled mosquitoes would reduce a person's risk of dengue or Zika virus infection. Scott served as the project leader for the Iquitos trial. “Earlier this year we determined that it had a big protective effect,” he related. “We are currently writing that manuscript.”
“Both of those grants ended earlier this year, but we still have a lot of work to do and will be busy for the next couple of years writing papers about those projects,” Scott said. He is currently working as a consultant in a follow-up study on spatial repellents for dengue prevention that will begin in Sri Lanka during 2021
Since retirement he has been--and continues to be--a member of the Management Committee for the NGO (non-governmental) Global Dengue and Aedes-Transmitted Diseases Consortium. He co-chairs a Lancet Commission on Aedes-transmitted viruses that is being organized by the NGO.
World Health Organization
“Working with WHO is important to me because at this stage of my career, being able to translate my science experience into improved quality of life, that is, improved public health policy, for other people, many of whom live in poverty, is the most meaningful thing I can do.”
Scott, who holds bachelor and master's degrees from Bowling Green (Ohio) State University, received his doctorate in ecology in 1981 from Pennsylvania State University and did postdoctoral research in epidemiology at Yale University School of Medicine's Arbovirus Research Unit, part of the Department of Epidemiology and Public Health. He served on the faculty of the Department of Entomology, University of Maryland, from 1983 to 1996 before joining the UC Davis entomology faculty as a professor of entomology and director of the Vector-Borne Disease Laboratory. He was acting director of the UC Davis Center for Vector-Borne Research from 1996 to 1999, and director of the UC Davis Arbovirus Research Unit (2001-2003). He was selected vice chair of the Department of Entomology in 2006, serving until 2008.
Highly honored by his peers, Scott won the coveted Harry Hoogstraal Medal from the American Society of Tropical Medicine and Hygiene in 2018. His other honors include fellow of three organizations: American Society of Tropical Medicine and Hygiene (2014), Entomological Society of America (2010), and the American Association for the Advancement of Science (2008). He was named a UC Davis distinguished professor in 2014. In 2015, he won the Charles W. Woodworth Award, the highest honor awarded by the Pacific Branch of the Entomological Society of America.

- Author: Kathy Keatley Garvey

Travelers know Iquitos as the "capital of the Peruvian Amazon" but scientists know it as a hot spot for dengue, a mosquito-borne viral disease with raging outbreaks in many tropical and subtropical countries.
Amy Morrison, stationed in Iquitos full-time, has directed dengue research activities there for the past 15 years. An epidemiologist who joined the UC Davis laboratory of medical entomologist Thomas Scott (now professor emeritus) in 1996, she's a project scientist and scientific director of the Naval Medical Research Unit No. 6 (NAMRU-6) Iquitos Laboratory.
Morrison is back in the states to present a UC Davis seminar on "Targeting Aedes Aegypti Adults for Dengue Control: Infection Experiments and Vector Control in Iquitos" from 4:10 to 5 p.m., Wednesday, Jan. 10, in 122 Briggs Hall, Kleiber Hall Drive.
Hosted by the UC Davis Department of Entomology and Nematology, it's the first in a series of winter seminars coordinated by assistant professor Rachel Vannette and Ph.D student Brendon Boudinot of the Phil Ward lab.
Dengue is a threat to global health, says Morrison, who holds a doctorate in epidemiology from Yale University and a master's degree in public health from UCLA. According to the World Health Organization (WHO), the incidence of dengue has increased 30-fold over the last 50 years and almost half of the world population is now at risk. It's ranked as "the most critical mosquito-borne viral disease in the world."

"Each year, an estimated 390 million dengue infections occur around the world," according to the World Mosquito Program. "Of these, 500,000 cases develop into dengue hemorrhagic fever, a more severe form of the disease, which results in up to 25,000 deaths annually worldwide."
Of the dengue project in Iquitos, Morrison says: "Comprehensive, longitudinal field studies that monitor both disease and vector populations for dengue viruses have been carried out since 1999 in Iquitos. In addition, to five large scale-vector control intervention trials, ongoing data collection has allowed the evaluation of Ministry of Health emergency vector control using indoor ULV space sprays with pyrethroids in concert with larviciding through multiple campaigns, as well as characterize local DENV (dengue virus) transmission dynamics through two and one novel DENV serotype and strain invasions into the city."
"Our research group has also been conducting contact cluster investigations on DENV-infected and febrile control individuals since 2008," Morrison relates. "These studies demonstrated that attack rates were consistent between houses where cases were first detected and recently visited contact houses independent of distance between these locations. Furthermore, contact cluster investigations allow us to identify viremic individuals across the spectrum of disease outcomes including inapparent infections."
"Using DENV positive individuals captured through these and other febrile surveillance protocols, we exposed laboratory reared (F2) Aedes aegypti mosquitoes directly on their arms or legs, and obtained blood samples with and without EDTA for exposure of mosquitoes in an artificial membrane feeder. After a 58-participant pilot study comparing feeding methods, we initiated a direct feeding protocol exposing participants (78 feeds in 31 participants to date). Feeding, survival, midgut infection and systemic dissemination are all higher using direct feeding than indirect feeding methods. Of 22 participants without detectable fluorescent focus assay titers in their serum at the time they were exposed to mosquitoes, 14 infected mosquitoes by at least one method."
"Although virus titer was a predictor of mosquito infection, mosquitoes became infected at low or undetectable titers and with subjects experiencing mild disease. We have evaluated insecticide-treated curtains and a novel lethal ovitrap (Attractive Lethal OviTrap = ALOT) for dengue control. Only the ALOT traps showed a significant impact on dengue incidence corresponding to a modest decrease in vector densities and a shift of the mosquito population age structure in the trap area to younger mosquitoes. Recent evaluations of indoor ULV interventions with pyrethroids suggest that ULV campaigns that reduce Aedes aegypti for at least 3 weeks through multiple fumigation cycles can mitigate DENV transmission during the same season."
Bottom line: "We argue that Aedes aegypti control should focus on interrupting transmission rather than long-term suppression at operationally unachievable levels and that emergency control should be applied at area-wide scales rather than reacting to individual DENV cases."


- Author: Kathy Keatley Garvey

The mosquito transmits the Zika virus, currently "the" hot medical topic.
But it also transmits dengue, yellow fever and chikungunya viruses.
Especially dengue.
“Dengue infects 400 million people worldwide each year, and 4 billion people or nearly half of the world's population are at risk for dengue,” says medical entomologist Thomas Scott, distinguished professor and now emeritus, UC Davis Department of Entomology and Nematology.
Scott, who has studied dengue more than 25 years and is recognized as the leading expert in the ecology and epidemiology of the disease, emphasizes that “There is no vaccine nor drug that is effective against this virus." There are four serotypes: DENV-1 through DENV-4.
Now for the groundbreaking news.
Scott and his colleagues just published a study in the Public Library of Science (PLOS), Neglected Tropical Diseases, that is sure to rock the world of everyone who has ever contracted dengue.

Well, yes, you can. "Lifetime of immunity" is false.
“Our most significant result from this study is that immunity to dengue viruses does not always provide perfect protection from reinfection,” Scott said. “The public health implications include evaluation of dengue vaccines, interpretation of a person's virus exposure history and susceptibility to new infections, and design of dengue surveillance programs.”
“Our data indicate that protection from homologous DENV re-infection may be incomplete in some circumstances, which provides context for the limited vaccine efficacy against DENV-2 in recent trials,” the research team wrote. “Further studies are warranted to confirm this phenomenon and to evaluate the potential role of incomplete homologous protection in DENV transmission dynamics.”
Former UC Davis researcher Steve Stoddard and senior author of the paper said it well:
“It has long been thought that infection with any one of the viruses produced lifetime immunity to that virus. This finding could help explain results of dengue vaccine trials that showed poor efficacy against one of the four serotype. It also has broad implications for vaccine development.”
The research team investigated the "validity of the fundamental assumption" by analyzing a large epidemic caused by a new strain of DENV-2 that invaded Iquitos, Peru, in 2010-2011, 15 years after the first outbreak of DENV-2 in the region.
Read more about the research on the UC Davis Department of Entomology and Nematology website and in PLOS, Neglected Tropical Diseases.

- Author: Kathy Keatley Garvey

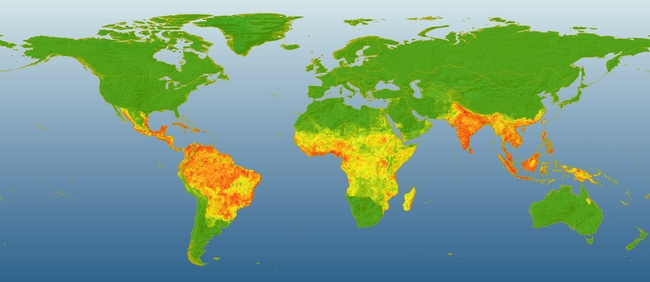
The mosquito-borne viral disease known as “breakbone fever,” is three times more prevalent than originally thought, according to a research paper published today in Nature and co-authored by dengue expert Thomas Scott of UC Davis.
In their research paper, titled “The Global Distribution and Burden of Dengue,” Scott and the 17 other team members estimated that 350 million people are infected each year--more than triple the World Health Organization’s current estimate of 50 to 100 million.
Professor Simon Hay of the University of Oxford led the research as part of the International Research Consortium on Dengue Risk Assessment, Management and Surveillance.
“Dengue takes an enormous toll on human health worldwide, with as many as 4 billion people at risk," said Scott, a UC Davis professor of entomology and worldwide expert on the epidemiology and prevention of dengue. He chairs the mosquito-borne disease modelling group in the Research and Policy for Infectious Disease Dynamics (RAPIDD) program of the Science and Technology Directory, Department of Homeland Security, Fogarty International Center, National Institutes of Health.
“The results of our study and infrastructure that created the dengue maps fill a critical gap in the battle against dengue,” said Scott, who maintains field research programs in Iquitos, Peru, and Khamphaeng Phet, Thailand. “Prior to this, without rigorously derived dengue estimates that can be continuously updated, it was not possible to know with confidence where and when to direct interventions for greatest potential impact or to objectively assess the effectiveness of regional and global control efforts. That kind of knowledge was among the most important missing information for developing enhanced dengue prevention programs.”

The highly infectious tropical and subtropical disease is spread by the bite of an infected female Aedes aegypti, a day-biting, limited flight-range mosquito that prefers human blood to develop its eggs. Dengue is caused by four distinct, but closely related, viruses. The most severe form of disease is life-threatening dengue hemorrhagic fever or DHF.
The researchers assembled known records of dengue occurrence worldwide and used a formal modelling framework to map the global distribution of dengue risk. They then paired the resulting risk map with detailed longitudinal information from dengue cohort studies and population surfaces to infer the public health burden of dengue in 2010.
“There are currently no licensed vaccines or specific therapeutics, and substantial vector control efforts have not stopped its rapid emergence and global spread,” the researchers wrote.
Dengue has now begun to appear along the southern border of the United States, including Texas. Florida has also reported cases of dengue.
Of the 96 million clinically apparent dengue infections, Asia bears 70 percent of the burden, the research paper revealed. India alone accounts for around one-third of all infections.