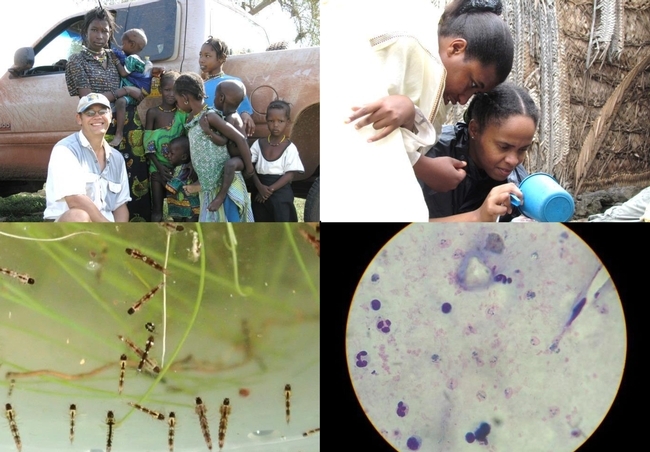
- Author: Kathy Keatley Garvey

"We are working with scientists and public health authorities in STP to establish the conditions that would facilitate an informed societal and government decision about a proposed release of Anopheles mosquitoes engineered to prevent transmission of the malaria parasite Plasmodium falciparum on the islands,” said principal investigator Gregory Lanzaro, director of the Vector Genetics Laboratory and a PMI professor.
This award will be used to extend their ongoing entomological, engagement and capacity building work through 2025.
“We are working in collaboration with the UC Irvine Malaria Initiative (UCIMI), a research consortium including scientists from UC Irvine, San Diego and Berkeley as well as Johns Hopkins University,” Lanzaro said. “We are working toward the application of advanced genetic tools aimed at the mosquito vector. It is our belief that this approach, used in conjunction with early malaria treatment and detection, can provide a cost effective, sustainable, and environmentally responsible program to ultimately eliminate malaria from Africa.”
Said Ana Kormos, engagement program manager and lead author of the proposal: “These funds provide the UCIMI program with support to strengthen our existing relationship-based approach to the co-development of this technology and ensures that our partners in STP lead the decision-making processes involved in all aspects of the research. This is a huge step forward in advancing a truly collaborative approach to translational research.”

The Vector Genetics Laboratory is engaged in research and training in the areas of population and molecular genetics, genomics and bioinformatics of insect vectors of human and animal disease. The website: “We have developed a program aimed at expanding knowledge that may be applied to improving control of disease vectors and that also addresses problems of interest in the field of evolutionary genetics. We are currently engaged in a range of projects, but our major research focus is on vectors of malaria in Africa."
Directors of the Vector Genetics Laboratory research programs are Lanzaro and Anthony "Anton" Cornel, a research entomologist with the UC Davis Department of Entomology and Nematology and director of the Mosquito Control Research Laboratory, Parlier.
New Tools. "The fight to reduce and possibly eliminate malaria continues and becomes especially challenging as efforts to reduce malaria morbidity have plateaued since 2015,” said Cornel. “Therefore, we must seriously consider new tools. One such tool is genetically modifying the major mosquito vector in the Afrotropics so that it cannot transmit malaria."
"The project aims to use genetically modified (GM) mosquito strategy to reduce and eliminate malaria from the Islands of São Tomé and Príncipe, as proof of concept, before using this technology on larger scales on mainland Africa,” Cornel said, adding that his role, as a field team co-investigator for UCIMI and VGL, is to work with Lanzaro and Pinto “to understand as much as we can about the behavior, population structure and population sizes of Anopheles coluzzi (the malaria vector) on these islands to design the most efficient strategy of releasing the genetically modified mosquitoes to have maximum effect.”
Malaria is an acute illness caused by Plasmodium parasites, which spread to humans through the bites of infected female Anopheles mosquitoes, according to the World Health Organization (WHO). In 2020, nearly half of the world's population was at risk of malaria. An estimated 241 million cases of malaria occurred worldwide in 2020, with 627,000 dying.
Tremendous Burden. Medical entomologist and geneticist Geoffrey Attardo of the UC Davis Department of Entomology and Nematology (who is not involved in this project), noted that “Malaria is a disease which creates a tremendous burden on people living in affected areas. In particular its impacts on the mortality in young children and pregnant women are devastating. Attempts to control this disease using traditional methods have been effective in recent years.”
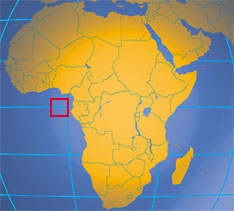
The island nation of São Tomé and Príncipe, population of 178,700 in 2016, is located about 200 miles west of Gabon on Africa's mainland. It shares maritime borders with Equatorial Guinea, Gabon, and Nigeria. The combined area of the archipelago is about five times the size of Washington, DC. The United States established diplomatic relations with São Tomé and Príncipe in 1976, following its independence from Portugal.
Open Philanthropy's mission, as noted on its website, is to “give as effectively as we can and share our findings openly so that anyone can build on our work. Through research and grant-making, we hope to learn how to make philanthropy go especially far in terms of improving lives. We're passionate about maximizing the impact of our giving, and we're excited to connect with other donors who share our passion.”
Resource:
São Tomé and Príncipe (nationsonline.org)


- Author: Kathy Keatley Garvey

The work, published in the current edition of Parasites & Vectors, a BioMed Central open-access medical journal, focuses on “determining how informative well-established genetic markers of resistance to pyrethroids are in predicting the resistance phenotype of individual mosquitoes of Aedes aegypti within a population,” said lead author Geoffrey Attardo, medical entomologist-geneticist in the UC Davis Department of Entomology and Nematology.
“Specifically, we generated mosquito colonies from invasive A. aegypti populations from four locations in the Central Valley (Dinuba, Clovis, Sanger and Kingsburg) and from collections in the Greater Los Angeles Area,” he said. “Mosquitoes from these populations have all demonstrated resistance to pyrethroid-type insecticides and we think this may be part of the reason why these mosquitoes have been so successful in spreading throughout California.”
A. aegypti transmits such viruses as dengue, Zika, chikungunya, and yellow fever. Despite California's aggressive surveillance and treatment efforts, this species presents a “significant challenge to local control agencies,” the nine-member team wrote in their research paper, “Frequency of Sodium Channel Genotypes and Association with Pyrethrum Knockdown Time in Populations of Californian Aedes aegypti.“
The paper is online and publicly accessible at https://bit.ly/3vmUxXR.
“What was interesting was that while all the mosquitoes from California show resistance to pyrethroids, there is a lot of variability from one individual to the next in terms of the level of resistance, even when they are carrying genetically identical resistance mutations,” Attardo said. “In particular, there seem to be two levels of resistance in these populations. The two levels seem to represent a resistant group and a super resistant group. However, the proportions of resistant/super-resistant differ in the sampled mosquitoes from population to population.”
Of particular interest was that mosquitoes carrying the resistance mutations at all five genetic locations were very resistant, he said. “However, there was also a large amount of unexplained variability in terms of the knockdown phenotypes demonstrated by mosquitoes of the same age and rearing conditions. We compared the knockdown times of mosquitoes positive for all five resistance mutations from different populations and found that these mutations account for only a proportion of the observed level of resistance. We believe that the unexplained variability is likely being mediated by the presence or absence of an undefined resistance mechanism.”

In launching the project, the researchers designed an assay “to test for the presence of mutations in the gene coding for the pyrethroid target protein, the voltage gated sodium channel (the para gene),” Attardo explained. “Detection of these mutations is used to monitor the level or resistance in populations. However, the actual link between the effect the genotype has on the phenotype of individual mosquitoes has not been looked at in detail. “
The scientists identified mutations from genetic sequences of Californian mosquitoes provided by co-author Yoosook Lee, a former UC Davis mosquito researcher now at the University of Florida-Florida Medical Entomology Laboratory, Vero Beach.
The authors also include research entomologist Anthony Cornel and staff research associate Katherine Brisco of the Mosquito Control Research Laboratory, Kearney Agriculture and Extension Center and UC Davis Department of Entomology and Nematology; and Lindsey Mack, Erin Taylor Kelly, Katherine Brisco, Kaiyuan Victoria Shen, Aamina Zahid, and Tess van Schoor, all with the UC Davis Department of Entomology and Nematology.
First, they tested the individual resistance phenotype of mosquitoes by placing them into bottles coated with the pyrethroid insecticide permethrin, and observed them to determine how long it takes for them to respond to the insecticide. Said Attardo: “This is a modified version of the assay used by the Center for Disease Control and Prevention to evaluate phenotypic resistance in groups of mosquitoes.”
Then they isolated the DNA from and performed a high-throughput genetic analysis on each individual to determine the composition of the five mutations in each individual. Next they looked at the resulting data to see how well knockdown time correlates with individual genotypes of mosquitoes.
Although A. aegypti was first detected in California in 2013, researchers believe that its arrival involved multiple introductions. Populations in Southern California are thought to have crossed the border from Mexico, while Central Valley populations may have been introduced, in part, from the southeastern United States.
“Upon detection in 2013, the Consolidated Mosquito Abatement District implemented an integrated vector control management strategy which involved extensive public education, thorough property inspections, sanitation, insecticide treatment at larval sources and residual barrier spraying with pyrethroids,” the authors wrote. Despite their efforts, the species successfully overwintered and continued to spread, implicating that it arrived in California with genetic mutations “conferring resistance to the type I pyrethroid insecticides applied for vector control in California.”
The project drew financial support from the Pacific Southwest Regional Center of Excellence for Vector-Borne Diseases, funded by the U.S. Centers for Disease Control and Prevention.


- Author: Kathy Keatley Garvey

The research, published in the Sept. 15 edition of PLOS Genetics, involved the study of Anopheles arabiensis, in Kilombero Valley in Tanzania. The mosquito is the primary vector of malaria in east Africa.
"We know that blood feeding preference among mosquitoes can be species specific,” said co-author and professor Greg Lanzaro, who leads the Vector Genetics Laboratory, UC Davis Department of Pathology, Microbiology and Immunology and is an affiliate of the UC Davis Department of Entomology and Nematology. “For example, there are mosquito species that specialize in feeding on amphibians or reptiles. We also know that many species are more catholic when choosing a meal and this can have important implications to human health—it's how some disease agents move between animals and humans.”

The publication is the work of a 13-member international team. Bradley Main, a researcher in the Vector Genetics Lab, is the lead author.
“Whether there is a genetic basis to feeding preferences in mosquitoes has long been debated,” said lead author Bradley Main, a researcher in the Vector Genetics Lab. “Using a population genomics approach, we have established an association between human feeding and a specific chromosomal rearrangement in the major east African malaria vector. This work paves the way for identifying specific genes that affect this critically important trait.”
Other co-authors, in addition to Lanzaro, are Anthony Cornel of the UC Davis Department of Entomology and Nematology faculty; researchers Yoosook Lee, Heather Ferguson, Travis Collier, Catelyn Nieman, Allison Weakley, all of the Vector Genetics Lab; Katharina Kreppel, Nicodem Govella and Anicet Kihonda of the Ifakara Health Institute, Ifakara, United Republic of Tanzania; and computer scientists Eleazar Eskin and Eun Yong Kang of UCLA.
In their summary, they wrote: “Malaria transmission is driven by the propensity for mosquito vectors to bite people, while its control depends on the tendency of mosquitoes to bite and rest in places where they will come into contact with insecticides. In many parts of Africa, where coverage with Long Lasting Insecticide Treated Nets is high, Anopheles arabiensis is the only remaining malaria vector. We sought to assess the potential for An. arabiensis to adapt its behavior to avoid control measures by investigating the genetic basis for its host choice and resting behavior. Blood fed An. arabiensis were collected resting indoors and outdoors in the Kilombero Valley, Tanzania. We sequenced a total of 48 genomes representing 4 phenotypes (human or cow fed, resting in or outdoors) and tested for genetic associations with each phenotype. Genomic analysis followed up by application of a novel molecular karyotyping assay which revealed a relationship between An. arabiensis that fed on cattle and the standard arrangement of the 3Ra inversion. This is strong support that An. arabiensis blood-feeding behavior has a substantial genetic component. Controlled host choice assays are needed to confirm a direct link between allelic variation within the 3Ra inversion and host preference.”
The publication, "The Genetic Basis of Host Preference and Resting Behavior in the Major African Malaria Vector, Anopheles arabiensis," is online at http://journals.plos.org/plosgenetics/article?id=10.1371/journal.pgen.1006303

- Author: Kathy Keatley Garvey

Male Aedes aegypti mosquitoes infected with a bacterium, Wolbachia pipientis, are being released in Clovis, Fresno County, where this mosquito was discovered in June 2013. Although this mosquito is now found in California, there has been no locally transmitted case of the Zika virus in the state.
The project, to determine dispersal and survival, began Monday, May 10.
“The daytime-biting mosquito, which feeds predominantly on humans, has spread to at least seven counties since its discovery in Clovis,” said Cornel, a mosquito researcher and faculty member with the UC Davis Department of Entomology and Nematology and the UC Kearney Agricultural Research and Extension Center, Parlier.
“The biting nuisance and potential of the mosquito Aedes aegypti to transmit Zika, Chikungunya and dengue viruses in California is cause for concern,” he said. “Efforts to curb its spread and reduce populations have not been very effective. Control efforts have included educating the public to remove standing water (source reduction) insecticide barrier sprays and bacterial larviciding.”
The collaborative effort involves Steve Mulligan, director of the Consolidated Mosquito Abatement District, based in Fresno County; Stephen Dobson of the University of Kentucky; MosquitoMate Inc, (mosquitomate.com); and UC Davis. The researchers will evaluate the population suppressive ability of the novel sterile insect technique, which is part of a comprehensive vector management approach.
Only Aedes aegypti is targeted. “When the Wolbachia-infected male mosquito mates with non-infected females, the result is “cytoplasmic incompatibility, which causes the female to lay infertile eggs that will not hatch,” Cornel explained.
“This approach requires the release of tens of thousands of Wolbachia-infected males into residential neighborhoods where this mosquito is a nuisance,” the medical entomologist said. “Releasing large numbers of males increases the chance that an introduced male will mate with the native females.”
Although residents will notice increased numbers of male mosquitoes, male mosquitoes do not bite and cannot transmit disease. Both U.S,federal and State regulatory agencies have approved the technique for evaluation of effectiveness.
“This sterile insect technique was evaluated in 2015 in Los Angeles to suppress another invasive mosquito, Aedes albopictus,” Cornel said, adding that the results from that trial look promising.
The Zika virus, now spreading throughout the Western hemisphere, was first identified in Uganda in 1947 in rhesus monkeys, according to the World Health Organization. It was subsequently identified in humans in 1952 in Uganda and the United Republic of Tanzania. Outbreaks of Zika virus disease have been recorded in Africa, the Americas, Asia and the Pacific.
Despite the mosquito's invasion into new areas of the United States, there are no reported cases of locally transmitted Zika virus in California or in the contiguous United States, according to the Centers for Disease Control and Prevention. The cases have all involved travelers returning home from countries plagued with disease outbreaks.
“We can't predict how far this mosquito will spread in California,” said Cornel, noting that its range has expanded “south of Fresno to San Diego. The farthest site north is Madera in the Central Valley, but it's also been found in the more coastal area of Menlo Park in San Mateo.”
It's troubling that the mosquito is becoming more and more resistant to pesticides, said Cornel, who collects, rears and researches mosquitoes from all over the world, including the United States, Mali, Cameroon, Comoros, Tanzania, South Africa and Brazil.
“We have found that Aedes aegypti have insecticide resistance genes which likely explains why the use of ultra-low volume and barrier spray applications for control have not worked as well as expected.”

- Author: Kathy Keatley Garvey

The event will take place in MU II (second floor) and is free and open to the public.
It's being held "to increase awareness among the general public about malaria, one of the world's oldest and deadliest diseases, as well as vector-borne problems at home in California," said medical entomologist Gregory Lanzaro, professor in the Department of Pathology, Microbiology and Immunology, UC Davis School of Veterinary Medicine.
Malaria "is a leading cause of death and disease in many developing countries, where young children and pregnant women are the groups most affected," the Center for Disease Control and Prevention (CDC) points out, citing these figures from the World Health Organization's World Malaria Report 2013 and the Global Malaria Action Plan:
- 3.4 billion people (half the world's population) live in areas at risk of malaria transmission in 106 countries and territories
- In 2012, malaria caused an estimated 207 million clinical episodes, and 627,000 deaths. An estimated 91% of deaths in 2010 were in the African Region.
The most vulnerable groups, CDC says, are young children, who have not yet developed partial immunity to malaria; pregnant woman, whose immunity is decreased by pregnancy, especially during the first and second pregnancies; and travelers or migrants coming from areas with little or no malaria transmission, who lack immunity.
The schedule for the UC Davis Malaria Awareness Day:
- 10 to 10:30 am.: Coffee/social/posters
- 10:30 to 10:50: "General Malaria Biology" by medical entomologist Gregory Lanzaro, professor, Department of Pathology, Microbiology and Immunology, UC Davis School of Veterinary Medicine.
- 10:50 to 11:20: Conducting Field Research in Rural Africa" by medical entomologist Anthony Cornel, associate professor, UC Davis Department of Entomology and Nematology and based at the UC Kearney Agriculture and Research Center, Parlier
- 11:10 to 11:30: "Malaria Parasites in the Mosquito" by molecular biologist Shirley Luckhart, professor, UC Davis Department of Medical Microbiology and Immunology and an adjunct professor in the Department of Entomology and Nematology
- 11:30 to 11:50: "Mosquito-Borne Viral Diseases" by medical entomologist Chris Barker, assistant adjunct professor and assistant research scientist, UC Davis Department of Pathology, Microbiology and Immunology
- 11:50 to 12:10: "Disease Transmission by Non-Mosquito Vectors" by epidemiologist/veterinarian and disease ecologist Janet Foley, professor, UC Davis Department of Medicine and Epidemiology
- 12:10 to 1:30: A free lunch will be provided, but reservations must be made by April 21 to Youki Kevin Yamasaki at ykyamasaki@ucdavis.edu.