- Author: Luis A Espino
It is time again for the Rice Diseases Survey. The objective of the Survey is to document the incidence of rice diseases in Sacramento Valley rice fields so that the Air Resources Board agrees to continue to allow the rice industry to burn up to 25% of the acreage. Every five years, the Board has to re-approve the straw burning program. One of the reasons for letting growers still burn is disease control. Therefore, it is important to show that diseases are still present in rice fields. This is the third Rice Diseases Survey Cooperative Extension is conducting. The first was in 2014 and the second in 2019.
A crew of four to five will be stopping in random fields in the major rice producing counties and inspecting for the presence of diseases in 30 sites in each field. We will look for stem rot, aggregate sheath spot, kernel smut, and blast. The precise location of each inspected field won't be disclosed, only the general location and County.
If you have any questions or concerns, contact me at laespino@ucanr.edu or 530-635-6234.
- Author: Luis A Espino
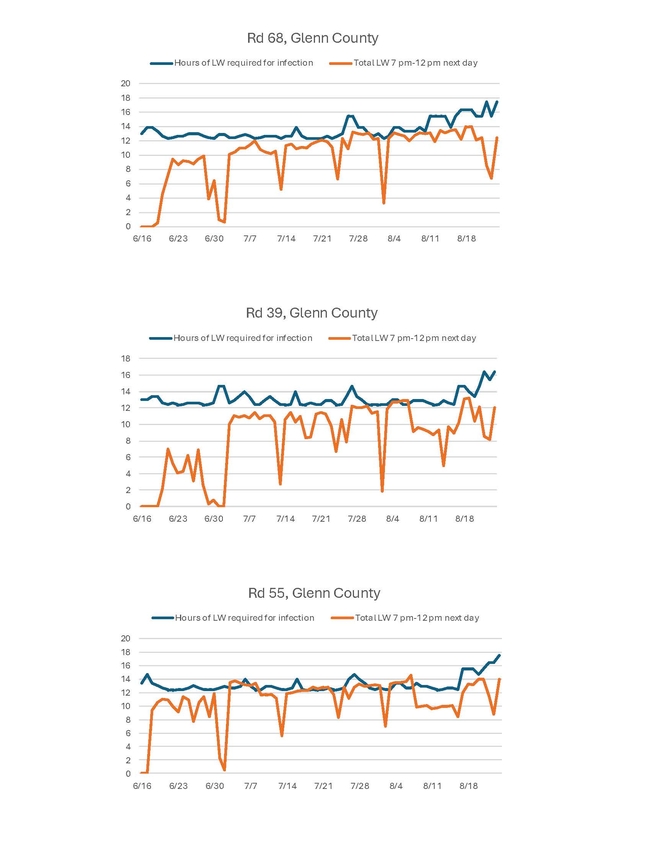
I just wanted to do a quick update on blast. I have not seen any other blast besides the one I reported on an earlier post. PCAs that I have spoken to have not seen blast either. With the cooler temperatures, conditions for blast are not as good. The hours of leaf wetness (LW) required for infection are higher when temperatures are cooler (see figures below). We'll see what happens as temperatures warm up again, but we know that as the crop matures, blast infections are less likely. However, I don't want to say we won't see any blast - last year we saw blast infect some fields late in the season. We'll keep monitoring.
- Author: Luis A Espino
Yesterday, I visited a farm with blast in Glenn County. In this case, the grower seeded M-210, and as an experiment, he planted some M-105 in a very small area at the time of clean up herbicide application. This is a location where blast is common. In fact, last year this location had severe neck blast.
Blast showed up on the M-105 but the M-210 is completely clean. The picture below shows some burned leaves and lesions that could be confused with herbicide burn. However, there are plenty of the typical diamond shaped lesions.
Small area of M-105 surrounded by M-210.
Blast burned down some of the M-105 leaves, resembling a herbicide burn. The M-210 does not have any lesions.
Typical blast lesions found around the M-105 burned leaves.
- Author: Sarah L Marsh
We are excited to announce the launch of a new rice podcast, "Thoughts on Rice", hosted by the UCCE Rice Advisors and found wherever you listen to podcasts.
This podcast is for growers, PCAs, consultants, and other industry professionals in the California rice industry. We'll primarily be focusing on the Sacramento Valley and Delta Region of California. We aim to deliver extension information relating to the California rice industry, but we are also looking for suggestions for topics that would be of interest to our stakeholders! Reach out to your local farm advisor for more information.
The podcast website can be found here.
The feedback poll can be found here:
- Author: Taiyu Guan
Date: August 28th, 2024
Location: Rice Experiment Station
(955 Butte City Hwy., Biggs, CA)
** No Registration Required! **
Meeting Agenda:
7:30 - 8:30 am Registration
- Posters and Demonstrations
8:30 - 9:15 am General Session
- CCRRF Annual Membership Meeting
- Rice Research Trust Report
- California Rice Industry Award
9:30 - Noon Field Tours of Rice Research
- Variety Development
- Disease and Insect Management
- Agronomy and Fertility
- Weed Management
12:00 (Noon) Lunch
2024-Invitation-to-Field-Day