- Author: Steven T. Koike
Situation: During the latter part of the 2015 Salinas Valley season (August through October), significant crop losses have been caused by Pythium wilt disease of lettuce. This disease appeared to be only recently introduced to coastal California, and prior to 2014 seemed to be of minor importance. However, during 2014 and 2015 seasons, the problem spread to a number of locations in the valley and caused perhaps 30% or more losses in some fields.
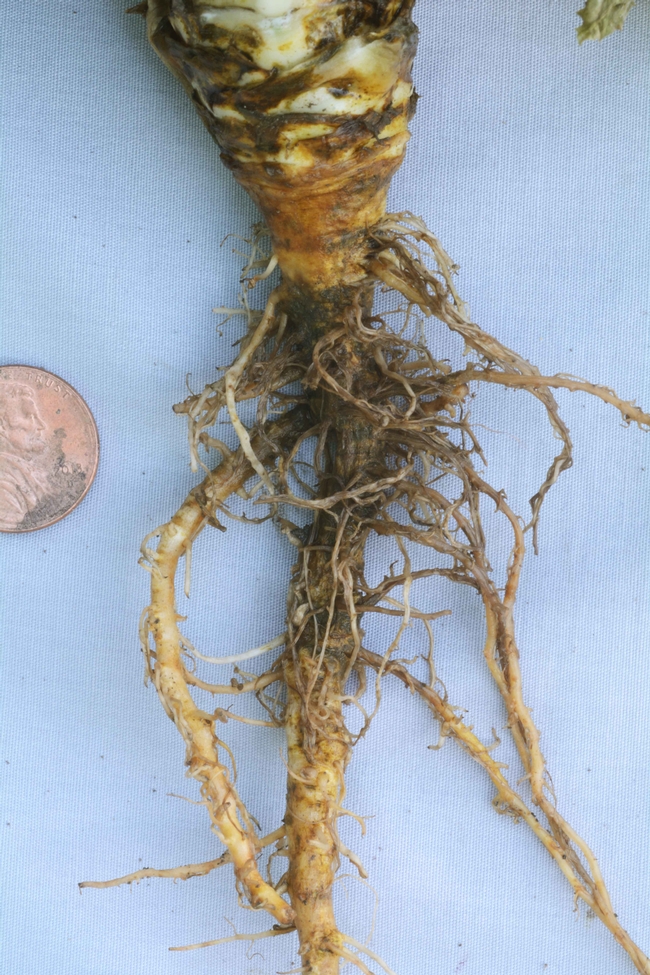
Symptoms: Above ground symptoms of Pythium wilt develop on lettuce that is at the rosette stage or older. In contrast to Pythium root rots of spinach and other vegetables, this lettuce pathogen does not cause damping-off of newly emerged, young lettuce seedlings. Infected plants will be stunted and lag behind healthy lettuce. As disease progresses, outer leaves will start to wilt during the warmer times of the day and eventually turn yellow before becoming brown and dead. In advanced stages, the entire foliar canopy likewise can wilt and senesce; such plants clearly are not harvestable. Below ground, the pathogen first attacks the small feeder roots of the lettuce, making them soft and brown gray in color. Late in disease development the taproot will also be darkly discolored and the entire root system can be rotted. Pythium wilt does not cause a rot of the lettuce crown. Pythium wilt has so far been confirmed on iceberg (crisphead), romaine, and greenleaf cultivars. See photos below.
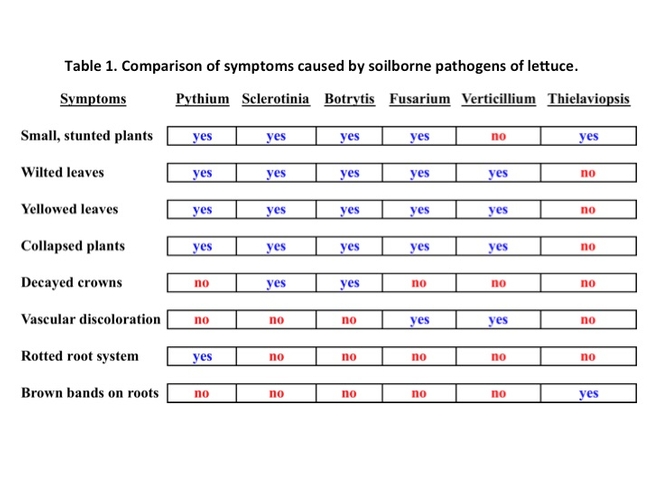
Diagnostic challenge: Because Pythium wilt causes a general wilting and collapse of lettuce foliage, this disease can be confused with other problems. Sclerotinia and Botrytis infections both can result in plant wilt and collapse. However, in these cases the symptoms result from crown infections; neither Sclerotinia nor Botrytis infect lettuce roots. Verticillium and Fusarium wilts, which also cause lettuce to collapse, will result in distinctive discolorations of the lettuce vascular tissues while leaving the roots intact. The other root disease new to Salinas Valley lettuce growers, black root rot (pathogen: Thielaviopsis basicola), causes dark bands to form on roots but does not result in the extensive feeder and tap root decay as seen with Pythium. Finally, foliar symptoms caused by Impatiens necrotic spot virus or Lettuce necrotic stunt virus can add further confusion to the diagnostic task because of the yellowing and browning of infected lettuce leaves. Accurate diagnosis of these lettuce diseases usually requires laboratory analysis. For assistance in diagnosing these problems, submit samples to the UC Cooperative Extension diagnostic lab in Salinas. See Table 1 for a comparison of symptoms caused by soilborne pathogenic fungi of lettuce.
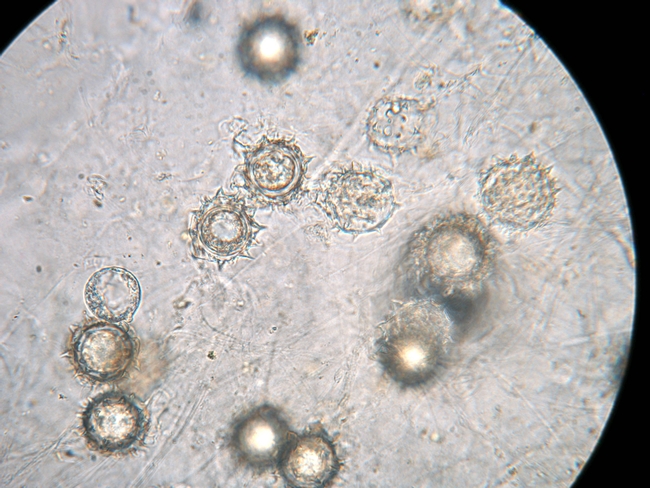
Causal pathogen: Pythium wilt is caused by Pythium uncinulatum. In addition to California, this lettuce pathogen has been reported from The Netherlands, Japan, and Arizona. California's first case of P. uncinulatum on lettuce was from the Coachella Valley in 1993. This pathogen was first found in the Salinas Valley in 2011. Pythium uncinulatum, like most Pythium species, produces swimming spores (zoospores) that are released and move within the water film in the soil. In addition to zoospores, the pathogen also produces a sexual spore (oospore) that is encased within a spiny outer covering (oogonium). It is the oospore that allows the pathogen to survive in the soil in the absence of susceptible plants. Pythium uncinulatum is host specific to lettuce and does not infect other vegetable crops such as broccoli, cabbage, carrot, onion, pepper, radish, spinach, or tomato.
Disease cycle: Pythium species are soil inhabitants and persist in most agricultural soils for extended periods of time, especially if soils are moist. Specific information on how Pythium uncinulatum might persist in coastal California soils is lacking; however, we assume this pathogen can persist in soil for a significant amount of time. The pathogen and its ability to infect lettuce are favored by wet soil conditions. Flowing water and movement of soil will spread the pathogen.
Disease management: The following management strategies should be considered when dealing with Pythium wilt disease. (1) Avoid planting lettuce into fields with a known history of the problem. (2) For infested fields, rotate to non-lettuce crops. Various research studies demonstrated that Pythium uncinulatum is host-specific to lettuce. (3) Implement field sanitation practices to minimize the movement of contaminated soil from infested to clean fields. (4) Be aware that surface water run-off from infested fields may contain the pathogen. Flooding events may also spread the pathogen to previously clean fields. (5) Prepare beds so that drainage of water is enhanced, since the pathogen is favored by wet soil conditions. (6) Manage the irrigation so that excessive soil moisture is avoided. (7) The effectiveness of fungicides for controlling Pythium wilt in California is currently unknown and will require field trials.
Photo 1. Romaine lettuce field severely infected with Pythium uncinulatum.
Photo 2. Greenleaf lettuce severely infected with Pythium uncinulatum.
Photo 3. Severely stunted lettuce plants affected by Pythium wilt disease. A healthy plant is on the left.
Photo 4. Dark, rotted feeder roots of lettuce infected with Pythium uncinulatum. Healthy roots are on the right.
Photo 5. In severe cases, Pythium uncinulatum will completely rot the lettuce taproot.
Photo 6. Pythium uncinulatum forms spiny structures called oospores that persist in the soil.
- Author: Richard Smith
1st Announcement- Save the Date
2015 Salinas Valley Weed School
Thursday, November 12
8:00 a.m. to 12:00 noon
Agricultural Center Conference Room
(1432 Abbott Street, Salinas)
This meeting will cover a number of new techniques for controlling weeds in vegetable crops. In addition the effect of weeds on bagrada bug populations and new mechanical tools will be discussed.
4.0 Continuing education credits have been applied for. Please call ahead for special accommodations.
For more information call Richard Smith (831) 759-7357
See attached file for full agenda.
2015 Weed School Agenda
- Author: Cheryl Reynolds
The UC Statewide Integrated Pest Management Program (UC IPM) put together a 26-page card set in English and Spanish on understanding pesticide labels. Intended for pesticide handlers, applicators, safety trainers, and pest control advisers (PCAs), the cards explain when to read the label, describe what kind of information can be found in each section of a pesticide label, and point out specific instruction areas so that applicators can apply pesticides safely and avoid illegal pesticide residues.
Traces of pesticide residue are normal and even expected after pesticides are applied to food crops, but by the time produce is ready to be sold, purchased, and consumed, residues are usually far below the legal limit.
In its latest report from 2013, the California Department of Pesticide Regulation (DPR) reported that there was little or no detectable pesticide residue in 97.8% of all California-grown produce. This demonstrates a strong pesticide regulation program and pesticide applicators that apply pesticides safely and legally. However, there have been instances in California where a pesticide not registered for a specific crop has been used unintentionally, resulting in illegal residues and eventually crop loss and destruction.
The Environmental Protection Agency (EPA) sets tolerances for the maximum amount of pesticide residue that can legally be allowed to remain on or in food.
DPR regularly monitors domestic and imported produce for pesticide residues and is considered the most extensive state residue-monitoring program in the nation.
The primary way pesticide applicators can assure that they make proper applications and avoid illegal pesticide residues is to follow the pesticide label. UC IPM's new card set was developed from information in the upcoming third edition of The Safe and Effective Use of Pesticides as well as Lisa Blecker, UC IPM's Pesticide Safety Education Program coordinator. Bound with a spiral coil, this eye-catching instructional card set was designed for both English-speakers and when flipped over, for Spanish-speaking audiences as well. UC IPM also plans to release a new online course on preventing illegal pesticide residues sometime late fall.
To download copies of the card set in English or in Spanish, see the UC IPM web site.

- Author: Shimat Villanassery Joseph
Save the Date!
Entomology Seminar 2015:
Tuesday, December 1, 2015
8:00 AM to 12:00 PM
County of Monterey Agricultural Center
Conference Room
1432 Abbott Street, Salinas, California
This seminar will focus on a broad range of topics dealing with arthropod pest management in coastal California, and current issues affecting growers, pest control advisors, and other agricultural professionals.
Registration/sign-in is from 7:30 to 8:00 AM. There is no fee for this meeting. Continuing education credits will be requested. Please call ahead (at least 24 hours) for arrangements for special needs; every effort will be made to accommodate full participation. For more information, contact Shimat Joseph (831-229-8985; 1432 Abbott Street, Salinas, California 93901).
Requirement from California DPR: Bring your license or certificate card to the meeting for verification when signing in for continuing education units.
- Author: Shimat Villanassery Joseph
Cabbage maggot (Delia radicum) is a serious insect pest of Brassica crops such as broccoli and cauliflower in the Central Coast of California. These crops are grown throughout the year; as a result cabbage maggot problems persist year long.Cabbage maggot eggs are primarily laid in the soil around the crown area of the plant. A single female fly can lay 300 eggs under laboratory conditions. The eggs hatch within 2-3 days and the maggots feed on the taproot for up to three weeks and can destroy the root system of the plant. The maggots pupate in the soil surrounding the root system and emerge into flies within 2-4 weeks. Severe cabbage maggot feeding injury to the roots cause yellowing, stunting even plant death.
Control of cabbage maggot on Brassica crops primarily involves the use of soil applied organophosphate insecticides such as chlorpyrifos and diazinon. However, the persistent use of organophosphate insecticides has resulted in high concentrations of the insecticide residues in the water bodies posing risks to non-target organisms and public health through contaminated water. Currently, use of organophosphate insecticides is strictly regulated by California Department of Pesticide Regulation. There is therefore an urgent need to determine the efficacy of alternate insecticides for cabbage maggot control.
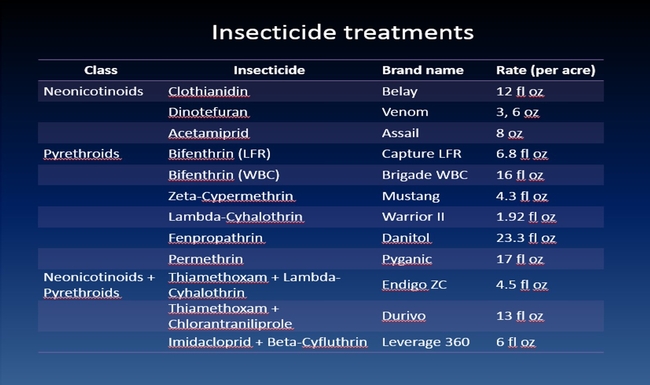
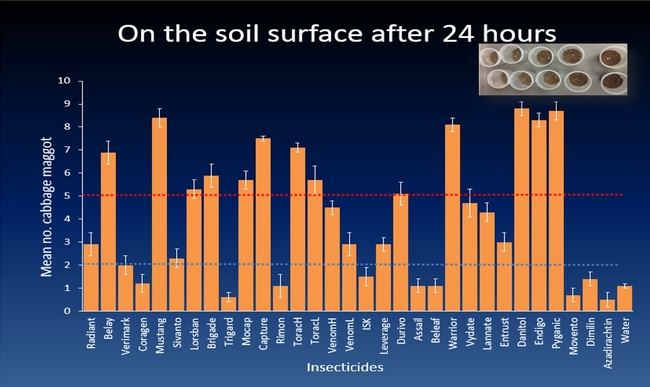
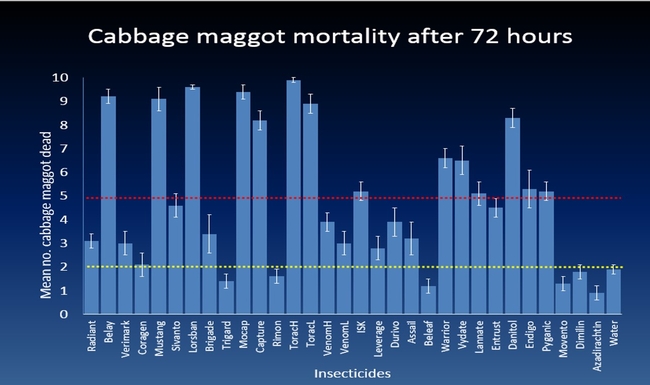
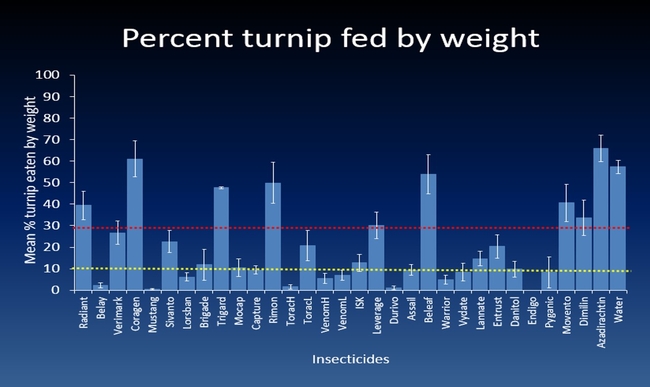
The efficacy of 29 insecticides was determined against cabbage maggot through a laboratory bioassay by exposing field collected maggots to insecticide treated soil immediately after application. Three parameters were used to evaluate efficacy (1) proportion of maggots on the soil surface after 24 h, (2) proportion of change in weight of turnip bait, and (3) dead maggots after 72 h. Based on the assays, 11 insecticides performed better and they were Mustang, Torac, Danitol, Belay, Capture, Warrior II, Lorsban, Mocap, Durivo, Pyganic and Vydate in the order of highest to lowest efficacy. Eight insecticides were selected based on superior efficacy to determine the length of residual activity on cabbage maggot larvae. The persistence of insecticide activity was greater with Capture, Torac and Belay than with other insecticides tested.
The mode of exposure of insecticides in this study was entirely by contact (through skin) and other modes of exposure such as ingestion (through mouth) or through respiratory holes (spiracles) were not investigated. Some of the insecticides tested in the study were insect growth regulators (IGRs) (Dimilin, Rimon, Trigard, and Aza-direct), which normally interfere with the growth and development of the insect and they showed a low efficacy against cabbage maggot larvae. Entrust (spinosad) showed a moderate efficacy possibly because the primary mode of exposure to Entrust is by ingestion. The diamide insecticides (Beleaf, Coragen and Verimark) have systemic activity as they move within the plant and likely away from the site of application. It is possible that the soil applied diamide insecticides are absorbed by the roots and translocated to the above ground plant parts with little effect on the feeding larvae in the tap roots.
This study was conducted under controlled conditions in the laboratory and the results may not be entirely consistent in field conditions. The Brassica fields in the California's Central Coast are profusely sprinkler irrigated up to three weeks after sowing to ensure uniform germination and proper establishment of plants. It is likely that applied insecticides are partially or completely leached out of the root zone area without providing anticipated maggot control. In this study, insecticides were drenched into the cup and none of the applied insecticide solution leached out. Therefore, it is likely that the insecticides were more effective in the laboratory assay than they would be in the field. Certain insecticides such as pyrethroids tend to bind to the soil organic matter. The organic matter in the California's Central Coast soils can be up to 4%, which could reduce the availability of soil applied pyrethroid insecticide to the root zone where cabbage maggot larvae typically colonize. In situations with poor insecticide spray coverage, invading cabbage maggot larvae are possibly exposed to no or sub-lethal doses of the soil applied insecticide and may be able to penetrate the soil and infest the roots. The air temperature in the field at the time of insecticide application may influence the efficacy of the applied insecticide. The efficacy of pyganic decreased as the temperature increased against onion maggot. This suggests that application of pyrethroid insecticides should be avoided during warmer periods of day.
Other field conditions that influence efficacy of insecticides are cabbage maggot incidence and frequency of invading cabbage maggot flies on Brassica crop in the Central Coast of California. The earliest peak of cabbage maggot infestation occur a month after sowing broccoli seeds and infestations can be continuous until harvest. Also, insecticides applied at sowing as a banded spray on the seed lines did not provide adequate cabbage maggot control based on the insecticide efficacy trials conducted in commercial broccoli fields. These findings suggest that delaying the insecticide application by 2-3 weeks after sowing is more likely to maximize maggot control. Because the cabbage maggot infestation can last several weeks, insecticides with extended persistence of efficacy would increase the value for cabbage maggot control. Overall, results show that Capture, Torac and Belay which performed effectively against cabbage maggot for a month after application. This indicates that insecticides used before the first peak of infestation may protect the younger stages of the Brassica plants allowing them to establish and tolerate milder cabbage maggot infestations thereafter.
In conclusion, 11 insecticides with high efficacy were identified for future investigation. Future studies will focus on determining the effects of application timing and delivery methods compatible with cabbage maggot incidence in both directly sown and transplanted Brassica crops in the Central Coast of California.
If you are interested in reading the details of this study, please click the link below to access the published article.