- Author: Michael D Cahn
- Author: Oleg Daugovish
- Author: Mark Bolda
Establishing strawberry transplants using drip has several potential advantages compared to overhead sprinklers. Irrigation run-off can be greatly reduced, which protects surface water quality. Some growers have found that they can save water using drip for transplant establishment, and save costs associated with using overhead sprinklers. Often nitrogen fertilizer can be spoon fed to the crop through the drip system earlier in the season than in fields established with sprinklers, thereby reducing the reliance on pre-plant fertilizers that may result in nitrate leaching losses.
Nevertheless, growers are concerned that irrigating transplants using mainly drip may result in less vigorous growth and more dead plants during the establishment period, and yields during the production season will be lower than crops established with sprinklers. One of the specific concerns is that the drip lines adjacent to plant rows may not be as effective in leaching salts from the root zone of young plants as overhead sprinklers. Another worry is that if the transplants are not properly planted and gaps exist between the root crown and the soil, moisture will not move toward and imbibe young roots, and the plant may be set back or die.
Last year was challenging for establishing strawberries is many fields on the Central Coast due to the lack of rain, which normally helps to leach salts that may accumulate around young strawberry transplants. However, these challenging conditions were perfect for comparing vigor and yield of strawberries established using drip and overhead sprinklers.
Field trial description
We conducted a demonstration trial at a ranch in North Salinas beginning November 13, 2013. Soil was a loam texture. The field was planted with UC Albion variety in 2 rows on 52-inch wide beds. Two plots, each of approximately 1-acre in size, were located adjacent to each other in one of the irrigation blocks. Transplants were established using drip in one of the plots and with overhead sprinklers in the other plot. The irrigation foreman made all decisions on how long and often to irrigate both plots. Overhead sprinklers were used for the first 2 irrigations in the drip plot to assure that the transplants were in good contact with the soil. All subsequent irrigations were made using 2 lines of drip tape per bed. In the plot established with overhead sprinklers, transplants were also irrigated twice using the drip system. The last sprinkler irrigation was on January 25th, after which both plots were irrigated with only drip.
Applied water was monitored using flow meters installed on the drip submain and on the sprinkler main line until the end of February. Soil moisture was evaluated in the upper 6 inches of soil next to the transplants using a volumetric moisture sensor at weekly intervals during establishment. Soil salinity was also periodically monitored to a 4 inch depth next to the transplants using a soil salinity sensor (Fig 1.), or by sampling soil and analyzing saturated paste extracts for salts. Plants were rated for vigor and evaluated for canopy cover until mid February (Fig 2.). Marketable fruit yield was evaluated between late April and mid July.
Figure 1. A 5TE decagon probe was used to measure bulk salinity near strawberry plants.
Figure 2. Transplants were periodically evaluated for canopy size by measuring plant width or using a multi-spectral NDVI camera.
Results
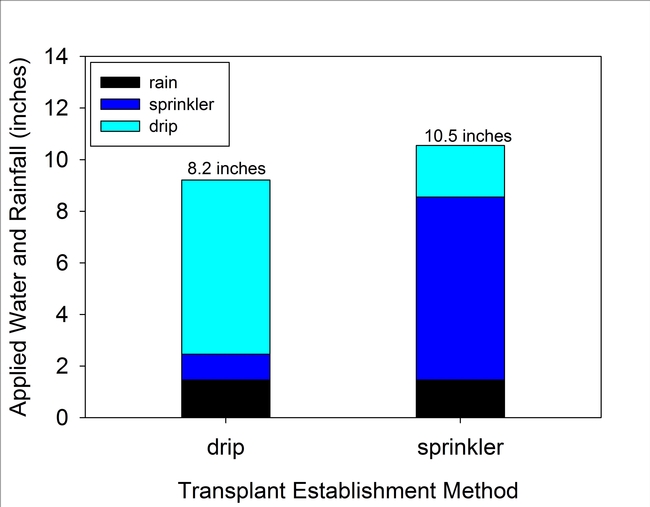
Applied water during transplant establishment was about 25% less for the drip treatment compared to the standard sprinkler treatment. As shown in Fig. 3, irrigation water applied to the drip treatment equaled 6.8 inches between Nov. 13 and Feb. 20th. During the same period, water applied in the sprinkler treatment equaled 9.1 inches. An additional 1.5 inches of rainfall were also measured during this period. Estimated evapotranspiration (ET) losses during establishment were 3.2 and 1.1 inches for the sprinkler and drip treatments, respectively. The lower estimated ET amount for drip was due to less wetting of the furrows than in the sprinkler treatment.
Figure 3. Irrigation water and rainfall for the drip and sprinkler treatments between Nov. 13 and Feb. 20th.
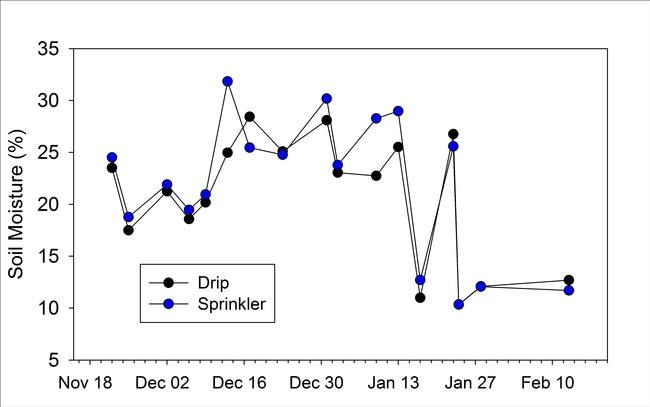
Soil moisture measured near the transplants was similar among the drip and sprinkler establishment treatments except for 4 dates between mid December and mid January when the sprinkler plot had higher soil moisture levels than the drip plot (Fig 4).
Figure 4. Volumetric soil moisture measured next to transplants for the drip and sprinkler treatments.
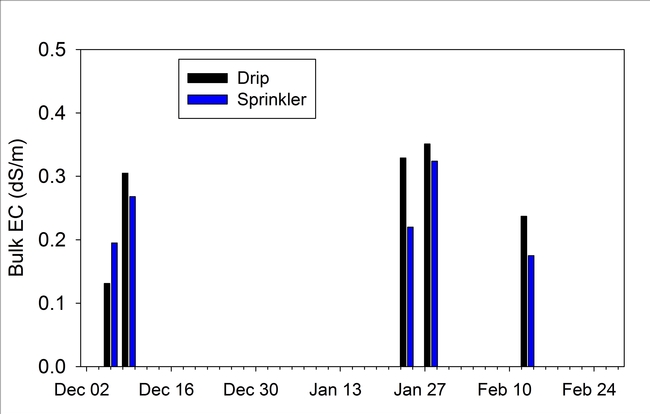
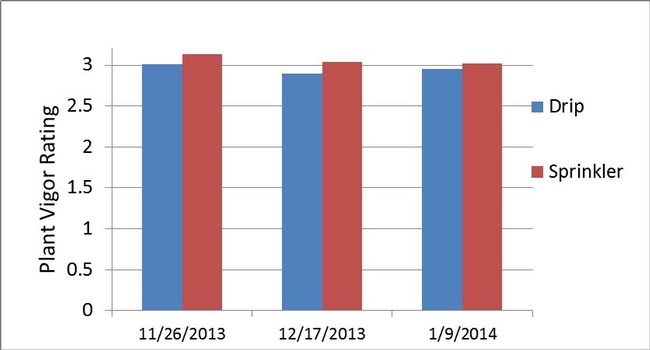
Bulk electrical conductivity (EC), an indirect measure of soil salinity, was slightly higher next to the drip established transplants than the sprinkler established transplants for 4 of 5 dates measured between December and early February (Fig. 4). Bulk EC values were generally low for both treatments, which was confirmed from the saturated paste extracts of soil sampled on January 24 2014 (3rd evaluation date). The EC of the saturated paste extract was 0.80 and 1.83 dS/m, respectively, for the sprinkler and drip established plots. Saturated pasted extract values below 2 dS/m would not be expected to harm strawberry plant growth. Plant vigor (Fig. 5) and canopy cover (Fig. 6) were not different between the sprinkler and drip establishment treatments. Likewise cumulative fruit yields (Fig. 7) were the same for the two methods of irrigation establishment.
Figure 5. Bulk electrical conductivity of the soil adjacent to strawberry transplants, measured using the Decagon 5TE probe.
Figure 6. Plant vigor of drip and sprinkler established transplants, where 0 equates to dead or dying plants, and 5 signifies all plants are very healthy.
Figure 7. Percentage of ground shaded by leaves, measured using a multi-spectral infra-red camera.
Figure 8. Cumulative marketable fruit yield for sprinkler and drip established plants.
Discussion and Conclusions
The results of this field trial demonstrated that drip can be successfully used to establish strawberry transplants during the winter on the Central Coast, even during drought conditions when rainfall is minimal. Marketable fruit yields were the same between the drip and sprinkler established plots. Additionally, 25% less water was used under drip than in the sprinkler plots during the initial establishment phase. Salinity was maintained at a sufficiently low level in the soil as to not impair transplant vigor and initial growth under drip.
Low water demand of plants during the late fall and early winter is an import factor that helped us successfully establish plants using drip. New transplants have few leaves and reference evapotranspiration, on average, is less than 0.07 inches per day. Since water demand is low, the main purpose of irrigation during establishment is to keep crown roots hydrated and to leach salts from the root zone. Using good planting techniques is critical to successfully using drip for establishment. Transplant roots need to be in contact with the soil and should not be “J” rooted. Also, soil salinity should be low as possible before planting. Preplant fertilizer bands should be located a sufficient distance from the transplant roots so that emerging new roots are not burned by fertilizer salts.
For this trial, we irrigated the drip treatment twice with overhead sprinklers to assure that the roots were in good contact with the soil. Under normal weather conditions on the Central Coast, rain often occurs between late November and February, which can also assist with the establishment of transplants by maintaining high soil moisture and leaching salts from the root zone of young plants.
Acknowledgements
We thank Dole and their employees for their help and partnership with this trial, and we thank Walmart for funding this project.
- Author: Steven T. Koike
- Author: Jim Correll
Denomination of Pfs: 15, a new race of downy mildew in spinach
Jim Correll, Steven Koike, Diederik Smilde, Jan de Visser
A new race of the downy mildew pathogen (Peronospora farinosa f. sp. spinaciae) on spinach was first identified in November 2012 in Imperial Valley, CA, U.S. This race overcomes the resistance of several important varieties. The isolate was designated UA4712 and was characterized with a standard set of differential varieties. Subsequently, isolates with the same reaction pattern on the differential set have been found in numerous locations. After careful evaluation of the significance of this development to the spinach industry, the International Working Group on Peronospora farinosa (IWGP) has denominated isolate UA4712 as race Pfs: 15.
Race Pfs: 15 gives the same disease reactions as Pfs: 4 on the standard set of differentials, but is able to overcome the resistance of race 1-14 resistant varieties such as Caladonia (see chart below). Also, Whale is susceptible to Pfs: 15 whereas it is resistant to Pfs: 4. Although this is a new race, a number of commonly used resistances are effective on this race.
New races and deviating strains of the Pfs: pathogen continue to appear in many spinach growing areas of the world. The IWGP is continuously monitoring the appearance of strains of the pathogen that deviate in virulence from the known races. In this way the IWGP aims to promote a consistent and clear communication between public and private entities, such as the seed industry, growers, scientists, and other interested parties about all resistance-breaking races that are persistent enough to survive over several years, occur in a wide area, and cause a significant economic impact.
The IWGP is located in The Netherlands and is administered by Plantum NL. The IWGP consists of spinach seed company representatives (Pop Vriend, Monsanto, RijkZwaan, Nunhems, Takii, Sakata, Bejo, Enza, Syngenta, and Advanseed) and Naktuinbouw, and is supported by research centers at the University of Arkansas and the University of California Cooperative Extension (Monterey County) in the U.S. Researchers all over the world are invited to join the IWGP initiative and use the common host differential set to identify new isolates.
For more information on this subject you can contact Jim Correll (jcorrell@uark.edu), Steven Koike (stkoike@ucdavis.edu), Diederik Smilde (d.smilde@naktuinbouw.nl), or the IWGP chairperson Jan de Visser (JandeVisser@popvriendseeds.nl).
Disease reactions of race 15 (UA4712) observed on spinach differentials by the IWGP compiled July 2014.
aDisease reactions observed in controlled inoculation tests by 11 different participants of the IWGP.
“ + ” indicates susceptible disease reaction; “ - “ indicates resistant disease reaction.
- Author: Shimat Villanassery Joseph
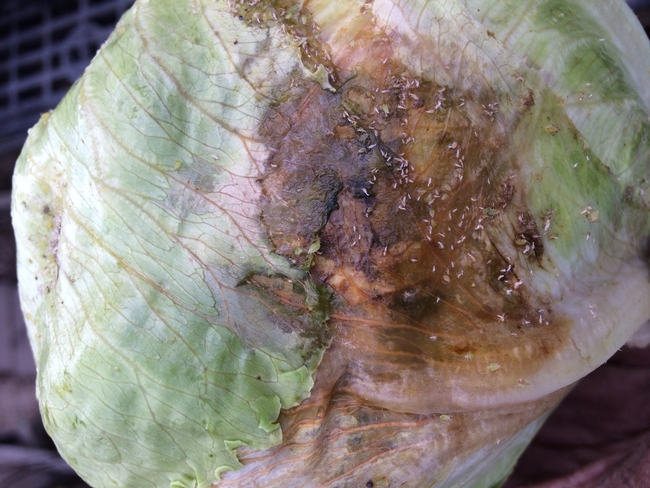
Recently, a widespread outbreak of foxglove aphid (Aulacorthum solani) (Figures 1, 2 and 3) has been reported in the Salinas Valley. Reports were primarily from Salinas to Soledad at this point. Growers have been losing several acres of lettuce to foxglove aphid since last one and half months.
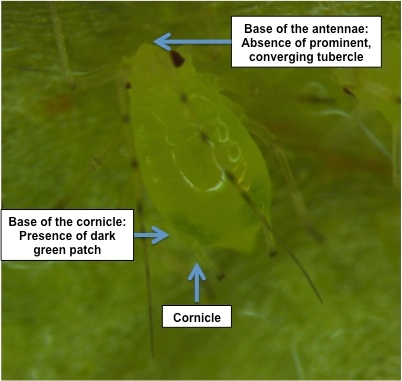
Foxglove aphid nymphs and wingless adults have a light to dark green patch at the base of the cornicle (Figures 4 and 5). The lettuce aphids (Nasonovia ribis-nigri) do not have any patch at the base of the cornicle. Cornicle is a tube-like organ at the back of the aphid as shown in the figures 4 and 5. The foxglove aphids observed were yellowish to green in color than pink or red. At the base of the antennae, foxglove aphid doesn't have any prominent converging tubercles (a projecting structure), which is typical for green peach aphid (Myzus persicae). The joins of legs and antennae of foxglove aphid are darker (dusky) than other regions. Winged adults of foxglove aphid and lettuce aphid are practically indistinguishable from each other.
Generally, aphids are capable of reproducing parthenogenetically meaning female aphids lay eggs without mating and all the eggs turn into females. Moreover, in warmer conditions like in summer, they pretty much give birth to young ones as the eggs hatch within the reproductive canal of the female. Typically, a single aphid give birth to about 50 to 100 nymphs in two weeks or about 10 nymphs every day, which could vary with environmental conditions such as temperature and humidity. Foxglove aphids can complete a generation in less than 2 weeks in the summer. Unlike lettuce aphid, foxglove aphid has a broad host range meaning it could survive on several host plants.
Detection of foxglove aphid early in the crop stage is critical. They initially infest the cap or outer leaves of head lettuce. Eventually, foxglove aphids move into the deeper layers of leaves then form colonies. Green peach aphids on the other hand, form colonies on the wrapper leaves from the onset. Based on anecdotal observations, foxglove aphid colonies have been observed in lettuce about 20 days to harvest. The lettuce crops during thinning stage often appeared clean (without foxglove aphids).
Management of foxglove aphid with insecticides has been monitored more closely than ever before. It is important to note that spirotetramat (Movento), which is widely used against aphids has to metabolize from applied form (spirotetramat) and convert into more toxic derivatives within the plant in order to be toxic or effective against aphids. Normally, it will take at least 10 days for this metabolic process and movement of derivatives into the growing tissues of the lettuce. Thus, two applications of spirotetramat (5 fl oz/acre each) should be timed at the onset of first foxglove infestation without delay. Late application of spirotetramat (e.g. 10 days before harvest) may not offer any control. Also, it is important to keep in mind that there is little use if the applications are made when high populations of foxglove aphid have been detected. Make sure that the insecticides such as acetamiprid (Assail), imidacloprid, thiamethoxam (Actara) are used in rotation. Back-to-back use of insecticides in same class or IRAC group (http://www.irac-online.org/teams/mode-of-action/) will increase the risk of development of aphid resistance to a particular class of insecticides. Once a super aphid has been created, there is no value in using insecticides from that class to manage aphids. The industry is currently seeking emergency registration of sulfoxaflor (Closer or Sequoia) for use to tackle foxglove aphid problem this season. Sulfoxaflor is systemic (moves within the plant), trans-laminar (move through leaves) and acts in hours once applied and is from a new insecticide class or IRAC group. Please email (Shimat Joseph: svjoseph@ucanr.edu) or call (831) 229 8589 if you have further questions.



- Author: Shimat Villanassery Joseph
Bagrada bug (Bagrada hilaris) (Figure 1), an invasive stink bug species native to old world countries of southern Africa, Middle East and Asia, was first detected in North America from Los Angeles Co, California in 2008, and is now established in brassica crop production regions of central coast of California.
Bagrada bug prefers cruciferous hosts (Family: Brassicaceae) including broccoli, cauliflower, cabbage, kale, arugula, or collards. Also, bagrada bug could survive on cruciferous weeds such wild radish, London rocket, short pod mustard, and shepherd's purse, as well as the insectary plant, sweet alyssum. Mustard weed species are very common in ditches, roadsides, and along the edges of agricultural fields in the central coast of California. Mustard cover crops such as white mustard and Indian mustard in particular, could harbor bagrada bug populations, which are not often monitored for insect pests.
Damage to brassica crops varies but can be severe. Severe economic loss has been reported when injury occurred during early developmental stages of the crop such as cotyledon or < 4 leaves stages. Injury on leaves appears initially as small puncture marks, which turn into white patches as leaves expand. In broccoli and cauliflower, the economic injury occurs when the bug feeding kills the apical meristematic tissue of young seedling, which later results in “multiple heads” or sometimes “blind head” (without a head). Severe feeding also depletes the nutrient reserves of the plant leading to desiccation or wilting. Other leafy brassica crops such as mizuna mustard or arugula are especially at risk from direct feeding damage of bagrada bug.
At this time, no baseline information on bagrada bug abundance, seasonal activity, or generations has been established in the coastal brassica crop production regions in California. Limited information exists to determine threshold based pest management decisions for bagrada bug. Monitoringfor bagrada bug during mid-day hours might increase the probability of finding them as the bugs typically hide and stay in the cracks and crevices or on the underside of leaves when the temperature is cooler. Strong attraction to specific color could be utilized when developing selective traps for monitoring bagrada bug in the field. Yellow traps are most commonly used for monitoring true bugs such as false chinch bugs, plant bugs, psyllids or stinkbugs. Yellow pyramid traps were used to monitor brown and dusky stink bugs.
This study was conducted to determine the influence of trap color on catches of bagrada bug. In fall 2013, an organically-managed broccoli field in San Ardo, CA was chosen for the study. Planting sweet alyssum is a common practice among organic growers in the Salinas Valley to harbor beneficial insects such as syrphid flies (hover flies) and lady beetles within the cole crop field. Strips of sweet alyssum were planted within the broccoli field. Cross-vane traps were constructed using corrugated plastic sheets and were painted with white, yellow, red, purple, or black paint (Figure 2). All the trap-base containers used in this study were light green colored. Traps were placed within the sweet alyssum plants (Figure 3).Bug captures were recorded for up to 5 weeks.
In total, 10,654 bagrada bug were captured and were predominantly adults (95%). Adult bagrada bug captures were influenced by trap color (Figure 4a). Black traps captured almost twice the number of adult bagrada bug than purple traps; however, adult catches were similar among purple, white or red traps. Catches of adults were higher in purple than in yellow traps. Similarly, the numbers of bagrada bug nymphs collected among various colored-traps were higher in black traps than other colored traps (Figure 4b). Future research will investigate the potential use of color preference with plant host odor to develop traps for field monitoring of bagrada bug
For those of you, who are interested to read more on it, please find the published article after clicking the link (below) and feel free to contact me (Shimat Joseph) at svjoseph@ucanr.edu or 831 759 7359.
http://cemonterey.ucanr.edu/files/195965.pdf