- Author: Ben Faber
For centuries, the prevailing science has indicated that all of the nitrogen on Earth available to plants comes from the atmosphere. But a study from the University of California, Davis, indicates that more than a quarter comes from Earth's bedrock.
The study, to be published April 6 in the journal Science, found that up to 26 percent of the nitrogen in natural ecosystems is sourced from rocks, with the remaining fraction from the atmosphere.
Before this study, the input of this nitrogen to the global land system was unknown. The discovery could greatly improve climate change projections, which rely on understanding the carbon cycle. This newly identified source of nitrogen could also feed the carbon cycle on land, allowing ecosystems to pull more emissions out of the atmosphere, the authors said.
"Our study shows that nitrogen weathering is a globally significant source of nutrition to soils and ecosystems worldwide," said co-lead author Ben Houlton, a professor in the UC Davis Department of Land, Air and Water Resources and director of the UC Davis Muir Institute. "This runs counter the centuries-long paradigm that has laid the foundation for the environmental sciences. We think that this nitrogen may allow forests and grasslands to sequester more fossil fuel CO2 emissions than previously thought."
WEATHERING IS KEY
Ecosystems need nitrogen and other nutrients to absorb carbon dioxide pollution, and there is a limited amount of it available from plants and soils. If a large amount of nitrogen comes from rocks, it helps explain how natural ecosystems like boreal forests are capable of taking up high levels of carbon dioxide.
But not just any rock can leach nitrogen. Rock nitrogen availability is determined by weathering, which can be physical, such as through tectonic movement, or chemical, such as when minerals react with rainwater.
That's primarily why rock nitrogen weathering varies across regions and landscapes. The study said that large areas of Africa are devoid of nitrogen-rich bedrock while northern latitudes have some of the highest levels of rock nitrogen weathering. Mountainous regions like the Himalayas and Andes are estimated to be significant sources of rock nitrogen weathering, similar to those regions' importance to global weathering rates and climate. Grasslands, tundra, deserts and woodlands also experience sizable rates of rock nitrogen weathering.
GEOLOGY AND CARBON SEQUESTRATION
Mapping nutrient profiles in rocks to their potential for carbon uptake could help drive conservation considerations. Areas with higher levels of rock nitrogen weathering may be able to sequester more carbon.
"Geology might have a huge control over which systems can take up carbon dioxide and which ones don't," Houlton said. "When thinking about carbon sequestration, the geology of the planet can help guide our decisions about what we're conserving."
MYSTERIOUS GAP
The work also elucidates the "case of the missing nitrogen." For decades, scientists have recognized that more nitrogen accumulates in soils and plants than can be explained by the atmosphere alone, but they could not pinpoint what was missing.
"We show that the paradox of nitrogen is written in stone," said co-leading author Scott Morford, a UC Davis graduate student at the time of the study. "There's enough nitrogen in the rocks, and it breaks down fast enough to explain the cases where there has been this mysterious gap."
In previous work, the research team analyzed samples of ancient rock collected from the Klamath Mountains of Northern California to find that the rocks and surrounding trees there held large amounts of nitrogen. With the current study, the authors built on that work, analyzing the planet's nitrogen balance, geochemical proxies and building a spatial nitrogen weathering model to assess rock nitrogen availability on a global scale.
The researchers say the work does not hold immediate implications for farmers and gardeners, who greatly rely on nitrogen in natural and synthetic forms to grow food. Past work has indicated that some background nitrate in groundwater can be traced back to rock sources, but further research is needed to better understand how much.
REWRITING TEXTBOOKS
"These results are going to require rewriting the textbooks," said Kendra McLauchlan, program director in the National Science Foundation's Division of Environmental Biology, which co-funded the research. "While there were hints that plants could use rock-derived nitrogen, this discovery shatters the paradigm that the ultimate source of available nitrogen is the atmosphere. Nitrogen is both the most important limiting nutrient on Earth and a dangerous pollutant, so it is important to understand the natural controls on its supply and demand. Humanity currently depends on atmospheric nitrogen to produce enough fertilizer to maintain world food supply. A discovery of this magnitude will open up a new era of research on this essential nutrient."
###
UC Davis Professor Randy Dahlgren in the Department of Land, Air and Water Resources co-authored the study.
The study was funded by the National Science Foundation's Division of Earth Sciences and its Division of Environmental Biology, as well as the Andrew W. Mellon Foundation.
Photo: The stuff that makes leaves green

- Author: Craig Kallsen
University of California (UC) researchers and private industry consultants have invested much effort in correlating optimal citrus tree growth, fruit quality and yield to concentrations of necessary plant nutrients in citrus (especially orange) leaf tissue. The grower can remove much of the guesswork of fertilization by adhering to UC recommendations of critical levels of nutrients in the tissues of appropriately sampled leaves. Optimal values for elements important in plant nutrition are presented on a dry-weight basis in Table 1. Adding them in appropriate rates by broadcasting to the soil, fertigating through the irrigation system or spraying them foliarly may correct concentrations of nutrients in the deficient or low range. Compared to the cost of fertilizers, and the loss of fruit yield and quality that can occur as a result of nutrient deficiencies or excesses, leaf tissue analysis is a bargain. At a minimum, the grower should monitor the nitrogen status of the grove through tissue sampling on an annual basis.
Leaves of the spring flush are sampled during the time period from about August 15 through October 15. Pick healthy, undamaged leaves that are 4-6 months old on non-fruiting branches. Select leaves that reflect the average size leaf for the spring flush and do not pick the terminal leaf of a branch. Typically 75 to 100 leaves from a uniform 20- acre block of citrus are sufficient for testing. Generally, the sampler will walk diagonally across the area to be sampled, and randomly pick leaves, one per tree. Leaves should be taken so that the final sample includes roughly the same number of leaves from each of the four quadrants of the tree canopy. Values in Table 1 will not reflect the nutritional status of the orchard if these sampling guidelines are not followed. Typically, citrus is able to store considerable quantities of nutrients in the tree. Sampling leaves from trees more frequently than once a year in the fall is usually unnecessary. A single annual sample in the fall provides ample time for detecting and correcting developing deficiencies.
Table 1. Mineral nutrition standards for leaves from mature orange trees based on dry-weight concentration of elements in 4 to 7 month old spring flush leaves from non-fruiting branch terminals.
|
element |
unit |
deficiency |
low |
optimum |
high |
excess |
|
|
|
|
|
|
|
|
|
N |
% |
2.2 |
2.2-2.4 |
2.5-2.7 |
2.7-2.8 |
3.0 |
|
P |
% |
0.9 |
0.9-0.11 |
0.12-0.16 |
0.17-0.29 |
0.3 |
|
K (Calif.*) |
% |
0.40 |
0.40-0.69 |
0.70-1.09 |
1.1-2.0 |
2.3 |
|
K (Florida*) |
% |
0.7 |
0.7-1.1 |
1.2-1.7 |
1.8-2.3 |
2.4 |
|
Ca |
% |
1.5 |
1.6-2.9 |
3.0-5.5 |
5.6-6.9 |
7.0 |
|
Mg |
% |
0.16 |
0.16-0.25 |
0.26-0.6 |
0.7-1.1 |
1.2 |
|
S |
% |
0.14 |
0.14-0.19 |
0.2-0.3 |
0.4-0.5 |
0.6 |
|
Cl |
% |
? |
? |
<0.03 |
0.4-0.6 |
0.7 |
|
Na |
% |
? |
? |
<0.16 |
0.17-0.24 |
0.25 |
|
B |
ppm |
21 |
21-30 |
31-100 |
101.260 |
260 |
|
Fe |
ppm |
36 |
36-59 |
60-120 |
130-200 |
250? |
|
Mn |
ppm |
16 |
16-24 |
25-200 |
300-500? |
1000 |
|
Zn |
ppm |
16 |
16-24 |
25-100 |
110-200 |
300 |
|
Cu |
ppm |
3.6 |
3.6-4.9 |
5 - 16 |
17-22? |
22 |
*California and Florida recommendations for K are sufficiently different that they are presented separately. The California standards are based on production of table navels and Valencias, and those for Florida were developed primarily for juice oranges like Valencia.
The sampled leaves should be placed in a paper bag, and protected from excessive heat (like in a hot trunk or cab) during the day. If possible, find a laboratory that will wash the leaves as part of their procedure instead of requiring the sampler to do this. Leaf samples can be held in the refrigerator (not the freezer) overnight. Leaves should be taken to the lab for washing and analysis as quickly as is feasible.
Often separate samples are taken within a block if areas exist that appear to have special nutrient problems. The temptation encountered in sampling areas with weak trees is to take the worst looking, most severely chlorotic or necrotic leaves on the tree. Selecting this type of leaf may be counter-productive in that the tree may have already reabsorbed most of the nutrients from these leaves before they were sampled. A leaf-tissue analysis based on leaves like this often results in a report of general starvation, and the true cause of the tree decline if the result of a single nutritional deficiency may not be obvious. Often in weak areas, it is beneficial to sample normal appearing or slightly affected leaves. If the problem is a deficiency, the nutrient will, generally, be deficient in the healthy-looking tissue as well.
Groves of early navels that are not normally treated with copper and lime as a fungicide should include an analysis for copper. Copper deficiency is a real possibility on trees growing in sandy, organic, or calcareous soils. For later harvested varieties, leaves should be sampled before fall fungicidal or nutritional sprays are applied because nutrients adhering to the exterior of leaves will give an inaccurate picture of the actual nutritional status of the tree.
Usually leaf samples taken from trees deficient in nitrogen will overestimate the true quantity of nitrogen storage in the trees. Trees deficient in nitrogen typically rob nitrogen from older leaves to use in the production of new leaves. Frequently, by the time fall leaf samples are collected in nitrogen deficient groves, these spent spring flush leaves have already fallen. Nitrogen deficient trees typically have thin-looking canopies as a result of this physiological response. Since the spring flush leaves are no longer present on the tree in the fall when leaves are sampled, younger leaves are often taken by mistake for analysis. These leaves are higher in nitrogen than the now missing spring flush leaves would have been and provide an inaccurately higher nitrogen status in the grove than actually exists.
Critical levels for leaf-nitrogen for some varieties of citrus, like the grapefruits, pummelos, pummelo x grapefruit hybrids and the mandarins, have not been investigated as well as those for oranges. However, the mineral nutrient requirements of most citrus varieties are probably similar to those for sweet oranges presented in Table 1, except for lemons, where the recommended nitrogen dry-weight percentage is in the range of 2.2- 2.4%.
A complete soil sample in conjunction with the leaf sample can provide valuable information on the native fertility of the soil with respect to some mineral nutrients and information on how best to amend the soil if necessary to improve uptake of fertilizers and improve water infiltration.
P.S. from Ben Faber
What has been said here about citrus is also generally true for avocado, although the nitrogen sufficiency levels are lower than for citrus. For a more detailed discussion see: http://www.californiaavocadogrowers.com/sites/default/files/documents/11-Final-Report-Issued-Giving-Tools-for-Fertilization-and-Salinity-Management-Winter-2016.pdf
Photo: Nitrogen deficient avocado leaf

- Author: Ben Faber
At a recent meeting the question came up about the fate of nitrogen fertilizer applied through the irrigation system. If it is applied as urea, how long does it take to convert it to nitrate? If applied as ammonium, how long does it take to convert to nitrate? Urea and nitrate pretty much move wherever water moves and is very susceptible to leaching. Because of the positive charge on ammonium, it is not as mobile as nitrate, but once bacteria transform it to nitrate, it moves with water.
This is an important question, since if more water is applied than is needed by the plant, the nitrate is going to move out of the root system and no longer be available to the plant and ends up heading to ground water. Reading the literature, growers get the sense that all this transformation takes time, maybe a long time.
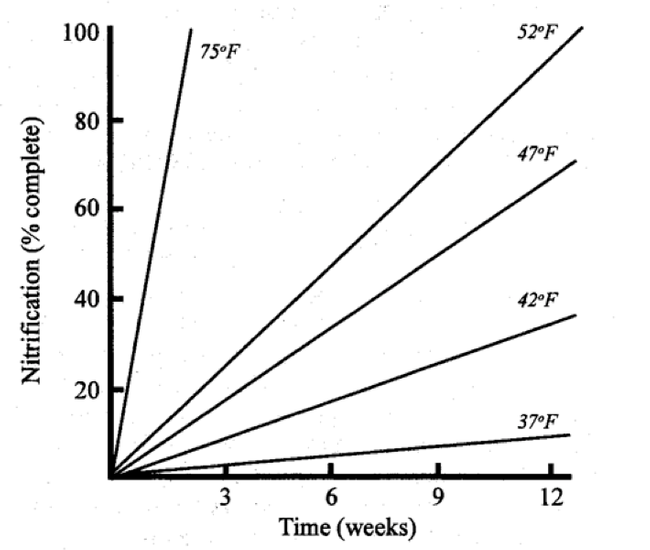
It turns out that soils in coastal California have a pretty rapid conversion of nitrogen. Francis Broadbent at UC Davis did a bunch of studies back in the 1950's and 60's and found enzyme hydrolysis of urea to ammonium occurring within hours. Other researchers have looked at nitrification, the conversion of ammonium to nitrate by soil bacteria, occurring within days and much of the conversion occurring within a week depending on soil temperature (see chart below).
So there is all this nitrate present and the key is what happens to it. It turns out that most plants when actively growing absorb nitrate at about 5 pounds of nitrogen per day. So with a 100% efficiency, applying 20 pounds of nitrogen, all of it would be taken up in four days. Of course, nothing in nature is that efficient. But the point is a big slug of nitrogen applied is not going to be taken up immediately and if more water is applied after that than is needed by the crop, it likely is pushed out of the avocado root zone.
Of course all the nitrogen a plant uses does not come from applied fertilizer. The bulk is coming from soil organic matter that is slowly decomposing. This nitrogen is being released at a rate that is probably in balance with the growth of the tree.
The applied fertilizer, however, is much more unstable and needs to be handled accordingly. The rule of thumb is to break the irrigation application into thirds. In the first third, run the irrigation to fill the lines and wet the soil. In the second third, run the fertilizer. This spreads it through the system and onto the ground. The last third is clear the irrigation system of the material and to move the fertilizer into the root zone. Then given time, the tree will take up the applied nitrogen. At the next irrigation then the bulk of that nitrogen will have been taken up and little will be pushed through the root system.
Low and High Nitrogen Avocado Leaves
Chart showing rapid conversion to nitrate with soil temperature

- Author: Ben Faber
This is a sad time to be an avocado. Winter's gone and temperatures are just ripe for flowering and the trees are going bust. So much so, that those sad leaves that have accumulated salts over the last year are being dropped and only flowers might be seen, especially on young trees. This is time for a little shot of nitrogen to encourage some new vegetative growth. Not a bunch, but a nudge. Several pounds per acre, something less than 10-15 pounds of N for a mature orchard and even less for a new orchard.
A commonly held belief is that if you apply nitrogen at the wrong time, it will push resources into vegetative growth at the expense of flower and fruit. This is somewhat true for annual plants that get most of their nutrients from outside sources (soil, air, fertilizer, water), but trees have a huge buffer in their storage organs (roots, stems, leaves, etc.). Most growth in trees occurs from this storage source and most importantly from photosynthesis and the sun. The more sun captured the more energy for flowering and fruit production.
So it is this competition for photosynthates that becomes the most limiting factor. When there is not enough to go around, the tree sheds fruit. If you see fruit dropping off a tree after applying a slug of fertilizer, it's a salt effect. Too much salt and it causes a water competition and the tree is stressed. It's not the nitrogen, but too much salt. With fertigation this is not so likely to happen as when dry fertilizers were applied and someone got too aggressive with the application
In fact a dose of nitrogen fertilizer is a good idea at this time when there are lots of flowers. This can encourage a flush of leaves that will protect the fruit that does set from sunburn and damage that would cause fruit to drop. A bit of nitrogen to encourage leaf replacement is a good approach to dealing with persea mite damage that occurred the previous season.
For further reading about the competition between vegetative and reproductive growth as affected by nitrogen (or little affected in fruit trees by nitrogen), D.O. Huett wrote a wonderful review of past research on this topic:
http://www.publish.csiro.au/?act=view_file&file_id=AR9960047.pdf
Also, if the trees have really defoliated, it might be time to do some whitewashing, south and west sides of branches, to prevent sunburn.
Images:
Avocado defoliated and ones in a balanced bloom


- Author: Ben Faber
Nutrient availability from organic sources has been considered “slow release” by many growers and advisers. This may be true in environments are colder and especially soils are cooler. Organic nutrients are dependent on microbes to break down materials and release those nutrients, and when soils are cold, microbes can't do their thing. Soils in much of agricultural California tend to be warm and lack the freezing conditions that occur in many soils in the continental US. Imagine how much microbial activity occurs in the Mid-West when soils cool down to 32 deg F at a four inch depth and deeper. The top layers of soil are where organic matter accumulates and where most microbial activity occurs. When soils cool below 50 deg F, nitrogen leaching becomes less common, because less activity is occurring which also coincides with much less plant growth.
Soils in coastal California rarely fall below 50 deg F in the surface layers, so microbial activity is ongoing, all year long. So the question is, how “slow acting” are organic fertilizers? A recent study by Tim Hartz, Richard Smith and Mark Gaskell looked at release rates of injectable organic fertilizer and found that much of the nutrient release occurs within about a week after application depending on the formulation and temperature during the study. The results conform to another study that they did where they evaluated the nitrogen release rates of dry formulations of organic fertilizers – compost, manures, feather meal, etc.
Aside from the issues of the higher costs of these materials and their potential clogging, there is the issue of application timing. In the case of avocados and citrus, adequate levels of nitrogen are needed in the trees going into to fruit set in order to optimize set. And then after fruit set, in order to maintain growth into the fast growth period, again nitrogen needs to be adequate. Using organic fertilizers with a rapid conversion to useable forms of nitrogen, means that application timing should coincide with these critical periods in tree phenology or growth cycle.
Using information on organic nutrient management based on work from cold soil climates needs to be carefully evaluated before applying it to California soils. One of the most common problems in organic production is nitrogen management. Part of the problem is the cost of supplemental nitrogen amendments, but also learning to anticipate when that applied nutrient becomes available to the plant. Developing better estimates for local release rates and patterns will better help manage organic nutrient sources.
Read more:
Nitrogen Availability from Liquid Organic Fertilizers by T.K. Hartz, R. Smith and M. Gaskell
http://horttech.ashspublications.org/content/20/1/169.full
Summary: Limited soil nitrogen (N) availability is a common problem in organic vegetable production that often necessitates additional N fertilization. The increasing use of drip irrigation has created a demand for liquid organic fertilizers that can be applied with irrigation. The N availability of three liquid organic fertilizers was evaluated in an incubation study and a greenhouse bioassay. Phytamin 801 contained fishery wastes and seabird guano, while Phytamin 421 and Biolyzer were formulated from plant materials. The fertilizers ranged from 26 to 60 g·kg−1 N, 8% to 21% of which was associated with particulate matter large enough to potentially be removed by drip irrigation system filtration. The fertilizers were incubated aerobically in two organically managed soils at constant moisture at 15 and 25 °C, and sampled for mineral N concentration after 1, 2, and 4 weeks. In the greenhouse study, these fertilizers and an inorganic fertilizer (ammonium sulfate) were applied to pots of the two organically managed soils with established fescue (Festuca arundinacea) turf; the N content of clippings was compared with that from unfertilized pots after 2 and 4 weeks of growth. Across soils and incubation temperatures, the N availability from Phytamin 801 ranged from 79% to 93% of the initial N content after 1 week, and 83% to 99% after 4 weeks. The plant-based fertilizers had significantly lower N availability, but after 4 weeks, had 48% to 92% of initial N in mineral form. Soil and incubation temperature had modest but significant effects on fertilizer N availability. Nitrification was rapid, with >90% of mineral N in nitrate form after 1 week of incubation at 25 °C, or 2 weeks at 15 °C. N recovery in fescue clippings 4 weeks after application averaged 60%, 38%, and 36% of initial N content for Phytamin 801, Phytamin 421, and Biolyzer, respectively, equivalent to or better than the N recovery from ammonium sulfate.




