- Author: Ben Faber
Fertilizers and Plant Nutrition Workshop, Ventura (English)
This workshop in English will present information for nursery and greenhouse growers, managers, and personnel on plant nutrition and fertilizer management with a focus on crops in containers. With demonstrations and hands-on activities. Topics discussed include:
• Plant nutrition
• Fertilizer types
• Uses of fertilizers
• Developing fertilizer programs
• Monitoring fertilizer status
Date:
August 9, 2022
8:00 am to 5:00 pm
Check-in starts at 7:30 am
Location:
UC Cooperative Extension Ventura County
669 County Square Dr. Suite 100
Ventura CA 93003
Registration:
$45 early registration before July 10 using the coupon code BZKMQHUTBJ at registration.
$55 after August 7th.
Box lunch, morning and afternoon refreshments provided
Cancellation Policy:
If you cannot attend on the day of the workshop, you may request a refund. Refunds may take 30 days or more to post.
For questions please contact:
Loren Oki lroki@ucdavis.edu
Organized and Sponsored by:
University of California Nursery and Floriculture Alliance
This event is made possible by funding from the CDFA Fertilizer Research and Education Program and support from the Plant California Alliance
Link:

Taller de Fertilizantes y Nutrición Vegetal, Ventura (Español)
Este taller en español presentará información para productores, gerentes y personal de viveros e invernaderos sobre nutrición vegetal y manejo de fertilizantes con un enfoque en cultivos en contenedores. Con demostraciones y actividades prácticas. Los temas discutidos incluyen:
• Nutrición vegetal
• Tipos de fertilizantes
• Usos de fertilizantes
• Desarrollo de programas de fertilizantes
• Supervisión del estado de los fertilizantes
Fecha:
10 de Agosto de 2022
8:00 am a 5:00 pm
El check-in comienza a las 7:30 am
Ubicación:
UC Cooperative Extension Ventura County
669 County Square Dr. Suite 100
Ventura CA 93003
Registro:
$45 por registro anticipado antes del 7 de Agosto utilizando el código de cupón NXADZYPUZC en el registro.
$55 después del 7 de Agosto.
Box lunch, refrigerios por la mañana y por la tarde proporcionados
Política de cancelación:
Si no puede asistir el día del taller, puede solicitar un reembolso. Los reembolsos pueden tardar 30 días o más en publicarse.
Si tiene preguntas, comuníquese con:
Loren Oki lroki@ucdavis.edu
Jared Sisneroz jasisneroz@ucdavis.edu
Organizado y patrocinado por:
University of California Nursery and Floriculture Alliance
Este evento es posible gracias al financiamiento de la CDFA Fertilizer Research and Education Program y apoyo de la Plant California Alliance
Link:
Developing a nitrogen fertilizer plan for olive orchards
Elizabeth J. Fichtner, Farm Advisor, UCCE Kings and Tulare Counties
Nitrogen management plans (NMP) for California olive orchards are essential for the Irrigated Lands Regulatory Program and can increase net return. A good NMP has the potential to increase yield, improve oil quality and mitigate biotic and abiotic stresses while reducing nitrogen losses from the orchard.
Olives differ from other orchard crops in California in that they are both evergreen and alternate bearing. Individual leaves may persist on the tree for two to three years. Leaf abscission is somewhat seasonal, with most leaf drop occurring in late Spring. Rapid shoot expansion occurs on non-bearing branches during the hottest part of the summer (July-August) on ‘Manzanillo' olives in California. The fruit on bearing branches limits current season vegetative growth. Olives bear fruit on the prior year's growth, and the alternate bearing cycle is characterized by extensive vegetative growth in one year followed by reproductive growth the following year (Figure 1). With bloom occurring in late April to mid-May, fruit set can be estimated in early July, allowing for consideration of crop load while interpreting foliar nutritional analysis in late July-early August.
Critical Nitrogen Values. Foliar nitrogen content in July/August should range from approximately 1.3-1.7% to maintain adequate plant health. The symptoms of nitrogen deficiency manifest when foliar nitrogen content drops to 1.1% nitrogen. As leaves become increasingly nitrogen deficient, foliar chlorosis progresses from yellow/green to yellow. Leaf abscission is common at nitrogen levels below 0.9%. Nitrogen deficiency in olive is associated with a reduced number of flowers per inflorescence, low fruit set, and reduced yield.
Excess nitrogen (>1.7%) adversely affects oil quality. Oil with low polyphenol concentration is associated with orchards exhibiting excess nitrogen fertility. Since polyphenols are the main antioxidant in olive oil, reduced polyphenol levels are associated with reduced oxidative stability.
Nitrogen content may impact orchard susceptibility to biotic and abiotic stresses. For example, while excess nitrogen content has been associated with increased tolerance to frost prior to dormancy, in spring (post-dormancy) it is associated with sensitivity to low temperatures. High nitrogen content has also been associated with increased susceptibility to peacock spot, a foliar fungal disease on olive.
Foliar Sampling for Nitrogen Analysis. By convention, foliar nutrient analysis is conducted in late July-early August in California. Fully-expanded leaves are collected from the middle to basal region of the current year's growth at a height of about 5-8 feet from the ground. To capture a general estimate of the nitrogen status of the orchard, samples should be taken from 15-30 trees, with approximately 5-8 leaf samples collected per tree. Leaves for analysis should only be collected from non-bearing branches. Growers may find it beneficial to make note of the ON and OFF status in the historical records of each block. The orchard bearing status, combined with anticipated yield and foliar analysis will guide decisions for nitrogen applications the following year.
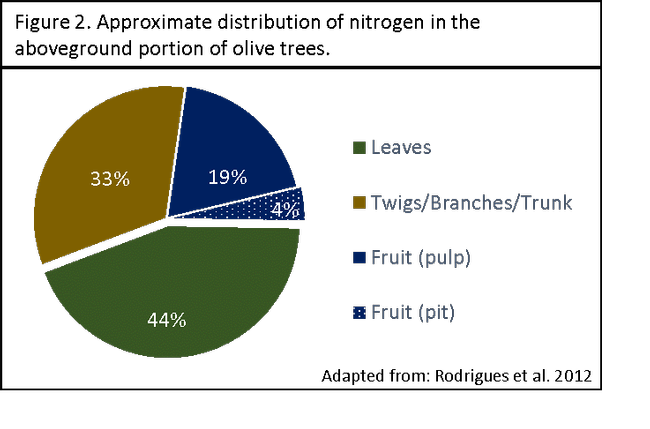
Distribution of nitrogen in the olive tree. Over 75% of the aboveground nitrogen in the olive tree is incorporated in the vegetative biomass (Figure 2). The twigs, secondary branches, main branches, and trunk account for approximately 33% of aboveground nitrogen (Figure 2). Twenty-three percent of the aboveground nitrogen is harbored in the fruit, with the majority in the pulp (19%) (Figure 2). Fruit is only an important nitrogen sink during the initial phase of growth. As fruit size increases, the N concentration decreases due to dilution.
Estimation of nitrogen removed from the orchard. The easiest component of orchard nitrogen loss to estimate is the nitrogen in the harvested fruit. A ton of harvested olives removes approximately 6-8 lbs of nitrogen from the orchard. The quantity of nitrogen in the fruit varies slightly between olive varieties (Table 1). Growers can use the Fruit Removal Nutrient Calculator for Olive on the California State University, Chico (CSU Chico) website to gain estimates of N removal by the three oil varieties (Arbequina, Arbosana, and Koroneiki), and the Manzanillo table olive. This tool was developed by Dr. Richard Rosecrance (Professor, CSU Chico) and Bill Krueger (Farm Advisor, UCCE). To access the Fruit Removal Nutrient Calculator for Olive, visit the following URL:
http://rrosecrance.yourweb.csuchico.edu/Model/OliveCalculator/OliveCalculator.html
Pruning may generate a second component of nitrogen loss from orchards. The best practice to mitigate nitrogen loss from pruning is to reincorporate the pruned material into the orchard floor by flail mowing. The nitrogen in this organic material will gradually become available to the trees through mineralization.
In mature orchards, the wood removed by annually pruning is approximately equal to the annual vegetative growth. Consequently, the input and removal of nitrogen in vegetative growth is cyclic and almost equal in mature orchards. In young orchards, nitrogen inputs are utilized to support vegetative growth and little nitrogen is removed from the orchard in prunings or crop. During this time nitrogen must be supplied to meet the demand to support vegetative growth. It is estimated that approximately 2.5 lbs nitrogen is required to produce 1,000 lbs. fresh weight of tree growth.
Nitrogen Use Efficiency. Not all the nitrogen supplied to the orchard from fertilizer and other inputs (ie. organic matter, irrigation water) is utilized for tree growth and crop production. A fraction of nitrogen is lost from the orchard ecosystem through processes such as runoff, leaching, and denitrification. Efficiency varies among orchards, with some orchard systems exhibiting higher nitrogen utilization rates than others. The efficiency generally varies from 60% - 90%. Higher values denote more efficient use of nitrogen inputs. To estimate the amount of nitrogen to supply an orchard, the demand is divided by the estimated efficiency. For example, if nitrogen demand is 50 lbs. per acre and efficiency is estimated at 0.8, then 62.5 lbs. of nitrogen per acre should be applied.
Summary. Nitrogen management plans are site-specific and designed to meet orchard and crop demand while reducing environmental losses. Nitrogen utilization is never 100% efficient. Nitrogen use efficiency can be maximized by minimizing losses from irrigation and fertilization practices while utilizing foliar analysis and knowledge of alternate bearing status to fine-tune applications.
Select References:
Fernández-Escobar, et al. 2011. Scientia Horticulturae 127:452–454.
Hartman, H.T. 1958. Cal Ag. Pgs 6-10.
Rodrigues, M.A. et al. 2012. Scientia Horticulturae 142:205-211.
- Author: Ben Faber
|
|
|
|
|
|
|
Two additional RAs were added to the NMP implementation schedule as part of the October 2020 Water Quality Management Plan update: RA 11 (McGrath Lake Coastal) and RA 16 (Boulder Creek). Both are shown in green with a deadline of the end of April 2021.
Note: This workshop is intended for growers, and their representatives, who are looking to learn more about nitrogen management planning and/or intend to self-certify their plans. If you are only looking to fulfill the annual 2-hour continuing education requirement, there is an event approved for education credit on April 16 (below) and additional events will be announced as they are scheduled throughout the year.
**6 Hours of VCAILG CEUs pending Regional Board approval
- Author: Ben Faber
The Fertilizer Research and Education Program has just published its 2020 proceedings. FREP was established in 1990 through legislative action to provide funding for research and education regarding the agronomically safe and environmentally sound use of fertilizer in California. The program is part of the California Department of Food and Agriculture (CDFA), Division of Inspection Services, within the Feed, Fertilizer, and Livestock Drugs Regulatory Services branch. At the website you can review the fertilizer guidelines for many of the crops grown in California.

- Author: Ben Faber
Please register for Nitrogen Management Plan Self-Certification Webinar on Tuesday and Wednesday, November 17/18, 2020 9:00 AM - 12:00 PM PST at:
https://attendee.gototraining.com/r/2352520986463929090
This workshop is intended for growers, and their representatives, who are looking to learn more about nitrogen management planning and/or intend to self-certify their plans. It is also a good education on how nitrogen works in our environment and how it can be managed. The program is sponsored by CA Department of Food and Agriculture, University of CA Cooperative Extension and the Ventura County Irrigated Lands Group.
Attendees must participate in both sessions to receive education credit and qualify to take the online certification test after the final webinar session.
After registering you will receive a confirmation email containing information about joining the training.
This workshop will open a half hour early at 8:30am, to allow attendees to test their connection and access the GoTo Training webinar link.
Email organizer: training@lwa.com
Image of nitrogen deficient avocado leaf on left