Author: Lynn M. Sosnoskie
The first step in developing an effective weed management program is being able to properly identify the weed species that are infesting a field. But why? Why is weed identification so important? Simply stated, because not all weeds are created equal. Species differ with respect to their emergence timing, life history traits, competitive interactions with the crop, potential to harm livestock, and sensitivity to available herbicides, among other characteristics. Consequently, the type and timing of weed control events need to be designed to target problematic species so that control is maximized, weed seed return to the soil seedbank is minimized, and crop yields are protected.
Unfortunately, weed identification is not a simple task, especially when it comes to members of the Asteraceae, one of the largest plant families in the world. Several species of Asteraceaeare found throughout California in many different agricultural and non-agricultural environments; these include: hairy fleabane (Erigeron (Conyza) bonariensis), horseweed (Erigeron (Conyza) canadensis), annual sowthistle (Sonchus oleraceus), prickly lettuce (Lactuca serriola), and common groundsel (Senecio vulgaris). This blog post has been developed to help you distinguish among these species.
Erigeron bonariensis – Hairy fleabane:
Hairy fleabane (also called asthma weed and flax-leaf fleabane)is an annual (sometimes biennial) herb that is not native to California. According to the USDA's PLANTS database (https://plants.usda.gov), the species can be found in California, Arizona, Nevada, Oregon, and Washington, and across the southern US to as far north as Virginia. Biotypes of fleabane can emerge fall through spring with the earlier emerging plants over-wintering as rosettes. Leaves are a grey-green in color, hairy, and are arranged alternate to each other up them stem. While the first leaves produced are roughly oval- to spatula-shaped, later leaves tend to be more linear with entire to weakly-toothed margins. Leaves of hairy fleabane are also twisted, crinkled or wavy in appearance. At bolting, hairy fleabane produces a many branched stem (0.5 to 3 feet [0.15 to 0.9 m] in height) upon which numerous cream- to yellow-colored, urn-shaped flowers are held. Lateral branches can be longer in length than the main stem. Flowering occurs, predominantly, mid-summer through fall. The resultant seedheads resemble those of dandelions, although they are much smaller in size (0.2 to 0.3 inches [5 to 7 cm] in diameter). Individual seeds possess a pappus (0.12 to 0.16 inches [3 to 4 mm] in length) on one end that facilitates wind-dispersal. Hairy fleabane plants produce taproots along with fibrous lateral roots. Hairy fleabane populations with resistance to glyphosate and with resistance to both glyphosate and paraquat have been confirmed in California.
Hairy Fleabane: http://ipm.ucanr.edu/PMG/WEEDS/hairy_fleabane.html
Hairy fleabane leaves are grey-greenish in color and are often wavy, crinkled, or twisted. Photo by L. M. Sosnoskie.
At bolting, hairy fleabane sends up a many branched stem upon which flowers are produced. From: Weeds of California and Other Western States by J. M. Ditomaso and E. A. Healy. Photo by J. M. DiTomaso.
Hairy fleabane seedheads. Individual seeds each possess a pappus, a tuft of hair that aids with wind dispersal, which gives the seedheads their fuzzy appearance. Photo by L. M. Sosnoskie.
Erigeron canadensis – Horseweed:
Horseweed, also known as marestail,is an annual (sometimes biennial) herb that is native to California and most other parts of North America. According to the USDA's PLANTS database, the species has been found in all 50 states and most of Canada. The pattern of horseweed germination and emergence is like that of hairy fleabane, were seedlings can emerge from the fall through early spring. At the seedling stage of development, horseweed and hairy fleabane resemble each other, morphologically; differences become more apparent past the 12 to 15 leaf stage. Not unlike hairy fleabane, horseweed leaves are alternate and are arranged in a rosette; however, horseweed leaves are typically a darker shade of green, as compared to fleabane. Upon bolting, plants send up a single, erect stem that can reach heights of 10 feet (3 m). Unlike hairy fleabane, lateral branches are shorter than the main branch. Cream- to yellow-colored, urn-shaped flowers are produced in dense panicles from mid-summer to fall. Like hairy fleabane, horseweed produces seeds that possess a pappus (0.10 to 0.12 inches [2.5 to 3 mm] in length) that facilitates wind dispersal. Whereas single hairy fleabane plants have been estimated to produce up to 20,000 seeds per plant, horseweed plants can produce more than 200,000. Horseweed plants produce taproots along with fibrous lateral roots. Horseweed populations with resistance to glyphosate and with resistance to both glyphosate and paraquat have been confirmed in California.
Horseweed: http://ipm.ucanr.edu/PMG/WEEDS/horseweed.html
Horseweed (L) and hairy fleabane (R) rosettes. Hairy fleabane leaves are grey-greenish in color and are often wavy, crinkled, or twisted as compared to horseweed. Photo by L. M. Sosnoskie.
Bolting horseweed. Notice the single erect non-branching stem. Photo by L. M. Sosnoskie.
Horseweed growth habit. At bolting, horseweed sends up a single, erect stem upon which flowers are produced. Horseweed stems can reach heights up to 10 feet. For comparison, Lynn Sosnoskie, an author of this post, is 5'3” tall. Photo by D. D. MacLean.
Sonchus oleraceus – Annual sowthistle:
Annual sowthistle is not native to North America but has become naturalized throughout the US and Canada. The first true leaf is round to egg-shaped although following leaves become spatulate with prickly margins. Older leaves are deeply lobed with the terminal lobe being large and triangular in shape. Stems are erect and hollow between nodes and can reach heights of 4 feet [1.2 m]. Leaves on the stems are clasping at the base. Broken or cut leaves and stems will exude a white sap. Because annual sowthistle can be both a summer and a winter annual, flowers can be produced year-round when conditions are favorable. Flowers (0.5 to 1 inch [1.2 to 2.5 cm] in diameter) resemble those of dandelions except they are a light yellow in color. Like other species in the Asteraceae family, the pappuses (0.2 to 0.3 inches [5 to 8 mm] in length) attached to individual seeds give the seedhead it's cotton ball-like appearance. Seeds are wind-dispersed. Annual sowthistle produces a short and think taproot with many lateral roots.
Annual sowthistle: http://ipm.ucanr.edu/PMG/WEEDS/annual_sowthistle.html
Annual sowthistle seedling. Note the spatula-shaped (spoon-shaped) leaves with finely-toothed margins. Photo by L. M. Sosnoskie.
Mature rosette of annual sowthistle. Leaves are deeply lobed with the terminal lobe being triangular in shape. Photo by L. M. Sosnoskie.
Leaves of annual sowthistle on flower stems are clasping at the base. Photo by L. M. Sosnoskie.
Lactuca serriola – Prickly lettuce:
Prickly lettuce is not native to North America but has become naturalized throughout the US and Canada. The species is classified as a winter and summer annual and sometimes a biennial. The first leaves are football-shaped; later leaves are elongated and club-shaped with weakly-toothed margins. The midrib vein is pronounced and covered with a row of prominent spines on the underside. Plants exist as a basal rosette (with alternating leaves) before sending up a single flower stalk that can reach heights of 6.5 feet (2 m). The stems are covered with stiff hairs that give them a spiny appearance and are branched at the terminal apex. Leaves that form on this stem can be deeply- and pinnately-lobed with rounded indentations; leaf bases are clasping. Stems and leaves exude a milky sap when cut or broken. Flowers (0.3 to 0.4 inches [8 to 10 mm] in diameter) are pale yellow; flowering occurs, typically, from April to October. Like other members of the Asteraceae, prickly lettuce seeds possess pappuses (0.16 to 0.2 (4 to 5 mm) in length) that facilitate wind dispersal. Plants produce deep tap roots with fibrous lateral roots.
Prickly lettuce: http://ipm.ucanr.edu/PMG/WEEDS/prickly_lettuce.html
Young prickly lettuce rosette. Photo by L. M. Sosnoskie
Spines on the underside of the midrib vein of a prickly lettuce leaf. Photo by L. M. Sosnoskie.
A mature Prickly lettuce stem exhibiting prickly/spiny stems and deeply lobed leaves with prominent midrib veins. From: Weeds of California and Other Western States by J. M. Ditomaso and E. A. Healy. Photo by J. M. DiTomaso
Senecio vulgaris – Common groundsel:
Common groundsel is not native to North America but has become naturalized throughout the US and Canada. The species is, predominantly, a winter annual and sometimes a summer annual. The youngest leaves are egg-shaped with shallowly-toothed edges, the base of the leaves may be purple on the underside. Following leaves are alternately arranged in a rosette, are usually more deeply (but irregularly) lobed, and may also be hairless to hairy. At maturity, plants may send up a single stem or else a series of branches from the base; stem heights can reach 2 feet (0.6 m). The plant has often been described as having a ‘ragged' or ‘scraggly' appearance. Common groundsel produces small, yellow flowers in clusters. Individual flowers are surrounded by green bracts with black tips. Flowering can occur throughout the year. Like other members of the Asteraceae, a pappus, a tuft of hair that aids with wind dispersal, is attached to each seed. Common groundsel plants produce small taproots with fibrous lateral roots. The presence of common groundsel can be problematic for alfalfa and forage producers; the species produces pyrrolizidine alkaloids, that can lead to liver damage in livestock following consumption. Even hay with a small amount of contamination can be problematic as pyrrolizidine alkaloid poisoning can accumulate in the livestock over months of feeding.
Common Groundsel: http://ipm.ucanr.edu/PMG/WEEDS/common_groundsel.html
Common groundsel leaves demonstrating deep, but irregular, lobed patterns. Photo by L. M. Sosnoskie.
Common groundsel at the bolting stage. Photo by L. M. Sosnoskie.
Mature groundsel plant displaying a many-branched habit, flowers in clusters, and fuzzy seedheads. Photo by L. M. Sosnoskie.
While similar in general appearance (i.e. alternate leaves arranged as a basal rosette, flowers produced on upright stems, and seeds with pappuses that allow for wind dispersal), hairy fleabane, horseweed, annual sowthistle, prickly lettuce, and common groundsel all possess unique morphological and phenological characteristics that differentiate them from one another. Properly identifying these and other species is the first step in developing a safe and effective weed management program in crop production systems. For example, the type and timing of herbicide applications are directly influenced by the emergence patterns of the target species as well as their sensitivity to available products. For instance, the presence of glyphosate-resistant weeds like hairy fleabane and horseweed may reduce the value of glyphosate as a stand alone active ingredient. Identifying the presence of poisonous plant species is also critical for some producers. Repeated consumption of common groundsel, can result in irreversible liver poisoning of livestock; consequently, a quick and proper ID of this species is important for limiting animal exposure to toxic alkaloids. Many tools are available to assist growers, PCAs, and other crop consultants with weed identification; this includes the reference materials listed below. Your local county farm advisors can also be an extremely useful resources for determining the identity of suspect species.
References:
Biology and management of horseweed and hairy fleabane in California. By: Anil Shrestha, Kurt Hembree, and Steven Wright. https://anrcatalog.ucanr.edu/pdf/8314.pdf
Glyphosate-resistant hairy fleabane documented in the Central Valley: By: Anil Shrestha, Brad Hanson, and Kurt Hembree. http://calag.ucanr.edu/archive/?type=pdf&article=ca.v062n03p116
Weeds of California and Other Western States. By: Joe DiTomaso and Evelyn Healy, https://anrcatalog.ucanr.edu/Details.aspx?itemNo=3488 (For purchase, link to online UC ANR store)


- Author: Ben Faber
Irrigation and Nutrient Management Field Day
Monday January 14, 2019
555 Las Varas Canyon Road Goleta 7:30 am - 11:30 pm, lunch is provided The event is FREE
but please register -
https://www.eventbrite.com/e/irrigation-nutrient-management-field-day-tickets-54248292102
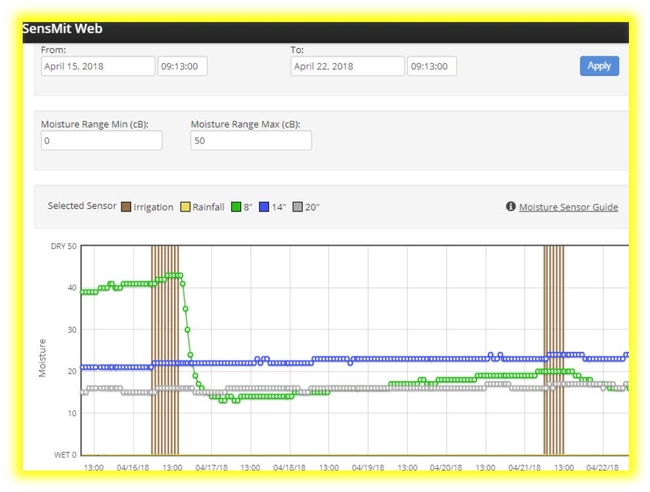
Learn all about soil moisture sensor systems -
• Using sensors to save water, energy, & fertilizer
• Monitoring & interpreting soil moisture data
• Checking & fixing common system problems
• System configurations & funding opportunities
• Practical and common sense considerations
Attendees get a
FREE Soil Nitrate
Quick Test Kit
The Cachuma Resource Conservation District, with funding from the Department of Water Resources, is
pleased to offer this FREE field day event in partnership with:
Parks Land and Cattle Company The Irrometer Company
Natural Resource Conservation Service
Picture: Soil Moisture Testing by the" Feel Method"
“Wood chip mulches will decrease soil nitrogen and spread pathogens” A Misunderstanding that is addressed below by:
Chalker-Scott, L. , Extension Specialist And Associate Professor, Washington State University
Downer, A.J., Farm Advisor, University of California
With chronic drought and/or record-breaking summer temperatures making it increasingly important to conserve water, many gardeners and groundkeepers are using landscape mulches. The ideal landscape mulch not only moderates soil temperature and conserves water, but also:
- reduces compaction;
- provides nutrients;
- enhances plant growth;
- provides habitat for beneficial insects;
- helps control weeds, pests and disease; and
- reduces the need for pesticides and fertilizers.
In addition, landscape mulches should be readily available, affordable, and easy to apply and replace. A review of the literature on landscape mulches (Chalker-Scott, 2007) determined that organic mulches are overall the best choice, with deep layers of coarse woody material providing most or all of the above-listed benefits. Arborist wood chips (created from leaves and branches chipped up by tree service companies) are a particularly good option as they are generally inexpensive and easy to obtain anywhere trees are managed.
Fortunately, none of these concerns are validated by research. Here are some brief explanations (Chalker-Scott, 2007) targeted to our audience:
- Wood chips will not draw nitrogen from the soil unless they are incorporated into it. When used as mulch, arborist chips have no effect on underlying soil nitrogen levels, except to increase them over time.
- Wood chip mulches, even those made from diseased trees, will not transmit pathogens to healthy plant roots. If diseased chips are incorporated into the soil they could infect plant roots, but field evidence of this is rare. Arborist chips that are stockpiled even for a few days undergo severe pathogen reduction through microbial attack within the pile (Downer et al., 2008).
- Wood chips, or any other organic mulch, will not change the pH of the soil. The soil volume is vast, and any acidification would occur only at the mulch-soil interface where it would quickly be neutralized.
- Wood chips, even those made from black walnut or cedar, will not kill landscape plants. There is no reliable evidence that chemical inhibition from decaying wood actually occurs in a landscape situation.
- Wood chip mulches do not lend themselves to tunnel building like landscape fabric and other sheet mulches do: they collapse. Termites do not eat wood chips unless they have no choice; they are negatively affected by some of the chemicals wood contains. In fact, arborist chip mulches house a number of beneficial insects and other species that naturally control pests.
For arborist wood chip mulches to be the most effective (Chalker-Scott, 2007), they should be:
- coarse – no less than ½” diameter – so water and air can move freely through them;
- applied as soon as possible after chipping both to maximize the materials available to microbes and to capture the nutrients released by their activity in the soil; and
- maintained at a depth of at least 4” to prevent weed growth.
Read on:

- Author: Ben Faber
Groundwater pumping may significantly affect growers' energy costs. As part of a California Energy Commission-funded research project, Lawrence Berkeley National Laboratory wants to hear directly from growers—via a short survey—about their experiences with the energy needed to pump groundwater. To learn more about the larger project, click here.
Your participation in this survey is greatly appreciated. We hope to understand more about the conditions you face on the ground so we can accurately convey your experiences with pumping groundwater to policymakers. We will be asking you some questions about your reliance on groundwater, past and planned well operation practices, and barriers to lowering the energy needed to pump groundwater. Results from this survey may inform future efforts to reduce energy use and costs for growers.
Your participation in this survey is completely anonymous. The survey software will not capture any personally identifiable information, including IP address. Individual anonymous responses will be stored in a secure location accessed only by a few authorized LBNL researchers, and will not be made public in any form. In publicly available reports, results will only be presented in aggregated form.
This survey should take around 20 minutes to complete. To preview the survey before taking it, a PDF version can be viewed here. If you are at least 18 years of age, own or operate a farm or ranch in California, and rely at least to some extent on groundwater for your farm or ranch operations, please click the link below to complete the questionnaire.
If you have questions about this survey or the larger project, or would prefer to respond to this survey over the phone, please contact us at gwenergy@lbl.gov, (510) 486-6839 [Heidi Fuchs, Survey Lead], or (510) 495-2865 [Helcio Blum, Project Lead]. For issues related to your rights as a research participant in this study (LBNL HSC 382H001-31AU19), please contact LBNL's Human Subjects Committee at (510) 486-5399.
Learn more and take the survey at
https://gwenergy.lbl.gov/growers
Questions? Contact us at gwenergy@lbl.gov or (510) 486-6839
Thank you very much for your time and participation!

Tuning up for Pruning Up--Care, Maintenance and Utilization of Hand Pruning Tools
J. Downer
University of California
Fall is passing into winter and the bare sticks in my deciduous fruit orchard are calling me to my annual fruit tree pruning chores. I can prune my entire orchard with very few tools: a good pair of bypass clippers, a similar set of loppers (optional) and a high quality “razor” or “tri edge” saw. Most tools require some maintenance especially the clippers and loppers. Sharpening is the usual need. Clippers are easily sharpened but modern saw blades can not be sharpened by gardeners and should be replaced. Sometimes it is just as easy to buy a new saw, replacing the old one when blade eventually dulls or is bent from over zealous use (illustration 1)
Illustration 1: Tri-edge saw blades are made from stainless steel and are not easily sharpened. When dull or bent they should be replaced
Before using your pruning tools inspect them for signs of damage. Blades should be sharp and unbent. Loppers should have their rubber “bumpers” intact otherwise your knuckles will be smashed after exerting force on a difficult branch. Sharp tools offer less resistance and actually decrease injury to users. One exception here is with the modern “tri-edge” or “razor” saws. These saws can cut so quickly that you may pass through the branch you are cutting and continue on to some part of your anatomy quickly ripping your flesh. I have suffered more cuts (some serious) from these saws than from any other gardening activity. They should be used with careful precision, not with the wild abandon and pruning fervor of the craven academic desperate for real world gardening experience. A thick long sleeved shirt and gloves will also help prevent cuts from hand pruning equipment.
Bypass clippers are so termed because the blade passes by the hook. To sharpen these, find the bevel on the edge of the clippers and align a small file to the same angle of this bevel, and file the bevel until you can feel the sharpness with your finger (Illustration 2). Never sharpen the back side of the bevel—this will create a gap, and every time you cut, a flap of tissue will remain. Back bevel sharpened clippers will require blade replacement or grinding until the back bevel is gone. The hook does not require sharpening, do not attempt to file it. Repeat this process with lopper blades.
Illustration 2: To sharpen bypass clipper blades follow the angle of the bevel. Do not sharpen the flat side of the blade
When you are done pruning for the day, wipe the blades of your clippers and loppers with an oil soaked rag or apply a few drops of oil and rub it into the blade. Most modern saws blades are made from stainless steel and require no oil protection.
As a Cooperative Extension Advisor, one of the most common questions I receive is: “Should I sanitize my clippers between cuts or between uses on various plants?”. Indeed, many publications, extension leaflets, gardening columns, and other sources make broad recommendations to sanitize clippers after every cut. Some articles even compare various products for their killing efficacy. Often blind recommendations are made to sanitize clippers when the pathogen is not even known or specified. It is not necessary to sanitize your clippers when pruning most garden plants and fruit trees. There are a few pathogens that are spread by dirty pruning equipment but published evidence that they are spread by hand pruning equipment (especially clippers) is nil. One exception is palm wilt caused by Fusarium oxysporum f.sp. canariensis which is easily spread by saws. Some of the canker fungi caused by Botryosphaeria can also be spread by pruning equipment. With many of these pathogens, a wound is required for infection so it may not be that the clippers are spreading disease so much as providing an entry point (infection court) so that pathogens have a way to enter.
In my garden, I have never, and will never need to sanitize clippers between cuts. However, conditions vary across the US, and in some places rain, humidity, and temperature are more favorable for disease development. To avoid spreading pathogens, prune during the dormant season, when the likelihood of pathogen activity is lowest. Apply dormant sprays containing copper to limit the onset of new fungal diseases that may enter pruning wounds. If you still feel you need to protect wounds from dirty clippers I like to use the flame from a plumber's torch to sanitize. A few seconds along the cutting edge front and back kills all pathogens (Illustration 3). Similar for a saw but efficacy is increased if the saw gullets are wiped clean with a cloth and then the flame applied. The only time I take these measures is when I know I am working with plants that can be inoculated with pathogens by pruning (which is rare).
Illustration 3: A plumber's torch will rapidly sanitize saws and blades when pathogens are present in plant tissues.
When pruning garden plants, there are a plethora of recommendations on how to make cuts. Rose experts have extolled the virtues of an angled cut so water runs away quickly, flush cuts used to be recommended by arborists as the highest quality cut. These examples are without research foundation. Cuts on woody plants should be angled to produce a circular exposure that is the smallest surface area possible. We abandoned flush cuts many years back because they cut into protective zones that limit decay in trees. Some gardeners feel compelled to cover their cuts with a pruning paint and there is a similar paucity of research to support this practice. Leave pruning wounds unpainted.



















