- Author: Ben Faber
WESTMINSTER, Colorado - An article in the most recent edition of the journal Weed Science shows that cover crops can play an important role in slowing the development of herbicide resistant weeds.
Researchers conducted field experiments in Pennsylvania to explore how cover cropping tactics influenced the management of horseweed in no-till grain crops. Seven cover-cropping treatments were used over two subsequent growing seasons.
There were several significant findings. In comparison to fallow control plots, cover crop treatments reduced horseweed density at the time of a pre-plant, burndown herbicide application by 52% in the first year and 86% in the next. This reduced the herbicide "workload" and lowered the selection pressure for resistant weeds. Cereal rye alone or in combination with forage radish was found to provide the most consistent horseweed suppression.
Importantly, winter hardy cover crops also reduced horseweed size inequality - meaning fewer large horseweed plants were found at the time of herbicide application. Researchers say this reduces the chance of a size-dependent fitness advantage for horseweed biotypes that develop herbicide resistance.
"Our hope is that understanding the complementary relationship between cover crops and herbicides can lead to new weed control strategies that slow the development of herbicide resistance," says John M. Wallace, Ph.D., of Pennsylvania State University.
Full text of the article "Cover crop effects on horseweed (Erigeron canadensis) density and size inequality at the time of herbicide exposure" is now available in Weed Science Volume 67, Issue 3.

- Author: Ben Faber
A garden can be a competitive environment. Plants and unseen microorganisms in the soil all need precious space to grow. And to gain that space, a microbe might produce and use chemicals that kill its plant competitors. But the microbe also needs immunity from its own poisons.
By looking for that protective shield in microorganisms, specifically the genes that can make it, a team of UCLA engineers and scientists discovered a new and potentially highly effective type of weed killer. This finding could lead to the first new class of commercial herbicides in more than 30 years, an important outcome as weeds continue to develop resistance to current herbicide regimens.
Using a technique that combines data science and genomics, the team found the new herbicide by searching the genes of thousands of fungi for one that might provide immunity against fungal poisons. This approach is known as "resistance gene-directed genome mining."
The study, which was published in Nature, also points to the potential for this genomics-driven approach to be used in medicine, with applications ranging from new antibiotics to advanced cancer-fighting drugs.
"Microorganisms are very smart at protecting themselves from the potent molecules they make to kill their enemies," said Yi Tang, the study's co-principal investigator and a UCLA professor of chemical and biomolecular engineering, and of chemistry and biochemistry. "The presence of these resistance genes provides a window into the functions of the molecules, and can allow us to discover these molecules and apply them to diverse applications in human health and agriculture."
For example, if a resistance gene that protects a microorganism from an anti-bacterial product is found, there's a possibility that the microorganism also has genes to produce that same anti-bacterial compound. That discovery could potentially lead to new antibacterial medicines.
The new herbicide acts by inhibiting the function of an enzyme that is necessary for plants' survival. The enzyme is a key catalyst in an important metabolic pathway that makes essential amino acids. When this pathway is disrupted, the plants die.
This pathway is not present in mammals, including humans, which is why it has been a common target in herbicide research and development. The new herbicide works on a different part of the pathway than current herbicides. A commercial product that uses it would require more research and regulatory approval.
"An exciting aspect of the work is that we not only discovered a new herbicide, but also its exact target in the plant, opening the possibility of modifying crops to be resistant to a commercial product based on this herbicide," said study co-principal investigator Steven Jacobsen, a professor of molecular, cell and developmental biology in the UCLA College and an investigator of the Howard Hughes Medical Institute. "We are looking to work with large agrochemical companies to develop this promising lead further."
To confirm the efficacy of the new herbicide, the UCLA team tested the fungus-produced product on a common plant used in lab studies called Arabidopsis. In experiments, the product killed the plants after they were sprayed with it. The researchers also implanted the resistance gene from the fungus into Arabidopsis genomes. The plants that had the resistance gene implanted in them were immune to the herbicide.
"The emergence of herbicide-resistance weeds is thwarting every herbicide class in use; in fact, there has not been a new type commercialized within the last 30 years," said Yan Yan, a UCLA chemical engineering graduate student who was a lead author of the paper. "We think this new, powerful herbicide -- combined with crops that are immune to it -- will complement urgent efforts in overcoming weed resistance."
Resistance-gene-directed discovery of a natural-product herbicide with a new mode of action
https://www.nature.com/articles/s41586-018-0319-4
https://www.eurekalert.org/pub_releases/2018-07/usso-rdn071318.php

- Author: Lynn M. Sosnoskie
Agronomy and Weed Science Advisor, Merced County
It's getting hot and dry in the Central Valley and the movement of equipment in and out of fields/orchards/vineyards has the potential to stir up a significant amount of dust. Among its other impacts to agriculture (soil erosion, tissue damage, reduced photosynthesis, etc...), wind blown dust can reduce the efficacy of glyphosate, which is an important tool for the management of weeds in trees and vines, along rights-of-ways, and in glyphosate-tolerant agronomic crops (e.g. corn, cotton, alfalfa) in CA.
The adoption of glyphosate has been facilitated, at least in part, by it's relative lack of soil activity (Miller et al. 2013; Zhou et al. 2006). Glyphosate can become tightly adsorbed to soil particles (depending on clay and organic matter content, pH, cation exchange capacity, etc...), thereby reducing the potential for crop injury via root uptake. The ability of glyphosate to bind to soil also contributes to it's reduced efficacy in certain situations. Specifically, dust that settles on weed leaf surfaces (Figure 1) can negatively impact glyphosate performance due to binding/inactivation.
Figure 1. Dirt on field bindweed (Convolvulus arvensis) leaves
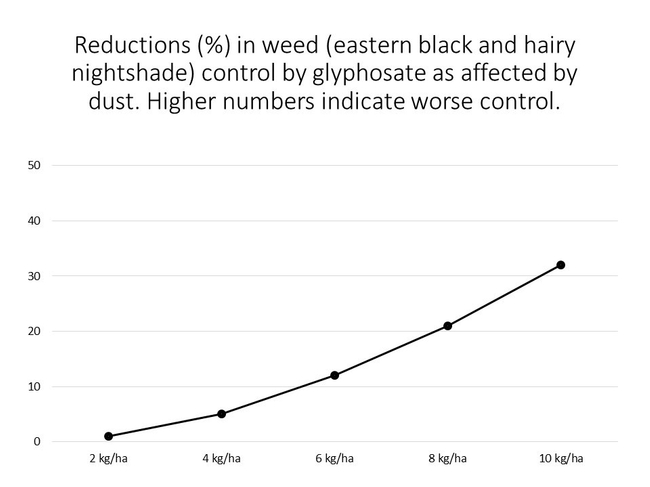
The detrimental effects of dust on glyphosate performance have been described by Zhou et al. (2006) and Boerboom et al. 2006). Results from Zhou et al. (2006) showed that dust applied to the surface of eastern black (Solanum ptychanthum) and hairy (Solanum sarrachoides) nightshades reduced weed control, with greater amounts of dust resulting in greater reductions in herbicide efficacy (Figure 2). In a study conducted by Boerboom et al. (2006), dust was deposited over the tops of plots of common lambsquarters (Chenopodium album); water was then used to remove the dust treatment from ½ of the plots prior to a glyphosate application. Similar to the results achieved by Zhou et al. (2006), the occurrence of dust visually reduced common lambsquarters control (relative to the plots where the dust had been washed away). Results from both sets of studies show that dust generation has the potential to significantly impact glyphosate performance'
Figure 2. Percent reduction in weed control by glyphosate as affected by the rate of a silty clay dust applied to the leaves of two nightshade species. Greater numbers on the Y-axis indicated greater reductions in control.
Adapted from Zhou et al. (2006) Weed Science 54:1132-1136.
A few closing thoughts about dust and it's impact on weed control:
- Be mindful of how soil disturbance (cultivation, farm traffic, etc...) affects dust production.
- Make glyphosate applications in advance of crop production events that are likely to generate substantial amounts of dust.
- Sprinkler irrigation may be able to remove dust from the leaves of weeds under some situations. Glyphosate applications should be made after the leaves have dried but before more dust can be deposited.
- Soil particles in spray water can also bind to glyphosate and reduce herbicide efficacy; only clean water should be used to fill spray tanks. (Not actually dust related, but a good practice to remember)
Citations:
Boerboom, C. et al. (2006) Factors affecting glyphosate control of common lambsquarters. Proceedings of the North Central Weed Science Society 61:54.
Miller, T. et al. (2013) Glyphosate stewardship: Maintaining the effectiveness of a widely used herbicide. ANR Publication 8492. https://anrcatalog.ucanr.edu/pdf/8492.pdf
Zhou, J. et al. (2006) Soil dust reduces glyphosate efficacy. Weed Science 54:1132-1136.
Note: An earlier version of this post is available at the following website: http://treefruit.wsu.edu/dust-can-affect-weed-control-with-glyphosate/
- Author: Ben Faber
A recent call about the poor control of marestail (horseweed, Conyza canadensis) to glyphosate (Roundup®) wasn't surprising, but that paraquat didnt do the trick was. It turns out that there is multiple resistance to the materials. If horseweed is resistant to glyphosate it is possibly going to be resistant to paraguat which also means that hairy fleabane which has glyphosate resistance could also show resistance to paraquat. A recent study reports on the increased Conyza resistance to paraquat (Distribution of Conyza sp. in Orchards of California and Response to Glyphosate and Paraquat, Moretti et al, https://doi.org/10.1614/WS-D-15-00174.1):
Resistance to glyphosate in hairy fleabane and horseweed is a problem in orchards and vineyards in California. Population genetic analyses suggest that glyphosate resistance evolved multiple times in both species, but it is unknown if resistance to other herbicides is also present. Two approaches of research were undertaken to further evaluate herbicide resistance in Conyza sp. in the perennial crop systems of California. In the initial study, the distribution of Conyza sp. in the Central Valley, using a semistructured field survey, was coupled with evaluation of the presence and level of glyphosate resistance in plants grown from field-collected seed. In a subsequent study, single-seed descendants representing distinct genetic groups were self-pollinated in the greenhouse and these accessions were evaluated for response to glyphosate and paraquat. Conyza sp. were commonly found throughout the Central Valley and glyphosate-resistant individuals were confirmed in all field collections of both species. The level of glyphosate resistance among field collections varied from 5- to 21-fold compared with 50% glyphosate resistance (GR50) of the susceptible, with exception of one region with a GR50 similar to the susceptible. When self-pollinated accessions from different genetic groups were screened, the level of glyphosate resistance, on the basis of GR50 values, ranged from 1.7- to 42.5-fold in hairy fleabane, and 5.9- to 40.3-fold in horseweed. Three accessions of hairy fleabane from different genetic groups were also resistant to paraquat (40.1- to 352.5-fold). One glyphosate-resistant horseweed accession was resistant to paraquat (322.8-fold), which is the first confirmed case in California. All paraquat-resistant accessions of Conyza sp. identified so far have also been resistant to glyphosate, probably because glyphosate resistance is already widespread in the state. Because glyphosate and paraquat resistances are found across a wide geographical range and in accessions from distinct genetic groups, multiple resistant Conyza sp. likely developed independently several times in California.

- Author: Ben Faber
So, this image comes across with the question of what is going on. It's a grapefruit, but what is going on with the leaves? It's happening to two grapefruit side by side or two different ages. It's not affecting other citrus nearby, and not a grapefruit several hundred feet away. Nothing like insect damage. It's not a nutrient deficiency because it's not following a clear pattern. It doesn't look like a chimera, which is common in citrus, because it's only a few outer leaves. It doesn't fit the pattern of an herbicide. It wouldn't be a contact or a drift spray because is both veinal and interveinal, and not strongly one or the other, so it's not consistent with a soil-applied herbicide either.
A plant pathologist and an herbologist (weed specialist) both asked if there had been a change in light -Different light exposure or more drought stress. Was a nearby tree removed or somehow the tree got more exposure than previously? Citrus Specialist, Peggy Mauk, nailed it, though. She said this type of coloration is characteristic of ‘Star Ruby' grapefruit when it is water stressed. And this can occur when the tree is more exposed to wind or light which is what happened here when a nearby hedge was pruned. The other citrus being less affected by such obvious symptoms, although probably stressed, as well.