- Author: Kathy Keatley Garvey

Enter Filipa Rijo-Ferreira, a UC Berkeley School of Public Health (BPH) assistant professor who specializes in parasitology and circadian rhythms.
She'll present a UC Davis Department of Entomology and Nematology seminar at 4:10 p.m., Wednesday, Oct. 19 on "Circadian Rhythms in Parasitic Diseases" in 122 Briggs Hall. Her seminar also will be virtual. The Zoom link is https://ucdavis.zoom.us/j/95882849672. Host is molecular geneticist and physiologist Joanna Chiu, professor and vice chair of the UC Davis Department of Entomology and Nematology.
"Malaria's main symptom is the periodic fevers experienced by patients, fevers that ‘come and go' at certain times of the day and are a consequence of synchronized parasite rhythms," Rijo-Ferreira says in her abstract. "In humans, circadian clocks regulate multiple aspects of physiology, including sleep-wake cycles, metabolism, and immune defense. Circadian biology leads to body rhythms experienced by the pathogens that infect humans. In addition to sensing host rhythms, we recently discovered that parasites which cause devastating health burdens such as malaria and sleeping sickness diseases also have their own intrinsic clocks. The clocks of parasites regulate core biological functions from metabolism to the cell cycle, and the discovery of the existence of their clocks serves as an opportunity to access the molecular mechanisms regulating their rhythmic biology."
A native of Lisbon, Portugal, Rijo-Ferreira joined the UC Berkeley Public Health faculty in January 2022. She describes herself as a "scientist passionate about the complex daily host-parasite interactions and how parasites evolved circadian clocks to anticipate environmental cycles." She recently authored "The Malaria Parasite Has an Intrinsic Clock," published in Science magazine.

"Our lab is interested in parasitic infections and we study them under the lenses of time of day," Rijo-Ferreira wrote on her lab website. "Our rhythmic world has been a driving force for organisms to evolve a molecular clock to anticipate such daily rhythms. Similarly, our own circadian biology leads to physiological rhythms that parasites experience.We study the single-celled parasites Plasmodium spp. that causes malaria, and Trypanosoma brucei that causes sleeping sickness. We employ technical approaches spanning from next-generation sequencing, to cellular and behavioral assays to investigate the interactions of these parasites with their hosts.Our work seeks to understand how circadian rhythms modulate host-parasite-vector interactions and identify opportunities in their rhythmic biology to treat parasitic infections
In an interview with BPH staff writer Eliza Partika, published in February 2022, she commented: "I am fascinated about our day and night cycles and how organisms evolved to anticipate them. I find it incredible that parasites, such as the ones that cause malaria, show a coordinated rhythmic pattern themselves, which underlies periodic fevers in infected individuals. Our research is aimed at understanding how this phenomenon is regulated molecularly, and how we can disrupt these rhythmic patterns to offset the infection."
"At BPH, we aim to set up a framework where we can explore the relationships between parasites, hosts, and the mosquitoes that serve as the vector of disease transmission, based on the time of day," Rijo-Ferreira related. "We hypothesize that the circadian rhythms of these three organisms need to be aligned in order for the parasite to cause an efficient infection. In fact, when rhythms are misaligned, there is a reduction in parasite levels. Thus, identifying the molecular players from host, parasite, and mosquito is essential to understanding this phenomenon and creating alternative strategies to manage deadly infections like malaria and sleeping sickness."
Rijo-Ferreira said she seeks to "bring to the attention the circadian aspect of infectious diseases and bring awareness of the potential benefits of time of day vaccination and drug treatment."
Emily Meineke, assistant professor of urban landscape entomology, UC Davis Department of Entomology and Nematology, coordinates the department's seminars for the 2022-23 academic year. All 11 seminars will take place both person and virtually at 4:10 p.m. on Wednesdays in Room 122 of Briggs Hall except for the Nov. 9th and Dec. 7th seminars, which will be virtual only, she said. (See list of seminars)
For further information on the seminars or to resolve any technical difficulties with Zoom, contact Meineke at ekmeineke@ucdavis.edu.

- Author: Kathy Keatley Garvey

"We are working with scientists and public health authorities in STP to establish the conditions that would facilitate an informed societal and government decision about a proposed release of Anopheles mosquitoes engineered to prevent transmission of the malaria parasite Plasmodium falciparum on the islands,” said principal investigator Gregory Lanzaro, director of the Vector Genetics Laboratory and a PMI professor.
This award will be used to extend their ongoing entomological, engagement and capacity building work through 2025.
“We are working in collaboration with the UC Irvine Malaria Initiative (UCIMI), a research consortium including scientists from UC Irvine, San Diego and Berkeley as well as Johns Hopkins University,” Lanzaro said. “We are working toward the application of advanced genetic tools aimed at the mosquito vector. It is our belief that this approach, used in conjunction with early malaria treatment and detection, can provide a cost effective, sustainable, and environmentally responsible program to ultimately eliminate malaria from Africa.”
Said Ana Kormos, engagement program manager and lead author of the proposal: “These funds provide the UCIMI program with support to strengthen our existing relationship-based approach to the co-development of this technology and ensures that our partners in STP lead the decision-making processes involved in all aspects of the research. This is a huge step forward in advancing a truly collaborative approach to translational research.”

The Vector Genetics Laboratory is engaged in research and training in the areas of population and molecular genetics, genomics and bioinformatics of insect vectors of human and animal disease. The website: “We have developed a program aimed at expanding knowledge that may be applied to improving control of disease vectors and that also addresses problems of interest in the field of evolutionary genetics. We are currently engaged in a range of projects, but our major research focus is on vectors of malaria in Africa."
Directors of the Vector Genetics Laboratory research programs are Lanzaro and Anthony "Anton" Cornel, a research entomologist with the UC Davis Department of Entomology and Nematology and director of the Mosquito Control Research Laboratory, Parlier.
New Tools. "The fight to reduce and possibly eliminate malaria continues and becomes especially challenging as efforts to reduce malaria morbidity have plateaued since 2015,” said Cornel. “Therefore, we must seriously consider new tools. One such tool is genetically modifying the major mosquito vector in the Afrotropics so that it cannot transmit malaria."
"The project aims to use genetically modified (GM) mosquito strategy to reduce and eliminate malaria from the Islands of São Tomé and Príncipe, as proof of concept, before using this technology on larger scales on mainland Africa,” Cornel said, adding that his role, as a field team co-investigator for UCIMI and VGL, is to work with Lanzaro and Pinto “to understand as much as we can about the behavior, population structure and population sizes of Anopheles coluzzi (the malaria vector) on these islands to design the most efficient strategy of releasing the genetically modified mosquitoes to have maximum effect.”
Malaria is an acute illness caused by Plasmodium parasites, which spread to humans through the bites of infected female Anopheles mosquitoes, according to the World Health Organization (WHO). In 2020, nearly half of the world's population was at risk of malaria. An estimated 241 million cases of malaria occurred worldwide in 2020, with 627,000 dying.
Tremendous Burden. Medical entomologist and geneticist Geoffrey Attardo of the UC Davis Department of Entomology and Nematology (who is not involved in this project), noted that “Malaria is a disease which creates a tremendous burden on people living in affected areas. In particular its impacts on the mortality in young children and pregnant women are devastating. Attempts to control this disease using traditional methods have been effective in recent years.”
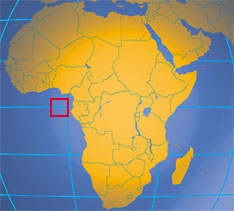
The island nation of São Tomé and Príncipe, population of 178,700 in 2016, is located about 200 miles west of Gabon on Africa's mainland. It shares maritime borders with Equatorial Guinea, Gabon, and Nigeria. The combined area of the archipelago is about five times the size of Washington, DC. The United States established diplomatic relations with São Tomé and Príncipe in 1976, following its independence from Portugal.
Open Philanthropy's mission, as noted on its website, is to “give as effectively as we can and share our findings openly so that anyone can build on our work. Through research and grant-making, we hope to learn how to make philanthropy go especially far in terms of improving lives. We're passionate about maximizing the impact of our giving, and we're excited to connect with other donors who share our passion.”
Resource:
São Tomé and Príncipe (nationsonline.org)


- Author: Kathy Keatley Garvey

In humans, the disease is commonly known as sleeping sickness: the parasite invades the central nervous system and disrupts the sleep cycle. If not treated, the disease can result in progressive mental deterioration, coma, systemic organ failure and death.
The newly published research in the journal Genome Biology compares and analyzes the genomes of six species of tsetse flies and could lead to better insights into disease prevention and control. “It was a behemoth project, spanning six to seven years,” said Attardo, an assistant professor in the Department of Entomology and Nematology. “This project represents the combined efforts of a consortium of 56 researchers throughout the United States, Europe, Africa and China.”
Tsetse flies, Glossina sp., are of great medical and economic importance, wrote Attardo and co-authors Adly M. M. Abd-Alla of the Insect Pest Control Laboratory, Division of Nuclear Techniques in Food and Agriculture, Vienna, Austria, and Serap Aksoy of the Yale School of Public Health, New Haven, Conn. They related that since the implementation of surveillance and record-keeping in the 20th century, “millions of people in sub-Saharan Africa” have died from sleeping sickness.
Tsetse flies, which resemble house flies, are distinguished from other Diptera by unique adaptations, including lactation and the birthing of live young, a vertebrate blood-specific diet by both sexes, and obligate bacterial symbiosis. The scientists targeted six Glossina genomes representing three sub-genera: Morsitans (G. morsitans morsitans, G. pallidipes, G. austeni), Palpalis (G. palpalis, G. fuscipes), and Fusca (G. brevipalpis) which represent different habitats, host preferences, and vectorial capacity.
“The aim of these studies,” the authors wrote, “was to generate and mine the genomic sequences of six species of tsetse flies with different ecological niches, host preferences, and vectorial capacities. The goals of the analyses performed here are to identify the novel genetic features specific to tsetse flies and to characterize the differences between the Glossina species to correlate the genetic changes with phenotypic differences in these divergent species.”
“Expanded genomic discoveries reveal the genetics underlying Glossina biology and provide a rich body of knowledge for basic science and disease control,” the scientists concluded. “They also provide insight into the evolutionary biology underlying novel adaptations and are relevant to applied aspects of vector control such as trap design and discovery of novel pest and disease control strategies.”
Attardo, who joined the UC Davis faculty in 2017 after serving 13 years with the Yale School of Public Health, said the massive research project involved “the complete sequencing and assembly of six Glossina species, including the two primary vectors of human African tryapnosomiasis, three major vectors of animal trypanosomiasis and one ancestral tsetse species which demonstrates some resistance to the species of trypanosomes responsible for human and some animal forms of the disease.”
The research, titled "Comparative Genomic Analysis of Six Glossina Genomes, Vectors of African Trypanosomes," offers:
- A clearer definition of the Glossina phylogenetic tree and placement of a controversial species.
- Identification of rapidly evolving regions of the tsetse genome relative to Drosophila.
- Identification of Glossina specific genes and their functions as well as expansions and contractions of gene families in tsetse relative to other flies.
“We discuss the functional implications of these changes and how they relate to tsetses' physiological adaptations and evolutionary history,” Attardo noted.
“We discovered that the rhodopsin gene family which is associated with vision/color detection shows conservation in motion detection and tracking associated genes.” Attardo said. “However, the gene coding for the protein that detects blue wavelengths is divergent relative to houseflies and shows the highest variance between Glossina species of all the rhodopsin genes. This is significant as the color blue is used as an attractant to bring tsetse into the traps used for control. It suggests that different species may be tuned/attracted to different wavelengths of blue.”
They also analyzed the genes associated with tsetse immunity and the relative differences in comparison with houseflies and fruit flies. “We see many immune genes missing in Glossina and increased copy numbers of genes associated with negative regulation of immune function. We think this may be associated with the evolution of obligate symbiosis as a way to protect their symbionts.”
“We also found extreme conservation of milk proteins between all sequenced species,” the UC Davis medical entomologist said. “On the flip side, male reproductive proteins (seminal proteins) appear to be very rapidly evolving relative to the rest of the genome. The copy numbers of these genes also change significantly between species.”
The scientists also found an overall reduction of olfactory associated genes and protein modifications specific to salivary proteins in the two species that vector human trypanosomiasis.
In 1995, the World Health Organization (WHO) estimated that 60 million people were at risk of sleeping sickness, with an estimated 300,000 new cases per year in Africa, and fewer than 30,000 cases diagnosed and treated. Due to increased control, only 3796 cases were reported in 2014, with less than 15,000 estimated cases, according to WHO statistics.
The parasitic disease “mostly affects poor populations living in remote rural areas of Africa,” according to WHO. “Untreated, it is usually fatal. Travelers also risk becoming infected if they venture through regions where the insect is common. Generally, the disease is not found in urban areas, although cases have been reported in suburban areas of big cities in some disease endemic countries.”
Several National Institutes of Health (NIH) grants, awarded to Attardo and Aksoy, funded the research. They also drew funding from the McDonnell Genome Institute at Washington University School of Medicine; the National Research Foundation, the Swiss National Science Foundation, and the Slovak Research and Development Agency.
Related Links:

- Author: Marissa Palin
Who will be moving to the new building? The answer is almost everyone! SAREP and the Ag Issues Center are not moving to the new building. The following units will move:
- REC administrative office
- Facilities
- EH&S
- Program Support Unit
- IPM
- CSIT
- Business Operations Center—Davis unit
- Contracts & Grants
- Staff Personnel Unit
- Academic Personnel Unit
- Youth, Families and Communities
- 4-H Youth Development
- EFNEP
- 4-H Foundation
- Master Gardeners
Everyone will have a new phone numbers once the move is made, so keep an eye out for contact changes come November!

