- Author: Kathy Keatley Garvey

The research, “Root-Knot Nematodes Produce Functional Mimics of Tyrosine-Sulfated Plant Peptides,” is published in the current edition of the Proceedings of the National Academy of Sciences (PNAS).
It's like hijacking plant development to facilitate parasitism, according to nematologist Shahid Siddique, an associate professor in the Davis Department of Entomology and Nematology and one of the corresponding authors of this study. “This finding showcases an amazing case of convergent evolution across three different types of organisms, revealing how diverse life forms can develop similar strategies for survival.”
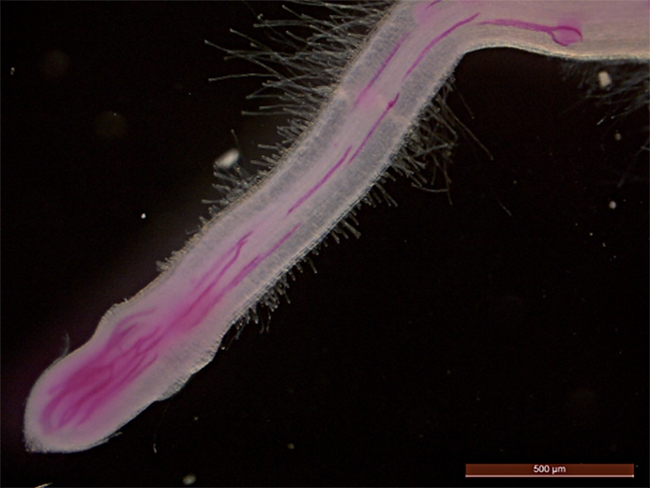
The root-knot nematode, which threatens global food security, is a small worm-like organism that is a highly evolved obligate parasite, or an organism that cannot survive without its host. It is known to infest some 2000 crops worldwide. “These parasites have a remarkable ability to establish elaborate feeding sites in roots, which are their only source of nutrients throughout their life cycle,” the authors wrote.
“Root-knot nematodes are a major threat to various crops, including fruit trees and vegetables,” Siddique said. “In California, tomatoes, almonds, and walnuts are among the major hosts susceptible to root-knot nematode infection.”
Siddique and UC Davis distinguished professor Pamela Ronald, a plant pathologist and geneticist in the Department of Plant Pathology and the Genome Center, are the joint corresponding authors. Joint first-authors are Henok Zemene Yemer, formerly of the Siddique lab and now with Gingko Bioworks, Emeryville, and Dee Dee Lu of the Ronald lab.

The team also included emerita professor Valerie Williamson of the former Department of Nematology; Maria Florencia Ercoli, postdoctoral fellow in the Ronald lab; Alison Coomer Blundell, a doctoral candidate in the Siddique lab; and Paulo Vieira of the USDA's Mycology and Nematology Genetic Diversity and Biology Laboratory, Beltsville, Md.
“Plant peptides containing sulfated tyrosine (PSY)-family peptides are peptide hormones that promote root growth via cell expansion and proliferation,” the authors explained. “A PSY-like peptide produced by a bacterial pathogen has been shown to contribute to bacterial virulence. Here, we discovered that PSY-like peptides are encoded by a group of plant-parasitic nematodes known as root-knot nematodes. These nematode-encoded PSY mimics facilitate the establishment of parasitism in the host plant. Our findings are an example of a functional plant peptide mimic encoded by a phytopathogenic bacterium (prokaryote) and a plant-parasitic nematode (an animal).”
The research involved gene expression analysis and parasitism of tomato and rice plants.
The project drew financial support from a collaborative grant awarded to Siddique and Ronald from the National Science Foundation's Division of Integrative Organismal Systems.
Siddique, a member of the UC Davis faculty since 2019, focuses his research on basic as well as applied aspects of interaction between parasitic nematodes and their host plants. “The long-term object of our research is not only to enhance our understanding of molecular aspects of plant–nematode interaction but also to use this knowledge to provide new resources for reducing the impact of nematodes on crop plants in California.”
Ronald, noted for her innovative work in crop genetics, especially rice, is recognized for her research in infectious disease biology and environmental stress tolerance. Thomson Reuters named her one of the world's most influential scientific minds and Scientfic American recognized her as among the world's 100 most influential people in biotechnology. In 2022 Ronald received the Wolf Prize in Agriculture.
The next steps? “Currently, we are working to understand the mechanism by which these peptides contribute to the nematode infection,” Siddique said. “This entails the characterization of receptors involved and gaining insights into transcriptional changes.”
- Author: Kathy Keatley Garvey

A team of nine researchers, including UC Davis biologist Scott Carroll, analyzed data over a six-year period and concluded that crop rotation works well in battling the notorious pest that annually causes $800 million in yield loss and $200 million in treatment costs.
“Answering this question was important not only to grower success but the agricultural economy, said Carroll, an associate of the UC Davis Department of Entomology and Nematology and owner of the Davis-based Institute for Contemporary Evolution. “Bt crops are far-and-away the single most important factor reducing soil and crop insecticide applications in the United States at present.”
When Bacillus thuringiensis (Bt) corn was introduced in 2003, the pest seemed under control. The genetically engineered corn is a transgenic, insecticidal crop that kills rootworm larvae but is harmless to humans.
However, when the pest began developing resistance to the Bt corn toxins, the U.S. Department of Agriculture recommended crop rotation as a method of control. Crop rotation, an age-old agricultural tactic, is a consistent and economical means of controlling rootworms the season following an outbreak. It reduces rootworm densities, and is considered more effective than insecticides.

“Corn rootworm is one of the nation's most devastating pests, giving a sense of urgency to protecting the efficacy of industrial pest control approaches with reduced non-target effects,” said Carroll, who studies basic and applied aspects of evolutionary biology. “Transgenic insecticidal Bt crops in the United States are cultivated under a very interesting socio-evolutionary model of resistance management that is mandated by the U.S. Environmental Protection Agency. Individual growers must implement resistance management--usually by devoting a small acreage to planting a 'refuge' of non-Bt crops in order to nurture a local reservoir population of Bt-susceptible pest insects.”
Carroll pointed out that the “outstanding productivity of Bt corn has led a portion of growers to reduce or eliminate their required refuge planting. Moreover, many time-tested practices for integrated pest management have fallen by the wayside as growers have found they could rely solely on the genetics of the seemingly invulnerable Bt varieties.”
“As predicted, Bt resistance evolution in corn rootworm has accelerated. In response to this dire risk, in 2016 EPA began mandating crop rotation as a complementary means of reducing the damage to Bt corn fields caused by resistant corn rootworms. Our working group analyzed the success of this traditional agricultural tactic to help sustain the efficacy of the high-tech Bt tactic.”
Carroll said that under the leadership of his colleague Yves Carrière at the University of Arizona, “our team analyzed six years of field data from 25 crop reporting districts in Illinois, Iowa and Minnesota—three states facing some of the most severe rootworm damage to Bt cornfields.
“The answer we found is that traditional crop rotation is working to protect the Bt corn fields from rootworm damage, including in areas that have seen the evolution of behavioral resistance to crop-rotation by rootworms.”
The bottom line, said Carrière, is this: "Farmers have to diversify their Bt crops and rotate. Diversify the landscape and the use of pest control methods. No one technology is the silver bullet.”
The project also included scientists from North Carolina State and McGill University, along with Carroll's colleague, Peter Jørgensen of the Stockholm Resistance Center.
While Jorgensen was pursuing his master's degree program at the University of Copenhagen and studying at UC Davis, he worked with Carroll and Sharon Strauss of the Department of Evolution and Ecology.
“This PNAS paper,” Carroll said, “is one of several that have developed from a pursuit Peter and I organized on 'Living with Resistance' at the National Socio-Environmental Synthesis Center in Annapolis, with the aim to explore more sustainable approaches to managing evolutionary challenges to health and food security.”
The abstract:
"Transgenic crops that produce insecticidal proteins from Bacillus thuringiensis (Bt) can suppress pests and reduce insecticide sprays, but their efficacy is reduced when pests evolve resistance. Although farmers plant refuges of non-Bt host plants to delay pest resistance, this tactic has not been sufficient against the western corn rootworm, Diabrotica virgifera virgifera. In the United States, some populations of this devastating pest have rapidly evolved practical resistance to Cry3 toxins and Cry34/35Ab, the only Bt toxins in commercially available corn that kill rootworms. Here, we analyzed data from 2011 to 2016 on Bt corn fields producing Cry3Bb alone that were severely damaged by this pest in 25 crop reporting districts of Illinois, Iowa, and Minnesota. The annual mean frequency of these problem fields was 29 fields (range 7 to 70) per million acres of Cry3Bb corn in 2011 to 2013, with a cost of $163 to $227 per damaged acre. The frequency of problem fields declined by 92% in 2014 to 2016 relative to 2011 to 2013 and was negatively associated with rotation of corn with soybean. The effectiveness of corn rotation for mitigating Bt resistance problems did not differ significantly between crop-reporting districts with versus without prevalent rotation-resistant rootworm populations. In some analyses, the frequency of problem fields was positively associated with planting of Cry3 corn and negatively associated with planting of Bt corn producing both a Cry3 toxin and Cry34/35Ab. The results highlight the central role of crop rotation for mitigating impacts of D. v. virgifera resistance to Bt corn."



- Author: Kathy Keatley Garvey

ASD, which impacts the nervous system, affects 1 percent of the U.S. population or 62.2 million globally. An estimated 64 percent and 91 percent of the population are at risk genetically.
The researchers' latest paper, “Maternal Glyphosate Exposure Causes Autism-Like Behaviors in Offspring through Increased Expression of Soluble Epoxide Hydrolase,” appears in the current edition of the Proceedings of the National Academy of Sciences (PNAS). Glyphosate is a broad-spectrum systemic herbicide and crop desiccant used to kill broadleaf weeds and grasses that compete with agricultural crops.
“In the research of autism, brain-gut-microbiota axis plays a key role in ASD from human studies,” said lead researcher and neurobiologist Kenji Hashimoto of the Division of Clinical Neuroscience, Chiba University Center for Forensic Mental Health, Chiba, Japan. “Accumulating evidence suggests abnormal composition of gut microbiota in subjects with autism. In this study, we found abnormal composition of gut microbiota in offspring after maternal glyphosate exposure. Thus, exposure of glyphosate during pregnancy may cause abnormal composition of gut microbiota in offspring, resulting in the risk for autism.'
The drug discovered in the Hammock lab inhibits sEH, a natural enzyme that regulates epoxy fatty acids, “which control blood pressure, fibrosis, immunity, tissue growth, depression, pain and inflammation to name a few processes,” said co-author Hammock, a distinguished professor with a joint appointment in the UC Davis Department of Entomology and Nematology and the UC Davis Comprehensive Cancer Center. Human clinical trials underway to see if the non-addictive drug relieves chronic pain.
“It is critical to appreciate that mouse models are not absolutely predictive of the human situation,” Hammock said.“Similarly, maternal immune stress is simply a model that gives behavioral changes in the offspring. That said, it is a relevant model of abnormalities in mental development in the offspring. This is a widely used model of the effect of maternal stress on the next generation that has been established in many species, including nonhuman primates. Sadly, maternal stress was shown tightly associated with the mental state of human children as well.”

“Such studies are important to generate hypotheses of environmental risk,” said Hammock, who meshes his expertise in chemistry, toxicology, biochemistry and entomology, in his 50-year research to find a non-addictive drug to control chronic pain. “Because we only saw maternal immune stress at exceptionally high doses of glyphosate, our data fail to support the hypothesis that glyphosate exposure causes autism with expected dietary, environmental or even occupational exposure.”
The research drew financial support from the Japan Society for the Promotion of Science (to Hashimoto); and the National Institute of Environmental Health Sciences (NIEHS) River Award (to Hammock), and NIEHS Superfund Program (to Hammock). Hammock has directed the UC Davis Superfund Program for nearly four decades.
The Hashimoto-directed Chiba group has shown that these drug candidates prevent and even reverse a variety of chronic diseases of the central nervous system in mice and human cells including ASD like behaviors.
The 16 co-authors include Hammock lab researchers Jun Yang, Sung Hee Hwang and Debin Wan.
Yang said that the researchers “hypothesized that the role of the sEH is important in the pathogenesis of ASD in offspring after maternal glyphosate exposure based on our previous finding that sEH plays a key role in the development of ASD-like behavioral abnormalities in juvenile offspring after maternal immune activation (MIA), a prenatal environmental factor.”
Said Hwang: “Some epidemiological studies suggest an association between glyphosate use in agriculture and increases in autism like disorders. The doses we used in mice were so high that we fail to support glyphosate epidemiological associations between the herbicide use and the cause ASD-like behaviors.
The PNAS abstract:
“Epidemiological studies suggest that exposure to herbicides during pregnancy might increase risk for autism spectrum disorder (ASD) in offspring. However, the mechanisms underlying the risk of ASD by herbicides such as glyphosate remain unclear. Soluble epoxide hydrolase (sEH) in the metabolism of polyunsaturated fatty acids is shown to play a key role in the development of ASD in offspring after maternal immune activation. Here, we found ASD-like behavioral abnormalities in juvenile offspring after maternal exposure to high levels of formulated glyphosate.
“Furthermore, we found increases in sEH in the prefrontal cortex (PFC), hippocampus, and striatum of juvenile offspring and oxylipin analysis showed decreased levels of epoxy-fatty acids such as 8(9)-EpETrE in the blood, PFC, hippocampus, and striatum of offspring after maternal glyphosate exposure, supporting increased activity of sEH in the offspring. Moreover, we report abnormal composition of gut microbiota and short chain fatty acids in fecal samples of juvenile offspring after maternal glyphosate exposure. Interestingly, oral administration of TPPU (an sEH inhibitor) to pregnant mothers from E5 to P21 prevented ASD-like behaviors such as social interaction deficits and increased grooming time in the juvenile offspring. These findings suggest that maternal exposure to high levels of glyphosate causes ASD-like behavioral abnormalities and abnormal composition of gut microbiota in juvenile offspring, and that increased activity of sEH might play a role in ASD-like behaviors in offspring after maternal glyphosate exposure. Therefore, sEH may represent a target for ASD in offspring after maternal stress from occupational exposure to contaminants.
“Maternal exposure to high levels of the herbicide glyphosate may increase the risk for autism spectrum disorder (ASD) in offspring; however, the underlying mechanisms remain largely unknown. Maternal glyphosate exposure during pregnancy and lactation caused ASD-like behavioral abnormalities and abnormal composition of gut microbiota in murine male offspring. Soluble epoxide hydrolase (sEH) in the brain of offspring after maternal glyphosate exposure was higher than controls. Treatment with an sEH inhibitor from pregnancy to weaning prevented the onset of ASD-like behavioral abnormalities in offspring after maternal glyphosate exposure. The glyphosate exposures used here exceed any reasonable dietary, environmental or occupational exposure, but they indicate that increased sEH plays a role in ASD-like behaviors in offspring.”
- Author: Kathy Keatley Garvey

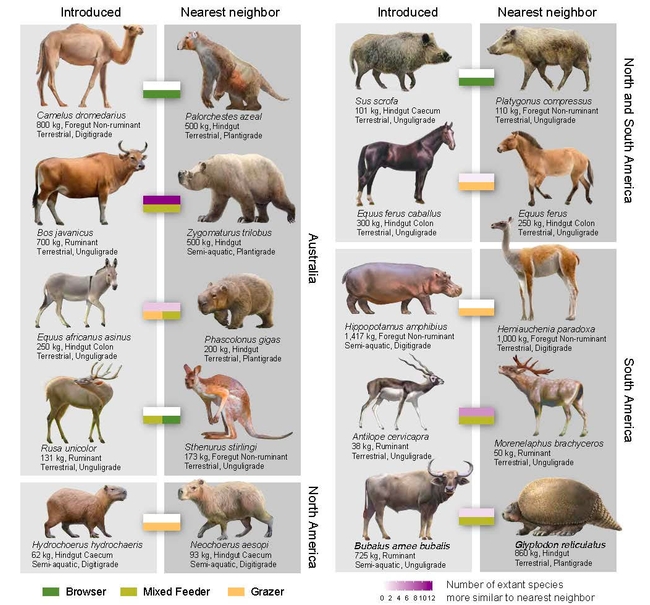
The research paper, “Introduced Herbivores Restore Late Pleistocene Ecological Functions” is the work of an 11-member international team led by Australian ecologist Erick Lundgren of the University of Technology, Sydney.
The authors pored over scientific literature; created a list of living and extinct herbivores over the last 126,000 years; and categorized them by their body size, anatomy, habitat, diet, and how their bodies digested the vegetation. Then they compared their lifestyles in overlapping regions.
Carroll, affiliated with the UC Davis Department of Entomology and Nematology, said one of the studies dealt with the abandoned hippos of Colombian drug lord Pablo Escobar (1949-1993), who purchased a male and three females in the 1980s from a California zoo and kept them in fields along the Magdalena River, northwestern Colombia. Without humans and other predators decimating them, the population today is 80 and is expected to reach 800 to 5000 by 2050.
The out-of-place hippos may be filling the exotic roles of extinct massive animals, such as giant llamas and rhinoceros-sized relatives, the ecologists said.
Said Carroll: “That paleontological analysis found that, amazingly, introduced herbivores– including Pablo Escobar's escaped Colombian hippos– often match the functional traits of extinct natives better than do those missing species' closest living native relatives. In this way, the ‘out-of-place' make the world more similar to the pre-extinction past. The ‘shoot-first- and-ask-questions later' approach as a maxim is as reckless as it sounds, and it's not going to sustain our life-saving drugs, nor the species we revere or ecosystems we rely on, into the future.”
“Many introduced herbivores restore trait combinations that have the capacity to influence ecosystem processes, such as wildfire and shrub expansion in drylands,” the team wrote.
As for feral hogs in North America, Carroll said their rooting increases tree growth and attracts bird flocks, like the ecological work of their extinct ancestors. Likewise, the feral horses and burros, known for their well-digging behavior, are replacing the original American horses, which went extinct 12,000 years ago.
In their abstract, the authors pointed out that humans “have caused extinctions of large-bodied mammalian herbivores over the past 100,000 years, leading to cascading changes in ecosystems. Conversely, introductions of herbivores have, in part, numerically compensated for extinction losses. However, the net outcome of the twin anthropogenic forces of extinction and introduction on herbivore assemblages has remained unknown. We found that a primary outcome of introductions has been the reintroduction of key ecological functions, making herbivore assemblages with nonnative species more similar to preextinction ones than native-only assemblages are. Our findings support calls for renewed research on introduced herbivore ecologies in light of paleoecological change and suggest that shifting focus from eradication to landscape and predator protection may have broader biodiversity benefits.”
Carroll, who also co-led an author group of the newly published “Coevolutionary Governance of Antibiotic and Pesticide Resistance” in the journal Trends in Ecology, said that the publications together “address both sides of the human-environment co-existence issue.”
“Reading the titles, you might not expect these two studies are two sides of the same coin,” Carroll said, “but for me they address both sides of the human-environment issue that most compels me: How can we create more workable, productive and respectful long-term relationships with other species? To help think about this as an evolutionary biologist, I divide the key challenges of human interactions with Nature into those that arise from competitor and parasite species that adapt too quickly for us to control, and those that arise in in our efforts to protect more valued species– like endangered large mammals– that adapt too slowly to survive human impacts.”
“Pesticide and drug resistance are nature's predictable resilience to our reliance on an escalating war of toxic eradication,” Carroll commented. “A broader understanding shows how we can develop our own behavior to instead cultivate susceptibility to control in species we fight, using both new and known practices for improved sanitation, locally diversified agriculture, and eating lower on the food chain to inflect their evolution in a positive direction. Similarly, after millennia of driving much of the Earth's giant mammal community to extinction, we need to step back from our reflex to extinguish the errant survivors to preserve a modern sense of what's natural, without stopping to consider how these new neighbors (commonly fading from their native lands) may restore ancient ecological functions our own ancestors extinguished not so long ago.”
Carroll emphasized that “neither of these studies dismisses the serious problems irruptive populations can cause for meeting our food, health and environmental needs, nor seeks to oversimplify complex challenges. But it's actually important to work against being limited by prejudicial generalizations that lead us to sort other species into ‘good' versus ‘bad' bins. This is a sensibility that ecologists in particular should strive to cultivate. To continue to feed and shelter ourselves and remain healthy while sharing the Earth with other species, we need to develop methods that respect the tremendous information and know-how inherent in each species. I want us to do a much better job of working with that intrinsic functional diversity and adaptive potential as our best resource for advancing resilient and biodiverse ecological systems into the future.”
Carroll and his wife, UC Davis ecologist Jenella Loye, own Carroll-Loye Biological Research, Davis. They engage in public health and environmental entomology and natural product development.
(Editor's Note: The lead author of Coevolutionary Governance of Antibiotic and Pesticide Resistance is Peter Søgaard Jørgensen, who during his University of Copenhagen graduate work, spent a year at Davis studying soapberry bug host adaptation in California with Scott Carroll. The duo led the multi-year international "Living with Resistance" pursuit at the National Science Foundation's National Socio-Environmental Synthesis Center. Carroll served as the senior author.)
- Author: Kathy Keatley Garvey

DAVIS--Newly published research in the Proceedings of the National Academy of Sciences (PNAS) indicates that a drug discovered and developed in the laboratory of Bruce Hammock,UC Davis Department of Entomology and Nematology, may have a major role in preventing and treating llnesses associated with obesity.
More than 43 percent of adults in the United States are obese, according to the Center for Disease Control and Prevention (CDC). Obesity increases the risk of coronary artery disease, stroke, type 2 diabetes, and certain kinds of cancer.
The drug, a soluble epoxide hydrolase (sEH) inhibitor, appears to regulate “obesity-induced intestinal barrier dysfunction and bacterial translocation,” the 12-member team of researchers from UC Davis, University of Massachusetts and University of Michigan discovered. The same non-opioid drug is being investigated in human clinical safety trials in Texas to see if it blocks chronic pain associated with diseases such as spinal cord injury, diabetes and inflammatory bowel disease.
The research, funded by multiple federal grants, is titled “Soluble Epoxide Hydrolase Is an Endogenous Regulator of Obesity-Induced Intestinal Barrier Dysfunction and Bacterial Translocation.”
“Obesity usually causes the loss of tight junctions and leaky gut,” said first author Yuxin Wang, a postdoctoral researcher who joined the Hammock lab in 2019 from the Department of Food Sciences, University of Massachusetts, Amherst. “In normal conditions, the gut mucosal barrier is like a defender to protect us from the ‘dirty things' in the lumen, such as bacteria and endotoxin. For obese individuals, the defender loses some function and leads to more ‘bad things' going into the circulation system, causing systemic or other organ disorders.”
Although intestinal dysfunction and other problems enhancing bacterial translocation underlies many human diseases, “the mechanisms remain largely unknown,” said Wang, who holds a doctorate in biochemistry and molecular biology from the Chinese Academy of Sciences. “What we found is sEH inhibition can repair the defender function (barrier function), decrease the ‘bad things' going into the blood (bacteria translocation), and reduce inflammation of fat.”
“Our research shows that sEH is a novel endogenous regulator of obesity-induced intestinal barrier dysfunction and bacterial translocation,” said corresponding author Guodong Zhang, a former researcher in the Hammock lab and now with the Food Science Department and Molecular and Cellular Biology Graduate Program at the University of Massachusetts. “To date, the underlying mechanisms for obesity-induced intestinal barrier dysfunction remain poorly understood. Therefore, our finding provides a novel conceptual approach to target barrier dysfunction and its resulting disorders with clinical/transitional importance.”

Corresponding author Hammock, a distinguished UC Davis professor who holds a joint appointment with the Department of Entomology and Nematology and the Comprehensive Cancer Center, praised Zhang's “amazing record while he was a postgraduate at UC Davis, and now in Food Science Department at the University of Massachusetts, where he recently received tenure.”
Zhang mentored two co-authors of the paper: Yuxin and Weicang Wang, both formerly of the Department of Food Science, University of Massachusetts and now with the Hammock lab.
“I feel so lucky that Yuxin and Weicang have joined my laboratory,” Hammock said. “The drugs studied in this PNAS paper are now in human clinical trials and on a path to replace opioid analgesics for pain treatment. I hope the continuing work of Guodong, Weicang and Yuxin will evaluate them as treatments for a variety of inflammatory bowel diseases.”
Andreas Baumler, professor and vice chair of research in the UC Davis Department of Medical Microbiology and Immunology, who was not affiliated with the study, said: “Obesity-induced gut leakage and bacterial translocation can be ameliorated by targeting microbes with antibiotics, suggesting that the microbiota contributes to disease. However, the work by Zhang and co-workers suggest that rather than targeting the microbes themselves, obesity-induced gut leakage and bacterial translocation can be normalized by silencing a host enzyme, which identifies host metabolism as an alternative therapeutic target.”
In addition to Hammock, Zhang, Yuxin and her husband Weicang, the other eight co-authors on the team are:
- Jun Yang, Sung Hee Hwang, and Debin Wan of the Hammock lab, UC Davis Department of Entomology and Nematology, and UC Davis Comprehensive Cancer Center
- Kin Sing Stephen Lee, formerly of the Hammock lab, and Maris Cinelli, both of the Department of Pharmacology and Toxicology, Michigan State University, Lansing
- Katherine Sanidad and Hang Xiao, Department of Food Science and the Molecular and Cellular Biology Graduate Program, University of Massachusetts, Amherst
- Daeyoung Kim, Department of Mathematics and Statistics, University of Massachusetts, Amherst
The abstract: “Intestinal barrier dysfunction, which leads to translocation of bacteria or toxic bacterial products from the gut into bloodstream and results in systemic inflammation, is a key pathogenic factor in many human diseases. However, the molecular mechanisms leading to intestinal barrier defects are not well understood, and there are currently no available therapeutic approaches to target intestinal barrier function. Here we show that soluble epoxide hydrolase (sEH) is an endogenous regulator of obesity-induced intestinal barrier dysfunction. We find that sEH is overexpressed in the colons of obese mice. In addition, pharmacologic inhibition or genetic ablation of sEH abolishes obesity-induced gut leakage, translocation of endotoxin lipopolysaccharide or bacteria, and bacterial invasion-induced adipose inflammation. Furthermore, systematic treatment with sEH-produced lipid metabolites, dihydroxyeicosatrienoic acids, induces bacterial translocation and colonic inflammation in mice. The actions of sEH are mediated by gut bacteria-dependent mechanisms, since inhibition or genetic ablation of sEH fails to attenuate obesity-induced gut leakage and adipose inflammation in mice lacking gut bacteria. Overall, these results support that sEH is a potential therapeutic target for obesity-induced intestinal barrier dysfunction, and that sEH inhibitors, which have been evaluated in human clinical trials targeting other human disorders, could be promising agents for prevention and/or treatment.”
The research was funded by grants from the National Institute of Food and Agriculture, U.S. Department of Food and Agriculture (USDA); National Cancer Institute; USDA Hatch Grant; National Institute of Environmental Health Sciences (NIEHS) Superfund Research Program; and a National Science Foundation.
According to the CDC, many of obesity-related conditions that lead to diseases are preventable. In 2008, the estimated annual medical cost of obesity in the United States tallied $147 billion. The medical cost for obese individuals averaged $1,429 higher than those of normal weight.
Contact: Bruce Hammock, bdhammock@ucdavis.edu



