- Author: Surendra K. Dara
The diamondback moth, Plutella xylostella (Lepidoptera: Plutellidae) is an important pest of broccoli, Brussels sprouts, cabbage, cauliflower, collards, kale, and other cruciferous crops. It exclusively feeds on cultivated and weedy crucifers and has a worldwide distribution. Larvae feed on foliage and growing parts of young plants or bore into the heads or flower buds resulting in skeletonization of leaves, stunting of the plants, or failure of head formation in some hosts. In warmer areas, the diamondback moth has up to 12 generations per year. While multiple species of parasitoids and predatory arthropods provide some level of natural control, insecticidal applications are a primary means of diamondback moth management. Although several synthetic and biological pesticides are effective against the diamondback moth, resistance to Bacillus thuringiensis (Ferré et al. 1991), abamectin (Pu et al. 2009), emamectin benzoate, indoxacarb, and spinosad (Zhao et al. 2006), pyrethroids and other pesticides (Leibee and Savage 1992; Endersby et al. 2011) has been well-known from around the world. Excessive use of any kind of pesticide leads to resistance problems (Dara 2020; also see a video presentation) to an individual pesticide or multiple pesticides. Integrated pest management (IPM) strategy encourages the use of various control options both for maintaining pest control efficacy and reducing the risk of resistance development (Dara 2019). Regularly monitoring the pest populations to make treatment decisions, rotating pesticides with different modes of action, exploring the potential of biocontrol agents, and other non-chemical control approaches such as mating disruption with pheromones are some of the IPM strategies for controlling the diamondback moth. While sex pheromones effectively used to manage several lepidopteran pests and are proven to be a critical IPM tool, mating disruption is not fully explored for controlling the diamondback moth. A study was conducted in Brussels sprouts to evaluate the efficacy of a sprayable pheromone against the diamondback moth and to enhance the current IPM strategies.
Methodology
The study was conducted on a 10-acre Brussels sprouts field in Santa Maria. Cultivar Marte was planted in early July 2020 with expected harvesting in mid to late December. A typical diamondback control program includes monitoring diamondback moth populations with the help of sticky traps and lures and applying various combinations of biological and synthetic pesticides at regular intervals. This study evaluated the efficacy of adding CheckMate DBM-F to the grower standard practice of monitoring the diamondback moth populations with traps and lures and applying pesticides. Treatments included i) grower standard pesticide program (Table 1) and ii) grower standard pesticide program with two applications of 3.1 fl oz of CheckMate DBM-F on 9 August and 11 September. Treatment materials were applied by a tractor-mounted sprayer using a 100 gpa spray volume and necessary buffering agents and surfactants. Each treatment was 5 ac and divided into four quadrants representing four replications. In the middle of each quadrant, one Suterra Wing Trap was set up with a Trécé Pherocon Diamondback Moth Lure. Lures were replaced once a month in early September and early October. Sticky liners of the traps were replaced every week to count the number of moths trapped. Traps were placed on 1, 12, and 24 August, 1, 11, 18, and 27 September, and 6 October and the moth counts were taken from respective traps on 8 and 20 August, 1, 11, 18, and 27 September, 6 and 15 October. CheckMate DBM-F was applied at 3.1 fl oz/ac on 9 August and 11 September. The number of larvae and their feeding damage on a scale of 0 to 4 (where 0=no damage, 1=light damage, 2=moderate damage, 3=high damage, 4=extensive/irrecoverable) were recorded from 25 random plants within each replication on 30 August and 6 and 18 October. Data were subjected to analysis of variance using Statistix software and significant means were separated using Tukey's HSD test. The retail value of various pesticides was also obtained to compare the cost of treatments.
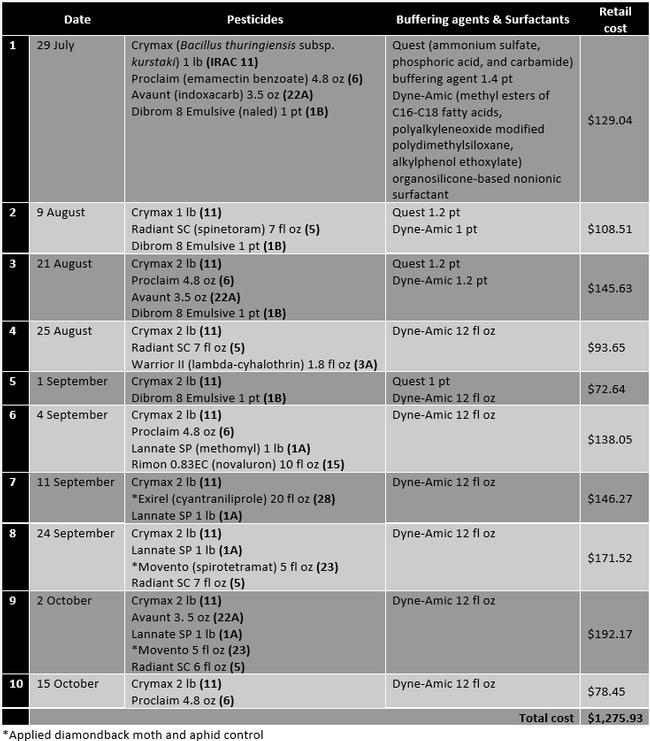
Table 1. Pesticides, buffering agents, and surfactants, their active ingredients, rates/ac (along with the IRAC mode of action groups), and retail pricing for those applied in the grower standard diamondback moth control program.
When CheckMate DBM-F [(Z)-11-Hexadecenal (3) , (Z) - 11 - Hexadecen-1-yl Acetate (1)] was applied the first time on 9 August, Dibrom 8 Emulsive was replaced with Warrior II, the buffering agent Quest was not used, and the surfactant Dyne-Amic was replaced with Induce (dimethylpolysiloxane) to avoid potential compatibility issues. The impact of this substitution is expected to be negligible within the scope of this study. The retail cost of 3.1 fl oz CheckMate DBM-F is $45.60. The cost of lures and traps would be about $4-8 per acre for a six-month crop like Brussels sprouts.
Results and Discussion
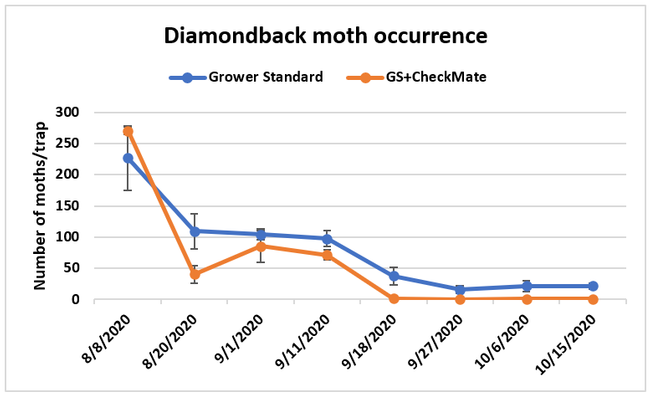
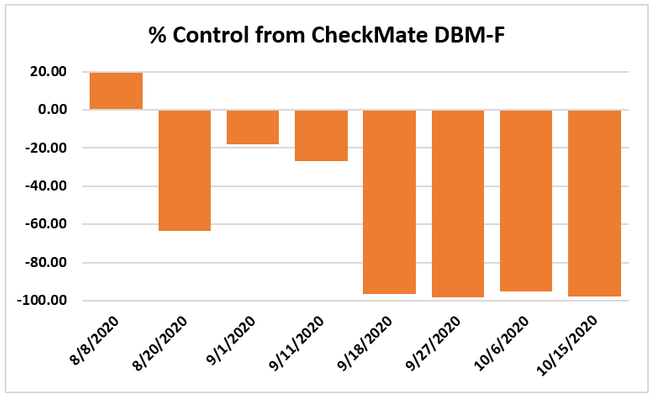
Moth populations: Traps in replication 4 in both treatments on 8 August and replication 1 in the grower standard were missing, probably knocked down by a tractor. The day before CheckMate DBM-F was first applied, the mean number of adult diamondback moths caught were 227 in the grower standard and 271 in the plots that would receive the pheromone application. There was a gradual decline in moth counts during the rest of the observation period in both treatments. However, the decline was higher in the plots that received CheckMate DBM-F. The number of moths per trap were about 19% higher in the pheromone-treated plots compared to the grower standard before the study but were nearly 98% lower by the end of the study. The reduction in moth populations from mating disruption was significant on 18 September (P =0.039) and 15 October (P = 0.006).
Larval populations: The mean number of larvae per 25 plants in a replication was zero on all observation dates except for 0.01 on 30 August in the plots that received CheckMate. Four insecticide applications by the time the study was initiated, and the remaining six applications effectively suppressed larval populations.
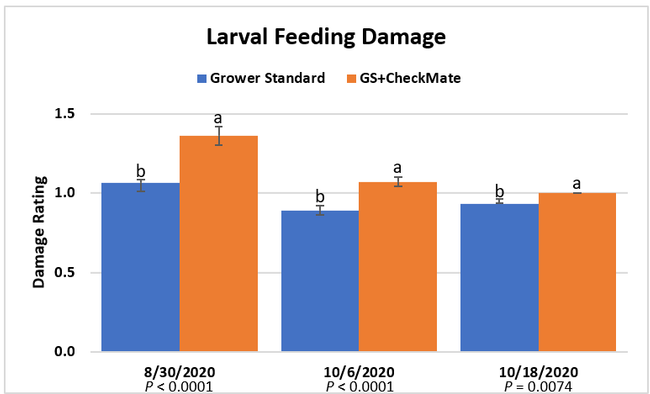
Damage ratings: Larval feeding damage ratings were consistently low (P < 0.0001) in the plants that did not receive CheckMate DBM-F. The damage was limited to the older leaves at the bottom of the plants and must have been from early feeding before the initiation of the study. The lack of larvae and the evidence of new feeding damage also confirm that the crop remained healthy and pest-free.
Since frequent pesticide applications effectively suppressed larval populations and prevented their feeding damage, the effectiveness of mating disruption in reducing yield losses could not be determined in this study. Since larval counts were not made weekly or between pesticide applications, those that were probably present between the pesticide applications could not be determined. Moths found in the traps probably developed from the larvae in the field or could have been those that flew in from other areas. However, lower moth populations in CheckMate DBM-F treatment demonstrated the overall influence of mating disruption and pest suppression.
It is common to make about 10-12 pesticide sprays during the 6-month crop cycle of Brussels sprouts. The cost of each application varied from about $73 to $192 depending on the materials used with an average cost of about $128 per application in this study. The cost of two CheckMate DBM-F applications is $91. If diamondback moth populations could be reduced with mating disruption, it is estimated that 2-3 pesticide applications could be eliminated. That results in $164 to $292 of saving for the pesticide costs and additional savings in the application costs per acre by investing $91 in the mating disruption. Since the diamondback moth can develop resistance to several chemical and natural pesticides, eliminating some applications as a result of mating disruption also contributes to resistance management along with potential negative impact of pesticides on the environment. Compared to other mating disruption strategies, a sprayable formulation compatible with other agricultural inputs is easier and cost-effective to use.
This study demonstrated that mating disruption with CheckMate DBM-F will significantly enhance the current IPM practices by reducing pest populations, contributing to insecticide resistance management, and reducing pest management costs. Additional studies, with fewer pesticide applications that allow larvae to survive and cause some damage, might further help understand the role of mating disruption where pest populations are not managed as effectively as in this field.
Watch a presentation of this study
Acknowledgments: Thanks to the PCA and the grower for their research collaboration, Tamas Zold for his technical assistance in data collection, Ingrid Schumann for market research of pesticide pricing, and Suterra for the financial support.
References
Dara, S. K. 2019. The new integrated pest management paradigm for the modern age. J. Int. Pest Manag. 10: 12.
Dara, S. K. 2020. Arthropod resistance to biopesticides. Organic Farmer 3 (4): 16-19.
Endersby, N. M., K. Viduka, S. W. Baxter, J. Saw, D. G. Heckel, and S. W. McKechnie. 2011. Widespread pyrethroid resistance in Australian diamondback moth, Plutella xylostella (L.), is related to multiple mutations in the para sodium channel gene. Bull. Entomol. Res. 101: 393.
Ferré, J., M. D., Real, J. Van Rie, S. Jansens, and M. Peferoen. 1991. Resistance to the Bacillus thuringiensis bioinsecticide in a field population of Plutella xylostella is due to a change in a midgut membrane receptor. Proc. Nat. Acad. Sci. 88: 5119-5123.
Leibee, G. L. and K. E. Savage. 1992. Evaluation of selected insecticides for control of diamondback moth and cabbage looper in cabbage in Central Florida with observations on insecticide resistance in the diamondback moth. Fla. Entomol. 75: 585-591.
Pu, X., Y. Yang, S. Wu, and Y. Wu. 2009. Characterisation of abamectin resistance in a field-evolved multiresistant population of Plutella xylostella. Pest Manag. Sci. 66: 371-378.
Zhao, J-Z., H. L. Collins, Y-X. Li, R.F.L. Mau, G. D. Thompson, M. Hertlein, J. T. Andaloro, R. Boykin, and A. M. Shelton. 2006. Monitoring of diamondback moth (Lepidoptera: Plutellidae) resistance to spinosad, indoxacarb, and emamectin benzoate. J. Econ. Entomol. 99: 176-181.
- Author: Surendra K. Dara
Several crown, fruit, and foliar diseases cause significant yield losses to strawberry. Gray mold or Botrytis fruit rot caused by Botrytis cinerea, mucor fruit rot by Mucor spp., and Rhizopus fruit rot by Rhizopus spp. are common fungal diseases in California. Botrytis cinerea is more prevalent and damaging fungus among these pathogens warranting regular fungicidal applications. Fungal spores survive in plant debris and soil and infection can occur before flower initiation. Both flowers and fruits are subjected to infection. Severely infected flowers fail to develop into fruits. Infection on developing or ripe fruit occurs as brown lesions, usually under calyxes. Infected areas rot and become dry and leathery under dry conditions or produce a thick, gray mat of spores under cool, moist conditions.
Mucor spp. invade the fruit through ruptured skin and cause leaky fruit rot. Under high humidity, profuse fungal growth of white, tough filaments with black spore-bearing structures is seen covering the fruit. In the case of Rhizopus fruit rot, discolored, water-soaked spots develop on fruit eventually leading to wilting. Similar to the Mucor fruit rot, Rhizopus rot also leads to leaky fruits and development of black spore-bearing structures on white mycelia under high humidity. Both pathogens survive in dead and decaying plant material and can persist in the field.
In a fall-planted conventional strawberry, growers usually make 12 or more fungicidal applications during a four-month period to control Botrytis and other fruit rots. Although fungicides with different modes of action are present and growers try to rotate them, fungicide resistance in B. cinerea is common and effective integrated disease strategies are necessary. Using biostimulants that might improve plant's ability to withstand diseases and alternating chemicals with biological fungicides could be some options to mitigate chemical fungicide resistance. Previous studies looked at the response of fruit diseases to various treatments that received biological soil amendments (Dara, 2020a), soil fungicides (Dara, 2020b), or chemical and biological fungicides (Dara, 2019). This study was conducted to evaluate the efficacy of some biological fungicides along with a chemical fungicide primarily against Botrytis fruit rot.
Methodology
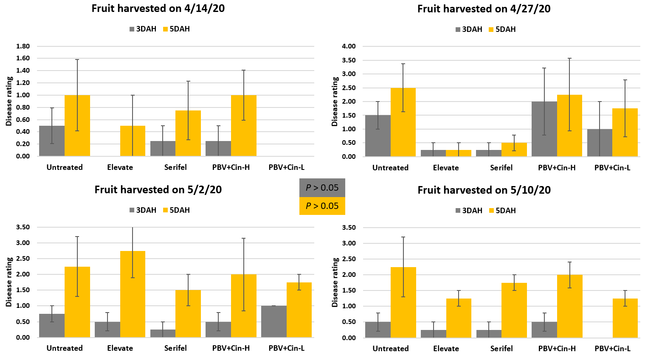
This study was conducted at a research strawberry field at the Shafter Research Station. Strawberry cultivar San Andreas was planted on 31 October 2019. Other than regular irrigation and fertigation, plants in this study were not treated with any agricultural inputs for agronomic or pest management purposes. Treatments included i) untreated control, ii) Elevate 50 WDG (fenhexamid) at 8 oz/ac, iii) Serifel (Bacillus amyloliquefaciens) at 8 oz/ac, iv) ProBlad Verde (Banda de Lupinus albus doce – BLAD, a polypeptide from sweet lupine) at 36 fl oz with Cinnerate (cinnamon oil) at 0.25% followed by ProBlad Verde at 36, 43, and 43 fl oz/ac on subsequent applications, and v) ProBlad Verde at 36 fl oz with Cinnerate at 0.25% followed by three subsequent applications of ProBlad Verde at 32 fl oz/ac. Each treatment had a 3.2' wide and 14' long plot with two rows of plants and replicated four times in a randomized complete block design. Treatments were applied using a CO2-pressurized backpack sprayer using a 45 gpa spray volume on 26 March, 2, 10, and 20 April 2020. Flowers and fruits were removed from all the plants before the first application. Fruit was harvested on 14 and 27 April and 2 and 10 May and stored in vented plastic containers for postharvest quality assessment. The severity of Botrytis and other fruit rots was recorded 3 and 5 days after harvest on a scale of 0 to 4 where 0=no disease, 1=1-25% fruit with fungal infection, 2=26-50% infection, 3=51-75%, and 4=76-100%. Compared to Botrytis fruit rot, other rots occurred as mixed infections at different times and it was not possible to accurately measure them separately. Data presented in this study primarily represent Botrytis fruit rot with other fruit rots included on some data sets. Data were subjected to analysis of variance using Statistix software to compare disease severity for individual harvest dates and their average.
Results
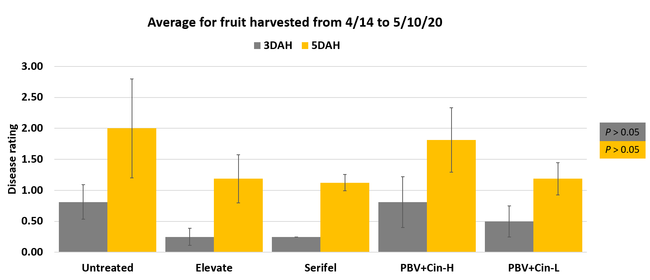
Fruit rots occurred from low to moderate levels during the observation period. Disease severity followed the usual trend with higher levels 5 days after harvest compared to 3 days after harvest. Compared to untreated control, disease severity was numerically lower in some treatments especially 3 days after harvest, but differences were not statistically significant (P > 0.05) when individual harvest dates or their average were considered. The average disease severity from four harvests was 0.25 in Elevate and Serifel, 0.50 in ProBlad Verde low rate with Cinnerate, and 0.81 in ProBlad Verde high rate with Cinnerate treatment and untreated control 3 days after harvest. The average disease severity was 1.13 for Serifel, 1.19 for Elevate and the low rate of ProBlad Verde with Cinnerate, 1.81 for the high rate of ProBlad Verde with Cinnerate, and 2.0 for untreated control 5 days after harvest. Although statistically significant differences could not be found among treatments, this study indicates the potential of non-chemical alternatives and warrants additional studies for further investigation.
Acknowledgements: Thanks to BASF and Sym-Agro for funding this study and Marjan Heidarian Dehkordi and Zach Woolpert for the technical assistance.
References
Dara, S. K. 2019. Five shades of gray mold control in strawberry: evaluating chemical, organic oil, botanical, bacterial, and fungal active ingredients. UCANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=30729
Dara, S. K. 2020a. Improving strawberry yields with biostimulants and nutrient supplements: a 2019-2020 study. UCANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=43631
Dara, S. K. 2020b. Impact of drip application of fungicides on strawberry health and yields. UCANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=43632
- Author: Surendra K. Dara
Strawberry, a high-value specialty crop in California, suffers from several soilborne, fruit, and foliar diseases. Verticillium wilt caused by Verticillium dahliae, Fusarium wilt caused by Fusarium oxysporum f. sp. fragariae, and Macrophomina crown rot or charcoal rot caused by Macrophomina phaseolina are major soilborne diseases that cause significant losses without proper control. Chemical fumigation, crop rotation with broccoli, nutrient and irrigation management to minimize plant stress, and non-chemical soil disinfestation are usual control strategies for these diseases. Botrytis fruit rot or gray mold caused by Botrytis cineaea is a common fruit disease requiring frequent fungicidal applications. Propagules of gray mold fungus survive in the soil and infect flowers and fruits. A study was conducted to evaluate the impact of drip application of various fungicides on improving strawberry health and enhancing fruit yields.
Methodology
This study was conducted in an experimental strawberry field at the Shafter Research Station during 2019-2020. Cultivar San Andreas was planted on 28 October 2019. No pre-plant fertilizer application was made in this non-fumigated field which had Fusarium wilt, Macrophomina crown rot, and Botrytis fruit rot in the previous year's strawberry planting. Each treatment was applied to a 300' long bed with single drip tape in the center and two rows of strawberry plants. Sprinkler irrigation was provided immediately after planting along with drip irrigation, which was provided one or more times weekly as needed for the rest of the experimental period. Each bed was divided into six 30' long plots, representing replications, with an 18' buffer in between. Between 6 November 2019 and 9 May 2020, 1.88 qt of 20-10-0 (a combination of 32-0-0 urea ammonium nitrate and 10-34-0 ammonium phosphate) and 1.32 qt of potassium thiosulfate was applied 20 times at weekly intervals through fertigation. Treatments were applied either as a transplant dip or through the drip system using a Dosatron fertilizer injector (model D14MZ2). The following treatments were evaluated in this study:
i) Untreated control: Neither transplants nor the planted crop was treated with any fungicides.
ii) Abound transplant dip: Transplants were dipped in 7 fl oz of Abound (azoxystrobin) fungicide in 100 gal of water for 4 min immediately prior to planting. Transplant dip in a fungicide is practiced by several growers to protect the crop from fungal diseases.
iii) Rhyme: Applied Rhyme (flutriafol) at 7 fl oz/ac immediately after and 30, 60, and 90 days after planting through the drip system.
iv) Velum Prime with Switch: Applied Velum Prime (fluopyram) at 6.5 fl oz/ac 14 and 28 days after planting followed by Switch 62.5 WG (cyprodinil + fludioxinil) at 14 oz/ac 42 days after planting through the drip system.
v) Rhyme with Switch: Four applications of Rhyme at 7 fl oz/ac were made 14, 28, 56, and 70 days after planting with a single application of Switch 62.5 WG 42 days after planting through the drip system.
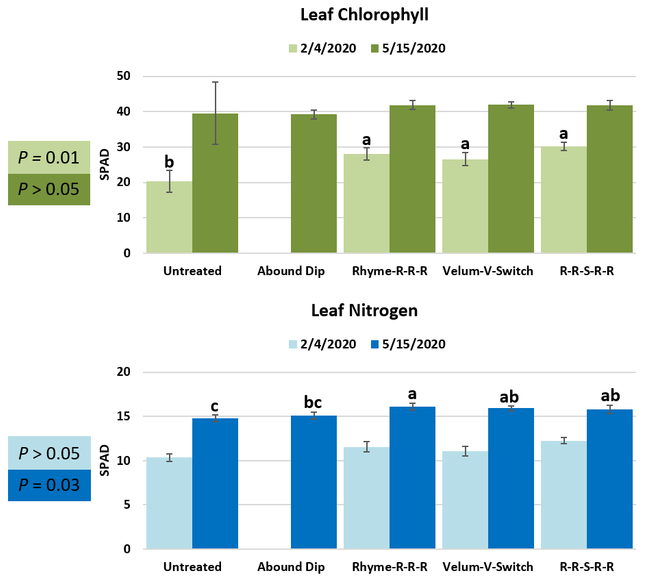
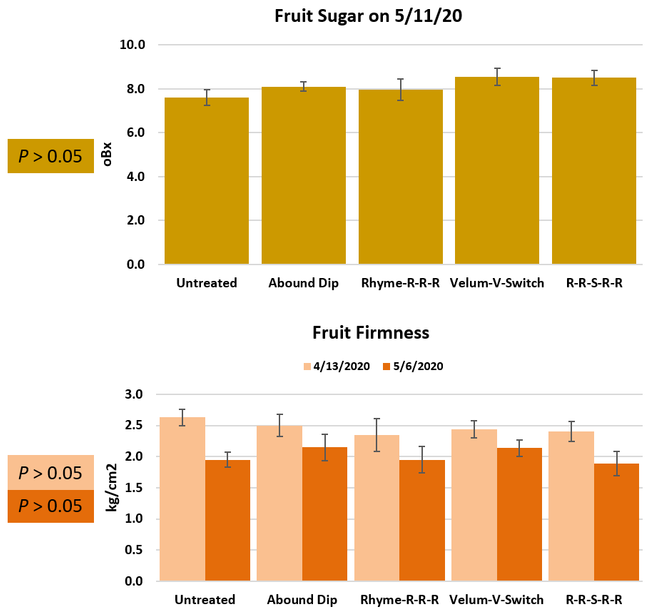
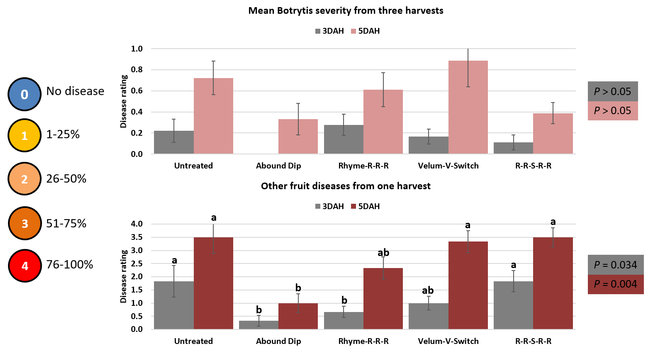
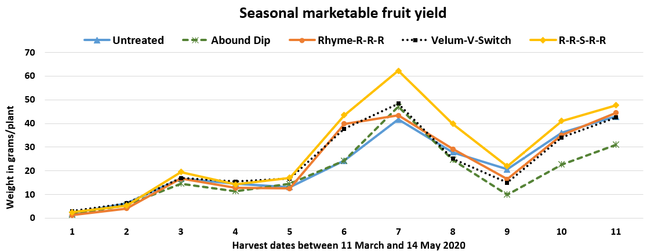
Parameters observed during the study included leaf chlorophyll and leaf nitrogen (with chlorophyll meter) in February and May; fruit sugar (with refractometer) in May; fruit firmness (with penetrometer) in April and May; severity of gray mold (caused by Botrytis cinereae) twice in March and once in May, and other fruit diseases (mucor fruit rot caused by Mucor spp. and Rhizopus fruit rot caused by Rhizopus spp.) once in May 3 and 5 days after harvest (on a scale of 0 to 4 where 0=no infection; 1=1-25%, 2=26-50%, 3=51-75% and 4=76-100% fungal growth); and fruit yield per plant from 11 weekly harvests between 11 March and 14 May 2020. Leaf chlorophyll and nitrogen data for the Abound dip treatment were not collected in February. Data were analyzed using analysis of variance in Statistix software and significant means were separated using the Least Significant Difference test.
Results and Discussion
Leaf chlorophyll content was significantly higher in plants that received drip application of fungicides compared to untreated plants in February while leaf nitrogen content was significantly higher in the same treatments during the May observation. There were no differences in fruit sugar or average fruit firmness among the treatments.
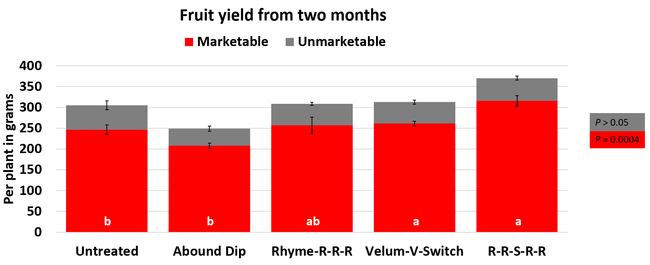
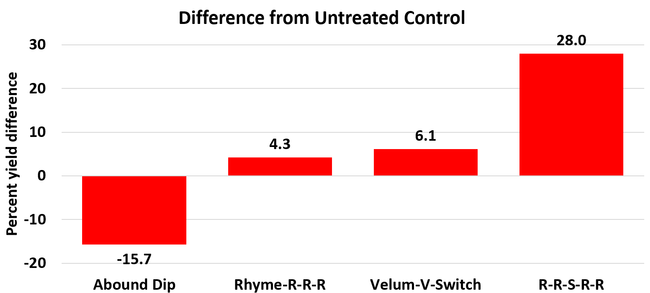
The average gray mold severity from three harvest dates was low and did not statistically differ among the treatments. However, the severity of other diseases was significantly different among various treatments with the lowest rating in Abound transplant dip on both 3 and 5 days after harvest and only 3 days after harvest in plants that received four applications of Rhyme. Unlike the previous year, visible symptoms of the soilborne diseases were not seen during the study period to evaluate the impact of the treatments. However, there were significant differences among treatments for the marketable fruit yield. The highest marketable yield was observed in the treatment that received Rhyme and Switch followed by Velum Prime and Switch and Rhyme alone. The lowest fruit yield was observed in Abound dip treatment. Unmarketable fruit (deformed or diseased) yield was similar among the treatments. Compared to the untreated control, Abound dip resulted in 16% less marketable yield and such a negative impact from transplant dip in fungicides has been seen in other studies (Dara and Peck, 2017 and 2018; Dara, 2020). Marketable fruit yield was 4-28% higher where fungicides were applied to the soil.
Although visible symptoms of soilborne diseases were absent during the study, periodic drip application of the fungicides probably suppressed the fungal inocula and associated stress and might have contributed to increased yields. The direct impact of fungicide treatments on soilborne pathogens was, however, not clear in this study due to the lack of disease symptoms. Considering the cost of chemical fumigation or soil disinfestation and the environmental impact of chemical fumigation, treating the soil with fungicides can be an economical option if they are effective. While this study presents some preliminary data, additional studies in non-fumigated fields in the presence of pathogens are necessary to consider soil fungicide treatment as a control option.
Acknowledgments: Thanks to FMC for funding this study and Marjan Heidarian Dehkordi and Tamas Zold for their technical assistance.
References
Dara, S. K. 2020. Improving strawberry yields with biostimulants and nutrient supplements: a 2019-2020 study. UCANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=43631
Dara, S. K. and D. Peck. 2017. Evaluating beneficial microbe-based products for their impact on strawberry plant growth, health, and fruit yield. UCANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=25122
Dara, S. K. and D. Peck. 2018. Evaluation of additive, soil amendment, and biostimulant products in Santa Maria strawberry. CAPCA Adviser, 21 (5): 44-50.
- Author: Surendra K. Dara
Balanced nutrient inputs are essential for optimal plant growth and yields. Depending on the soil, crop, and environmental conditions, certain nutritional supplements further enhance crop performance. While macro- and micro-nutrients are necessary for plant growth and optimal yields, biostimulants play multiple roles by increasing the bioavailability of nutrients, improving nutrient and water absorption, protecting plants from pestiferous organisms either through direct antagonism or by triggering plants defense mechanisms (Berg, 2009; Dara, 2019a). In addition to improving health and yields, biostimulants are also known to increase nutritional quality (Parađiković et al., 2011; Fierentino et al., 2018). Multiple field studies in California demonstrated the potential of biostimulants and soil amendments in improving yields in tomato (Dara, 2019b; Dara and Lewis, 2019) and strawberry (Dara and Peck, 2018; Dara, 2019a). As the knowledge of biostimulants and their potential for sustainable agriculture is expanding, there has been a steady introduction of biostimulant products in the market warranting additional studies. A study was conducted to evaluate the potential of different biostimulant materials on strawberry growth, health, and fruit yields.
Methodology
This study was conducted in an experimental strawberry field at the Shafter Research Station during 2019-2020. Cultivar San Andreas was planted on 29 October 2019. No pre-plant fertilizer application was made in this non-fumigated field which had both Fusarium oxysporum and Macrophomina phaseolina infections in previous year's strawberry planting. Each treatment was applied to a 300' long bed with single drip tape in the center and two rows of strawberry plants. Sprinkler irrigation was provided immediately after planting along with drip irrigation, which was provided one or more times weekly as needed for the rest of the experimental period. Each bed was divided into six 30' long plots, representing replications, with an 18' buffer in between. This study included both biostimulant and nutrient supplements, but this article presents data from the biostimulant treatments only. Treatments were applied either as fertigation through the drip system using a Dosatron or sprayed over the plants with a handheld garden sprayer. The following treatments were evaluated in this study:
i) Grower Standard (GS): Between 6 November 2019 and 9 May 2020, 1.88 qt of 20-10-0 (a combination of 32-0-0 urea ammonium nitrate and 10-34-0 ammonium phosphate) and 1.32 qt of potassium thiosulfate was applied 20 times at weekly intervals through fertigation. This fertilizer program was used as the standard for all treatments except for the addition of biostimulant materials.
ii) GS + Abound: Transplants were dipped in 7 fl oz of Abound (azoxystrobin) fungicide in 100 gal of water for 4 min immediately prior to planting. Transplant dip in a fungicide is practiced by several growers to protect from fungal diseases and is considered as another standard in this study.
iii) GS + Locus program: Applied Str10 (Wickerhamomyces sp.) at 5 fl oz/ac with molasses at 10 fl oz/ac immediately after planting and Rhizolizer (Trichoderma harzianum and Bacillus amyloliquefaciens) at 3 fl oz with a food source blend at 10 fl oz 2 weeks after Str10 application through the drip system. Repeated the same pattern starting from mid-February 2020. From February to May, applied 6 fl oz/ac of Rhizolizer with 20 fl oz/ac of food source once a month. Str10 is an unregistered product with yeast that is expected to help with nutrient uptake and phosphorous mobilization for improved plant vigor and yield. Rhizolizer is expected to solubilize soil nutrients and improve crop growth and yield.
iv) GS + Redox program: Starting from about one month after planting, diKaP (0-31-50 NPK) was applied as a foliar spray at 2 lb in 50 gpa every two weeks. In addition to potassium and phosphorus, diKaP also contains proprietary soluable carbon compounds that improve antioxidant production leading to increased plant respiration and tolerance to abiotic stress.
v) Bio Huma Netics (BHN) program: Transplants were dipped in 10 gal of water with 6.4 fl oz of BreakOut (4-14-2 NPK), 1.28 fl oz of Promax (thyme oil), 1.28 fl oz of Vitol (8-16-4 NPK with iron, manganese, sulfur, and zinc), and 1.28 fl oz of Zap (8-0-0 N with iron, manganese, sulfur and zinc) for 4 min immediately prior to planting. Custom blends of macro- and micro-nutrients (Ultra Precision A and B) were prepared based on soil (pre-planting) and plant tissue analyses and applied as a substitute to the grower standard fertility program. Ultra Precision A during the first 30 days after planting and Ultra Precision B for the rest of the study period were applied at weekly intervals at 1.6 gal/bed for a total of 12 times (compared to 20 fertigation events for the grower standard program). Ultra Precision blends were made with Super Phos/Phos-Max, Super Potassium, X-Tend, Nitric acid, Calcium, 44 Mag, BreakOut, Vitol, Max Pak, Iro-Max, Activol, Comol, and Surf-Max that provided N, P, and K along with boron, calcium, cobalt, copper, iron, magnesium, manganese, molybdenum, and sulfur.
vi) GS + BioWorks program 1: Applied 32 fl oz of ON-Gard (based on soy protein hydrolysate) every two weeks through the drip system from planting until canopy develops and then applied as a foliar spray in 50 gpa. ON-Gard is expected to increase the nutrient use efficiency and decrease abiotic stress to the plants.
vii) GS + BioWorks program 2: Applied 32 fl oz of ON-Gard (soy protein-based) every two weeks through the drip system from planting until canopy develops and then sprayed in 50 gpa. Also applied RootShield Plus WP (T. harzianum and T. virens) at 2 lb/ac through drip immediately after planting and 1 lb/ac at the end of November and again at the end of December 2019. RootShield is a biofungicide expected to protect strawberry from phytopathogens and improve water and nutrient uptake.
viii) GS + Fauna Soil Production (FSP) program: Applied CropSignal at 10 gpa six days prior to planting and at 5 gpa 30 after transplanting through the drip system. CropSignal is a carbon-based nutrient formula containing botanical extracts and along with cobalt, copper, manganese, and zinc and is expected to support the growth and diversity of beneficial aerobic soil microbes for improved soil structure, water retention, nutrient cycling, and plant protection.
ix) GS + Stoller program 1: Applied Stoller Root Feed Dry (9-0-5 NPK with boron, calcium, magnesium, and molybdenum) at 10 lb/ac every 10 days starting from 19 February 2020 and Stoller Grow (4-0-3 NPK with copper, magnesium, manganese, and zinc) at 8 fl oz/ac once on 27 February 2020 through the drip system. Stoller Root Feed Dry is expected to promote continuous root growth by maintaining nutritional balance while Stoller Grow is expected to increase growth efficiency and abiotic stress tolerance.
x) GS + Stoller program 2: Applied Harvest More Urea Mate (5-10-27 NPK with boron, calcium, cobalt, copper, magnesium, manganese, molybdenum, and zinc) at 10 lb/ac along with Stoller Crop Mix (algal extract with boron and calcium) at 8 fl oz/ac every 10 days starting from 19 February 2020 and Stoller Grow at 8 fl oz/ac once on 27 February 2020 through the drip system. Harvest More Urea Mate is expected to provide optimal plant growth while Stoller Crop Mix is expected to maintain the nutritional balance and improve crop vigor and yields.
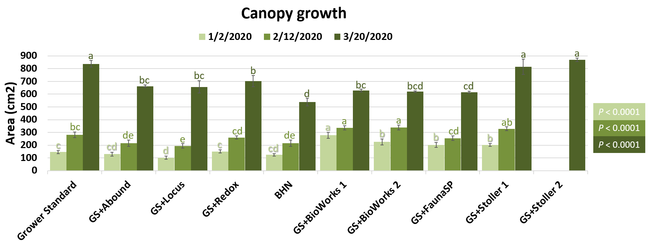
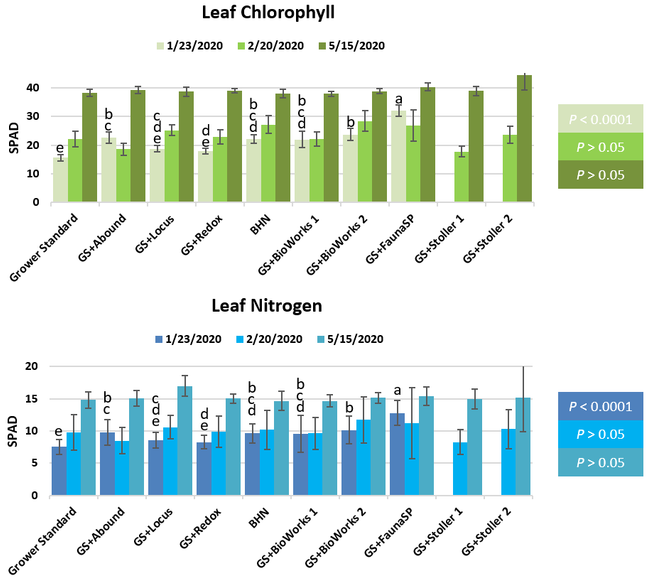
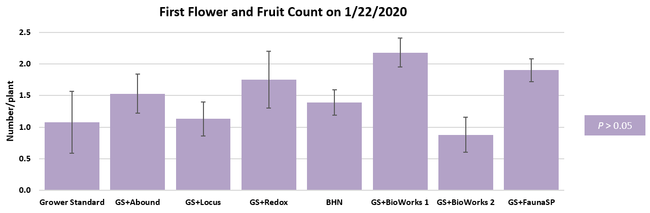
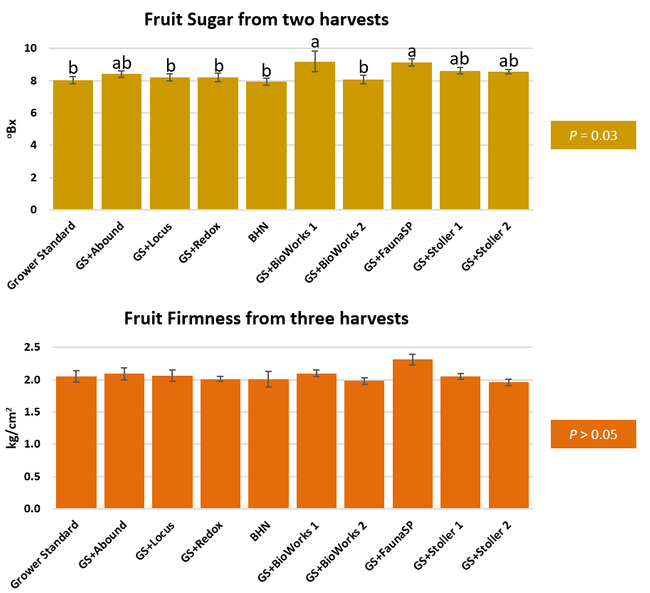
Parameters observed during the study included canopy growth (area of the canopy) in January, February, and March; first flower and fruit count in January; leaf chlorophyll and leaf nitrogen (with chlorophyll meter) in January, February, and May; fruit sugar (with refractometer) in March and May; fruit firmness (with penetrometer) in March, April, and May; severity of gray mold (caused by Botrytis cinereae) and other fruit diseases (mucor fruit rot caused by Mucor spp. and Rhizopus fruit rot caused by Rhizopus spp.) 3 and 5 days after harvest (on a scale of 0 to 4 where 0=no infection; 1=1-25%, 2=26-50%, 3=51-75% and 4=76-100% fungal growth) in March and May; sensitivity to heat stress (expressed as the number of dead and dying plants) in May; and fruit yield per plant from 11 weekly harvests between 11 March and 14 May 2020. Data were analyzed using analysis of variance in Statistix software and significant means were separated using the Least Significant Difference test.
Results and Discussion
The impact of treatments varied on various measured parameters. The interactions among plants, available nutrients, beneficial and pathogenic microorganisms in the crop environment, the influence of environmental factors, and how all these biotic and abiotic factors ultimately impact the crop health and yields are very complex. The scope of this study was only to measure the impact of biostimulants and nutrient supplements on growth, health, and yield parameters and not to investigate those complex interactions.
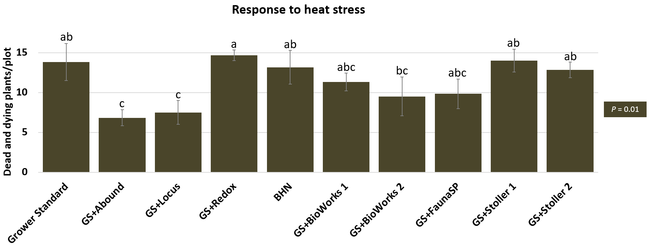
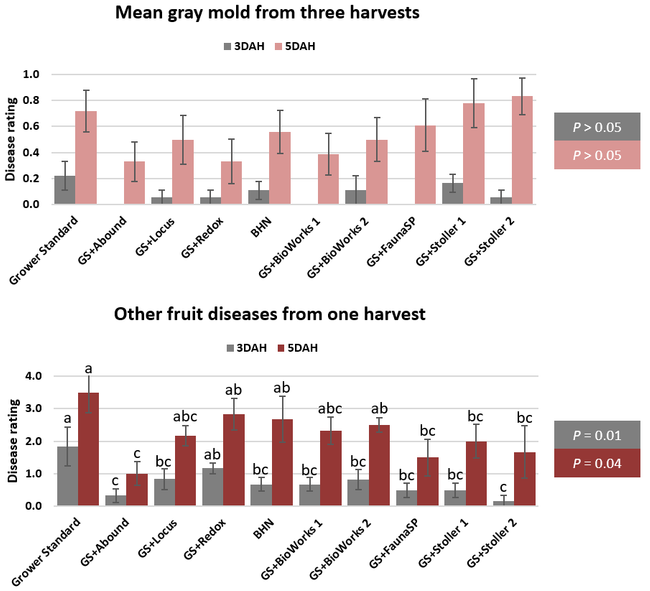
The canopy size does not always correspond with yields but could be indicative of stresses and how the plant is responding to them in the presence of treatment materials. Plants in some treatments had significantly larger canopy size in January and February, but plants in the grower standard and both Stoller programs were significantly larger than the rest by March. Leaf chlorophyll and nitrogen contents were significantly different among treatments only in January where the grower standard plants had the lowest and the plants that received CropSignal had the highest. When the counts of the first onset of flowers and developing fruits were taken in January, plants that received the BioWorks program that only received ON-Gard had the highest number followed by the CropSignal and Abound treatments. Stoller treatments were not included in the study at this time, so data for leaf chlorophyll, nitrogen, and first flower and fruit counts were not available in January. Average fruit sugar was the highest in BioWorks program with ON-Gard alone followed by FSP's Crop Signal, both Stoller programs, and the Abound treatments. There was no statistically significant difference in the average fruit firmness among the treatments. Severity of the gray mold, which occurred at low levels during the observation period, also did not statistically differ among the treatments. However, the severity of other diseases was significantly different among various treatments with the highest level in fruits from the grower standard. Temperatures were unusually high during the last week of May and several plants exhibited heat stress and started to die. The number of dead or dying plants on 28 May was the lowest in Locus and Abound treatments.
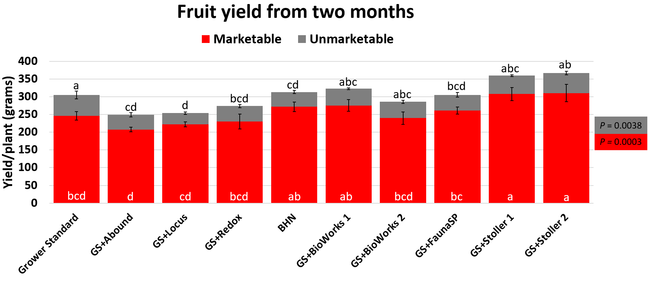
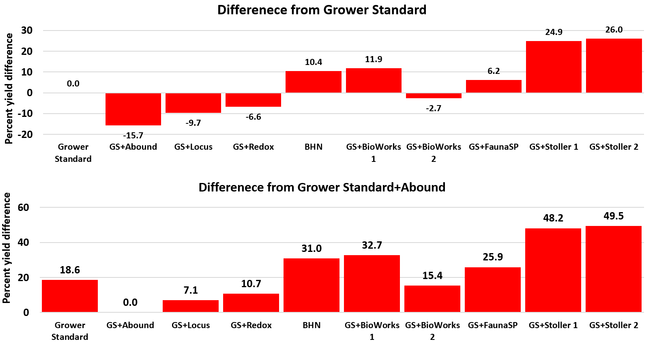
There were significant differences in marketable and unmarketable fruit yields among treatments. Highest marketable yields were seen in both Stoller treatments followed by BioWorks program with ON-Gard alone, BHN, and other treatments. Transplant dip in a fungicide seems to have a negative impact on fruit yields as observed in the current study or earlier studies (Dara and Peck, 2017 and 2018; Peck unpublished data). While the grower standard had the highest amount of unmarketable fruits, the Locus treatment had the lowest in this study. Fruit yield and some of the observed parameters appeared to be better in the grower standard compared to some treatments, which has also been seen in some earlier strawberry studies. While biostimulants can help plants under some stresses, providing sufficient macro- and micro-nutrients seems to be critical for higher fruit yields as seen with Stoller and BHN treatments. It is important to note that BHN materials were applied only 12 times compared to 20 applications of the grower standard treatment or other treatments that were applied on top of the grower standard treatment. It is also important to note that when ON-Gard was used alone, it also improved the marketable fruit yields by nearly 12% compared to the grower standard. When marketable fruit yield in the Abound treatment was considered, all treatments performed better 7-50% higher yields. Sometimes natural balance of the nutrients, organic matter, and microbial community in the soil might result in optimal yields in the absence of pathogens or other stressors. However, it is very common to use fungicidal treatments or add biological or supplemental nutrition to protect from potential threats and improving yields. These results help understand the impact of various biostimulants and supplements and warrant the need to continue such studies under various environmental, crop, and soil conditions.
Acknowledgments: Thanks to Bio Huma Netics, BioWorks, Inc., Fauna Soil Production, Locus Agricultural Solutions, Redox Ag, and Stoller for the financial support of the study and Marjan Heidarian Dehkordi and Tamas Zold for their technical assistance.
References
Berg, G. 2009. Plant-microbe interactions promoting plant growth and health: perspectives for controlled use of microorganisms in agriculture. Appl. Microbiol. Biotechnol. 84: 11-18.
Dara, S. K. 2019a. Improving strawberry yields with biostimulants: a 2018-2019 study. UCANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=31096
Dara, S. K. 2019b. Effect of microbial and botanical biostimulants with nutrients on tomato yield. CAPCA Adviser, 22(5): 40-45.
Dara, S. K. and D. Peck. 2017. Evaluating beneficial microbe-based products for their impact on strawberry plant growth, health, and fruit yield. UCANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=25122
Dara, S. K. and D. Peck. 2018. Evaluation of additive, soil amendment, and biostimulant products in Santa Maria strawberry. CAPCA Adviser, 21 (5): 44-50.
Dara, S. K. and E. Lewis. 2019. Evaluating biostimulant and nutrient inputs to improve tomato yields and crop health. Progressive Crop Consultant 4(5): 38-42.
Fiorentino, N., V. Ventorino, S. L. Woo, O. Pepe, A. De Rosa, L. Gioia, I. Romano, N. Lombardi, M. Napolitano, G. Colla, and Y. Rouphael. 2018. Trichoderma-based biostimulants modulate rhizosphere microbial populations and improve N uptake efficiency, yield, and nutritional quality of leafy vegetables. Frontiers in Plant Sci. 9: 743.
Parađiković, N., T. Vinković, I. V. Vrček, I. Žuntar, M. Bojić, and M. Medić-Šarić. 2011. Effect of natural biostimulants on yield and nutritional quality: an example of sweet yellow pepper (Capsicum annuum L.) plants. J. Sci. Food. Agric. 91: 2146-2152.
- Author: Surendra K. Dara
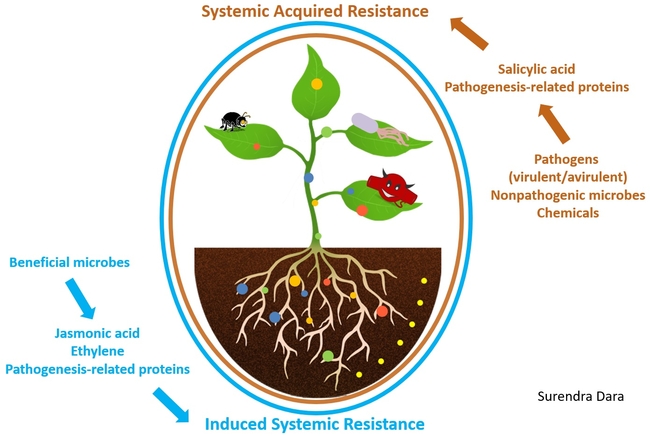
Biostimulants are beneficial microorganisms or substances that can be used in crop production to improve plants' immune responses and their ability to perform well under biotic and abiotic stresses. Biostimulants induce plant resistance to stress factors through systemic acquired resistance or induced systemic resistance. When plants are exposed to virulent and avirulent pathogens, non-pathogenic microorganisms, and some chemicals, the systemic acquired resistance mechanism is activated through the salicylic acid pathway triggering the production of pathogenesis-related proteins. On the other hand, when plants are exposed to beneficial microbes, the induced systemic resistance mechanism is activated through the jasmonic acid and ethylene pathways. The jasmonic acid pathway also leads to pathogenesis-related protein production in plants. In other words, when plants are exposed to pathogens, non-pathogens, or other compounds, various defense genes are activated through two major immune responses, helping plants fight the real infection or prepare them for potential infection. Beneficial microbes and non-microbial biostimulants are like vaccines that prepare plants for potential health problems.
Earlier studies in tomato (Dara and Lewis, 2018; Dara, 2019a) and strawberry (Dara and Peck, 2018; Dara, 2019b) demonstrated varying levels of benefits to crop health and yield improvements from a variety of botanical, microbial, or mineral biostimulants and other supplements. Some of the evaluated products resulted in significant yield improvement in both tomatoes and strawberries compared to the grower standard practices. There are several biostimulant products in the market with a variety of active ingredients, and some also have major plant nutrients such as nitrogen, phosphorus, and potassium. Depending on the crop, growing conditions, potential risk of pests and diseases, and other factors, growers can use one or more of these products. A study was conducted to evaluate the impact of various biostimulants on the yield, quality, and shelf life of strawberries.
Methodology
Strawberry cultivar San Andreas was planted late November 2018 and treatments were administered at the time of planting or soon after, depending on the protocol. Each treatment had a 290' long strawberry bed where 10' of the bed at each end was left out as a buffer. Then, six 30' long plots, each representing a replication, were marked within each bed with an 18' buffer between the plots. Since the test products needed to be applied through the drip system, an entire bed was allocated for each treatment, except for the standard program that had one bed on either side of the experimental block, and plots were marked within each bed for data collection. The following treatment regimens were used in the study:
1. Standard Program (SP): Major nutrients were provided in the form of Urea Ammonium Nitrate Solution 32-0-0, Ammonium Polyphosphate Solution, and Potassium Thiosulfate (KTS 0-0-25). Nitrogen, phosphorus, and potassium were applied before planting in November 2018 at 170, 60, and 130 lb/acre, respectively. From 15 January to 9 May 2019, a total of 26 lb of nitrogen, 13 lb of phosphorus, and 26 lb of potassium were applied through 13 periodic applications.
2. SP + Terramera Program: Formulation labeled as Experimental A (cold-pressed neem 70%) was applied at 1.2% vol/vol immediately after planting. Additional applications were made starting from 2 weeks after planting once every two weeks until the end of February (six times), followed by 13 weekly applications from the beginning of March.
3. SP + Locus Low Rate Program: This program contained Rhizolizer soil amendment (Trichoderma harzianum 1X108 CFU/ml and Bacillus amyloliquefaciens 1X109 CFU/ml) at 3 fl oz/acre, humic acid at 13.5 fl oz/acre, and kelp at 6.8 fl oz/acre. The first application was made within 15 days and at 30 days after planting followed by once in February, March, and April 2019.
4. SP + Locus High Rate Program: This program contained Rhizolizer soil amendment (Trichoderma harzianum 1X108 CFU/ml and Bacillus amyloliquefaciens 1X109 CFU/ml) at 6 fl oz/acre, humic acid at 13.5 fl oz/acre, and kelp at 6.8 fl oz/acre. The first application was made within 15 days and at 30 days after planting followed by once in February, March, and April 2019.
5. SP + BioGro Program: Transplants were treated with Premium Plant BB (Beauveria bassiana 1.1%) by spraying 2 fl oz/acre (1.29 ml in 850 ml of water). About 7 weeks after planting, 30 gpa of Plant-X Rhizo-Pro (botanical extracts), 2 gpa of CHB Premium 21 (humic acid blend), 3 gpa of CHB Premium 6 (3% humic acids), and 5 gpa of NUE Flourish 4-12-0 were applied. Starting from mid-February 2019, 15 gpa of Plant-X Rhizo-Pro, 1 gpa of CHB Premium 21, and 2 gpa of CHB Premium 6 were applied four times every 2 weeks until the end of March. Starting from 5 April 2019, 8 weekly applications of 10 gpa of Plant-X Rhizo-Pro, 1 gpa of CHB Premium 21, 2 gpa of Premium 6, and 4 gpa of NUE Flourish 4-12-0 were made until 26 May 2019.
6. SP + Actagro Program: Structure 7-21-0 at 3 gpa and Liquid Humus 0-0-4 with 22% organic acids at 1 gpa were first applied within 1 week of planting and then three more times every 2 weeks until the end of December 2018. Additional monthly applications were made from the end of January to the end of April 2019.
All the fertilizers and treatment materials were applied through the drip system using the Dosatron (Model D14MZ2) equipment. The following parameters were measured during the experimental period from January to May 2019.
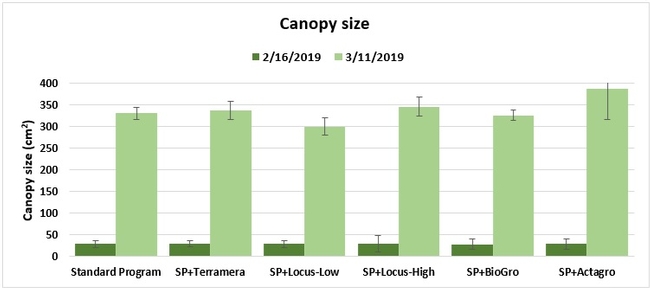
Canopy: The size of the plant canopy was determined on 21 January and again on 17 February 2019 by measuring the spread of the canopy across and along the length of the bed from 16 random plants within each plot, and calculating the area.
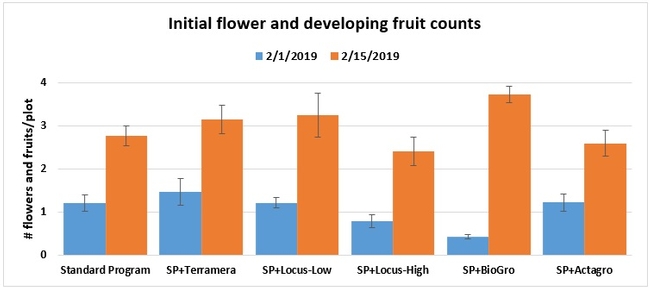
Initial flowering and fruiting: When flowering initiated, the number of flowers and developing fruits was counted from 16 random plants within each plot on 1 and 16 February 2019.
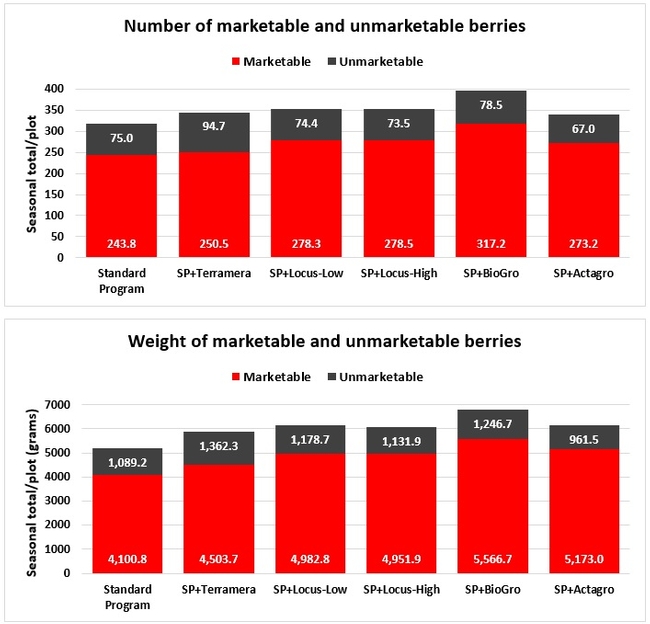
Fruit yield: Fruit was harvested weekly from every plant within each plot from 3 March to 26 May 2019 on 11 dates and the number and weight of the marketable and unmarketable fruit was determined. Due to a technical error, some of the yield data from an additional date (29 March) were lost and excluded from the analysis.
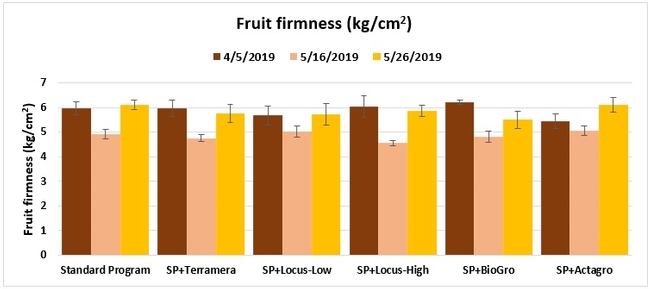
Fruit firmness: The firmness of two marketable fruit from each of five random plants per plot was measured using a penetrometer on 5 April, and 16 and 26 May 2019.
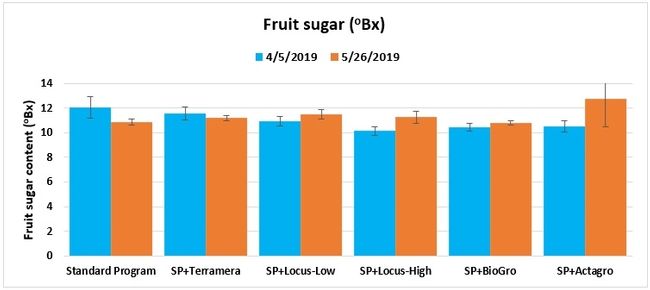
Fruit sugar content: The sugar content from one marketable fruit from each of 10 plants per plot was measured using a refractometer on 5 April and 26 May 2019.
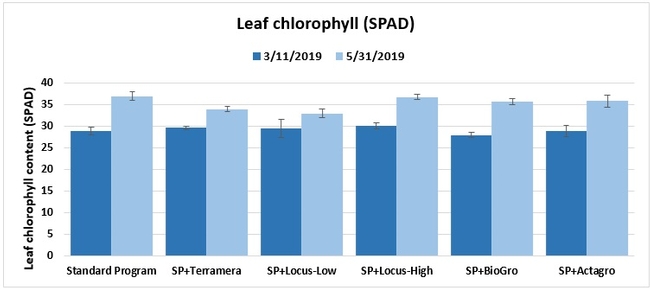
Leaf chlorophyll content: On 11 March and 31 May 2019, the chlorophyll content of one mature leaf from each of five random plants per plot was measured using a chlorophyll meter.
Postharvest disease: Marketable fruit harvested on 21 and 28 April, and 5 and 26 May 2019 was kept at the room temperature in perforated plastic containers (clamshells) and the growth of gray mold (Botrytis cinerea) or Rhizopus fruit rot fungus (Rhizopus spp.) was measured on a scale of 0 to 4 (where 0=no fungus, 1=1-25%, 2=26-50%, 3=51-75%, and 4=76-100% fungal growth) 3 and 5 days after each harvest.
Data were analyzed using analysis of variance in Statistix software and significant means were separated using the Least Significant Difference means separation test.
Results and Discussion
Statistically significant differences among treatments were seen for the seasonal total number of unmarketable berries (P = 0.0172), the initial flower and fruit numbers on 1 February (P < 0.0014), the leaf chlorophyll content on 31 May (P = 0.0144), and the disease rating 3 days after the 28 April harvest (P = 0.0065).
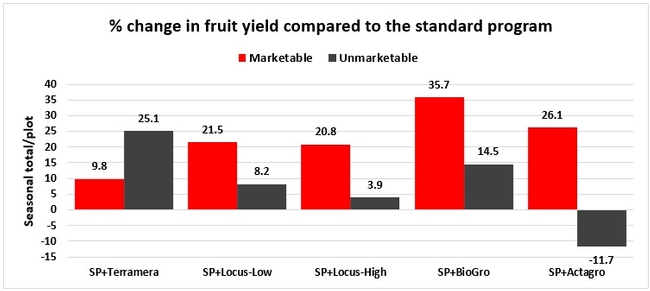
Treatments did not differ (P > 0.05) in any other measured parameters of the plant, fruit quality, or yield. However, the total seasonal fruit yield was 13 to 31% higher and the total marketable fruit yield was 10 to 36% higher in various treatment programs compared to the standard program. The seasonal total of unmarketable fruit yield was also 4 to 25% higher in treatment programs than the standard program except that there were nearly 12% fewer unmarketable berries in the Actagro program compared to the standard program.
While treatments did not statistically differ for many of the measured parameters, numerical differences in marketable fruit yield could be helpful for some understanding of the potential of these biostimulants. Additional studies with larger treatment plots would be useful for generating additional data.
Acknowledgments
Thanks to Dr. Jenita Thinakaran for the assistance at the start of the study, Hamza Khairi for his technical assistance throughout the study, the field staff at the Shafter Research Station for the crop maintenance, NorCal Nursery for the strawberry transplants, and Actagro, BioGro, Locus, and Terramera for their collaboration and financial support
References
Dara, S. K. 2019a. Improving tomato yield with nutrient materials containing microbial and botanical biostimulants. eJournal of Entomology and Biologicals, 6 June 2019 https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=30448
Dara, S. K. 2019b. Evaluating the efficacy of anti-stress supplements on strawberry yield and quality. eJournal of Entomology and Biologicals, 10 August 2019 https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=31044
Dara, S. K. and D. Peck. 2018. Microbial and bioactive soil amendments for improving strawberry crop growth, health, and fruit yields: a 2017-2018 study eJournal of Entomology and Biologicals, 3 August 2018 https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=27891
Dara, S. K. and E. Lewis. 2018. Impact of nutrient and biostimulant materials on tomato crop health and yield. eJournal of Entomology and Biologicals, 9 January 2019 https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=26054