- Author: Kathy Keatley Garvey

The predators and their prey were all in costumes, of course:
- The queen bee: UC Davis distinguished professor Lynn Kimsey, director of the Bohart Museum
- The praying mantis: Tabatha Yang, education and outreach coordinator of the Bohart Museum
- The green darner dragonfly: Christofer Brothers, a UC Davis doctoral candidate researching dragonflies
- The monarch: Barbara Heinsch, a Bohart Museum volunteer, who arrived with her entomologist-husband, Mike Pitcairn, retired senior environmental scientist, supervisor, California Department of Food and Agriculture (CDFA). He wore his CDFA lab coat and swung an insect net.
And the guy in the ghillie suit serving beverages (that would be forensic entomologist Robert "Bob" Kimsey of the UC Davis Department of Entomology and Nematology) is keenly interested in flies, but he didn't net the fly.
UC Davis entomology alumna Ivana Li, a biology lab manager at UC Davis, catered the event and arrived with her dog, Juniper, dressed as a taco. Lynn Kimsey cut a carrot cake, decorated with tiny carrots and large googly eyes.
Some attendees, including Joanna Chiu, professor and chair of the Department of Entomology and Nematology; UC Davis doctoral alumna Fran Keller, professor at Folsom Lake College; Bohart Museum associate Greg Karofelas; UC Davis doctoral alumnus Dick Meyer (who studied with the late Richard Bohart); and entomology student Kaitai Liu, arrived as themselves, sans Halloween costumes.
The Bohart Museum, located in Room 1124 of the Academic Surge Building, 455 Crocker Lane, houses a global collection of eight million insect specimens, plus a live insect petting zoo and a gift shop. Founded in 1946 by the late UC Davis professor Richard Bohart, it has been directed by Kimsey, his former doctoral student, since 1990. (See more Halloween images on the UC Davis Department of Entomology and Nematology website)
Next Open House on Monarchs. The Bohart's next open open house, set from 1 to 4 p.m., Saturday, Nov. 4, is on monarchs.
The event, free and family friendly, will be held in Room 1124 of the Academic Surge Building, Crocker Lane. This is an opportunity for attendees to ask questions about monarchs (Danaus plexippus) and native vs. non-native milkweed, among other topics.
The scientists will include:
- UC Davis distinguished professor emeritus Art Shapiroof the Department of Evolution and Ecology, who has studied butterfly populations in central California since 1972 and maintains a research website, Art's Butterfly World.
- UC Davis emeritus professor Hugh Dingle, a worldwide authority on animal migration, including monarchs. He is the author of Migration: The Biology of Life on the Move (Oxford University Press), a sequel to the first edition published in 1996. See news story on the UC Davis Entomology and Nematology website.
- UC Davis professor Louie Yang, who does research on monarchs. Due to parental duties, he may be able to attend only the last part of the open house. See news story about his work.
- UC Davis professor Elizabeth Crone of the Department of Evolution and Ecology, formerly of Tufts University, who researches monarchs. See news story about the declining monarch population on the UC Davis Entomology and Nematology website.
- UC Davis postdoctoral fellow Aramee Diethelm of the Elizabeth Crone lab. She holds a doctorate from the University of Nevada, Reno. Both her Ph.D. and postdoctoral work are on monarch butterflies. As a doctoral student, she investigated the phytochemical landscape of milkweed (Asclepias) species across northern Nevada and the effects of this variation on western monarch (Danaus plexippus) butterfly performance. See her research posted on Google Scholar, and her blog on "Drought Influences Monarch Host Plant Selection."
Shapiro points out that the monarch "is NOT a focal species in my research and I am NOT a monarch expert. On the other hand, I have a unique breeding-season census data set starting in 1999. The only other census data are for the overwintering roosts on the coast. It has become apparent that the two data sets do not always agree." Shapiro said he'd talk briefly about this at the open house.
The Bohart Museum, founded in 1946, houses a global collection of eight million insect specimens; a live insect petting zoo; and a gift shop. It is located in Room 1124 of the Academic Surge Building, 455 Crocker Lane. For more information, access the website or email bmuseum@ucdavis.edu.


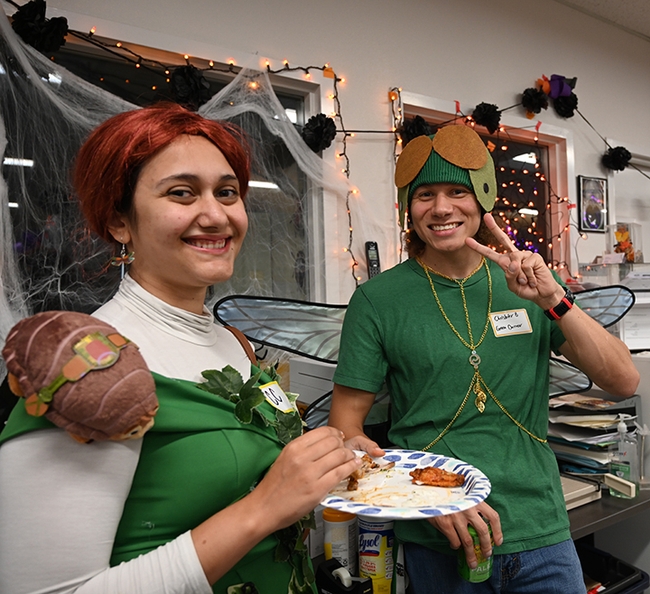



- Author: Kathy Keatley Garvey

Treat, please, says this jumping spider.
It's time to jump in to help the 13th annual UC Davis Biodiversity Museum Day crowdunding project.
It's a Super Science Day to be held Saturday, Feb. 10 on the UC Davis campus.
It's a free and family friendly event. You can visit such biological museums or collections as the Phaff Yeast Culture Collection, Bohart Museum of Entomology, Museum of Wildlife and Fish Biology, Arboretum and Public Garden, California Raptor Center, Earth and Planetary Sciences Paleontology Collections, Botanical Conservatory, Center for Plant Diversity, Nematode Collections, Marine Invertebrate Teaching Collection, and the Department of Anthropology Museum.
And you can talk to the scientists one-on-one.
The crowdfunding project ends at 11:59 p.m., Monday, Oct. 30. You can donate here. You can donate in memory of a loved one, or, for example, to honor your favorite scientist or favorite insect or spider.
"Donations will help us sustain this free science event, and enable our student interns to take science outreach to a whole new level," according to Biodiversity Museum Day chair Tabatha Yang, education and outreach coordinator for the Bohart Museum of Entomology." The goal of our event is to connect people from all walks of life to science and the biodiversity surrounding them."
Donation levels start at $5.
Don't want to buy candy corn and other treats for Halloween? Another kind of treat will help fund a Super Science Day.
- Author: Kathy Keatley Garvey

Then you'll want to attend the Bohart Museum of Entomology open house on Saturday, Nov. 4 from 1 to 4 p.m. in Room 1124 of the Academic Surge Building, 455 Crocker Lane. Scientists will be there to answer your questions.
The event is free and family friendly and a great opportunity to learn more about Danaus plexippus.
The scientists will include:
- UC Davis distinguished professor emeritus Art Shapiro of the Department of Evolution and Ecology, who has studied butterfly populations in central California since 1972 and maintains a research website, Art's Butterfly World.
- UC Davis emeritus professor Hugh Dingle, a worldwide authority on animal migration, including monarchs. He is the author of Migration: The Biology of Life on the Move (Oxford University Press), a sequel to the first edition published in 1996. See news story on the UC Davis Entomology and Nematology website.
- UC Davis professor Louie Yang, who does research on monarchs. Due to parental duties, he may be able to attend only the last part of the open house. See news story about his work.
- UC Davis professor Elizabeth Crone of the Department of Evolution and Ecology, formerly of Tufts University, who researches monarchs. See news story about the declining monarch population on the UC Davis Entomology and Nematology website
Shapiro, a Lepidopterist, points out that the monarch "is NOT a focal species in my research and I am NOT a monarch expert. On the other hand, I have a unique breeding-season census data set starting in 1999. The only other census data are for the overwintering roosts on the coast. It has become apparent that the two data sets do not always agree." Shapiro said he'd talk briefly about this at the open house.
"The iconic black and orange monarch butterfly is known for its astonishing long-distance annual migration and reliance on milkweed as its obligate larval host plant," according to a post on the California Department of Fish and Wildlife (CDFW). "Though genetically similar, there are two subpopulations of monarchs in North America, with the eastern population overwintering in Mexico and breeding in the midwestern states, and the western population overwintering in coastal California and fanning out across the west from Arizona to Idaho. Outside the U.S., there are at least 74 known populations of resident, non-migratory monarchs that have established around the world in the past 200 years, all with origins in North America (Nial et al. 2019)."
"Both North American migratory populations have declined over the past twenty years due to a suite of interrelated factors including habitat loss in breeding and overwintering sites, habitat degradation, disease, pesticide exposure, and climate change," CDFW says. "Recently the western population has experienced dramatic swings, for a low of less than 2,000 in 2020-21 to over 200,000 in 2021-22 (Xerces Society Western Monarch Count). While it is unclear which of the many factors are driving these dynamics, insect population commonly fluctuate from year to year. The overall downward trend remains concerning, particularly if the threats are not ameliorated. Though more research is needed, a stable population for western monarchs is likely closer to the historic averages in the 1980's, which are estimated to have ranged between one to four million overwintering butterflies."
"n 2014, monarchs were petitioned to be listed under the federal Endangered Species Act. In December 2020, the U.S. Fish and Wildlife Service found that listing was warranted but precluded by other listing actions on its National Priority List. The monarch is currently slated to be listed in 2024."
The monarch population is in trouble. Says the Xerces Society for Invertebrate Conservation in a post on its website: "In the 1990s, hundreds of millions of monarchs made the epic flight each fall from the northern plains of the U.S. and Canada to sites in the oyamel fir forests in central Mexico, and more than a million monarchs overwintered in forested groves on the California coast. Now, researchers and community scientists estimate that only a fraction of the population remains—a decline of approximately 70% has been seen in central Mexico and a decline of >90% has been seen in California."
Resources/Further Reading:
- Monarch Butterfly, California Department of Fish and Wildlife
- Research Permits, CDFW
- Western Monarch Mllkweed Mapper
- Integrated Monarch Monitoring Program, Monarch Joint Venture
- Western Monarch Count, Xerces Society for Invertebrate Conservation
- Monarch Butterfly Conservation, Xerces Society
- Spreading Milkweed, Not Myths, U.S. Fish and Wildlife Service
- Habitat Corridor Project, Views on milkweed
- Tropical Milkweed Doesn't Deserve the Bad Rap, Bug Squad blog, views on the ban of tropical milkweed by UC Davis emeriti professors Art Shapiro and Hugh Dingle, and Washington State University entomologist David James, who studies migratory monarchs.


- Author: Kathy Keatley Garvey

They learned that "medical entomologist is the study of arthropods (such as insects and ticks) that spread pathogens that cause human disease. It is also important to study insects and arthropods that spread diseases to other animals! This field o study is called veterinary entomology. Some diseases affect both humans and animals. This is called a zoonotic disease." (from Bohart Museum poster)
They asked questions. They observed "the vampires" through microscopes. And they left with first-hand information.
The presenters included:
-
Robert "Bob" Kimsey, forensic entomologist, Department of Entomology and Nematology, who answered questions about medical entomology.
-
Luz Maria Robles, public information officer, Sacramento-Yolo Mosquito and Vector Control District, who discussed and displayed mosquitoes and how to keep yourself safe. See https://www.fightthebite.net/
- Carla-Cristina "CC" Melo Edwards, doctoral student and mosquito researcher in the laboratory of medical entomologist-geneticist Geoffrey Attardo, associate professor of entomology, UC Davis Department of Entomology and Nematology, who fielded questions about mosquitoes. Attardo displayed enlarged images of mosquitoes, including a blood-fed Aedes aegypti, and a female and male Culex tarsalis.
- Moriah Garrison, senior entomologist and research coordinator with Carroll-Loye Biological Research (CLBR), (owned by doctoral scientists Scott Carroll and Jenella Loye, affiliated with the Department of Entomology and Nematology), displayed live ticks and mosquitoes.
- Nazzy Pakpour, UC Davis alumna, Novozymes scientist and author, displayed her newly published children's book, Please Don't Bite Me
For the occasion, UC Davis alumnus Kevin Murakoshi, gifted the Bohart Museum a trio of origami sculptures: a tick, an engorged tick and a bedbug. At an earlier open house, he presented the museum with origami sculptures of praying mantises. "They're beautiful," said UC Davis distinguished professor Lynn Kimsey, director of the Bohart Museum. "We're going to display them in our hallway."
The museum houses a global collection of eight million insect specimens, plus a live insect petting zoo (including Madasgascar hissing cockroaches and walking sticks), and a gift shop. It is located in Room 1124 of the Academic Surge Building, 455 Crocker Lane. UC Davis.
The next open house, themed "Monarchs," is set for Saturday, Nov. 4 from 1 to 4 p.m. All open houses are free and family friendly and include a family arts-and-crafts activity. For more information, contact the Bohart Museum at bmuseum@ucdavis.edu or telephone (530-752-0493.
(Part 2 of the open house will be published Friday, Sept. 29)







- Author: Kathy Keatley Garvey
When the Bohart Museum of Entomology hosted an open house, "An Evening at the Museum," on Saturday, July 22, showcasing moths and flies, the moth experts were there to celebrate National Moth Week, July 22-30 and the fly experts had just attended the 10th International Dipterology Congress, held July 16-21, in Reno.
But just as the Bohart Museum houses a global collection of 8 million insect specimens, international scientists were there representing a number of countries, including Greece, Ukraine, Iran and Spain, as well as the United States.
Among the fly experts participating in the Bohart Museum's open house was tephritid fruit fly expert Nikos Papadopoulos of Greece, professor of applied entomology, who directs the laboratory of Entomology and Agricultural Zoology at the University of Thessaly. He shared a video ontephritid flies and answered questions. Professor Papadopoulos obtained his PhD in 1999 (Aristotle University of Thessaloniki), and did postdoctoral work (2001-2003) at UC Davis, before joining the University of Thessaly. He collaborates with UC Davis distinguished professor James R. Carey. (See his biosketch)
Postdoctoral researcher Severyn Korneyev, a Ukrainian entomologist who studies flies, showed specimens and fielded questions. He holds a joint postdoc position with the UC Davis Department of Entomology and the California Department of Food and Agriculture. A member of the Ukrainian Entomological Society and the Entomological Society of America, Korneyev specializes in the systematics and taxonomy of the true fruit flies (Diptera: Tephritidae). His expertise includes morphological and molecular diagnostics, collection management, and the field collecting of insects. (See Bug Squad blog)
Professor Miguel Angel Miranda of the University of the Balearic Islands (UBI), Spain, showed specimens and led insect-drawing workshops. A zoologist, entomologist and noted insect illustrator, he currently teaches zoology, parasitology, and biotechnology applied for pest control. He is a member of UBI's Applied Zoology and Animal Conservation Research Group or ZAP. See Bug Squad blog)
Iranian-American scientist Nazzy Pakpour, who received her bachelor's degree in entomology from UC Davis and her doctorate in microbiology, virology, and parasitology from the University of Pennsylvania, read from her newly published children's book, Please Don't Bite Me. Her UC Davis connections including serving as a postdoctoral scholar in the mosquito research lab of Professor Shirley Luckhart, now with the University of Idaho.
The Bohart Museum, established in 1946 by noted entomologist Richard M. Bohart of UC Davis, is directed by UC Davis distinguished professor Lynn Kimsey. It is located in Room 1124 of the Academic Surge Building, 455 Crocker Lane. The insect museum is open to the public for summer hours from 2 to 5 p.m. on Tuesdays. It also maintains a live insect petting zoo (Madagascar hissing cockroaches, stick insect and tarantulas) and an insect-themed gift shop.










