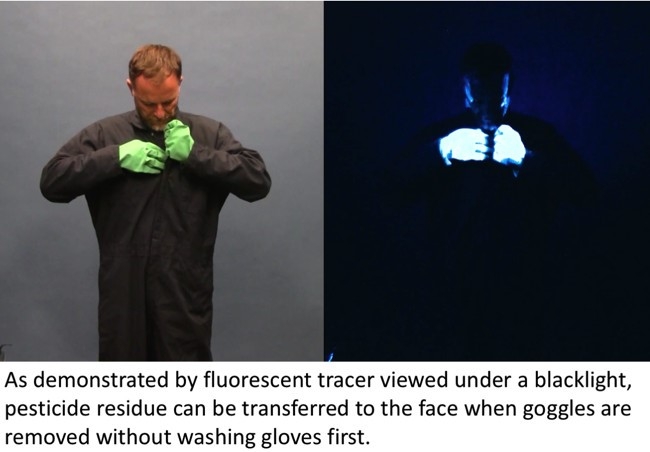
- Author: Cheryl Reynolds
Spring is in full swing and summer is right around the corner. If you work in agricultural, turf, landscape, or structural settings, you are probably at your busiest. If you handle pesticides as part of your work, you most likely wear some sort of personal protective equipment (PPE). However, do you know if you are wearing the right type for the job that you do? Wearing the appropriate PPE, taking it off the right way, and correctly cleaning it prevents unnecessary pesticide exposure to yourself and others. Learn the steps so you don't expose your family members or those around you to pesticide residues by viewing a brand new online course on Proper Selection, Use, and Removal of Personal Protective Equipment from the UC Statewide IPM Program (UC IPM).
The courseis approved by the California Department of Pesticide Regulation (DPR) for 1.5 hours in the Laws and Regulations category. This course is designed for all pesticide handlers with the goal to provide them with information on pesticide labels and the California Code of Regulations (CCR) to help them select, wear, remove, and dispose of or store PPE.
In California, all pesticide handlers (applicators, mixers, loaders, those who transport pesticides, or those who fix application equipment) are legally required to wear PPE. However, in order to get the most protection from PPE, it must be used correctly. Violations involving the incorrect use of PPE were the second most commonly reported type of agricultural-use violation in 2017 as reported by DPR (PDF).
The new PPE online course opens with a scenario describing a real example of an accident reported to DPR that led to an incident of pesticide exposure because the correct eye protection was not worn. The content that follows is divided into six instructional modules, highlighting types of PPE, how to select it, and when certain items should be worn. Answer short questions about the different types of PPE. Open pesticide labels to learn how to select the right PPE and learn when certain items should be worn. Short how-to videos and animated sequences demonstrate the proper way to put on or remove items such as gloves, coveralls, respirators, and eyewear. You must pass a final test with 70% or higher to receive your certificate of completion and continuing education hours.
If this is the year to renew your license with DPR, get a jumpstart on it. Take this new course and all the other UC IPM online courses to refresh your knowledge and get the CEUs you need. There is a $30 fee for taking Proper Selection, Use, and Removal of Personal Protective Equipment. You are welcome to view the content for free on YouTube, but without the activities, final exam, and continuing education credit. For more information about license renewal, visit DPR.
Some screenshots from this course are shown below.

- Author: Ian Grettenberger
- Author: Charlie Pickett
- Author: Brian Hogg
- Author: Frank G Zalom
- View More...
With warming weather come contemplations of bagrada bug. This is the time in the past when we start to find bagrada bug populations on flourishing shortpod mustard plants or patches of pepperweed in the Salinas Valley. We thought this would be a good time to update everyone on continuing work associated with long-term management of the bagrada bug, as we begin another year of a field study looking for resident natural enemies that target bagrada bug eggs in California. We also wanted to remind everybody to keep an eye out for bagrada bug and provide a quick reminder about where to look and what to look for. If you do find substantial bagrada bug populations or have problems with bagrada bug damage in your fields in the Salinas Valley, please reach out to Alejandro Del-Pozo (adelpozo@ucanr.edu, 831-759-7359). We'd love to get a better handle on the situation in the area to better serve you. We (Alejandro) have been scouting for bagrada bug on weeds near San Ardo for the past three weeks. Thus far, there have been no bugs or damage.
First, for an update on our work on biological control. As part of a grant from California Department of Food and Agriculture's federally funded Specialty Crops Block Grant Program, and support from Farm Bill funds, a number of researchers (CDFA, USDA-ARS, UC Davis, and UC Riverside) are surveying for resident parasitoid wasps that attack bagrada bug eggs. The long-term goal is to support the introduction of parasitoids from the bagrada bug's native range (e.g., Pakistan) in a classical biological control program that will suppress this pest across the landscape. As far as stink bugs go, the bagrada bug is a peculiar beast when it comes to laying its eggs. Rather than sticking them to leaves in clumps like other stink bugs, they instead lay the majority of them in the soil (Fig. 1, video below).
Video of a bagrada bug laying an egg into the soil
Before we introduce any parasitoids, we first need to know what parasitoids and predators already attack bagrada bug eggs in California. To address this, we are placing eggs glued to cards in the field both in the plant canopy and on the ground and in both patches of weeds (non-crop areas) and in cruciferous crop fields (Figs. 2-4). We put these sentinel eggs (generally freeze-killed) out for several days and then bring them back into the lab to allow any parasitoids in the eggs to develop and emerge. We also check the eggs for signs of predation because natural enemies such as ants, earwigs, or ground beetles could feed on the eggs and destroy them. Our survey sites cover a region from the Sacramento/North Bay area south to San Ardo and Fresno (Fig. 5).



What have we found? Thus far (2017), we have found very little parasitism by natural enemies in the Central Coast and in the Central Valley. Higher levels of parasitism have been recorded in southern California. We have recovered parasitized eggs from only four of the hundreds of cards we have deployed in central and northern California. We recovered one species, a member of the genus Ooencyrtus, from one card in the Davis area (Fig. 6, three individuals), another Ooencyrtus individual from near Winters, and Trissolcus erugatus (one individual) from the Petaluma area. For one of the cards at the same Davis site, eggs were parasitized, but adult wasps did not successfully emerge. Notably, work further south by Dr. Thomas Perring at UC Riverside has discovered greater levels of parasitism. Predation varied at our sites and ranged from none at all to nearly all eggs destroyed (Fig. 7). Time-lapse cameras at a few of our sites helped capture these predators in action (videos below). Many of these eggs were easily accessible on the cards (vs. buried in soil or crevices), so this is likely an optimistic estimate of natural levels of predation on bagrada bug eggs. However, covering eggs with 1 or 2mm of sand for some cards did not cause predation to drop off. There are clearly predators out there in the landscape that are willing and able to eat bagrada bug eggs.

Earwig feeding on sentinel egg
Rove beetle feeding on sentinel egg
Ants feeding on sentinel eggs

What to do with respect to scouting? We have recommended in the past that cruciferous weeds near fields be scouted for bagrada bug (see this previous post for more information about the association between bagrada bug and non-crop hosts). Now is the time of year to start doing this to get an idea of what kind of bagrada bug pressure you might be facing. We don't know exactly how far bagrada bugs will travel to invade fields, but checking likely areas within several hundred yards to a half a mile will give you a good idea of what is happening nearby. Shortpod mustard and perennial pepperweed are the two species to pay attention to, so look for areas with these plants. You want to mainly look for damage (Figs 8 and 9), but also for the insects themselves. Damage on leaves is often much easier to find than bagrada bugs. This is especially true at low population densities, although finding bagrada bugs will confirm it is not simply harlequin bug damage (damage is similar). For damage, you can scout at any time of the day. For the bugs, the warmer the better because they become more active at higher temps. See the weed post linked above for pictures of what these plants look like if you are not already well-acquainted with them and to see additional pictures of damage on weeds. Fresh damage consists of still-green starbursts, which will turn white as it ages. Obviously also keep an eye on your newly planted crops for damage, but this nearly goes without saying.
- Author: Alejandro Del Pozo-Valdivia
I have been receiving phone calls about the presence of “red aphids” in organic romaine lettuce that is resistant to the lettuce aphid, Nasonovia ribis-nigri (Hemiptera: Aphididae, Fig. 1). The lettuce aphid is considered a difficult pest to manage. This aphid can be green as well as red, and will be usually found in large colonies inside the heart of the lettuce head. Being inside the lettuce head protects these aphids from predators and parasitoids; and their presence is considered a contaminant when heads are harvested.

However, none of the samples that were submitted to the UC Cooperative Extension diagnostic laboratory were identified as the lettuce aphid. What are those “red aphids”?
First of all, aphids could have different color morphs within the same species. Therefore, relying only on color to identify aphids is not the ideal character to tell apart these pest species.
Submitted “red” aphid samples were identified as either: 1) the potato aphid, Macrosiphum euphorbiae, and 2) the foxglove aphid, Aulacorthum solani. Potato aphid has both the green and the red color morphs (Fig. 2). On the other hand, the foxglove aphid is usually green (Fig. 3); but infections of a naturally occurring Entomophthora fungus will make the specimens look like reddish. This sort of red coloration is due to the sporulation of the fungus from the dead aphid stuck on the leaves (Fig. 4).



If you are managing organic lettuce with resistance to the lettuce aphid and you find red aphids, it is important to consider other features beside color. Additional information on how to identify aphid species infesting lettuce can be found at http://ipm.ucanr.edu/PMG/selectnewpest.lettuce.html. If you would like to have a second opinion on your identification, you can always call and/or send your sample to the UC Cooperative Extension office in Salinas.
- Author: Richard Smith
- Author: Timothy K Hartz
- Author: Daniel Geisseler
- Author: Patricia Love
Summary
These studies provided insight into nitrogen (N) management of leafy green vegetables. Organic fertilizer evaluations showed that 4-4-2 and 12-0-0 released about a third of the N when left on the soil surface, and 54 and 86% over the course of the cropping season when incorporated into the soil, respectively. Phosphorus release from 4-4-2 was low due to the high pH's of soils at all sites. Quantities of N applied as fertilizer ranged from 1.4 to 3.9 times crop N uptake, but discounting the amount of N that actually mineralizes from the organic fertilizer, N applications ranged from 0.4 to 1.9 times crop N uptake. This observation indicates that many of these crops relied on other sources of N to meet crop needs. Quantities of N mineralized from soil organic matter were modest for each site, and the quantity of N in irrigation water was also low at 9 of the 10 sites. Residual quantities of mineral N in soil were high at most sites and may have been a key source of N for crop growth. Preplant measurements of residual mineral N using the nitrate quick test may be a useful means of understanding if fertilizer rates can be reduced.
Introduction
Organic production in Monterey County was valued at $365 million in 2016, which was 8.6% of total agricultural value. Organic agriculture has a prominent role in Monterey County agriculture, but many aspects of organic production are still poorly understood and are in need of more research. The transformations of N that occur in soil, and organic amendments and fertilizers that ultimately produce plant available forms of N (nitrate and ammonium, a.k.a. mineral nitrogen) are only understood in a general way. In the 2016 and 2017 seasons we conducted evaluations of N mineralization (release of mineral N from soil organic matter) from soils on organic farms and from organic fertilizers. Evaluations included determination of the mineralization of soil organic matter over the course of the cropping cycle of baby vegetables and full-term romaine and broccoli. In addition, evaluations were conducted of mineralization of organic fertilizers in production fields as well as laboratory incubations.
Predicting response to applied N
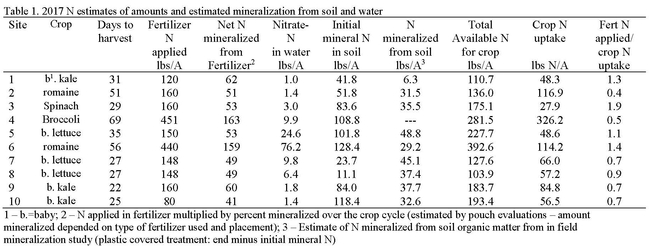
In 2016 and 2017, twenty N mineralization trials were conducted in the Salinas Valley with cooperating growers in commercial production fields in diverse locations and with a variety of soil types. Field evaluations consisted of establishing replicated unfertilized areas in each field. Within each unfertilized plot, we had areas with and without plants, as well as an area covered with plastic mulch; these areas allowed us to estimate the amount of N mineralized from soil organic matter without the confounding factors of crop uptake and leaching. Unfertilized areas were compared with adjacent fertilized plots. In 2017, the amount of N mineralized in field evaluations ranged from 6.3 to 45.1 lbs N/A over the course of the crop cycle and was generally modest in comparison to crop uptake (Table 1). In addition, net N mineralized from applied organic fertilizer was less than crop uptake at 6 of the 10 sites. Nitrate-N in the irrigation water was low at all sites except site 6.
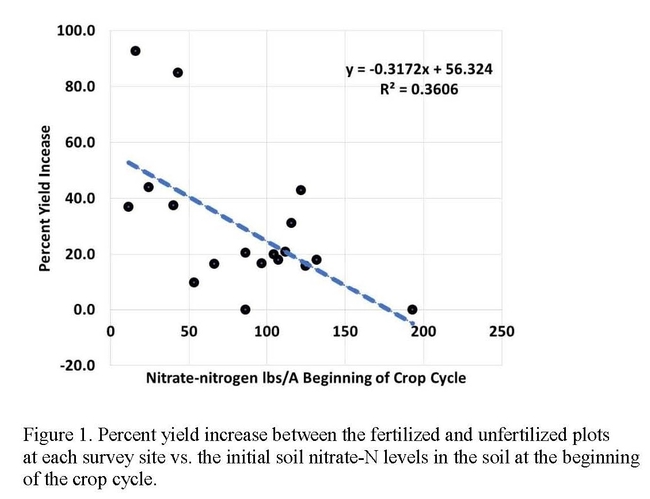
In comparison, residual soil nitrate-N at the beginning of the cropping cycle was quite high at all but two sites. Given the short crop cycle for most of the crops evaluated in these studies and the lag in the releases of mineralize N from organic fertilizers, the question arises, is a measure of nitrate-N at the beginning of the crop cycle a useful indication of the amount of N available for crop growth. For baby vegetables that mature in approximately 30 days, a preplant evaluation of the available pool of nitrate is the only reasonable time to take this measurement because the lag time in the release of nitrate from organic fertilizers makes it impractical to test later in the crop cycle. The disadvantage of relying upon a preplant evaluation of soil nitrate is that leaching with the germination water could move the measured nitrate beyond the reach of the developing crop. We observed modest yield increases even at sites with 80 to 130 lbs of residual nitrate-N/A prior to planting (Figure 1). From these studies, it appears that the utility of the preplant nitrate-N test is not in being able to skip a fertilizer application, but rather with adjusting the quantity of fertilizer applied. How much the quantities of applied N can be reduced is a question that needs further study.
Evaluations of organic fertilizers
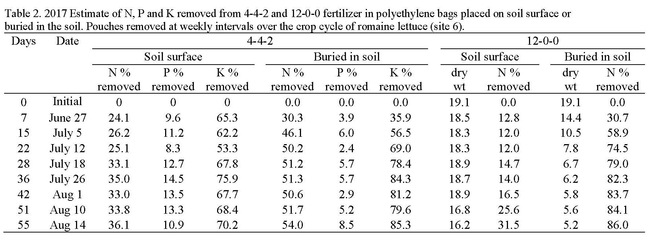
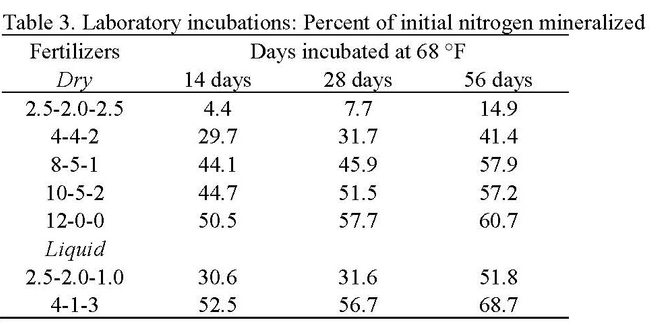
Organic fertilizers used in the production of cool season vegetables are applied by either incorporation into the soil or applied to the top of the bed (a.k.a. drop-on-top) and incorporated with sprinkler irrigation. We mimicked these application methods in studies in which we placed organic fertilizers in polyethylene pouches (See photos below) and applying them to the soil in two ways: 1) burying them 3 inches deep to simulate incorporated applications and 2) by placing the pouches on the soil surface to simulate surface applications. The 2017 evaluations were conducted in a commercial production field and the pouches were placed in the field at the beginning of the crop cycle. Four pouches were collected weekly and the contents were dried, weighed and analyzed for N (12-0-0 only) and for phosphorus (P) and potassium (K) (4-4-2); the rate of disappearance of nutrients from the pouches estimated the rate of plant-availability. Organic fertilizers used were 4-4-2 (chicken, meat and bone meal) and 12-0-0 (feather meal). The pouches were placed in the soil at the beginning of the crop cycle of full term romaine on June 19 and the final set was removed on August 14. Surface applied 4-4-2 released 24.1% of its N in the first week of the evaluation and then slowly released a total of 36.1% over the next 48 days (Table 2). Buried 4-4-2 released 30.3% of its N in the first week and a total of 54.0% over the next 48 days indicating that burying the fertilizer released greater quantities of N faster than surface applications. Surface applied 12-0-0 released 12.8% of its N in the first week of the evaluation and released a total of 31.5% over the next 48 days. Buried 12-0-0 released 30.7% of its N in the first week and a total of 86.0% over the next 48 days. Burying 12-0-0 had a more dramatic impact on the release pattern of N than for 4-4-2. The amount of P released from 4-4-2 in surface and buried applications was 10.9 and 8.5%, respectively which was low. The amount of K released from the surface and buried applications was 70.2 and 85.3%, respectively, which was high.
Theseevaluations show a rapid release of N from the organic fertilizer over the first 1 to 2 weeks after application. After that time, the rate of release of N slows. Burying the material increased the rate of N release and particularly so with 12-0-0, indicating that fertilizer with a higher N concentration release N faster. It is possible that the pouch evaluations overestimate the N release rate because of movement of small particles of fertilizer that filter out of the pouch; this idea is supported by observations in a laboratory evaluation conducted at UC Davis that showed a 20-30% lower rate of N release of 4-4-2 and 12-0-0 over the same period of time (Table 3).
The amount of N applied as fertilizer ranged from 1.4 to 3.9 times crop N uptake. However, counting only the fraction of organic fertilizer N assumed to be mineralized, N applications ranged from 0.4 to 1.9 times crop N uptake. The net amount of fertilizer N made available from the organic fertilizer was less than crop uptake in 6 out of the 10 fields. This data would suggest that the organic crops were commonly under fertilized, but tissue N evaluations at harvest indicate that only one of 20 fields evaluated might have been low in nitrogen. It appears that the crops were getting their needs met from the other sources of N: residual pool of nitrate, in-season mineralization and nitrate in the irrigation water.
Soil pH at the evaluation sites ranged from 7.4 to 8.1, indicating that measurement of soil Olsen P was the appropriate means of measuring available P. Soil P values at the sites ranged from 21 to 51 ppm with a mean of 38. These P values are similar to values encountered in comparable commercial production fields (mean of 37 ppm P). Quantities of elemental P applied to these crops ranged from 35 to 86 lbs P/A (80 to 200 lb P2O5/A), which is quite high. Six of the ten sites have been in organic production for more than 10 years, and assuming similar fertilizer practices occurred over this period of time, it is surprising that the levels of P in the soil have not risen to higher levels. We attribute this lack of soil P enrichment to the fact that P in the form of bone meal remains unavailable in high pH soil. The fertilizer pouch study confirmed that only a small fraction of the P in the 4-4-2 fertilizer was released from the pellets.


- Author: Jim Correll
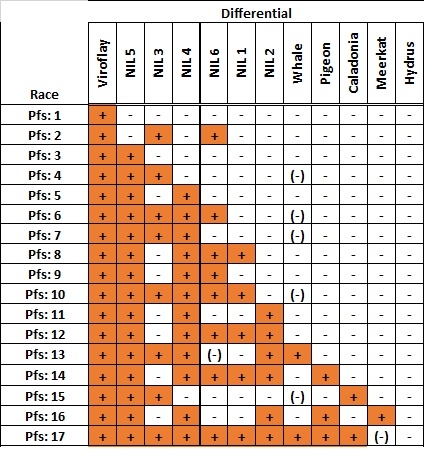
A new race of the downy mildew pathogen (Peronospora farinosa f. sp. spinaciae = P. effusa) on spinach was first identified in 2014 in Yuma, Arizona, U.S. This race was able to overcome the resistance of important spinach varieties. First, one isolate (UA1014), was characterized on a standard set of differential varieties. Subsequently, isolates with the same reaction pattern on these differential varieties have been found in numerous locations and each year since 2014. After careful evaluation of the significance of this development to the spinach industry, the International Working Group on Peronospora in spinach (IWGP) has denominated isolate US1602 as Pfs: 17. This isolate is available at Naktuinbouw in The Netherlands.
Race Pfs: 17 is able to infect the differentials NIL1-6, Whale, Pigeon, and Caladonia. Meerkat has intermediate resistance. The reaction of Meerkat can be variable in seedling tests. Hydrus is resistant.
The IWGP is continuously monitoring the appearance of strains of the pathogen that deviate in virulence from the known races. In this way the IWGP aims to promote a consistent and clear communication between public and private entities, such as the seed industry, growers, scientists, and other interested parties about all resistance-breaking races that are persistent enough to survive over several years, occur in a wide area, and cause a significant economic impact.
The IWGP is located in The Netherlands and is administered by Plantum NL. The IWGP consists of spinach seed company representatives (Pop Vriend, Monsanto, RijkZwaan, Bayer, Takii, Sakata, Bejo, Enza, Syngenta, and Advanseed) and Naktuinbouw, and is supported by research centers at the University of Arkansas. Spinach researchers over the world are invited to join the IWGP initiative and use the common host differential set to identify new isolates.
For more information on this subject you can contact Jim Correll (jcorrell@uark.edu), Diederik Smilde (d.smilde@naktuinbouw.nl), or the IWGP chairperson Jan de Visser (JandeVisser@popvriendseeds.nl).
Attached:
Table with disease resistance reactions of spinach downy mildew races on IWGP differentials. Differentials and type isolates are available at Naktuinbouw in The Netherlands.