- Author: Mark Bolda
- Contributor: Alejandro Del Pozo-Valdivia
- Author: Shimat Villanassery Joseph
- Author: Mark Bolda
Lygus bug (Lygus hesperus) (Fig. 1) is a major pest of strawberry in the Central Coast. Lygus bug populations develop on weed hosts surrounding the strawberry fields such as wild radish, common groundsel, lupines, and mustards (Zalom et al. 2012). Time to time, adults migrate into the strawberry fields and lay eggs. Eggs hatch, and molt through five nymphal stages before molting into adults. Lygus bug feeding on the developing embryos affects the normal development of tissues surrounding the embryo (Handley and Pollard 1993) and affected fruits are misshapen often referred as “catfaced fruit” (Fig. 2) which are deemed unmarketable. Although both nymphs and adults can cause catface injury, nymphs are considered more destructive than adults. The young fruits up to ~10 days after petal fall are considered vulnerable to economic injury from lygus bug feeding (Zalom et al. 2012).
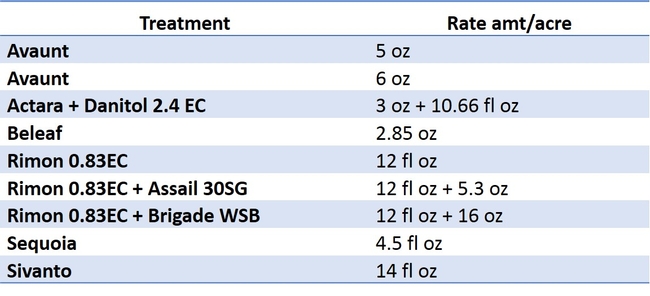
Chemical control continues to be an effective tool for lygus bug control and growers are always seeking effective and softer insecticides for its control. A replicated trial comparing the efficacy of insecticide treatments against lygus bug was conducted in first-year strawberry ‘San Andreas' in Watsonville, CA in 2016. The details on insecticide products and rates used in the trial are shown in Table 1. The insecticides were applied twice at 10 day interval using commercial tractor mounted sprayer. The water volume used for both the applications was 150 gal per acre and was applied at 140 psi. Dyne-Amic (surfactant) was added at 0.25% v/v to all the treatments. Insect samples were collected using regular sized Rubbermaid container by hitting 20 flowering strawberry plants with lid. In addition, 60 fruits were sampled from each plot to determine catface injury.
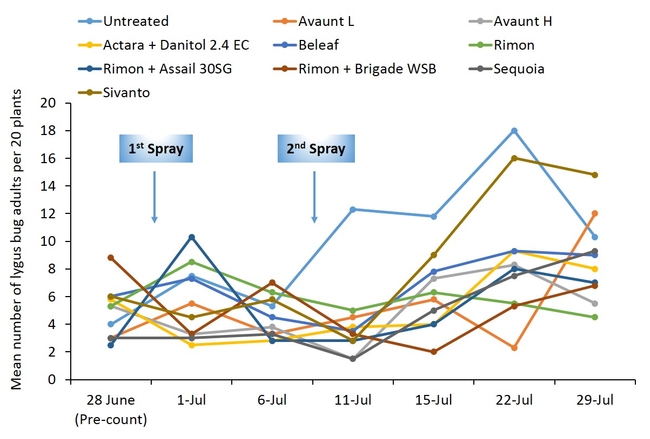
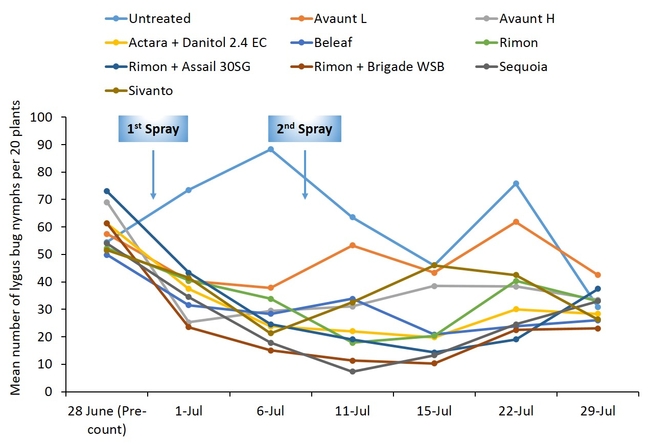
Pre-count sample did not show any difference in number of adult and nymphal lygus bugs among treatments (Figs. 3 and 4). Overall, all the insecticide treatments reduced the number of lygus bug adults and nymphs compared with untreated plants. The combination treatments using pyrethroid insecticides such as Danitol and Brigade suppressed lygus bugs and general predators such as bigeyed bug, minute pirate bug, and damsel bug as well as spiders (Figs. 5-8). Data show that reduced-risk insecticides, Rimon and Beleaf suppressed lygus bug nymphs as well. Sequoia, not yet registered on strawberry, provided a decent lygus bug control. Sivanto initially provided a good suppression of adults and nymphs but could not adequately sustain the control for more than a week. Two rates of Avaunt (unregistered insecticide on strawberry) was included in this experiment and were comparable to other effective insecticides in this experiment.
Insecticide use certainly reduced catface injury on strawberry fruit. Number of fruits with catface injury was lower in all the insecticide treated plants than untreated except the lower rate of Avaunt (Fig. 9). Catface injury on fruits treated with Sequoia was lower than untreated but not different from other insecticides (except lower rate of Avaunt).
References
Handley, D. T., and J. E. Pollard. 1993. Microscopic examination of tarnished plant bug (Heteroptera: Miridae) feeding damage to strawberry. J. Econ. Entomol. 86: 505-510.
Zalom, F. G., M. P. Bolda, S. K. Dara, and S. Joseph. 2012. Strawberry: Lygus bug. UC Pest Management Guidelines, UC ANR Publication 3468. http://www.ipm.ucdavis.edu/PMG/r734300111.html

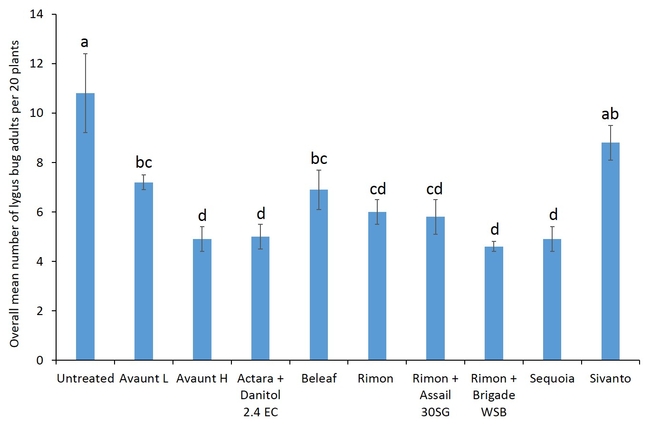
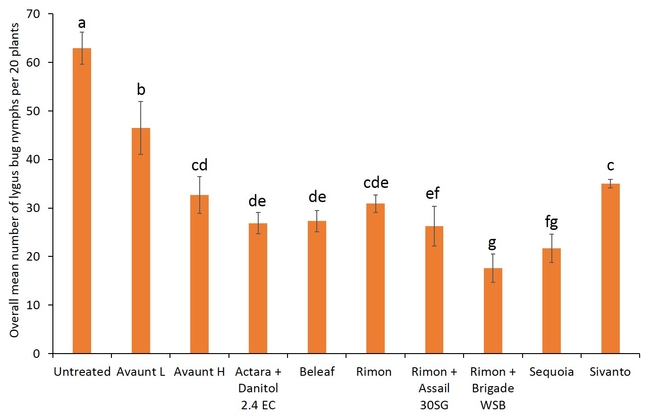
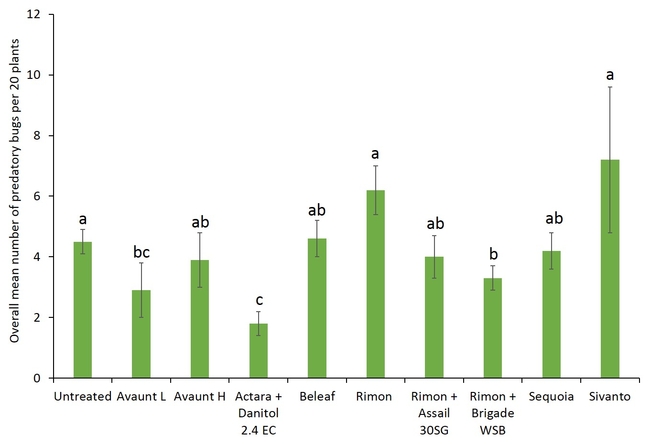
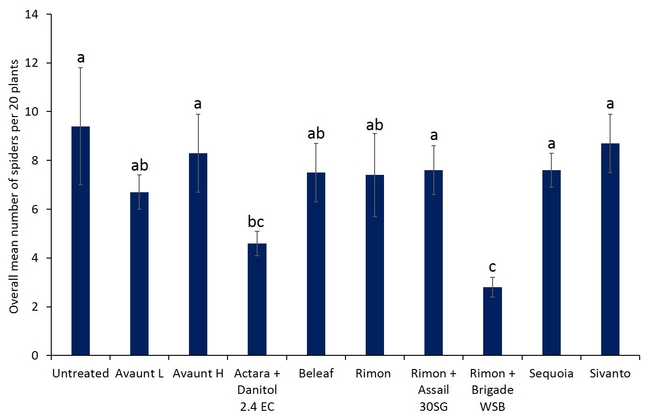

- Author: Shimat Villanassery Joseph
- Author: Mark Bolda
Two spotted spider mite (Fig. 1A) is a major pest of strawberry and caneberries. The spider mites lay 0.14 mm eggs (in diameter) on the undersides of strawberry leaves. Once the eggs hatch and the larvae go through three stages and molt into adult males, and reproductive females. As the name suggests, they are called two spotted spider mite because of one or more dark spots on the bottom half of each side of the body. They are typically dull or yellow colored organism but during the winter months, they undergo diapause (a resting stage) and appear as reddish or orange color (Fig. 1B) which often is confused and misidentified as carmine mite or presumably a new mite never seen before. Carmine spider mite is a bright red colored mite (Fig. 1C) but do not have spots on the either side of the body. They rarely cause economic injury to strawberry. They occurring during the winter and spring months in the Salinas/Watsonville strawberry fields. Predatory mites are also greyish, pale reddish or orange colored in general but they are shiny and moves very fast on the leaves (Video).
For further reading click the Pest Management Guidelines for mites in strawberry (http://www.ipm.ucdavis.edu/PMG/r734400111.html)
- Author: Shimat Villanassery Joseph
- Author: Mark Bolda
Lygus bug (Lygus herperus) (Figure 1a) continues to be a major pest of strawberry in northern Central Coast of California. The feeding injury on the young developing fruits results in catfaced fruits (Figure 1b) rendering them unmarketable. Management of lygus bug in strawberry is always been a challenge because of lack of effective insecticides with desirable attributes such as short pre-harvest intervals (PHIs). Moreover, the insecticides registered on strawberry for lygus bug control have been used over and over in the same season and it is likely that lygus bug developed resistance to those insecticides.
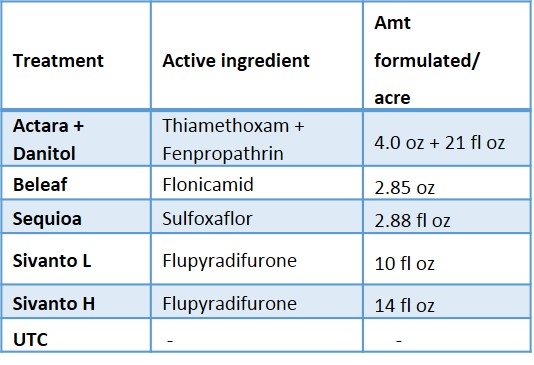
In 2015, an insecticide trial was conducted to evaluate the efficacy of newer insecticides registered on lygus bug. The details on insecticide products, active ingredients and rates are shown in Table 1. The newer insecticides tested were Sivanto and Sequoia. Sivanto is registered on strawberry with the maximum rate 14 fl oz per acre. Sequoia is not registered at this moment and rate tested is 2.88 fl oz per acre which is lower than 4.5 fl oz or 5.75 fl oz per acre tested in the previous years. The study was a replicated (5 replications) and the treatments were randomized. The plot size was ten 65-feet long beds which is fairly large for insecticide trial in a commercial strawberry field. First broadcast-spray application of insecticides was done on 13 June 2015 followed by a second broadcast-spray application on 20 June 2015. The insecticides were applied using commercial tractor mounted sprayer. The water volume used for both the applications was 200 gal/ acre. Dynamic (surfactant) was added at 0.25% v/v.
Beat-trays were used to sample insect populations (Figure 2). Twenty strawberry plants were sampled and the sampling consists of five strikes per plant with the lid of a regular sized Rubbermaid container. Sampling was done a day before application then at 3 and 7 days after first application then 3, 7, 14, 21, and 28 days after second application. The insect samples were bagged, transported to the laboratory and stored in the freezer for later evaluation in the laboratory. The samples were evaluated for all nymph stages and adult of lygus bug, thrips, predators (damsel bug, minute pirate bug, bigeyed bug, rove beetle, and spiders) (Figure 3) and parasitoids. In addition, 100 fruits were randomly sampled from each plot at 28-days after second insecticide application. The fruits were evaluated for lygus bug injury or “catface” and other unmarketable symptoms such as rot, spit strawberries etc.
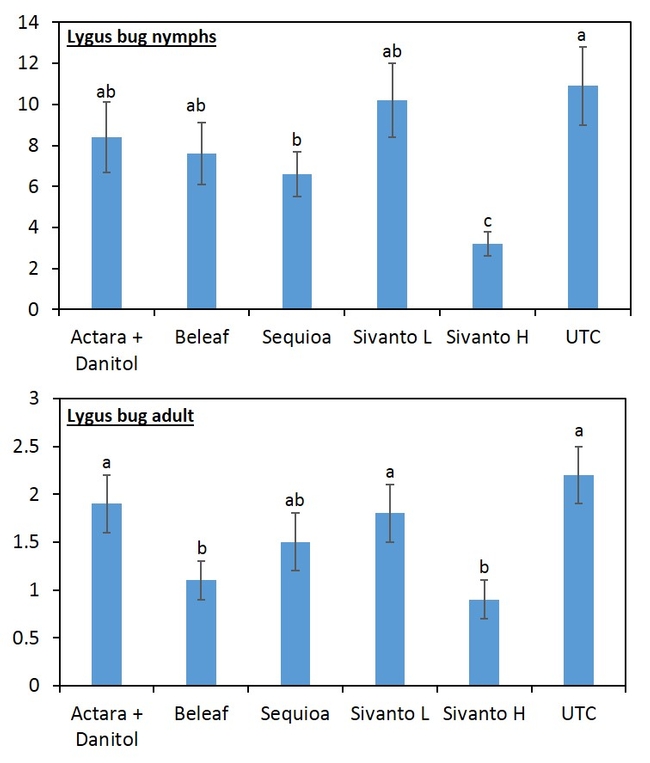
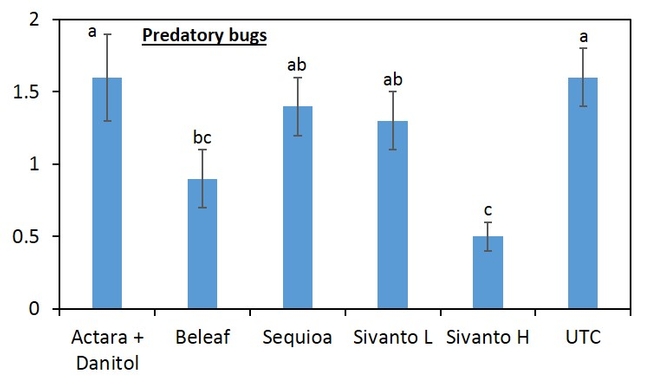
When all the data were combined, number of lygus bug nymphs were lower in the higher rate of Sivanto and Sequoia than in untreated check treatment (Figure 4). Lower number of lygus bug adult was captured in higher rate of Sivanto and Beleaf than in untreated check. Similarly, number of predatory bugs was lower in the higher rate of Sivanto than in other treatments. Spiders captured were similar among treatments (Figure 5).
On fruit evaluation, there was no difference in number of fruits with catface injury or those marketable fruits among the insecticide treatments, although numerically, number of fruit with catface injury was lower in the higher rate of Sivanto treatment than in other treatments.
Overall, it appears that Sivanto at 14 fl oz per acre performed better than other treatments against lygus bug. Sequoia and Beleaf also showed evidence of lygus bug suppression. However, Sivanto at 10 fl oz per acre did not suppress lygus bug. The representative industry standard -- combined treatment of Actara and Danitol did not show any evidence of lygus bug suppression in this study.
If you are interested in reading the complete report, please click the link below.



- Author: Michael D Cahn
- Author: Oleg Daugovish
- Author: Mark Bolda
Establishing strawberry transplants using drip has several potential advantages compared to overhead sprinklers. Irrigation run-off can be greatly reduced, which protects surface water quality. Some growers have found that they can save water using drip for transplant establishment, and save costs associated with using overhead sprinklers. Often nitrogen fertilizer can be spoon fed to the crop through the drip system earlier in the season than in fields established with sprinklers, thereby reducing the reliance on pre-plant fertilizers that may result in nitrate leaching losses.
Nevertheless, growers are concerned that irrigating transplants using mainly drip may result in less vigorous growth and more dead plants during the establishment period, and yields during the production season will be lower than crops established with sprinklers. One of the specific concerns is that the drip lines adjacent to plant rows may not be as effective in leaching salts from the root zone of young plants as overhead sprinklers. Another worry is that if the transplants are not properly planted and gaps exist between the root crown and the soil, moisture will not move toward and imbibe young roots, and the plant may be set back or die.
Last year was challenging for establishing strawberries is many fields on the Central Coast due to the lack of rain, which normally helps to leach salts that may accumulate around young strawberry transplants. However, these challenging conditions were perfect for comparing vigor and yield of strawberries established using drip and overhead sprinklers.
Field trial description
We conducted a demonstration trial at a ranch in North Salinas beginning November 13, 2013. Soil was a loam texture. The field was planted with UC Albion variety in 2 rows on 52-inch wide beds. Two plots, each of approximately 1-acre in size, were located adjacent to each other in one of the irrigation blocks. Transplants were established using drip in one of the plots and with overhead sprinklers in the other plot. The irrigation foreman made all decisions on how long and often to irrigate both plots. Overhead sprinklers were used for the first 2 irrigations in the drip plot to assure that the transplants were in good contact with the soil. All subsequent irrigations were made using 2 lines of drip tape per bed. In the plot established with overhead sprinklers, transplants were also irrigated twice using the drip system. The last sprinkler irrigation was on January 25th, after which both plots were irrigated with only drip.
Applied water was monitored using flow meters installed on the drip submain and on the sprinkler main line until the end of February. Soil moisture was evaluated in the upper 6 inches of soil next to the transplants using a volumetric moisture sensor at weekly intervals during establishment. Soil salinity was also periodically monitored to a 4 inch depth next to the transplants using a soil salinity sensor (Fig 1.), or by sampling soil and analyzing saturated paste extracts for salts. Plants were rated for vigor and evaluated for canopy cover until mid February (Fig 2.). Marketable fruit yield was evaluated between late April and mid July.
Figure 1. A 5TE decagon probe was used to measure bulk salinity near strawberry plants.
Figure 2. Transplants were periodically evaluated for canopy size by measuring plant width or using a multi-spectral NDVI camera.
Results
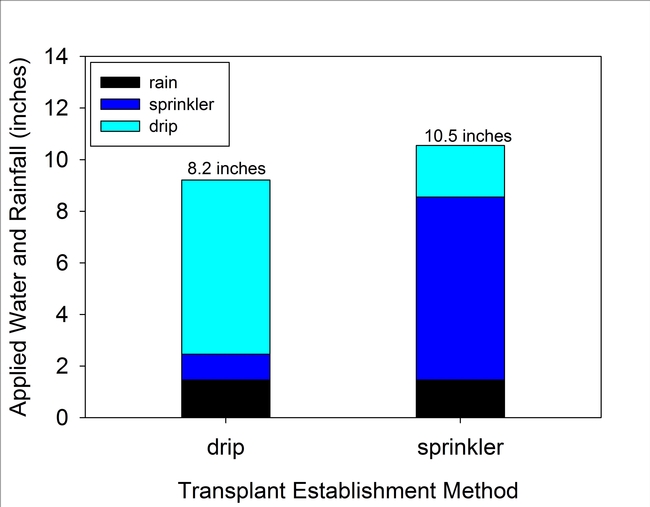
Applied water during transplant establishment was about 25% less for the drip treatment compared to the standard sprinkler treatment. As shown in Fig. 3, irrigation water applied to the drip treatment equaled 6.8 inches between Nov. 13 and Feb. 20th. During the same period, water applied in the sprinkler treatment equaled 9.1 inches. An additional 1.5 inches of rainfall were also measured during this period. Estimated evapotranspiration (ET) losses during establishment were 3.2 and 1.1 inches for the sprinkler and drip treatments, respectively. The lower estimated ET amount for drip was due to less wetting of the furrows than in the sprinkler treatment.
Figure 3. Irrigation water and rainfall for the drip and sprinkler treatments between Nov. 13 and Feb. 20th.
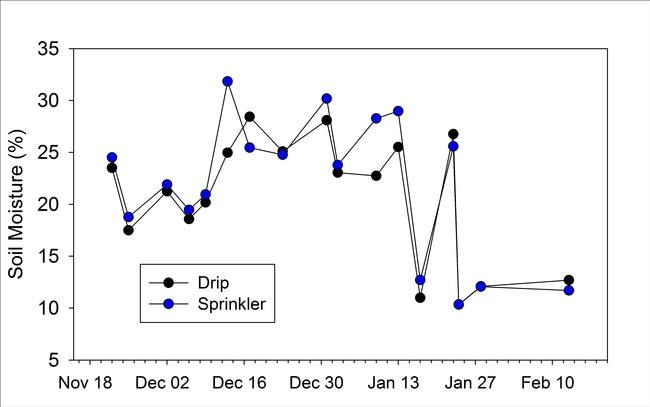
Soil moisture measured near the transplants was similar among the drip and sprinkler establishment treatments except for 4 dates between mid December and mid January when the sprinkler plot had higher soil moisture levels than the drip plot (Fig 4).
Figure 4. Volumetric soil moisture measured next to transplants for the drip and sprinkler treatments.
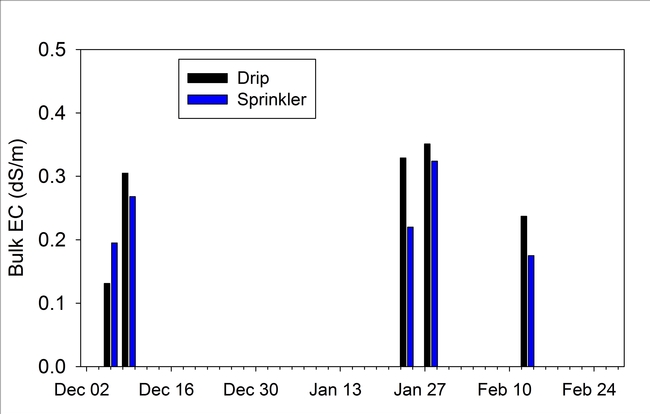
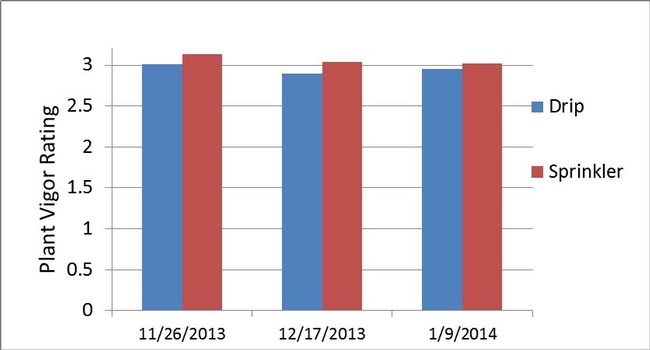
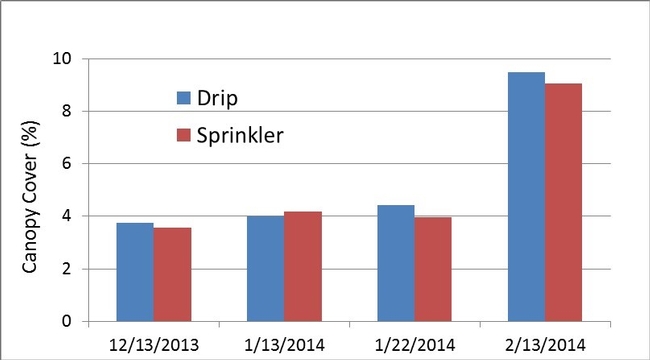
Bulk electrical conductivity (EC), an indirect measure of soil salinity, was slightly higher next to the drip established transplants than the sprinkler established transplants for 4 of 5 dates measured between December and early February (Fig. 4). Bulk EC values were generally low for both treatments, which was confirmed from the saturated paste extracts of soil sampled on January 24 2014 (3rd evaluation date). The EC of the saturated paste extract was 0.80 and 1.83 dS/m, respectively, for the sprinkler and drip established plots. Saturated pasted extract values below 2 dS/m would not be expected to harm strawberry plant growth. Plant vigor (Fig. 5) and canopy cover (Fig. 6) were not different between the sprinkler and drip establishment treatments. Likewise cumulative fruit yields (Fig. 7) were the same for the two methods of irrigation establishment.
Figure 5. Bulk electrical conductivity of the soil adjacent to strawberry transplants, measured using the Decagon 5TE probe.
Figure 6. Plant vigor of drip and sprinkler established transplants, where 0 equates to dead or dying plants, and 5 signifies all plants are very healthy.
Figure 7. Percentage of ground shaded by leaves, measured using a multi-spectral infra-red camera.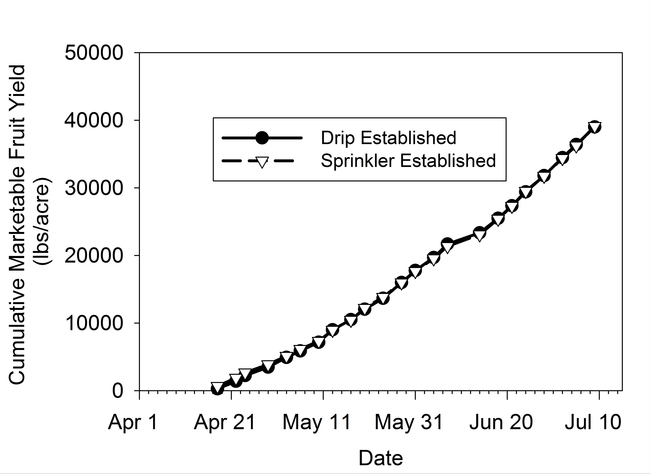
Figure 8. Cumulative marketable fruit yield for sprinkler and drip established plants.
Discussion and Conclusions
The results of this field trial demonstrated that drip can be successfully used to establish strawberry transplants during the winter on the Central Coast, even during drought conditions when rainfall is minimal. Marketable fruit yields were the same between the drip and sprinkler established plots. Additionally, 25% less water was used under drip than in the sprinkler plots during the initial establishment phase. Salinity was maintained at a sufficiently low level in the soil as to not impair transplant vigor and initial growth under drip.
Low water demand of plants during the late fall and early winter is an import factor that helped us successfully establish plants using drip. New transplants have few leaves and reference evapotranspiration, on average, is less than 0.07 inches per day. Since water demand is low, the main purpose of irrigation during establishment is to keep crown roots hydrated and to leach salts from the root zone. Using good planting techniques is critical to successfully using drip for establishment. Transplant roots need to be in contact with the soil and should not be “J” rooted. Also, soil salinity should be low as possible before planting. Preplant fertilizer bands should be located a sufficient distance from the transplant roots so that emerging new roots are not burned by fertilizer salts.
For this trial, we irrigated the drip treatment twice with overhead sprinklers to assure that the roots were in good contact with the soil. Under normal weather conditions on the Central Coast, rain often occurs between late November and February, which can also assist with the establishment of transplants by maintaining high soil moisture and leaching salts from the root zone of young plants.
Acknowledgements
We thank Dole and their employees for their help and partnership with this trial, and we thank Walmart for funding this project.